Considerations for the Selection of Suitable Energy Sources Needed for Future World-Wide Energy Consumption.
Received: 20-Oct-2011 / Accepted Date: 28-Mar-2012 / Published Date: 18-Apr-2012
Abstract
The major part of worldwide energy production concerns fossil energy. Therefore, concomitant carbon dioxide production is a problem. What are suitable alternatives? Here we present considerations for selection of energy sources needed for future worldwide energy consumption. Plants are collectors and converters of sunlight and are indispensable in the carbon cycle. This cycle converts sunlight to chemical energy, and next frees this energy by biochemical oxidation in humans and other organisms, or by chemical oxidation. Currently, the carbon cycle is strongly disturbed by abundant use of fossil fuels. Sunlight- and wind energy-collecting and -converting systems seem to be the best energy-generating systems in near-by future, but in far future also nuclear fusion will become important. In addition, the more man collects non-carbon-related energy and uses available natural non-fossil organic materials for energy generation, the more he gets the energy demand under control and will reduce use of fossil fuel.
Keywords: Solar energy; Wind energy; Fossil energy; Nuclear energy; Carbon cycle; Fermentations; Carbon dioxide
19769Introduction
The problems
At present the need for energy is enormous [12]. At one hand people want to perform many activities and at the other they want to move by car, train or plane. Consequently, to achieve that we use as energy sources mainly fossil fuels, such as mineral hydrocarbons, varying from methane to kerosene. At combustion a lot of energy is freed, but simultaneously also massive amounts of CO2 arise [7].
After World War II the United States have constructed many fission reactors that strongly contributed to less CO2 increase. Because of problems regarding storage of radioactive waste the United States had stopped construction of new nuclear fission reactors about 30 years ago. In order to reduce future American CO2 production, in 2010 Barack Obama has presented a “climate change bill” to the Senate in his State of the Union. Obama said: “[. . . ] Thatmeans building a new generation of safe, clean nuclear power plants in this country. It means making tough decisions about opening new offshore areas for oil and gas development. It means continued investment in advanced biofuels and clean coal technologies [. . . ].”
Here, we present an overview concerning coherence of various energy sources and what it means for future worldwide energy consumption.
Energy laws
Important in this respect are the “energy laws” in nature. They tell us, that energy is never lost, and that energy can change from the one form to the other one. For example, radiation energy can transform to electric and chemical energy. Einstein’s law (E=mc2) says that matter can be transformed to energy and thus broadens the mentioned laws.
Energy coherence in nature
Combining these laws with insights of biological, microbiological, and biochemical processes, makes that all transformations that occur in the field of energy generation may be seen as parts of the complete energy flow on earth. From that view we present both the total energy flow and new visions in order to resist future problems regarding both energy generation, CO2 production and food consumption.
Sources Of Energy
Primary and secondary energy sources
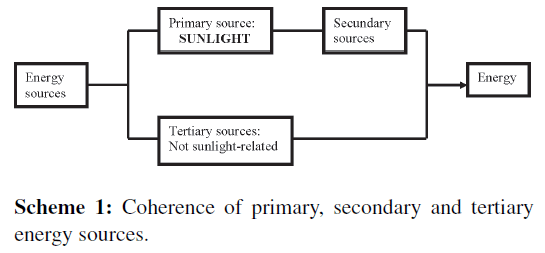
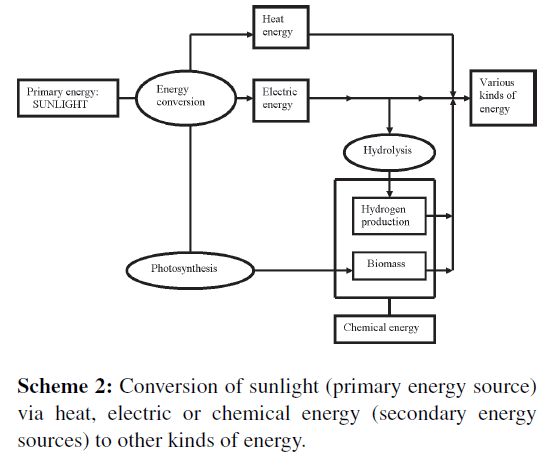
Already billion years sunlight is the primary source (Scheme 1) that creates all secondary energy sources, especially heat, electric, and chemical energy (Scheme 2), and all kinds of derived energy that we are using currently, such as wind energy, altitude-energy achieved by means of watermills, waterfalls, and storage lakes, or chemical energy in sugars, alcohol, and fossil oil.
