Editorial Open Access
Challenges in Searching for Zika Therapeutics
Yuguang Zhao*Division of Structural Biology, Oxford University, UK
- *Corresponding Author:
- Yuguang Zhao
Division of Structural Biology
University of Oxford
The Wellcome Trust Centre for Human Genetics
Headington, Oxford, OX3 7BN, UK
Tel: +44-0-1865 287551
E-mail: yuguang@strubi.ox.ac.uk
Received date: May 27, 2016; Accepted date: May 31, 2016; Published date: June 06, 2016
Citation: Zhao Y (2016) Challenges in Searching for Zika Therapeutics. J Chem Biol Ther 2:e102. doi: 10.4172/2572-0406.1000e102
Copyright: © 2016 Zhao Y. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Chemical Biology & Therapeutics
Zika virus was discovered nearly 70 years ago, but received little attention prior to 2015, as the virus infection causes no or only a mild, self-limiting illness without need for treatment. However, large scale outbreaks in South and Central America and the Caribbean in 2015 strongly suggested Zika epidemic may have associations with the increased cases of birth defect called microcephaly. Microcephaly is a condition in which a person’s head is significantly smaller than normal for their age and sex, and occurs most often when a baby’s brain has not developed properly during pregnancy. The microcephalus babies may have life-long intellectual or physical disabilities. Now, a compilation of evidences solidified the link between the Zika virus infection in pregnant mothers and microcephaly of their babies. As the Zika virus has ‘explosive’ pandemic potential in Africa, Southeast Asia, the Pacific Islands and the Americas, the World Health Organization (WHO) declared Zika virus a Public Health Emergency of International Concern in early 2016.
Zika virus is a positive single stranded RNA virus with a genome size of about 11 kb nucleotides long. This single transcript gives a polypeptide which processed into 3 structural proteins (capsid [C], premembrane/membrane [prM] and envelope [E]) and 7 non structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, NS5), as shown schematically on Figure 1A. The structural proteins form the viral particles. The non-structural proteins may contribute to viral replication, virion assembly and modulation of the host immune system. For example, the viral RNA polymerase NS5 is not only used for the viral RNA replication, but it also can promote the proteasomal degradation of host signal transducer and activator of transcription 2 (STAT2) to inhibit type I interferon (IFN) signalling and thus antagonize the host antiviral response [1]. Zika is mainly transmitted by the mosquito species Aedes aegypti or Aedes albopictus. It may also be transmitted by blood transfusion or unprotected sex. Once the Zika virus get into the bloodstream of a pregnant woman, the virus may cross the placenta and infect the foetus’s neural progenitor cells (NPC), which form the brain’s outer layer. Damaged NPC creates fewer neurons, leading to decreased brain volume [2,3]. It is suggested that even if a pregnant women is exposed to a low dose of the virus for a short period of time, it may still damage the baby’s brain development, especially in the first trimester of pregnancy. In experimental model of cultured human embryonic stem cell-derived cerebral organoids, Zika can shrink the size of the organoids significantly [4]. It was demonstrated that Zika viral RNA can activate the Toll-like receptor 3 (TLR3), and lead to down steam pathways modulating neurogenesis and apoptosis [2]. The central nervous system (CNS) developmental axon guidance molecules, netrin-1 (NTN1) and receptor tyrosine kinase ephrin type-B receptor 2 (EphB2) may particularly be relevant to Zika pathogenesis [2].
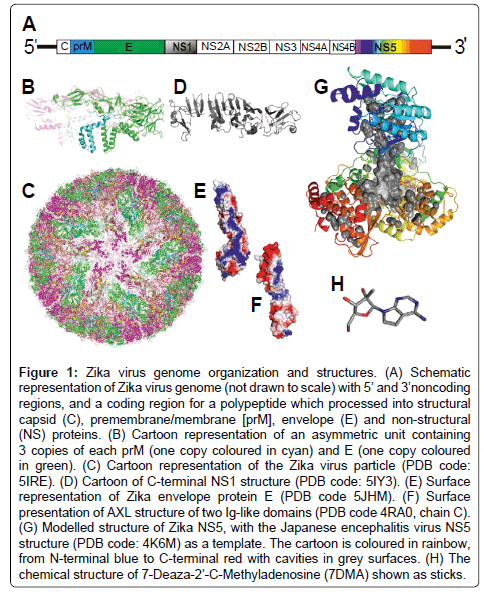
Figure 1: Zika virus genome organization and structures. (A) Schematic representation of Zika virus genome (not drawn to scale) with 5’ and 3’noncoding regions, and a coding region for a polypeptide which processed into structural capsid(C), premembrane/membrane[prM], envelope(E) and non-structural (NS) proteins. (B) Cartoon representation of an asymmetric unit containing 3 copies of each prM (one copy coloured in cyan) and E (one copy coloured in green). (C) Cartoon representation of the Zika virus particle (PDB code: 5IRE). (D) Cartoon of C-terminal NS1 structure (PDB code: 5IY3). (E) Surface representation of Zika envelope protein E (PDB code 5JHM). (F) Surface presentation of AXL structure of two Ig-like domains (PDB code 4RA0, chain C). (G) Modelled structure of Zika NS5, with the Japanese encephalitis virus NS5 structure (PDB code: 4K6M) as a template. The cartoon is coloured in rainbow, from N-terminal blue to C-terminal red with cavities in grey surfaces. (H) The chemical structure of 7-Deaza-2’-C-Methyladenosine (7DMA) shown as sticks.
