Congenital Adenomatoid Cystic Malformation: Expectant Management Outcomes
Received: 19-Aug-2018 / Accepted Date: 06-Sep-2018 / Published Date: 13-Sep-2018 DOI: 10.4172/2376-127X.1000387
Keywords: Congenital adenomatoid cystic malformation; Prenatal diagnosis; Perinatal outcome
Introduction
Congenital Adenomatoid Cystic Malformation (CCAM) is a relatively rare abnormality of lung development, with a reported incidence of 1/25,000 pregnancies [1,2]. This anomaly results from an arrest of lung development between the 7th and 15th weeks of gestation during the pseudoglandular stage and is characterized by overgrowth of terminal bronchi without the corresponding alveoli [3-7]. The typical ultrasonographic finding is a cystic mass that could partially or totally occupy the fetal hemithorax, with less than 3% of the cases having bilateral involvement [1,7]. The natural history of CCAM is variable, most cases reach a plateau during the third trimester showing a decrease in size relative to the overall size of the fetus, while some others may even present ultrasonographic regression, an increase in lesion size is uncommon [3,8-11]. CCAM prognosis is usually good with a global survival rate higher than 90% [3]. The purpose of this paper is to describe the evolution and perinatal outcome of CCAM according to prenatal sonographic features in expectantly managed fetuses over the course of 22 years in our institution.
Materials and Methods
A retrospective review of 27 prenatally diagnosed CCAM cases at the Maternal-Fetal Medicine department of the National Institute of Perinatology was conducted. Each case underwent an initial comprehensive ultrasound evaluation performed by Maternal-Fetal Medicine specialist of the department in which CCAM diagnosis and classification was established. Serial scans were scheduled according to lesion size and the presence of complications. Pregnancy resolution, newborn integral care and most of the surgical procedures were also performed within this institution. Information related to prenatal diagnosis and perinatal results were collected from clinical charts and registered in a database, which was analyzed with the SPSS 20.0 statistics software using descriptive statistics.
Results
A total of 27 CCAM prenatally diagnosed cases were identified, 14 of which were diagnosed from 2012-2017. All cases were referrals from other Institutions. The average maternal age was 26 years old. Mean gestational age at the time of diagnosis was 26.5 weeks. In 96.2% of the cases, at least two evaluations were performed prior to delivery, 69.2% had three evaluations, 40.7% had four evaluations and only 22.25% had five or more (Table 1).
| Maternal age (years) a | 26 (± 6.0) |
| Parity b | 1 (1-8) |
| Gestational age at the time of diagnosis a | 26.5 (± 4.1) |
| Gestational age at birth a | 37.1 (± 3.1) |
| Birth weight, g a | 2,817 (± 598) |
| Apgar score (1 min) b | 8 (4-9) |
| Apgar score (5 min) b | 9 (8-9) |
| Silverman Anderson b | 2 (2-4) |
Table 1: Obstetric and neonatal characteristics (n= 27); *Data displayed as mean and standard deviation (SD) a and some are displayed as mode and (rank) b
Lesions were left sided in most cases (55.6%). According to Stocker ´s classification, there were eight type 1 (29.6%), ten type 2 (30%) and nine type 3 (33.4%) cases of CCAM [12]. The lesions had a mean maximum diameter of 36.1 mm for type 1, 31.6 mm for type 2 and 33.9 mm for type 3 or microcystic. No bilateral lesions were found during the review period of the study. Sixteen fetuses (59.2%) had an associated mediastinal shift, which was observed since the initial evaluation. Additional anomalies were found in six cases (22.2%): Intrauterine growth restriction was diagnosed in three cases, myelomeningocele was found in one case and subaortic ventricular communication in another. Hydrops, defined as fluid accumulation in two or more fetal body cavities, was observed in two cases (7.4%). The first case of hydrops was reported in the year of 1995 and was found in a followup scan of a fetus diagnosed with type 1 CCAM at 33.1 weeks of gestation. In the second case reported in 2004, hydrops was present at the initial evaluation of a 27 weeks fetus diagnosed with type 3 CCAM. In other cases, isolated ascites (7.4%), pleural effusion (11.1%), pericardial effusion (11.1%) and polyhydramnios (22.2%) were reported. None of the cases were subjected to invasive procedures or treated with steroids (Tables 2 and 3).