The total energy absorbed by Earth atmosphere, oceans, and land masses is approximately 3,850,000 exajoules per year. In 2002 this meant more energy in one hour than the world used in one year [6]. In other words: the solar energy absorbed by the Earth surface is roughly about 10,000-fold the energy that we consume on this planet. This makes clear one of our energy problems: how can we increase the yield of energy conversion of solar energy for human consumption?
Solar radiation energy heats sea water and atmosphere and consequently both high-pressure and low-pressure areas will arise. The most important effect of this atmospheric activity is the wind that at present is only marginally used by sailing ships and wind mills. A secondary effect is the rain that falls onto the earth and next, as water, flows to the sea. Thanks to the gravity of earth this water has obtained potential energy that is first transformed to kinetic energy and next by human activity to electric energy. Whereas a major part of the potential energy does not become available as water flows to sea, a minor part does via water mills, waterfalls, and storage lakes.
Sunlight is transformed by the photosynthetic process into chemical energy and “canned” into glucose molecules. Most familiar photosynthetic organisms are the green plants. Although plants are seen all over the world, they still collect only 0.1% of the solar energy that reaches the planet Earth [11]. Beside these plants also green algae and blue algae (= cyanobacteria) are strongly involved in photosynthesis (Scheme 2). It has been estimated [9] that two-thirds of the total photosynthesis on Earth must be ascribed to the total oceanic [algal] photosynthesis.
Wood, a photosynthetic biomass product, is very important for current global live. Another part of (older) biomass has been synthesized million years ago and transformed to fossil carbon products, such as coal, gasoline, and gas. Till now, we have mainly used these kinds of biomass as sources for energy production.
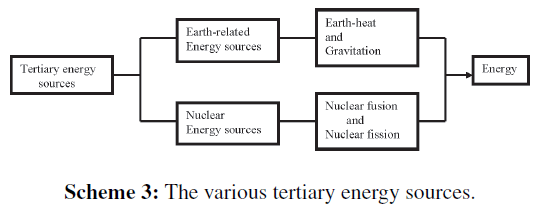
Tertiary energy sources
Tertiary energy sources are directly or indirectly not related to solar radiation (Scheme 3), e.g., the heat that comes from the inner side of our planet, i.e., from the superhot core. The outer side is rather cold and solid, but at several instances the hot content reaches to the earth surface via active volcanoes as red-hot lava or hot water. Icelanders exploit this glowing hot water (geysers) as energy source. In other countries, people have learned to less rigorously heat cold water deep in the less hot crust for exploitation.
Energy has also been generated from tide-differences of coastal water, that arise due to lunar gravity exerted on our globe. Differences of water levels may sometimes be rather small, but as it concerns enormous masses of water, it may still be profitable.
Nuclear energy
Two forms of nuclear energy can be distinguished (Scheme 3). The most well-known form concerns fission energy that arises from a small amount of atomic mass at the decline of radioactive atoms. Fission energy is released at explosions of nuclear fission bombs (A-bombs) with as fission elements “actinides,” i.e., heavy, radioactive metals, such as uranium and plutonium. Mankind has learned both to control release of this energy, especially in nuclear fission reactors, and to transform this energy via heat to electric energy. Disadvantage of this technology is (i) the storage of dangerous radioactive waste for more than 100,000 years, and (ii) the possibility that unpredictable extremely dangerous disastersmay occur, such as in Harrisburg (1979), Tsjernobyl (1986), and Fukushima (due to a tsunami that was caused by an earthquake [March 2011]).
