Case Report Open Access
Cardiac Cirrhosis May Present Ten Years after Pericardiectomy for Chronic Constrictive Pericarditis
Madhumita Premkumar1*, Devaraja Rangegowda1, Amrish Sawhney1, Awinash Sinha1, Tanmay Vyas1, Saloni Nitin Desai3, Shrruti Grover2, Joshi YK1 and Chhagan Bihari2
1Department of Hepatology, Institute of Liver and Biliary Sciences, Vasant Kunj, New Delhi, India
2Department of Pathology, Institute of Liver and Biliary Sciences, Vasant Kunj, New Delhi, India
3Department of Radiodiagnosis, Institute of Liver and Biliary Sciences, Vasant Kunj, New Delhi, India
- Corresponding Author:
- Dr. Madhumita Premkumar
Senior Resident, Department of Hepatology
ILBS, New Delhi 110070, India
Tel: 919540951061
E-mail: drmadhumitap@gmail.com
Received Date: July 14, 2015 Accepted Date: September 14, 2015 Published Date:September 20, 2015
Citation:Premkumar M, Rangegowda D, Sawhney A, Sinha A, Vyas T, et al. (2015) Cardiac Cirrhosis May Present Ten Years after Pericardiectomy for Chronic Constrictive Pericarditis. J Gastrointest Dig Syst S13: S13-001. doi:10.4172/2161-069X.1000S13-002
Copyright: ©2015, Premkumar M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
A 51 year old male, presented to us with progressive abdominal distention and jaundice since 1 month, and a single episode of hematemesis. He had undergone pericardiectomy 10 years back for chronic constrictive pericarditis (CCP) and had completed a course of anti-tubercular therapy following the surgery. Physical examination revealed normal vital signs and mild icterus, moderate ascites, splenomegaly, and mild ankle edema. On endoscopy, he found to have large esophageal varices and endoscopic variceal ligation (EVL) was performed. Thyroid function tests, hepatitis serologies, autoimmune assays (ANA, AMA) ASMA, ferritin, ceruloplasmin, and α1-AT, level were unremarkable. Liver biopsy showed cirrhosis. After extensive evaluation, a diagnosis of cardiac cirrhosis was made.
Keywords
Cardiac cirrhosis; Chronic constrictive pericarditis
Introduction
Constrictive pericarditis is a rare consequence of the chronic inflammation of the pericardium, leading to an impaired filling of the ventricles and reduced ventricular function, thus presenting as heart failure [1]. The symptoms of right heart failure overlap with those of decompensated liver disease. Hence a high index of suspicion for recognizing and diagnosing these cases is imperative. Early diagnosis and management of underlying cardiac disease is important because this is potentially reversible cause of cirrhosis [2]. We will briefly review the literature on cardiac causes of liver cirrhosis, and also discuss the factors causing persistence of subclinical cardiac disease after corrective surgery in CCP.
Case Report
We present a case of a 51 year old male who developed decompensated liver disease ten years after undergoing pericardiectomy for CCP. He was diagnosed with CCP ten years ago when he presented with dyspnea on exertion, ascites requiring frequent taps, low grade fever and exercise intolerance. Thoracic imaging and showed pericardial calcifications which ultimately led to the diagnosis of constrictive pericarditis. Hence he was advised pericardiectomy and the operative findings revealed thickened pericardium with calcification. The surgical biopsy specimen showed fibrinous pericarditis with calcification. It was negative for acid fast bacillus, but in view of high index of suspicion for tuberculosis, he received 10 months of anti-tubercular therapy (ATT). His symptoms entirely resolved within 6 months of surgery, and he was not not on follow up. He did not have diabetes, hypertension or thyroid disease and had no obvious symptoms for almost a decade. He had no history of alcohol consumption or toxin exposure.