| Left hemithorax % (n) a | 55.6 (15) |
| Right hemithorax % (n) a | 44.4 (12) |
| Type 1 CCAM (Stocker) % (n) a | 29.6 (8) |
| Type 2 CCAM (Stocker) % (n) a | 37 (10) |
| Type 3 CCAM (Stocker) % (n) a | 33.4 (9) |
| Mediastinal shift % (n) a | 59.2 (16) |
| Hydrops % (n) a | 7.4 (2) |
| Ascites % (n) a | 7.4 (2) |
| Pleural effusion % (n) a | 11.1 (3) |
| Pericardial effusion % (n) a | 11.1 (3) |
| Polyhydramnios % (n) a | 22.2 (6) |
| Stable lesion % (n) a | 63 (17) |
| Complete regression % (n) a | 18.5 (5) |
| Partial regression % (n) a | 14.8 (4) |
| Progression % (n) a | 1 (3.7%) |
Table 2: Prenatal lesions characteristics; *Data displayed as percentage and (total number) a
| Lesion Characteristics | Type 1 CCAM | Type 2 CCAM | Type 3 CCAM |
|---|---|---|---|
| (n=8) | (n=10) | (n=9) | |
| Maximum diameter, mm (SD) a | 36.1 (10.9) | 31.6 (21.2) | 33.9 (11.6) |
| Stable evolution % (n) b | 62.5 (5) | 80 (8) | 44.4 (4) |
| Complete regression % (n) b | 0 (0) | 10 (1) | 44.4 (4) |
| Partial regression % (n) b | 25 (2) | 10 (1) | 11.2 (1) |
| Progression % (n) b | 12.5 (1) | 0 (0) | 0 (0) |
Table 3: Lesion characteristics according to Stocker´s Classification (n=27); *Data displayed as mean and standard deviation (SD) a or as percentage and (total number) b
Lesion dimensions remained stable throughout gestation in most cases (63%), however, ultrasonographic regression was observed in nine fetuses (33.3%). Mean gestational age at the time of diagnosis of the ultrasonographic regression cases was 24.5 weeks and 34.5 weeks by the time regression was documented. Of these cases, five showed complete regression and four partial regression (defined as a 50% or more decrease in size when compared to the initial measure). The complete ultrasonographic regression group consisted of four type 3 CCAM cases and one type 2. The mean lesion maximum diameter of the complete regression group was 26.8 mm. The Partial regression group included two type 1 CCAM cases, one type 2 and one classified as type 3 CCAM. The reported mean lesion maximum diameter for the partial regression group was 27.9 mm. A postnatal pulmonary lesion was observed in one of the complete ultrasonographic regression cases. In this case, type 2 CCAM was diagnosed at 21 weeks of gestation with an initial maximum diameter of 18.8 mm, lesion dimensions gradually decreased throughout gestation until reported undetectable at 34 weeks. However, a pulmonary lesion identified as type 2 CCAM was observed at birth. Lobectomy was performed and the diagnosis was confirmed by pathologic evaluation. Only one case demonstrated continued CCAM growth throughout the gestational period.
