Mini Review Open Access
Chemical Transdifferentiation: a New Strategy in the Fight agai nst Neurodegenerative Disease
Yuewen Tang and Lin Cheng*
State Key Laboratory of Medical Genomics, Shanghai Institute of Hematology, Ruijin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China
- Corresponding Author:
- Lin Cheng
State Key Laboratory of Medical Genomics, Shanghai Institute of Hematology
Ruijin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine
Shanghai 200025, China
Tel: +86-21-64370045
E-mail: cl11754@rjh.com.cn
Received date: March 04, 2016; Accepted date: April 07, 2016; Published date: April 14, 2016
Citation: Tang Y, Cheng L (2016) Chemical Transdifferentiation: a New Strategy in the Fight against Neurodegenerative Disease. J Alzheimers Dis Parkinsonism 6:231. doi:10.4172/2161-0460.1000231
Copyright: © 2016 Tang Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Alzheimers Disease & Parkinsonism
Cell transdifferentiation of somatic cells directly into desirable cells has sprung up in recent years, which also has provided an alternative cell source for cell therapy in regenerative medicine, comparing with cell reprogramming. However, the conventional strategy for generating target cells through transdifferentiation was mainly based on viruses expressing exogenous transcription factors, which was unavoidably related with safety issues when translated into clinical application. Small molecules with more advantages in manipulating cell fate changes has attracted attentions of researchers in regenerative medicine. Especially the advent of chemically-induced pluripotent stem cells [1] opens a new era of regeneration and later kindles the light of pursuing diverse chemical cocktails in generating different types of cells, which we refer to as chemical transdifferentition.
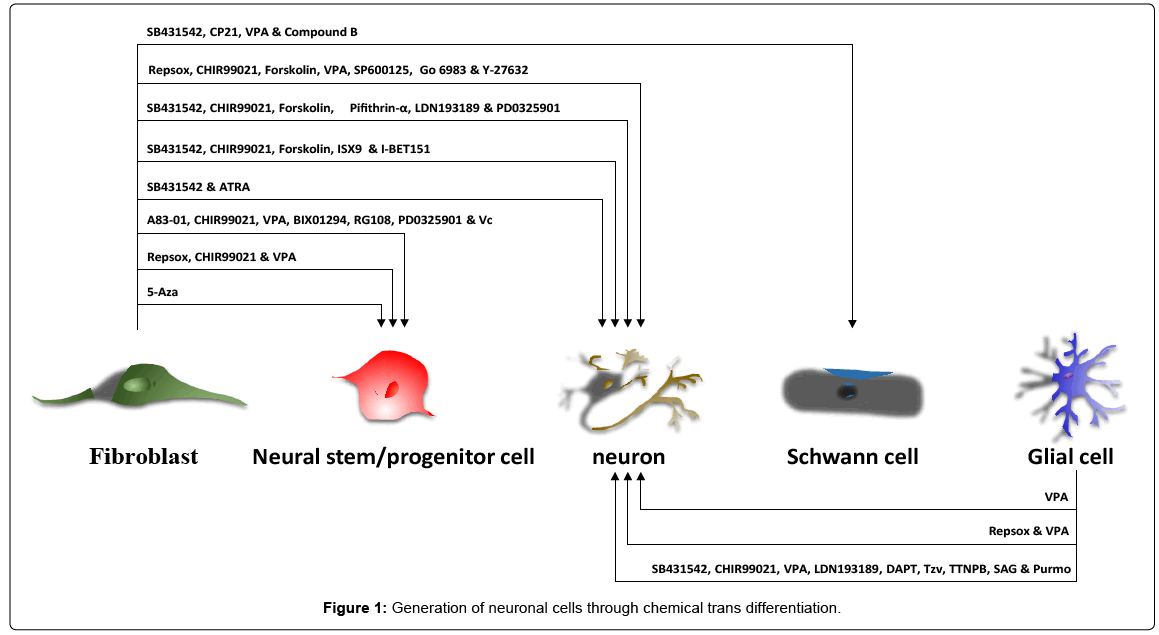
At this point, generation of neuronal cells leads the way so far, due to highly requirement of novel treatments for neurological diseases. We have used cocktail of VPA, CHIR99021, and Repsox (VCR) to convert mouse fibroblasts and human urinary cells into neural progenitor cells under hypoxia [2]. Without hypoxia, Han et al. added more chemical compounds with VCR to generate neural stem cells from mouse fibroblasts, which includes VPA, CHIR99021, A83-01, BIX01294, RG108, PD0325901, and Vitamin C [3]. Interestingly, Baharvand et al. induced human fibroblasts in suspension culture into neural progenitor cells only in the presence of 5-Aza [4]. Based on our VCR induction protocol, Hu et al. added small molecules known to promote neural progenitor cell differentiation to directly convert normal and Alzheimer’s disease human fibroblasts into neuronal cells. The additional compounds are Forskolin, SP600125, GO6983, and Y-27632 [5]. Besides, functional neurons can also be induced from human fibroblasts by alternative recipe containing SB431542, CHIR99021, Forskolin, Pifithrin-α, LDN193189 and PD0325901 [6]. For initial cells from mouse, Li et al. demonstrated that cocktail of SB43152, CHIR99201, Forskolin, I-BET151, and ISX9 can induce mouse fibroblasts into TUJ1 positive neurons [7]. For induction of more specific-subtype of neuron, Xu et al. cultured mouse fibroblasts in conditional medium from olfactory ensheathing cells with SB431542 and Retinoic acid, then generated GABAergic neurons [8].