Structural biology, aimed at the study of molecular mechanisms at near atomic level, may help in understanding the Zika virus’ pathogenesis and provide molecular basis for drug design or improving the effectiveness of available drugs. The Zika virus cryo-EM structures have been determined [5,6], as illustrated in Figure 1B and 1C. The protein prM and E are visible in the virus particle structure. 3 copies of prM/E form an asymmetric unit (Figure 1B) and 60 copies of such units assemble the whole virus particle (Figure 1C). The envelope E glycoprotein is the major protein responsible for receptor binding and fusion of host cells, and thus may be serve as a good target for the development of therapeutics. The crystal structure of E and its complex of a neutralizing antibody has been determined by X-ray crystallography [7]. The receptors for the E glycoprotein have not been clearly defined. A receptor tyrosine kinase AXL (AXL named from the Greek word ‘Anexelekto’, meaning uncontrolled) has been proposed to be one of the candidate for the Zika’s entry receptor [8], and it is highly expressed in neural stem cells. The single span transmembrane AXL has a large extracellular part containing two Ig-like and two fibronectin domains with the cellular part containing a protein kinase domain. The AXL plays an important role in inhibition of TLR signalling. Interestingly, the Zika virus induces NPC damage by promoting the TLR3 signalling, suggesting a good link between AXL receptor and Zika’s pathogenesis. The structure of the two Ig-like domain of AXL is available [9], as shown in Figure 1F. It is interesting to note that the AXL two Ig-like domain exhibits a good surface charge complementary to that of viral E protein (Figure 1E). The biochemical data showing the direct interaction of AXL and Zike E protein remains to be demonstrated. If they interact directly, the complex structure would provide valuable information for drug designing. For the Zika’s nonstructural protein structures, the c-terminal fragment of NS1 has been obtained [10], as shown in Figure 1D. The structure for NS5, a RNA polymerase has not yet been determined, however, it can be modelled with the Japanese encephalitis virus NS5 structure (PDB code: 4K6M) [11]. The modelled structure shows large cavities as shown in Figure 1G. These cavities may be used for docking of potential inhibitors, especially nucleotide analogues. One nucleotide analogue, 7-Deaza- 2’-C-Methyladenosine (7DMA, chemical structure shown in Figure 1H), originally used for inhibition of the close related Hepatitis C virus (HCV) RNA polymerase, has been shown effectively inhibit the Zika virus replication in mouse model [12]. In order to design powerful specific inhibitors, the actual structure in complex with inhibitors will be required.
Developing brand-new drug takes an enormous amount of time, money and effort. The potential strategies to reduce the time frame, decrease costs and improve success rates may be drug repurposing. Many Food and Drug Administration (FDA) approved drugs have been developed or are being developed for treatment of diseases not initially purposed. These drugs have good safety records, and could be readily used for clinical trials. Much like in the case for Ebola, some anticancer drugs from selective estrogen receptor modulators (SERM) were discovered to be able to inhibit Ebola infection in animal models [13]. More recently, the complex structure of a SERM compound with Ebola glycoprotein was achieved in our laboratory. It provides valuable information for further development of powerful anti-ebola drugs (Zhao Y, Ren J et al., to be published in Nature). Searching for repurposed drugs for Zika has just started. The American National Centre for Advancing Translational Sciences (NCATS) is taking the leading role in this field. It is claimed that one compound has been identified which is capable of blocking the Zika induced caspase 3 activity, and thus stops the viral induced apoptosis in the infected cells. Additionally, advances in reverse genetics of Zika virus may also facilitate the large scale drug screening. For example, an infectious Zika virus cDNA clone was engineered with an insertion of a Renilla luciferase reporter gene. This recombinant virus allows rapid luciferase assay be used for large scale drug screening. The compound, NITD008, has been identified by this assay [14]. NITD008 is very similar to 7DMA, both are nucleotide analogues targeting the viral RNA polymerase NS5. There is a long way to go before we could know if these potential compounds could actually be used for the treatment of Zika infection in pregnant women.
Vaccination is a common way of fighting viral diseases. Vaccines for other members of same flavivirus family is available, for example, yellow fever and Japanese encephalitis vaccines. To develop a Zika vaccine, the recombinant virus-like particles (VLP) may be a good choice. This can be achieved by recombinant expression of prM/E genes with a part of C sequence. The resulting VLP contains no virus non-structural genes, and thus, is a safe form of vaccine which retains full antigenicity.