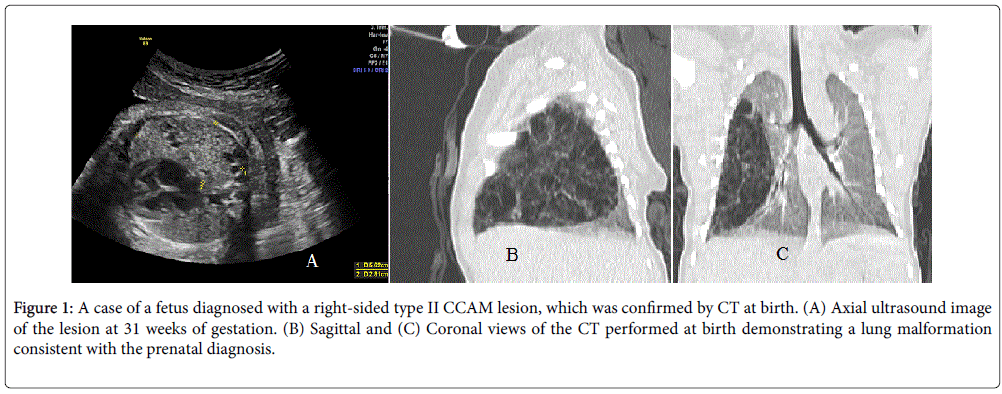
There were no elective pregnancy terminations. Fetal demise was reported in two cases, one at 28.4 weeks and another one at 34.6 weeks of gestation. In both cases of fetal demise hydrops was identified earlier in gestational period. Mean gestational age at birth was 37.1 weeks, mode of delivery was decided according to obstetric indications. The mean birth weight was 2,817 g (± 598). Most newborns required only usual neonatal care, while nine cases (33.3%) required mechanical ventilation. Prenatal diagnosis of CCAM was confirmed by image studies, pathology evaluation or both in all cases. Computed Tomography (CT) or magnetic resonance imaging (MRI) confirmed the diagnosis in 62.9% of the cases. Elective lobectomy was performed in 16 cases (59.2%) and pathology confirmation of prenatal diagnosis was available for all but two cases as those surgical procedures were performed at an outside institution. A hybrid lesion with mixed CCAM-bronchopulmonary sequestration was reported among the pathology confirmed cases. After birth, two cases showed progressive deterioration which lead to early neonatal death. In one case the newborn´s condition deteriorated within hours of surgery and death occurred on day two after delivery. The second case of neonatal death presented with deterioration shortly after delivery and death was reported on day five after delivery. Follow-up data were not available for these cases (Tables 1 and 4) (Figure 1).
Figure 1: A case of a fetus diagnosed with a right-sided type II CCAM lesion, which was confirmed by CT at birth. (A) Axial ultrasound image of the lesion at 31 weeks of gestation. (B) Sagittal and (C) Coronal views of the CT performed at birth demonstrating a lung malformation consistent with the prenatal diagnosis.
| Live newborns % (n) a | 96.2 (25) |
| Assisted mechanical ventilation % (n) a | 33.3 (9) |
| Diagnosis confirmation (Imaging) % (n) a | 62.9 (17) |
| Lobectomy % (n) a | 59.2 (16) |
| Diagnosis confirmation (Hystopathological examination) % (n) a | 51.8 (14) |
| Neonatal death (n) a | 7.4 (2) |
Table 4: Postnatal follow-up (n=27); *Data displayed as percentage and (total number) a
Discussion
Prenatal diagnosis of CCAM is uncommon, with a reported incidence of 1 in 25,000 pregnancies [2]. During the study period the observed incidence was 1 in 2,500, which could be explained by the fact that the study was conducted at a tertiary referral center. Advances in ultrasonographic technology have permitted a more frequent and earlier prenatal diagnosis of CCAM, with most cases being currently diagnosed at the morphologic scan at 18-22 weeks gestation [13]. This has enabled the identification of prenatal prognostic markers, which may guide the clinical approach and management of these fetuses. In our cohort, the mean gestational age at the time of diagnosis was 26.5 weeks as all cases were referrals from other institutions.