Another form of nuclear energy concerns fusion energy that arises during nuclear fusion of deuterium and tritium to helium. With a higher out-put also here atomic mass is transformed to energy. It is difficult to start this process that occurs during the explosion of a hydrogen fusion bomb (Hbomb), and therefore a nuclear fission bomb is needed as detonator for ignition of an H-bomb. This process is not yet under control (ITER-Tokamak project in France).
Use Of Solar Energy
Especially in sunlight-rich areas, such as steppes and deserts, sunlight may be focused so strongly by means of concave mirrors, that this form of energy is available for certain melting, cooking, and evaporation processes. Sunlight (Scheme 2) is increasingly collected by specific solar energy collectors and used for generating electric current [4]. Electric energy is also used for conversion of water to molecular hydrogen and oxygen. In this case, sunlight is first converted to electric energy and next to chemical energy that is stored in the clean fuel, H2.
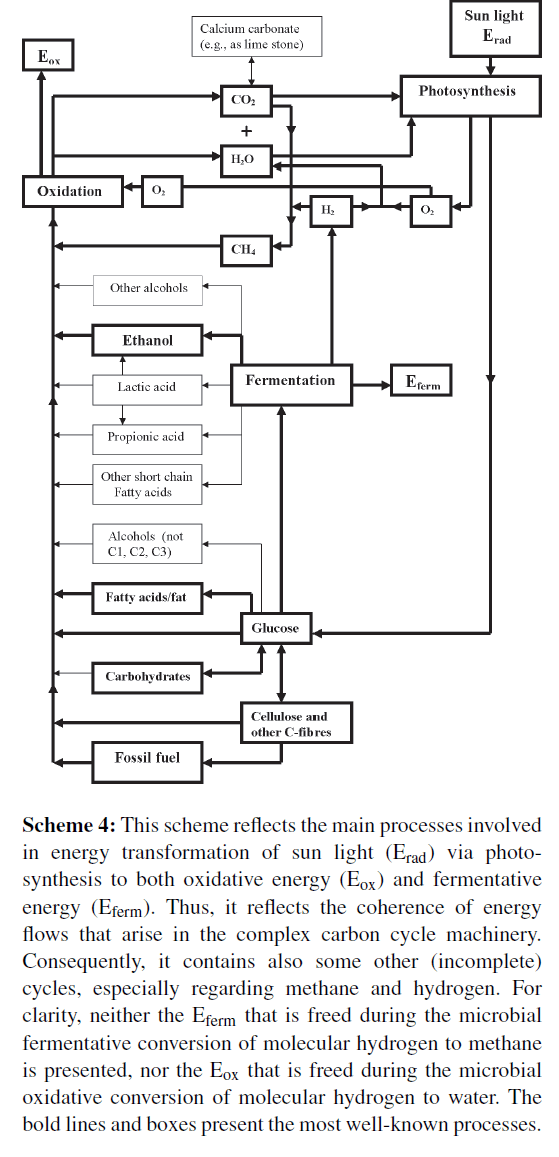
The oldest universal system for collecting solar energy is the photosynthesis (Scheme 2). Two chlorophyll systems convert sunlight to chemical energy that is stored in glucose. The process requires CO2, water, sunlight, and leaves of green plants. Glucose is often polymerized to starch, a nutritional substance, or to cellulose, a base component of wood. Photosynthesis can be considered as a system that passes on solar energy to a system in which carbon is important as energy transporter: when the caddie (CO2) is empty, it is reloaded and transformed to biomass (mainly sugars, fatty acids, and proteins) via photosynthesis (Scheme 4).
Scheme 4: This scheme reflects the main processes involved in energy transformation of sun light (Erad) via photosynthesis to both oxidative energy (Eox) and fermentative energy (Eferm). Thus, it reflects the coherence of energy flows that arise in the complex carbon cycle machinery. Consequently, it contains also some other (incomplete) cycles, especially regarding methane and hydrogen. For clarity, neither the Eferm that is freed during the microbial fermentative conversion of molecular hydrogen to methane is presented, nor the Eox that is freed during the microbial oxidative conversion of molecular hydrogen to water. The bold lines and boxes present the most well-known processes.