All the above neural cell transdifferentiation reported so far was initiated mainly from fibroblasts both in mouse and human. Due to neural cells and fibroblasts belong to distinct germ layer ectoderm and mesoderm individually, generated neural cells often remain the epigenetic memory of starting fibroblasts, which indicates incomplete transformation between cells and also hurdles the clinical translation of these chemically induced cells. Considering the proximity in lineage distance among neuronal cells, astrocytes are one of the ideal starting candidate cell type for generating neural stem/progenitor cells or neurons. Recently, we found that combination of VPA and Repsox was able to convert mouse astrocytes into neuro blasts and neurons in vitro [9]. Beyond that, Zhang et al. demonstrated that cultured human astrocytes can also be induced efficiently into functional neurons under sequential exposure to cocktail of SB431542, CHIR99021, VPA, LDN193189, DAPT, Tzv, TTNPB, SAG, and Purmo [10].
Both Alzheimer’ Disease (AD) and Parkinson’s Disease (PD) belong to neurodegenerative disorder, partially due to losses of functional neurons. Transplantation of isolated neural stem cells or differentiated sub-specific neurons is proven to be helpful in alleviating symptoms in rodent models of AD and PD [11]. However, cell transplantation has been limited to cell sources. Chemical transdifferentiation of easily accessible somatic cells into neuronal cells in vitro successfully help solving this issue, which will provide unlimited and clinically safe cells for individual patients. Except for that, the other issues related with transplantation of engineered cells into central nervous system are delivery strategy and integration efficiency. To bypass these obstacles, transdifferentiation in vivo of resident cells into functional cells to repair damaged tissues is thought to be the ultimate goal in regenerative medicine. For neurodegenerative disease, glial cells, especially astrocytes proliferate, accompanying by losses of nearby neurons after damage. Thus, many groups have worked on delivery of viruses expressing exogenous transcription factors in vivo to convert local astrocytes into neural stem cells or neuro blasts or neurons [12-16]. Without introduction of viruses delivering exogenous genes, which are also involved in safety issues, the cocktails of small molecules even cocktail of pharmaceutical drugs identified in astrocytic-neuronal conversion in vitro assay might be applied by local delivery or systematic administration to achieve in transdifferentiation in situ. It is worth to mention that glial cells can be converted into neurons in vivo after brain injury at low efficiency by VPA alone [17], which is a drug to treat epilepsy and bipolar disorder. Although the side effect of the small molecules needs to be fully considered before clinical application, chemical transdifferentiation brings us promising prospects to the treatments of neurodegenerative disease, including AD and PD [18] (Figure 1).
References
- Hou P, Li Y, Zhang X, Liu C, Guan J, et al. (2013) Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science 341: 651-654.
