Research Article Open Access
Catalytic Reduction of 4-Nitrophenol Using Biogenic Silver Nanoparticles Derived from Papaya (Carica papaya) Peel extract
CH Prasad, K Srinivasulu and P Venkateswarlu*
Department of Chemistry, Sri Venkateswara University, Tirupati, Andhra Pradesh, India
- *Corresponding Author:
- P Venkateswarlu
Biopolymers and Thermo Physical Laboratories
Department of Chemistry, Sri Venkateswara University
Tirupati-517 502, Andhra Pradesh, India
Tel: +91-9393600444
E-mail: ponneri.venkateswarlu@gmail.com
Received date: October 05, 2015; Accepted date: October 14, 2015; Published date: October 21, 2015
Citation: Prasad CH, Srinivasulu K, Venkateswarlu P (2015) Catalytic Reduction of 4-Nitrophenol Using Biogenic Silver Nanoparticles Derived from Papaya (Carica papaya) Peel extract. Ind Chem Open Access 1:104. doi: 10.4172/2469-9764.1000104
Copyright: © 2015 Prasad CH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Industrial Chemistry
Abstract
A facile and green method is described for the synthesis of Silver (Ag) nanoparticles (NPs) from the extract of Papaya Peel as capping and reducing agent. The green synthesized Ag NPs were characterized by diverse techniques such as powder X-ray diffraction (XRD), UV-visible, Fourier transform-infrared spectroscopy (FTIR) and Transmission electron microscopy (TEM) coupled with X-ray energy dispersive spectroscopy (EDS) techniques. These clearly reveal that the structure of the synthesized silver nanoparticles was face centered cubic. The nanoparticles obtained from Papaya Peel extract were spherical shape with an average diameter of 3-5 nm. Furthermore, the catalytic activity of synthesized Ag NPs in the reduction of 4-nitrophenol (4-NP) was studied by UVvis absorption spectroscopy. The synthesized Ag NPs have a good catalytic activity on the reduction of 4-nitrophenol (4-NP) by Papaya Peel extract which is confirmed by the decrease in absorbance maximum values of 4-nitrophenol (4-NP) with respect to time using UV-vis absorption spectroscopy. An efficient reduction of 4-nitrophenol (4-NP) to 4-aminophenol (4-AP) in the presence of Ag NPs and NaBH4 was observed and was found to depend upon the nanoparticle size or the peel extract concentration used for synthesis.
Keywords
Ag NPs; X-ray diffraction; Transmission electron microscopy; Catalytic activity
Introduction
Silver nanoparticles (NPs) have shown remarkable potential for numerous applications in chemical, electronic, medical, and biological fields due to their distinctive properties, when compared to their bulk counterparts [1-3]. Several physical properties of metal can be tailored for a specific application by controlling their shape, size, and morphology [4,5]. Consequently, there has been a growing need to replace the chemical synthetic procedures with clean, nontoxic, and environmentally suitable “green chemistry” methods. An environmentally benign solvent and eco-friendly capping and reducing agents are the three fundamental elements for a completely green synthesis technique. Accordingly, many researchers have turned toward biological systems such as microorganisms and plants to draw inspiration for green technologies [6-13]. Metal nanoparticles have proven to be the excellent catalysts for inorganic synthesis, pollution treatments and fuel cell, due to their quantum effect, high surface-tovolume ratio and surface energy [14-16]. In particular, the catalytic properties of the nanoparticles of noble metals such as gold [17-20], platinum [21-23] and palladium [24,25] have been extensively studied in recent years. Relatively, only a few investigations have been focused on the catalytic properties of the silver nanoparticles, although silver metal has already been applied as catalysts for commercial production of ethylene oxide [26,27]. The peel extracts function for bio-reduction of silver ions to yield metallic nanoparticles. Here, we report facile and cost effective biogenic synthesis of Ag NPs using aqueous extract of papaya peel and to investigate the bio molecules responsible for the synthesis of Ag NPs. Papaya botanical name Carica papaya, is an oblong tropical fruit. Papaya (Carica papaya L.) or pawpaw belongs to the family of the Caricaceae. This fruit, native to the tropics of America is now spread throughout the world [28]. Solo, Formose, Sunset, Golden and Sunrise are the most common varieties [29]. The fresh fruit is attractive to consumers due to its striking odours, high vitamin content (i.e., vitamin A and C) and high fiber content [30]. Moreover, the fruit’s dietary value, papaya stems, leaves and fruits contain high levels of proteins and vitamins which are used in the elaboration of cosmetics and medications. Further, the catalytic activity of synthesized Ag NPs in the reduction of 4-nitrophenol (4-NP) was studied by UVvis absorption spectroscopy.