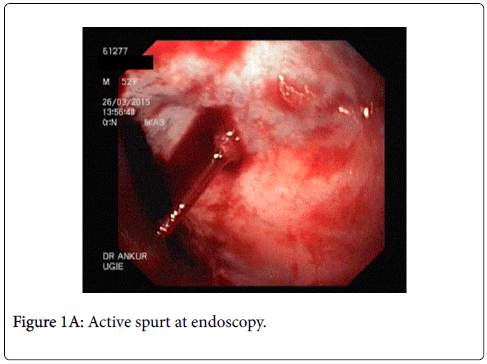
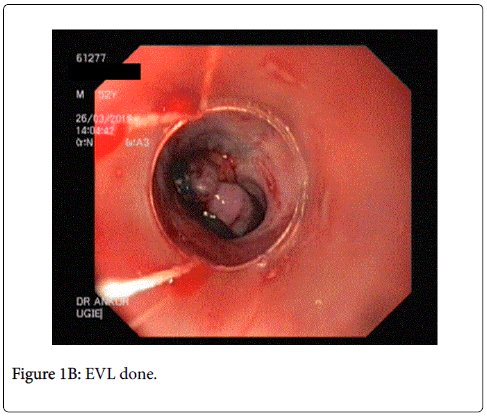
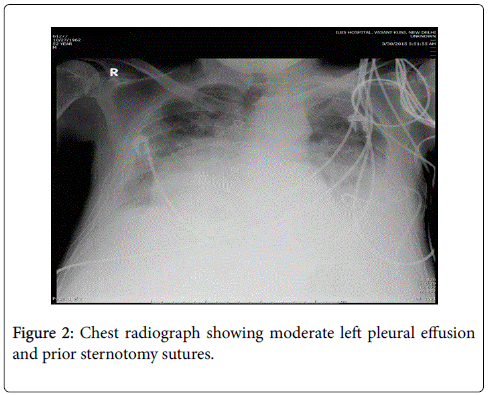
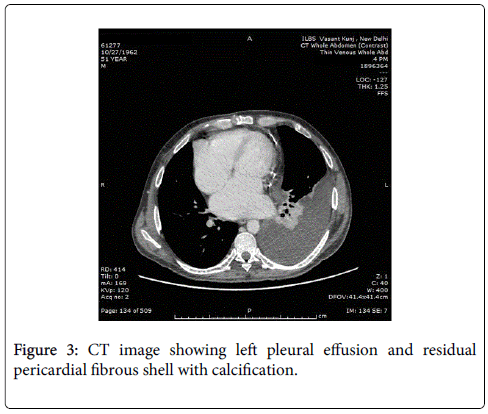
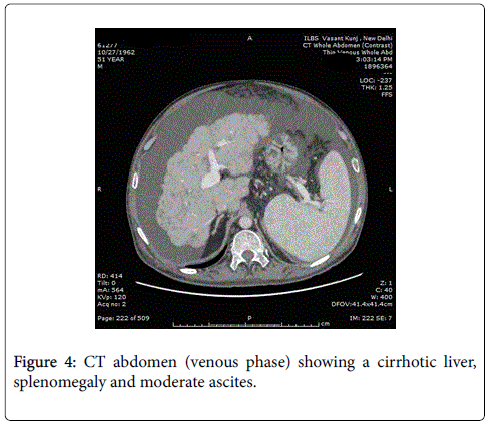
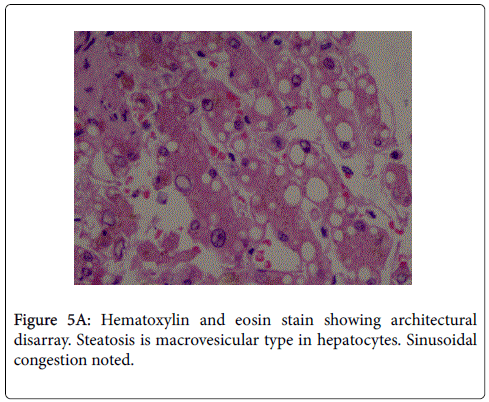
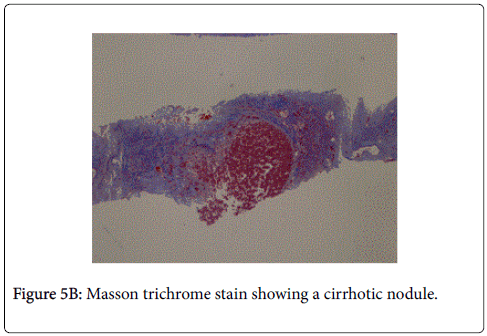
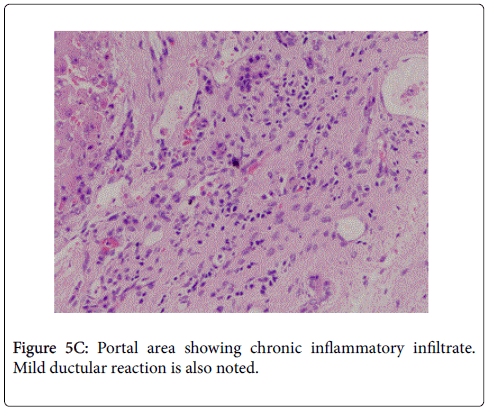
The patient presented to us in the emergency department as he had 2 episodes of hematemesis. He also reported progressive abdominal distention and non cholestatic jaundice since a month. Physical examination revealed normal vital signs, mild icterus, moderate ascites, splenomegaly, and mild ankle edema. He did not have a raised jugular venous pressure or a pericardial knock on clinical examination. A complete diagnostic evaluation was done to elucidate the cause of liver disease. His initial lab investigations showed a mild normocytic normochromic anemia. Liver function test showed a bilirubin of 4.8 g/dl with aspartate transaminase 287 IU/l, and alanine transaminase 185 IU/l, which gradually settled over time to 58 and 45 IU/l respectively. Thyroid function tests, hepatitis serologies, autoimmune assays (ANA, AMA) ASMA, ferritin, ceruloplasmin, and α1-AT, level were unremarkable. On esophagogastroduodenoscopy, he was found to have grade 3 esophageal varices with an active spurt and endoscopic variceal ligation (EVL) was performed (Figures 1A and 1B). The liver function tests were abnormal with mildly elevated liver enzymes, bilirubin of 3.4 mg/dl and hypoalbuminemia. INR was 1.79.USG abdomen showed a shrunken liver with gross ascites without any pericardial effusion. The chest radiograph showed a moderate left pleural effusion (Figure 2). About 800 ml of pleural fluid was removed which showed a high SPAG (serum pleural albumin gradient) of 1.4, protein 1.8 gm/dl and no evidence of infection. About 3 l of ascitic fluid was removed. Ascitic fluid analysis showed 200 cells, predominantly lymphocytes, with 1.4 g/dl protein and SAAG (serum ascitic albumin gradient) of 1.6. Computed tomography scan showed that the liver measured approx. 