Right side predominance has been reported for CCAM lesions [1]. In our study, however, 55.6% of lesions were located in the left hemithorax. Type 2 CCAM was the most prevalent lesion type (37%) in this cohort, this differs from literature data where type 3 CCAM has been reported as the most frequent (50%) [3,11,13]. Main CCAM prenatal prognostic markers include: Size and evolution of the mass, mediastinal shift, development of hydrops and the finding of additional anomalies [8,14,15]. In our series, lesion dimensions remained stable throughout gestation in most cases (63%), ultrasonographic regression was observed in 33.3% of cases and only one case showed continued CCAM growth. A mediastinal shift was observed in more than half of the cases (59.2%), in agreement with other series [13]. CCAM may result in compression of the heart and great vessels ultimately leading to fetal hydrops, a known highly predictive factor for a negative outcome, with a reported associated mortality rate higher than 90% when managed expectantly [5,16,17]. In our cohort, hydrops was diagnosed in two fetuses (7.4%) that subsequently presented with intrauterine demise, a rate similar to that reported by other authors [3]. Associated anomalies are unusual but occur more often with type 2 CCAM in our study additional anomalies were observed in six cases (22.2%) half of which corresponded to type 2 CCAM [3,17]. There were no elective pregnancy terminations in our series, which could be explained by the influence of cultural aspect and religion beliefs in our country.
Several reports have shown significant or complete ultrasonographic regression in up to 30-60% of CCAM cases, this variability could be explained by the lack of universal criteria for lesion regression [9,10,18-20]. It is well documented that CCAM growth generally peaks around 18-25 weeks of gestation, while spontaneous regression has regularly been described between 28-37 weeks [3,5,8,10,19]. The precise mechanisms leading to CCAM regression remain largely unknown, it has been hypothesized that it may be secondary to outgrowing of the vascular supply of the CCAM or alternatively to spontaneous resolution of an underlying bronchial obstruction [3,9,10,16,21]. Ultrasonographic regression was observed in nine cases (33.3%) in our series, five cases showed complete regression and four partial regression, in agreement with other authors data [20]. The mean reported gestational age at the time of regression was 34.1 weeks, which is consistent with other reports [9]. Whereas in our cohort just one of the complete ultrasonographic regression cases (11%) showed a postnatal pulmonary lesion identified as CCAM, other investigators have reported this finding in up to 25-40% of the cases [9,10]. This phenomenon could be explained by the fact that lung lesions without mediastinal shift often become isoechoic and indistinguishable from the adjacent normal lung parenchyma during the late saccular stage of lung development [10,22]. In our study, 44.4% of the lesions that showed regression were categorized as type 3 CCAM, similar findings have been reported in two other series where resolution was more likely to occur when lesions were classified as microcystic [9,10]. Maternal betamethasone administration has shown to decrease growth and promote regression of CCAM lesions [5,8,19,23,24]. Reported CCAM regression rate after giving the standard dose of betamethasone is around 60%. This effect may involve down regulation of abnormal gene expression within the lesion and accelerated maturation of CCAM tissue [5,16]. Steroids were not given in any of the cases examined in this series, however, observed regression rate did not differ from that reported by other authors [19,20].
Although intrauterine fetal therapy has not been accepted as a universal treatment for CCAM, it has been considered as first-line therapy for fetuses that develop hydrops, especially in those cases related to type 1 CCAM with a dominant cyst [5,6,16]. Reported post inutero therapy complications are preterm labor, premature rupture of membranes, chorioamnionitis, catheter displacement, malfunction, occlusion and thoracic deformities [25]. No intrauterine interventions were performed in our study. However, when analyzing the two reported hydrops cases, one corresponded to the year of 1995 when fetal interventions were not available and the other was a type 3 CCAM for which most interventions are not feasible [26].