Role of carbon dioxide
Carbon dioxide is formed in an exothermic reaction with O2 (Scheme 4). During normal combustion energy is freed as light energy (flames) and heat. However, in the human body oxidative processes are very well controlled and consequently, released energy stored in special energy-carriers (e.g., in ATP molecules). Finally, all energy stored in glucose is transformed in well controlled steps to heat, a process in which CO2 arises again and again.
Micro-organisms, especially moulds and bacteria, gain a lot of aerobic energy from wood or wooden material. Each year immense quantities of plant rests are totally converted into CO2 by these organisms. The question even arises: who is the largest CO2 producer, mankind or the aerobic microorganisms, e.g., the aerobic moulds? Nevertheless, because of their high energy usage humans are essentially contributing to increased CO2 concentration in the air.
Carbon Cycle And Energy
Both the reducing processes, by which light energy is converted to chemical energy and simultaneously along a biochemical pathway (with CO2 as initial substance) canned in glucose (as final substance), and the oxidative processes, in which this energy is made free from carbohydrates for several purposes, form the hard core of the carbon cycle (Scheme 4).
Oxidized carbon (CO2) is converted to reduced carbon and vice versa. Complete conversion of reduced carbon to CO2 may occur in one process, but this ought absolutely not always to occur so. During microbial fermentation glucose is converted without oxygen. Such conversion provides the fermenting bacteria with only 2 ATP per glucose molecule. The fermentation products, lactic acid and ethanol, still contain 18 ATP per molecule. This energy is released during oxidative conversion to CO2.
After discovery of fire people started consuming stored energy in carbon. Now people massively do so for industry and transport, especially regarding fossil fuel (coal, peat, natural gas, and crude oils), and they feel that problems are arising, at least in local situations.
The mentioned fuels are energy reserves that are not actively involved in the carbon cycle and therefore we are very rapidly exhausting these reserves. The past 100 years energy consumption has enormously increased and a consequent massive CO2 emission cannot be denied. It is not excluded that the buffering capacity of the “green world” is insufficient. Still it is difficult to measure CO2 concentrations in such a way that reliable global pictures may arise. Complicating factors are (i) global photosynthesis by the total “green world,” i.e., the oceanic green algae included, (ii) CO2 uptake by the gigantic oceans, and (iii) CaCO3 precipitations on the bottom of these oceans. Nevertheless, people feel that the gigantic CO2 emission must be stopped. This has led to global environmental health conferences and has also contributed to the documentary film “An Inconvenient Truth,” an initiative of Nobel Prize winner Al Gore about CO2 and global warming. Questions that arise here are:
• What part of produced CO2 is taken up by the immense oceans?
• What part is precipitated as solid CaCO3?
• What part is converted to biomass by oceanic green and blue algae?
• Can oceanic algae introduce the CO2 from fossil fuel again into the carbon cycle?
• Shortly, can oceanic algae nullify by photosynthesis the world-wide CO2 production?
Fermentations
Bio-ethanol
Ethanol is increasingly used as bio-fuel. Although bioethanol may be produced by fermentative bacteria, in the current practice it stems mainly, if not all, from yeastmediated fermentation of sugar. An essential step in this fermentation is the instantaneous conversion of pyruvic acid via acetyl-CoA to ethanol and CO2. Some years ago in France bio-ethanol has been produced from superfluous wine, but this was an exceptional case. Normally, the sugar that is used for the mentioned fermentation processes, mainly comes from sugar cane or even from corn that should be used for human food consumption (first-generation fuel production). In October 2010 the Congressional Research Service reported that if the entire record U.S. corn crop was used to make ethanol, it would replace only 18% of the country’s gasoline consumption. Otherwise, making the 13 billion gallons of corn ethanol in 2010 consumed roughly 40% of the nation’s corn crop, cultivated on 32 million acres of farmland, thus pushing up food prices [2]. In this way bio-ethanol production is essentially contributing to a world malnourishment problem, especially in poor developing countries. In 2007 the WHO reported that the malnourished people in the world number more than 3.7 billion, at that moment the largest number of malnourished people and proportion ever reported [14].