11.5-cm and showed a cirrhotic architecture with lobulated margins, and relatively hypertrophied left and caudate lobes. It showed interlacing and confluent fibrosis. No evidence of any arterial enhancing focal lesion or intrahepatic biliary radicle dilatation was noted. Hepatic veins and the spleno-portal axis showed normal contrast opacification. Both lung fields revealed no significant focal pulmonary parenchymal lesion. No evidence of pericardial effusion was seen. However the pericardial rim showed mild thickening but no overt evidence of a constrictive pathology (Figures 3 and 4). This also correlated with the echocardiographic findings of our patient. There was no evidence of pericardial effusion or constriction, but a calcified rim was visualized. Both atria were dilated but normal ventricular function was seen with no regional wall motion abnormality. After he was stabilized and ascites and effusion controlled on diuretics, he underwent right heart catheterization with showed a hepatic vein pressure gradient (HVPG) of 16 mmHg, right atrial pressure of 8 mmHg, pulmonary artery pressure of 13 mmHg, and pulmonary capillary wedged pressure of 14 mmHg. Transjugular liver biopsy (TJLB) showed sinusoidal dilation and congestive changes in addition to the presence of f3 fibrosis, indicating the cirrhosis had an underlying cardiac etiology (Figures 5A-5C). He was referred to another centre for a cardio-thoracic surgical review but there was no evidence of active constriction and conservative management was advised. Thereafter he was placed on the liver transplant waiting list. He developed an episode of spontaneous bacterial peritonitis and was managed with diuretics and antibiotics. Though his ascites and effusion was controlled, he developed coagulopathy and episodic hepatic encephalopathy. The patient succumbed to progressive liver failure as he could not find a matching liver donor.