Experimental Section
Preparation of leaf extract from Papaya peel
Papaya peel is thoroughly rinsed with double distilled water to remove the fine dust particles and then the Papaya Peel is dried under shade at room temperature for 24 h under dust free condition. The dried Papaya peel was grinded with a mortar and pestle to make a powder. An amount of 10 g of Papaya Peel powder is mixed in to 100 mL double distilled water and refluxed for 1 h, at 80ºC until the color of aqueous extract solution changes from watery to pale yellow. The resultant composition is cooled to room temperature and filtered with a Whatman No. 1 filter paper and the final extract is stored at 4ºC for further use.
Synthesis of silver nanoparticles
30 mL of 1 mM aqueous solution of silver nitrate was taken in Erlenmeyer flask and then 5.0 mL and 10 mL of Papaya Peel extract was added to the above solution separately at room temperature and stirred for 1 h. Then the reaction flask was kept at room temperature for Overnight. Finally, the color of solution changed from pale yellow to dark brown color was developed which indicates formation of Ag NPs. As the present method for the synthesis of Ag NPs with Papaya Peel extract in aqueous solution was made without any additional hazardous chemicals, this pathway satisfies pure green eco-friendly process.
Characterization techniques
UV-Vis absorption spectra of the transparent colloid solution were performed on UV-Vis Spectrometer (Shimedzu 2400 UV-Vis double beam model) at a resolution of 1 nm in 200 - 800 nm wavelength range. The FT-IR spectra of silver nanoparticles and Papaya peel extract was carried out with a Thermo Nicolet FTIR-200 thermo electron corporation. The phase purities of as synthesized compounds were checked by XRD technique. The X-ray diffraction measurements were recorded on a Seifert 3003 TT X- ray diffract meter with Cu Kα radiation with a wave length of 1.52 Å. The morphology and diameter of silver nanoparticles were determined with a Phillips, TECHNAI FEI 12 transmission electron microscope (TEM) and the quantitative elemental analysis of the nanoparticles were carried out an Oxford instruments Inca Penta FET x 3 Energy dispersive spectrum (EDS).
Results and Discussion
UV-visible analysis of Ag NPs
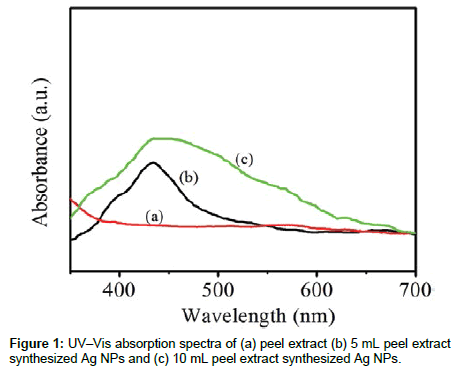
In order to monitor the formation and stability of silver nanoparticles, the absorption spectra of the synthesized silver nanoparticles were recorded against water. Figure 1 represents the UV- Vis absorption spectra for colloidal Ag NPs synthesized using different quantities Peel extract of Papaya 5 mL and 10 mL. It is clearly observable that there is no absorption in visible region for Papaya Peel extract sample. As well as small absorption band at 439 nm starts appearing in the absorption spectra of the prepared sample (5 mL sample). This band grew and blue shifted from 439 nm to 435 nm with increase in the Papaya Peel extract amount. However, the color of solution changed from pale yellow to yellowish brown depending on the extract concentration indicating silver nanoparticles formation as the color change observed is due to excitation of surface Plasmon vibration in the silver nanoparticles.
FT-IR characterization
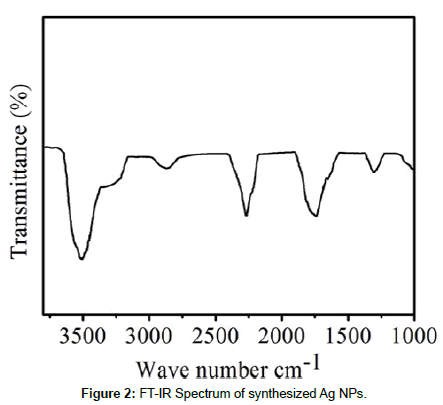
Figure 2 shows the FT-IR spectrum of Papaya Peel extract. The FT-IR spectrum was carried out to identify the possible bio molecules responsible for capping and efficient stabilization of the metal nanoparticles synthesized by Papaya Peel extract. The FT-IR spectrum showed the presence of three different peaks at 3510, 1740, 1300 cm-1. The FT-IR spectrum reveal the presence of different functional groups like - OH, C–O–C, amide linkages and –COO–, these may be between amino acid residues in protein and synthesized Ag NPs. The bands in the region of 3510 cm-1 were assigned to hydroxyl stretching of poly phenols, whereas the peak at 1740 cm-1 was associated with stretch is assigned to the carboxylic acids, ketones, aldehydes and ester compounds. The FT-IR results indicate the presence of poly phenols and other bio molecules in the Papaya Peel extract and these bio molecules reduced Ag+ to Ag0 as well as stabilizing Ag NPs.