In a similar way, bio-ethanol may also be achieved from plants with a high carbohydrate content. Currently, agriculture and horticulture is especially used for the harvest of fruits and seeds. However, plants produce far more polysaccharides, especially cellulose fibers (lignocellulosic parts). In the future we have to concentrate more and more on harvesting seeds and fruit for human consumption and other vegetable materials, such as leaves, stems (such as straw), branches and other detritus, for production of bio-ethanol as bio-fuel [13].
This fermentative bio-ethanol production should occur after de-polymerization of cellulose polysaccharide fibers in plants by means of highly active cellulase (especially obtained from moulds) to perform de-polymerization of the fibers [10].
Other fermentations
Fermentation processes depend primarily on the microorganisms involved. For instance, some micro-organism will ferment carbohydrates to propionic acid and/or propanol, whereas other ones will ferment them to butyric acid and/or butanol.
Ethanol is not an ideal fuel, since it has a lower energy density than gasoline and since its hygroscopicity poses a problem for storage and distribution. Higher alcohols (C4 and C5) have better energy densities, are not hygroscopic and are less volatile. In addition, branched-chain alcohols have higher octane numbers compared with their straightchain counterparts [13]. For this reason non-fermentative pathways have become starting-points for metabolic engineering regarding next-generation biofuels [1,8]. In these non-fermentative processes only the last step remains the same as in the fermentative ones: the reduction of a carbonyl oxygen to a hydroxyl group.
Methane and hydrogen
Energy, stored in biological material, e.g., farm- and sewer detritus, may be freed by specific anaerobic bacteria, and stored in molecular hydrogen or methane. Although the energy content of these gases can easily be freed for heat production by ignition in presence of O2, also specific oxidative bacteria are able to free this energy.
Deposition of carbon
What is currently going wrong in the carbon cycle? Part of the CO2 is converted to (bi)carbonate and as such present in rivers, seas and oceans. In turn, carbonate can be precipitated by calcium ions as calcium carbonate that is seen as Italian marble or as the “white cliffs” of Dover. At present, industries have started to condense and deposit produced CO2 in deep cavities in the earth’s crust, from which natural gas has been removed. A better alternative might be deposition of solid precipitated calcium or iron carbonates in old coal or ore mines.
A better and more significant manner, however, is to store abundant CO2 as durable wood of tropical hardwood trees, which can grow for a long time and thus can collect a lot of CO2.
Discussion and conclusions
Many people feel that mankind should strongly reduce CO2 production by diminishing use of fossil fuel. We should do a much greater appeal to the next “alternative” energy sources: (i) solar energy to be used for direct and indirect [3] production of electricity as well as for production of hydrogen, (ii) wind energy generated by means of windmills, (iii) electric energy produced by means of waterfalls, dam lakes, tide impact or dash of waves, (iv) earth heat, and (v) nuclear fusion energy. Nuclear fission energy is not preferred as alternative energy source mainly because of the storage problem of nuclear waste. Since generation of nuclear fusion energy does not produce radioactive waste, theoretically it seems suitable as future energy source, but its introduction is still far away. Increased use of all mentioned alternative energy sources will lead to considerable CO2 reduction.
We must realize, that because of the growing world population the need for energy will increase. Simultaneously, the production of CO2 will also increase, if we continue to use fossil fuel as main energy source. As long as we are not sure about exact impact of the present level of CO2 production, we have to limit use of fossil fuel. Even then it will last many years from now before CO2 overproduction is under control. Whatsoever, complete elimination of fossil fuels in next future is absolutely wishful thinking. Therefore, we have to make optimal use of all energy sources in our surrounding nature.
Additionally, we must collect CO2 as much as possible. Felling of tropical forest must be stopped, not because the lungs of the world are affected – the real lungs are the oceans –, but especially to fixate CO2 for decades as durable hardwood. Although it is good to keep our tropical forest intact, it is better to increase them. New cultivation means ongoing preservation of CO2 for decades. Meanwhile use of fossil fuel may be diminished and more and more other energy be used.
Future use of energy that is not coupled to the carboncycle will increasingly become important. Use of modern artificial solar energy collectors and giant wind-mills will most strongly increase, but use of other non-carbon energy sources will also increase, e.g., water force, and (as soon as possible) nuclear fusion.