The majority of fetuses diagnosed with CCAM have a favorable outcome, with an overall survival rate of 90% [16,19]. In our cohort, the survival rate was 96.2% with a reported mean gestational age and weight at birth of 37.1 weeks and 2,817 g (± 598) respectively. Furthermore mechanically assisted ventilation was necessary in only nine cases (33.3%), all data in line with that reported in previous studies [9]. There is an ongoing debate about the ideal management of newborns with asymptomatic CCAMs [27]. While some authors advocate resection in all cases, due to the risk of malignancy (blastoma, bronchoalveolar carcinoma) and infection, recent trends would favor expectant management with serial CT follow-up, reserving surgical treatment for those cases where respiratory deterioration or infectious complications are present [17,21,27]. In our series, lobectomy was performed in 55.5% of the cases, a higher percentage than those found in recent publications that could be explained by the broad study period in which clinical approach and management of these cases may have been modified [1].
In the present study, we sought to better define the outcomes of expectant management in fetuses diagnosed with CCAM in an effort to enhance prenatal counseling in low-income settings where fetal interventions are limited. Although our results are reassuring showing favorable outcomes in most cases, we acknowledge that the retrospective design and long observation period of the study limit the strength of the data. Continued experience with these rare lesions at centers worldwide will hopefully give further insight into the evolution of CCAM and clarify the precise need for surgical fetal intervention in the years to come.
Conclusion
CCAM is a relatively rare abnormality of lung development, which can be accurately diagnosed by prenatal ultrasound. Conservative management is appropriate in most cases, with a favorable outcome and a chance of regression of up to one-third of the cases. Favorable perinatal results observed in this series are reassuring for lowincome settings where fetal interventions are limited.
Acknowledgements
The authors wish to acknowledge the staff of the maternal-fetal medicine, genetics and pathology departments of the National Institute of Perinatology where this study took place for their wonderful everyday job.
References
- Ducombe GJ, Dickinson JE, Kikiros CS (2002) Prenatal diagnosis and management of congenital cystic adenomatoid malformation of the lung. Am J Obstet Gynecol 187: 950-954.
- Laberge J, Flageole H, Pugash D, Khalife S, Blair G, et al. (2001) Outcome of the prenatally diagnosed congenital cystic adenomatoid lung malformation: A Canadian experience. Fetal Diagn Ther 16: 178-186.
- Gajeswka-Knapik K, Impey L (2015) Congenital lung lesions: Prenatal diagnosis and intervention. Semin Pediatr Surg 24: 156-159.
- De Santis M, Masini L, Noia G, Cavaliere A, Oliva N, et al. (2000) Congenital cystic adenomatoid malformation of the lung: Antenatal ultrasound findings and fetal-neonatal outcome. Fifteen years of experience. Fetal Diagn Ther 15: 246-250.
- Khalek N, Johnson MP (2013) Management of prenatally diagnosed lung lesions. Semin Pediatr Surg 22: 24-29.
- Wilson R, Baxter J, Johnson M, King M, Kasperski S, et al. (2004) Thoracoamniotic shunts: Fetal treatment of pleural effusions and congenital cystic adenomatoid malformations. Fetal Diagn Ther 19: 413-420.
- Davenport M, Eber E (2012) Long term respiratory outcomes of congenital thoracic malformations. Semin Fetal Neonatal Med 17: 99-104.
- Cass D, Olutoye O, Cassady C, Moise K, Johnson A, et al. (2011) Prenatal diagnosis and outcome of fetal lung masses. J Pediatr Surg 46: 292-298.
- Hadchouel A, Benachi A, Revillon Y, Rousseau V, Martinovic J, et al. (2011) Factors associated with partial and complete regression of fetal lung lesions. Ultrasound Obstet Gynecol 38: 88-93.
- Kunisaki S, Ehrenberg-Buchner S, Dillman J, Smith E, Mychaliska G, et al (2015) Vanishing fetal lung malformations: Prenatal sonographic characteristics and postnatal outcomes. J Pediatr Surg 50: 978-982.
- Kunisaki S, Barnewolt C, Estroff J, Ward V, Nemes L, et al (2007) Large fetal congenital cystic adenomatoid malformations: Growth trends and patient survival. J Pediatr Surg 42: 404-410.