Powder XRD analysis
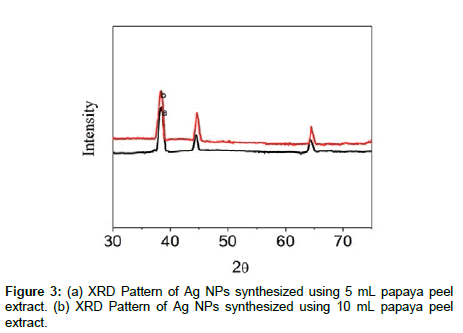
Figure 3 shows the powder XRD patterns of dried silver nanoparticles synthesized using 5 mL and 10 mL peel extract of papaya at room temperature. The green synthesized Ag NPs were highly crystalline with diffraction peaks could be clearly assigned to the facecentered cubic phase of metallic silver. In addition XRD analysis showed three distinct diffraction peaks at 2θ values of 38.1º, 44.3° and 64.5° were indexed to the (111), (200)and (220)reflections of the fcc structure of metallic silver. The XRD patterns in of Ag NPs obtained were similar to the results reported earlier [15]. The average particle size of the silver nanoparticles formed in the bio reduction process was determined using Debye-scherrers formula D=0.89 λ/β cosθ, where D is the average particle size, λ is the wavelength of the X-ray, β is the full width at half maximum intensity of the diffraction peak and θ is diffraction angle of the (111) plane of cubic silver nanoparticles and the calculated particle size ∼10 nm, which is quite close to TEM result. The lattice constant calculated by the formula: 1/d2=1/a2 (h2+k2+l2) where ‘a’ was lattice parameter, dhkl was the interplanar separation corresponding to miller indices h, k, l and the calculated lattice constant from this pattern was a=4.081A°. It clearly exhibited the green synthesized silver nanoparticles are nanocrystalline in nature.
TEM analysis
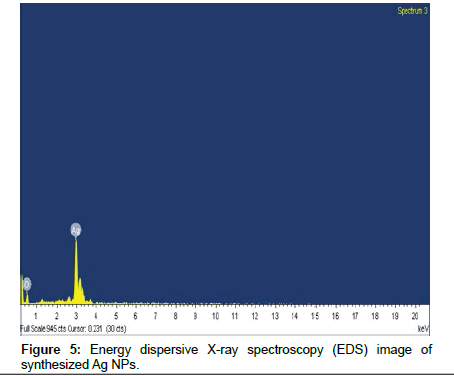
The high resolution study of the nanoparticles using HRTEM revealed that the Ag NPs are poly dispersed and spherical in shape which appears to be characteristic of Ag NPs prepared in peel extract of papaya. TEM images of the precipitated solid phase obtained after termination of the reaction between the papayas aqueous peel extract and AgNO3 solution are show in Figure 4. The TEM micrograph of the synthesized Ag NPs sizes at 3 to 5 nm. The result of energy dispersive spectroscopy (EDX) analysis was shown in Figure 5. This confirmed the significant presence of elemental silver. The above results indicate the spherical shape and elemental silver formed by a facile manner.