Currently, there is no FDA approved vaccine or drugs for the Zika virus infection. Searching for Zika therapeutics for pregnant woman is especially challenging, as foetuses are particularly vulnerable to substances that may influence their development. For the time being, the personal protection from mosquito bite is the most effective way of preventing Zika infection. Some mosquito repellent, such as N, N-diethyl-meta-toluamide (DEET) can be safely used for pregnant women [15]. With the rapid progress in understanding molecular mechanisms of Zika pathogenesis, and joined efforts from both academic research and commercial development, the therapeutics for Zika will be available in the near future.
Acknowledgments
This is a contribution from the UK Instruct Centre and supported by BIOSTRUCT-X (grant 283570). The Wellcome Trust Centre for Human Genetics is supported by the Wellcome Trust (grant 090532/Z/09/Z).
References
- Grant A, Ponia SS, Tripathi S, Balasubramaniam V, Miorin L, et al. (2016) Zikavirus targets human stat2 to inhibit type i interferon signaling. Cell Host Microbe.
- Dang J, Tiwari SK, Lichinchi G, Qin Y, Patil VS, et al. (2016) Zikavirus depletes neural progenitors in human cerebral organoids through activation of the innate immune receptor TLR3. Cell Stem Cell.
- Tang H, Hammack C, Ogden SC, Wen Z, Qian X, et al. (2016) Zikavirus infects human cortical neural progenitors and attenuates their growth. Cell Stem Cell 18: 587-590.
- Garcez PP, Loiola EC, da Costa RM, Higa LM, Trindade P, et al. (2016) Zika virus impairs growth in human neurospheres and brain organoids. Science 352: 816-818.
- Kostyuchenko VA, Lim EX, Zhang S, Fibriansah G, Ng TS, et al. (2016) Structure of the thermally stable Zika virus. Nature 533: 425-428.
- Sirohi D, Chen Z, Sun L, Klose T, Pierson TC, et al. (2016) The 3.8 Åresolution cryo-EM structure of Zika virus. Science 352: 467-470.
- Dai L, Song J, Lu X, Deng YQ, Musyoki AM, et al. (2016) Structures of the Zikavirus envelope protein and its complex with a flavivirus broadly protective antibody. Cell Host Microbe 19: 696-704
- Nowakowski TJ, Pollen AA, Di Lullo E, Sandoval-Espinosa C, Bershteyn M, et al. (2016) Expression analysis highlights AXL as a candidateZikavirus entry receptor in neural stem cells. Cell Stem Cell 18: 591-596.
- Kariolis MS, Miao YR, Jones DS 2nd, Kapur S, Mathews II, et al. (2014) An engineered Axl 'decoy receptor' effectively silences the Gas6-Axl signaling axis. Nat Chem Biol 10: 977-983.
- Song H, Qi J, Haywood J, Shi Y, Gao GF (2016) Zika virus NS1 structure reveals diversity of electrostatic surfaces among flaviviruses. Nat Struct Mol Biol 23: 456-458.
- Lu G, Gong P (2013) Crystal Structure of the full-length Japanese encephalitis virus NS5 reveals a conserved methyltransferase-polymerase interface. PLoS Pathog 9: e1003549.
- Zmurko J, Marques RE, Schols D, Verbeken E, Kaptein SJ, et al. (2016) The Viral polymerase inhibitor 7-deaza-2'-c-methyladenosine is a potent inhibitor of in vitro Zika virus replication and delays disease progression in a robust mouse infection model. PLoS Negl Trop Dis 10: e0004695.
- Johansen LM, Brannan JM, Delos SE, Shoemaker CJ, Stossel A, et al. (2013) FDA-approved selective estrogen receptor modulators inhibit Ebola virus infection. Sci Transl Med 5: 190ra179.
- Shan C, Xie X, Muruato AE, Rossi SL, Roundy CM, et al. (2016) Aninfectious cDNAclone of Zikavirus to study viral virulence, mosquito transmission, and antiviral inhibitors. Cell Host Microbe.
- Kline MW, Schutze GE (2016) What pediatricians and other clinicians should know about Zikavirus. JAMA Pediatr 170: 309-310.
Relevant Topics
- Analytical Methods
- Biocatalysis and biotransformation
- Bioinorganic Chemistry
- Biological chemistry
- Bioorganic chemistry
- Chemical Biology of Tetracyclines
- Chemotherapeutic Agents
- Enzyme Inhibitor
- Mechanisms of DNA Damage and Repair
- Nanomaterials For Imaging and Drug Delivery
- Natural Product Biosynthesis
- Nucleic Acid Analogs
- Spectroscopic Probes
Recommended Journals
Article Tools
Article Usage
- Total views: 12014
- [From(publication date):
November-2016 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 10977
- PDF downloads : 1037