Discussion
This case illustrates the importance of diagnosing a cardiac cause in the work-up of cirrhosis when the more common etiologies have been excluded after extensive clinical evaluation. Constrictive pericarditis occurs when a thickened fibrotic pericardium, of whatever cause, impedes normal diastolic filling and usually involves the parietal pericardium [3]. Acute and subacute forms of pericarditis may deposit fibrin and lead to exudation of a pericardial effusion. In turn this leads to pericardial inflammation, chronic fibrotic scarring, calcification, and restricted cardiac filling [4]. In India the most common cause of constrictive pericarditis is tuberculosis [5]. Other causes include pericardial involvement from cardiac trauma (including surgery), mediastinal irradiation, infiltrative disease like sarcoidosis, infectious diseases, neoplasms, and renal failure. Although acute pericarditis from most causes may bring about constrictive pericarditis the diagnosis of constrictive pericarditis and secondary congestive hepatopathy/cirrhosis remains a challenge. In one case, the diagnosis of constrictive pericarditis was made only after liver transplantation was performed for presumed cryptogenic cirrhosis [6]. Diagnosis of CCP is confounded by the fact that the clinical picture is very similar to primary hepatic dysfunction. Conversely, in a patient presenting with liver disease, the presence of dyspnea largely points to a diagnosis of hepatopulmonary syndrome, portopulmonary hypertension, cirrhotic cardiomyopathy, hepatic hydrothorax, or anemia [7]. Furthermore, the presence of ascites is a cardinal manifestation of portal hypertension. The patient presented to us after development of a variceal bleed and ascites secondary to portal hypertension. At first a recurrence of CCP was thought to be the reason for his symptoms. He suffered from dyspnea, fatigue, orthopnea, pedal edema, and ascites, all of which could be explained by CCP. Clinical clues to the correct diagnosis include elevated jugular venous pressure and pericardial knock in constrictive pericarditis, which were absent in our case. Moreover the development of portal hypertension, variceal bleed, and a normal echocardiography then steered the diagnosis towards cirrhosis. Hence the diagnostic dilemma persists and CCP should be excluded in all cases of unexplained cirrhosis, regardless of atypical hepatic histology [8].
Our patient had a classical presentation with CCP ten years ago. He received timely surgery, and was asymptomatic for ten years before presenting with cirrhosis. Long-term survival after pericardiectomy depends on the underlying cause. Post-pericardiectomy survival rates were reported to be 71% and 52% at 5 and 10 years, respectively [9]. The long-term prognosis with medical therapy alone is poor. Idiopathic constrictive pericarditis has the best prognosis (88% survival at 7 years), followed by constriction due to cardiac surgery (66% at 7 years). The worst post-surgical prognosis occurs in postradiation constrictive pericarditis (27% survival at 7 years), probably due to an associated neoplastic etiology [9,10]. Predictors of poor outcome in surgically managed constrictive pericarditis are advanced age, poor renal function, abnormal left ventricular systolic or diastolic function, high pulmonary artery systolic pressure, lower serum sodium level, worsening NYHA (New York Health Association) classification, and, as noted above, radiation therapy as the cause of the constrictive pericarditis [10]. None of these risk factors existed in our case. It appears there was insidious onset of increased right sided pressures leading to cirrhosis over a period of ten years.
This was confirmed by performing a transjugular liver biopsy (TJLB) with measurement of right heart pressures. Increased venous pressure caused by right ventricular dysfunction leads to atrophy of the hepatocytes and causes perisinusoidal edema that can impair diffusion of oxygen and nutrients to the hepatocytes [11]. As mentioned earlier the right heart pressures were largely normal with only mild elevation of pulmonary capillary wedge pressure (PCWP). As a result, mild jaundice, abnormalities in liver enzymes, and functional change in hepatic drug metabolisms ensues. On gross examination the congestive liver is enlarged, with a purple or reddish hue with prominent hepatic veins. The cut surface shows the classic nutmeg appearance, with an alternating pattern of hemorrhage and necrosis of zone 3 with the normal or slightly steatotic areas in zones 1 and 2. Microscopically, the hallmark features of hepatic venous hypertension are prominence of the central veins, central vein hemorrhage, and sinusoidal engorgement [12,13]. Untreated, long-standing congestion can lead to cardiac fibrosis and, ultimately cardiac cirrhosis
Complete pericardiectomy is the definitive therapy and is a potential cure for CCP. Results are generally better if the procedure is performed earlier in the course, when less calcification is present and when the chance of abnormal myocardium or advanced heart failure is reduced. The timing of surgical intervention is controversial, but many experts recommend a trial of medical therapy for noncalcific pericardial constriction, and pericardiectomy in nonresponders after 4 to 8 weeks of ATT. In patients, with recurrence of disease, unrecognized diastolic dysfunction or recurrent mediastinal scarring may result in progression of right-side heart failure rather than incomplete initial pericardiectomy [14]. The recurrence of constriction physiology due to exuberant post-operative fibrosis after complete pericardiectomy in non-tuberculous pericarditis is rarely reported in the literature. The management supports repeat surgery and the potential value of steroid administration [15]. In another case, a patient with cirrhosis secondary to CCP developed hepatocellular carcinoma due to long standing disease, further emphasizing the need for early diagnosis and treatment [16].