Evaluation of catalytic activity of Ag NPs
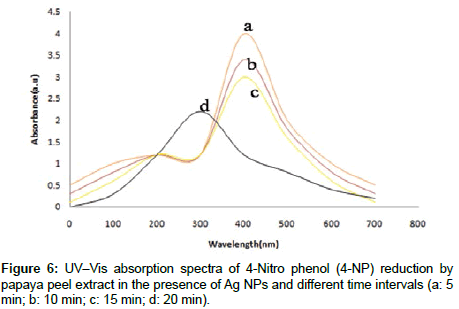
The biogenic Ag NPs were make use of as a catalyst for the reduction of 4-NP by NaBH4. In order to find out the catalytic activity of Ag NPs, three typical reactions were carried out in a 3.5 mL capacity Quartz curette. In the first reaction, 1.50 mL (1mM) of 4-NP was mixed with 1.50 mL of water. In the second reaction, addition of 1 mg of solid NaBH4 to the cuvette containing 1.50 mL (1 mM) of 4-NP and 1.50 mL of water was carried out. In the third reaction, 0.5 mL of biogenic Ag NPs was mixed with the solution obtained from the second reaction. All the three reactions were monitored by Shimedzu 2400 UVVis double beam model. The reduction reaction of 4-NP to 4-AP was monitored by UV–vis Spectrophotometer. Here Figure 6 shows the UV-vis spectra of the reduction of 4-NP by NaBH4 in presence of Ag NPs as a catalyst. From the figure, it could be observed that on addition of NaBH4 the λ max of 4-NP was 400 nm with appearance of bright yellow color. This was due to the formation of sodium phenolate [31] and no change was observed in the Absorption maxima with respect to different time intervals. On addition of biogenic Ag NPs (0.5 mL) to the solution, reduction of 4-NP became rapid which was evident from the fast de colorization of bright yellow Color and the appearance of λ max at 296 nm which was characteristic of 4-AP [32,33]. These results demonstrated the catalytic activity of biogenic Ag NPs on the reduction of 4-NP by NaBH4.
Conclusion
Papaya peel extract have been effectively used for the synthesis of silver nanoparticles. We have demonstrated the use of a natural renewable and low cost bio-reducing agent. These nanoparticles are found to be highly crystalline as evidenced by the peaks in the powder XRD pattern corresponding to Bragg reflections from the (111), (200) and (220) planes of the FCC structure. The average size of the particles is found to be 3-5 nm from TEM image analysis. The biogenic Ag NPs showed high catalytic on reduction of anthropogenic pollutant 4-NP by NaBH4 which has been explained on the basis of Langmuir- Hinshelwood model. Here the present study has brought to light a novel method for reduction of anthropogenic pollutant viz., 4-NP using biogenic Ag NPs. Overall, this method very good eco-friendly and nontoxic source for the synthesis of Ag NPs as compared to the conventional physical and chemical methods. And also the obtained catalytic results are well better than the previous reports.
Acknowledgements
The authors thank to IIT Madras and IICT Hyderabad for providing the instrumentation facility. One of the author Ch. Prasad is grateful acknowledge to CSIR-UGC for financial assistance under JRF.
References
- Ozin AG (1992) Nanochemistry: Synthesis in diminishing dimensions. Adv Mater 4: 612-649.
- Daniel CM, Astruc D (2004) Gold nanoparticles: Assembly, supramolecular chemistry, quantum size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Rev 104: 293-346.
- Kholoud MM, El-Nour A, Eftaiha AA, Al-Warthan A, Ammar AA (2010) Synthesis and applications of silver nanoparticles. Arab J Chem 3: 135-140.
- Coe S, Woo KW, Bawendi M, Bulovic V (2002) Electroluminescence from single monolayers of nanocrystals in molecular organic devices. Nature 420: 800-803.
- Kelly LK, Coronado E, Zhao LL, Schatz CL (2003) The Optical Properties of Metal Nanoparticles: The Influence of Size, Shape, and Dielectric Environment. J Phys Chem B 107: 668-677.
- Kalishwaralal K, Deepak V, Pandian KRBS, Kottaisamy M, Barathmanikantha S, et al. (2010) Biosynthesis of silver and gold nanoparticles using Brevibacterium casei.Colloids Surf B Biointerfaces 77: 257-262
- Mukherjee P, Senapati S, Mandal D, Ahmad A, Khan IM, et al. (2002) Extracellular Synthesis of Gold Nanoparticles by the Fungus Fusarium oxysporum. ChemBioChem 3: 461-463.
- Shaligram SN, Bule M, Bhambure R, Singhal SR, Singh KS, et al. (2009) Biosynthesis of silver nanoparticles using aqueous extract from the compacting producing fungi. Process Biochem 44: 939-943.
- Gericke M, Pinches A (2006) Microbial production of gold nanoparticles. Gold Bull 39:22-28.
- Ahmad A, Senapati S, Khan IM, Kumar R, Sastry M (2003) Extracellular Biosynthesis of MonodisperseGold Nanoparticles by a Novel Extremophilic Actinomycete, Thermomonospora sp Langmuir 19: 3550-3553.
- Chandran PS, Chaudhary M, Pasrichaa R, Ahmad A, Sastry M (2006) Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotechnol Prog 22: 577-583.
- Shankar SS, Rai A, Ahmad A, Sastry M (2004) Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. J Colloid Interface Sci 275: 496-502.