- Stocker J, Madewell J, Drake R (1977) Congenital cystic adenomatoid malformation of the lung. Classification and morphologic spectrum. Hum Pathol 8: 155-159.
- Hsieh CC, Chao AS, Chang YL, Kuo DM, Hsieh TT, et al. (2005) Outcome of congenital cystic adenomatoid malformation of the lung after antenatal diagnosis. Int J Gynaecol Obstet 89: 99-102.
- Correia-Pinto J, Gonzaga S, Huang Y, Rottier R (2010) Congenital lung lesions-underlying molecular mechanisms. Semin Pediatr Surg 19: 171-179.
- Bunduki B, Ruoano R, Da-Silva M, Miguelez J, Miyadahira S, et al. (2000) Prognostic factors associated with congenital cystic adenomatoid malformation of the lung. Prenat Diagn 20: 459-464.
- Witlox R, Lopriore E, Oepkes D (2011) Prenatal interventions for fetal lung lesions. Prenat Diagn 31: 628-636.
- Zozzaro-Smith P (2013) Ultrasonography for fetal lung masses. Ultrasound Clin 8: 49-54.
- Monni G, Paladini D, Ibba R, Teodoro A, Zoppi M, et al. (2000) Prenatal ultrasound diagnosis of congenital cystic adenomatoid malformation of the lung: A report of 26 cases and review of the literatura. Ultrasound Obstet Gynecol 16: 159-162.
- Mann S, Wilson R, Bebbington M, Adzick N, Johnson M (2007) Antenatal diagnosis and management of congenital cystic adenomatoid malformation. Semin Fetal Neonatal Med 12: 477-481.
- Cavoretto P, Molina F, Poggi S, Davenport M, Nicolaides KH (2008) Prenatal diagnosis and outcome of echogenic fetal lung lesions. Ultrasound Obstet Gynecol 32: 769-783.
- Laberge J, Bratu I, Flageole H (2004) The management of asymptomatic congenital lung malformations. Paediatr Respir Rev 5: S305-312.
- Adzick NS, Harrison MR, Crombleholme TM, Flake AW, Howell LJ (1998) Fetal lung lesions: Management and outcome. Am J Obstet Gynecol 179: 884-889.
- Paranteau WH, Wilson RD, Liechty KW, Johnson MP, Bebbington MW, et al. (2007) Effect of maternal betamethasone administration on prenatal congenital cystic adenomatoid malformation growth and fetal survival. Fetal Diagn Ther 22: 365-371.
- Curran PF, Jelin EB, Rand L, Hirose S, Feldstein VA, et al. (2010) Prenatal steroids for microcystic congenital cystic adenomatoid malformations. J Pediatr Surg 45: 145-150.
- Merchant A, Parenteau W, Wilson R, Johnson M, Bebbington M, et al. (2007) Postnatal chestwall deformities after fetal thoracoamniotic shunting for congenital cystic adenomatoid malformation. Fetal Diagn Ther 22: 435-439.
- Litwinska M, Litwinska E, Janiak K, Piaseczna-Piotrowska A, Gulczynska E, et al. (2017) Thoracoamniotic shunts in macrocystic lung lesions: Case series and review of the literature. Fetal Diagn Ther 41: 179-183.
- Puligandla PS, Laberge JM (2012) Congenital Lung Lesions. Clin Perinatol 39: 331-147.
Citation: Gallegos SA, Sibaja MJR, Gaona JMG, Calvo JAR, Torrez BV, et al. (2018) Congenital Adenomatoid Cystic Malformation: Expectant Management Outcomes. J Preg Child Health 5: 387. DOI: 10.4172/2376-127X.1000387
Copyright: © 2018 Gallegos SA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 4433
- [From(publication date): 0-2018 - Sep 03, 2025]
- Breakdown by view type
- HTML page views: 3540
- PDF downloads: 893