The most well-known distinct sunlight collecting system is the green plant, an essential part of the carbon cycle. This natural system is indispensable for food production. However, we have to make more use of “waste” products of green plants. This will definitively lower the need of fossil fuel and leave more fruits and seeds for human consumption. Thus, optimal recycling of biomass keeps the carbon-cycle closed and consequently our global biosphere sound.
In 2002 according to the FAO world agricultural land supplies more than 99% of all world food (all cereal grains included [80%], and thus also corn for bio-ethanol production), whereas aquatic ecosystems supply less than 1% [5]. Why should not we start harvesting (“mining”) oceanic algae as new crude fuel instead of mining fossil fuel? At this moment these algae seem to be the eternal energy phoenix of the future: they are a cheap continuous living source from which a part might be taken to function as fuel. After energy yielding the products, such as CO2, are taken up by the oceans and via photosynthesis converted again to new algae biomass.
In 2011 David Biello wrote: “The breakthroughs needed to replace oil with plant-based fuels are proving difficult to achieve” [2]. In his opinion this regards achieving bioethanol produced from wood chips and waste (cellulosic ethanol) rather than achieving it from the edible corn kernel. By 2011 the U.S. was supposed to be producing 100 million gallons of cellulosic ethanol a year, but instead already in 2010 the 2011-goal was rolled back to just 6.5 million gallons, and it is unclear whether even that amount has been reached [2]. As long as for the production of suitable fuel more fuel is needed than can be produced, the process is useless. Currently it must be questioned if the production processes are not too refined, and if one has to look for cheaper and quite different processes. The advantage of extracting algae at large scale from the ocean means that the step of conditioned culturing has been passed and that compact algae mass has to be handled as starting material in processes that are comparable with refinement of crude oil. Regarding the next phase(s), is it possible to create by oxygen-free thermolysis energy-rich products directly from crude biomass, that are next partially separated by distillation-like processes?
References
- S. Atsumi, T. Hanai, and J. C. Liao, Non-fermentative pathways for synthesis of branched-chain higher alcohols as biofuels, Nature, 451 (2008), 86–89.
- D. Biello, The false promise of biofuels, Sci Am, 305 (2011), 58– 65.
- S. Cheng and B. E. Logan, Sustainable and efficient biohydrogen production via electrohydrogenesis, Proc Natl Acad Sci U S A, 104 (2007), 18871–18873.
- V. Cleave, Solar power: a flight to remember, Nature, 451 (2008), 884–886.
- Food and Agriculture Organization of the United Nations, Food balance sheets, FAO, Rome, 2002.
- P. Hunter, The impact of CO2. The global rise in the levels of CO2 is good for trees, bad for grasses and terrible for corals, EMBO Rep, 8 (2007), 1104–1106.
- J. D. Keasling and H. Chou, Metabolic engineering delivers next- generation biofuels, Nat Biotechnol, 26 (2008), 298–299.
- A. P. Kuznetsov and M. E. Vinogradov, On photosynthesis and organic carbon, carbon dioxide, and oxygen flows in the ocean, Biol Bull, 28 (2001), 84–87.
- D. Pimentel, T. Patzek, and G. Cecil, Ethanol production: energy, economic, and environmental losses, Rev Environ Contam Toxicol, 189 (2007), 25–41.
- L. Ruth, Bio or bust? The economic and ecological cost of biofuels, EMBO Rep, 9 (2008), 130–133.
- E. Waltz, Cellulosic ethanol booms despite unproven business models, Nat Biotechnol, 26 (2008), 8–9.
- World Health Organization, Malnutrition worldwide, WHO, Geneva, 2006.
Citation: Bongaerts GPA (2012) Considerations for the Selection of Suitable Energy Sources Needed for FutureWorld-Wide Energy Consumption. InnovativeEnergy Policies 2: 105.
Copyright: ©2012 Bongaerts GPA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Usage
- Total views: 16368
- [From(publication date): 12-2012 - Aug 29, 2025]
- Breakdown by view type
- HTML page views: 11785
- PDF downloads: 4583