- Bar H, Bhui KD, Sahoo PG, Sarkar P, Pyne S,et al. (2009) Green synthesis of silver nanoparticles using seed extract of Jatropha curcas. Colloids Surf. A: Physico Chem Eng Aspects 348: 212.
- Green synthesis of silver nanoparticles using seed extract of Jatropha curcas
- Narayanan R, El-Sayed AM (2005) Catalysis with Transition Metal Nanoparticles in Colloidal Solution: Nanoparticle Shape Dependence and Stability. J Phys Chem B 109: 12663.
- Astruc D, Lu F, Aranzaes RJ (2005) Nanoparticles as recyclable catalysts: the frontier between homogeneous and heterogeneous catalysis. Angew Chem Int 44: 7852-7872.
- Haruta M (2003) When gold is not noble: catalysis by nanoparticles. The Japan Chemical Journal 3: 75-85.
- Tsunoyama H, Sakurai H, Ichikuni N, Negishi Y, Tsukuda T (2004) Colloidal gold nanoparticles as catalyst for carbon-carbon bond formation: application to aerobic homocoupling of phenylboronic acid in water. Langmuir 20: 11293-11296.
- Manea F, Houillon BF, Pasquato L, Scrimin P (2004) Nanozymes: Gold-Nanoparticle Based Trans phosphorylation Catalysts. Angew Chem Int Ed 43: 6165-6169.
- Liu J, Qin G, Raveendran P, Ikushima Y (2006)Facile “Green” Synthesis, Characterization, and Catalytic Function of β-D-Glucose-Stabilized Au Nanocrystals. Chem Eur J 12: 2131-2138.
- Praharaj S, Nath S, Ghosh SK, Kundu S, Pal T (2004) Immobilization and recovery of au nanoparticles from anion exchange resin: resin-bound nanoparticle matrix as a catalyst for the reduction of 4-nitrophenol. Langmuir 20: 9889-9892.
- Parker FS, Frost DC, Telling M, Albers P, Lopez M,et al. (2006) Characterisation of the adsorption sites of hydrogen on Pt/C fuel cell catalysts. Catalysis Today 114: 418.
- PrabhuramJ, WangX, Hui LC, Hsing MI(2003) Synthesis and Characterization of Surfactant-Stabilized Pt/C Nano catalysts for Fuel Cell Applications. J Phys Chem B107:11057-11064.
- Liu Z, Gan ML, Hong L, Chen W, Lee YJ (2005) Carbon-supported Pt nanoparticles as catalysts for proton exchange membrane fuel cells. J Power Sources13:73-78.
- Narayanan R, El-Sayed AM (2005) Carbon-supported spherical palladium nanoparticles as potential recyclable catalysts for the Suzuki reaction.Journal of Catalysis 234: 348-355
- Morkel M, Rupprechter G, Freund HJ (2005)Finite size effects on supported Pd nanoparticles: Interaction of hydrogen with CO and C2H4. Science 588: 209-219.
- Yu HG (2006) Density functional theory study of ethylene partial oxidation on Ag7 clusters. Chemical Physics Letters 431: 236-240.
- Shiraishi Y, ToshimaN (1999) Colloidal silver catalysts for oxidation of ethylene. Journal of Molecular Catalysis A: Chemical 141: 187-192.
- Teixeira da Silva JA, Rashid Z, Tan D, Sivakumar DN, Gera A, et al.(2007) Papaya (Carica papaya L.) biology and biotechnology. Tree and Forestry Science and Biotechnology 1: 47-73.
- Paull RE, Nishijima W, Reyes M, Cavaletto C (1997) Postharvest handling andlosses during marketing of papaya (Carica papaya L.). Postharvest Biology and Technology 11:165.
- Almora K, Pino JA, Hernandez M, Duarte C, Gonzalez J, et al. (2004) Evaluationof volatiles from ripening papaya (Carica papaya L., var. Maradolroja). Food Chemistry 86: 127-130.
- Wunder S, Polzer F, Lu Y, Mei Y (2010) Kinetic Analysis of Catalytic Reduction of 4-Nitrophenol by Metallic Nanoparticles Immobilized in Spherical Polyelectrolyte Brushes. J Phys Chem C 114: 8814.
- Premkumar T, Lee K, Geckeler KE (2011) Shape-tailoring and catalytic function of anisotropic gold nanostructures.Nano scale Research Letters6:547.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 17194
- [From(publication date):
October-2015 - Aug 19, 2025] - Breakdown by view type
- HTML page views : 12226
- PDF downloads : 4968