Conclusion
Our case highlights the importance of careful follow up of patients with cardiac disease who may progress to cirrhosis. Conversely it stresses the need to consider a cardiac etiology in the work-up of cirrhosis especially when the most common causes are excluded The presence of pericardial calcification in a patient with cirrhosis should suggest the possibility of constrictive pericarditis and cardiac cirrhosis and prompt cardiac MR/CT imaging and cardiac catheterization.
References
- Alvarez AM, Mukherjee D (2011) Liver abnormalities in cardiac diseases and heart failure.Int J Angiol 20: 135-142.
- Sherlock S (1951) The liver in heart failure; relation of anatomical, functional, and circulatory changes.Br Heart J 13: 273-293.
- Maisch B, Seferovic PM, Ristic AD, Erbel R, Rienmüller R, et al. (2004) Guidelines on the diagnosis and management of pericardial diseases executive summary; The Task force on the diagnosis and management of pericardial diseases of the European society of cardiology.Eur Heart J 25: 587-610.
- Sheth AA, Lim JK (2008) Liver disease from asymptomatic constrictive pericarditis.J ClinGastroenterol 42: 956-958.
- Goel PK, Moorthy N (2011) Tubercular chronic calcific constrictive pericarditis.Heart Views 12: 40-41.
- Alkaddour A, Vega KJ, Shujaat A (2014) An unusual case of cirrhosis.Case Rep Gastrointest Med 2014: 670176.
- Van der Merwe S, Dens J, Daenen W, Desmet V, Fevery J (2000) Pericardial disease is often not recognised as a cause of chronic severe ascites.J Hepatol 32: 164-169.
- Bertog SC, Thambidorai SK, Parakh K, Schoenhagen P, Ozduran V, et al. (2004) Constrictive pericarditis: etiology and cause-specific survival after pericardiectomy.J Am CollCardiol 43: 1445-1452.
- Imazio M, Brucato A, Maestroni S, Cumetti D, Belli R, et al. (2011) Risk of constrictive pericarditis after acute pericarditis.Circulation 124: 1270-1275.
- Griffin BP, Topol EJ (2004) Pericardial Disease. Manual of Cardiovascular Medicine. (2ndedn) Philadelphia, Pennsylvania: Lippincott Williams and Wilkins 372-396.
- Safran AP, Schaffner F (1967) Chronic passive congestion of the liver in man. Electron microscopic study of cell atrophy and intralobular fibrosis.Am J Pathol 50: 447-463.
- Dunn GD, Hayes P, Breen KJ, Schenker S (1973) The liver in congestive heart failure: a review.Am J Med Sci 265: 174-189.
- Kirsch M, Fleshler B (1992) Deceptive liver histology delays diagnosis of cardiac ascites.South Med J 85: 1151-1152.
- Cho YH, Schaff HV, Dearani JA, Daly RC, Park SJ, et al. (2012) Completion pericardiectomy for recurrent constrictive pericarditis: importance of timing of recurrence on late clinical outcome of operation.Ann ThoracSurg 93: 1236-1240.
- Kashani IA, Higgins CB, Utley JR (1983) Inflammatory constriction following complete pericardiectomy in tuberculous constrictive pericarditis.ClinPediatr (Phila) 22: 219-21.
- Song PS, Koh KC, Yoo BC, Paik SW, Lee JH, et al. (2005) [A case of hepatocellular carcinoma complicating cardiac cirrhosis caused by constrictive pericarditis].Korean J Gastroenterol 45: 436-440.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 15764
- [From(publication date):
specialissue-2015 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 11175
- PDF downloads : 4589