- Cheng L, Hu W, Qiu B, Zhao J, Yu Y, et al. (2014) Generation of neural progenitor cells by chemical cocktails and hypoxia. Cell Research 24: 665-679.
- Han YC, Lim Y, Duffieldl MD, Li H, Liu J, et al. (2016) Direct Reprogramming of Mouse Fibroblasts to Neural Stem Cells by Small Molecules. Stem Cells Int 2016: 4304916.
- Mirakhori F, Zeynali B, Kiani S, Baharvand H (2015) Brief azacytidine step allows the conversion of suspension human fibroblasts into neural progenitor-like cells. Cell J 17: 153-158.
- Hu W, Qiu B, Guan W, Wang Q, Wang M, et al. (2015) Direct Conversion of Normal and Alzheimer's Disease Human Fibroblasts into Neuronal Cells by Small Molecules. Cell Stem Cell 17: 204-212.
- Dai P, Harada Y, Takamatsu T (2015) Highly efficient direct conversion of human fibroblasts to neuronal cells by chemical compounds. J ClinBiochemNutr 56: 166-170.
- Li X,Zuo X, Jing J, Ma Y, Wang J, et al. (2015) Small-Molecule-Driven Direct Reprogramming of Mouse Fibroblasts into Functional Neurons. Cell Stem Cell 17: 195-203.
- Xu H, Wang Y, He Z, Yang H, Gao WQ (2015) Direct conversion of mouse fibroblasts to GABAergic neurons with combined medium without the introduction of transcription factors or miRNAs. Cell Cycle 14: 2451-2460.
- Cheng L, Gao L, Guan W, Mao J, et al. (2015) Direct conversion of astrocytes into neuronal cells by drug cocktail. Cell Res 25: 1269-1272.
- Zhang L, Yin JC, Yeh H, Ma NX, Lee G, et al. (2015) Small Molecules Efficiently Reprogram Human Astroglial Cells into Functional Neurons. Cell Stem Cell 17: 735-747.
- Dantuma E, Merchant S, Sugaya K (2010) Stem cells for the treatment of neurodegenerative diseases. Stem Cell Res Ther 1: 37.
- Heinrich C, Blum R, Gascon S, Masserdotti G, Tripathi P, et al. (2010) Directing astroglia from the cerebral cortex into subtype specific functional neurons. PLoS biology 8: e1000373.
- Guo Z, Zhang L, Wu Z, Chen Y, Wang F, et al. (2014) In vivo direct reprogramming of reactive glial cells into functional neurons after brain injury and in an Alzheimer's disease model. Cell Stem Cell 14: 188-202.
- Niu W, Zang T, Smith DK, Vue TY, Zou Y, et al. (2015) SOX2 reprograms resident astrocytes into neural progenitors in the adult brain. Stem Cell Reports 4: 780-794.
- Niu W, Zang T, Zou Y, Fang S, Smith DK, et al. (2013) In vivo reprogramming of astrocytes to neuroblasts in the adult brain. Nat Cell Biol 15: 1164-1175.
- Liu Y, Miao Q, Yuan J, Han S, Zhang P, et al. (2015) Ascl1 Converts Dorsal Midbrain Astrocytes into Functional Neurons In Vivo. J Neurosci35: 9336-9355.
- Ghasemi-Kasman M, Hajikaram M, Baharvand H, Javan M (2015) MicroRNA-Mediated In Vitro and In Vivo Direct Conversion of Astrocytes to Neuroblasts. PLoS One 10: e0127878.
- Cheng L (2014) Novel strategy for treating neural disease. Sci China Life Sci 57: 947-948.
Relevant Topics
- Advanced Parkinson Treatment
- Advances in Alzheimers Therapy
- Alzheimers Medicine
- Alzheimers Products & Market Analysis
- Alzheimers Symptoms
- Degenerative Disorders
- Diagnostic Alzheimer
- Parkinson
- Parkinsonism Diagnosis
- Parkinsonism Gene Therapy
- Parkinsonism Stages and Treatment
- Stem cell Treatment Parkinson
Recommended Journals
Article Tools
Article Usage
- Total views: 12619
- [From(publication date):
June-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 11551
- PDF downloads : 1068