Review Article Open Access
Global Photosynthesis and its Regulatory Role in Natural Carbon Cycle
Alexander A Ivlev*
Russian State Agrarian University of K.A. Timiryazev, Moscow, Russia
- *Corresponding Author:
- Prof. Alexander A Ivlev
Russian State Agrarian University of K.A. Timiryazev
Moscow, Russia
Tel: +7 (499) 976-0480
E-mail: aa.ivlev@list.ru
Received date: June 07, 2017; Accepted date: June 15, 2017; Published date: June 28, 2017
Citation: Ivlev AA (2017) Global Photosynthesis and its Regulatory Role in Natural Carbon Cycle. J Ecosyst Ecography 7: 233. doi: 10.4172/2157-7625.1000233
Copyright: © 2017 Ivlev AA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
Main features of global photosynthesis operating in global carbon cycle are considered. Global photosynthesis has all the basic features of normal photosynthesis of C3 type, excepting those that characterize ontogenetic changes. They include: the existence reciprocal processes assimilation and photorespiration, the possession of the key enzyme Rubisco having carboxylase/oxygenase activity, the existence of oscillatory mechanism switching over assimilation to photorespiration and back, carbon isotope fractionation in CO2 assimilation and photorespiration with opposite signs of isotope effects and some others. Considering these features of global photosynthesis, its functioning in global carbon cycle is analyzed. The photosynthetic mechanism forming carbon isotope composition of “living matter” and sedimentary organic matter is suggested. It is shown that environmental conditions of photosynthesis in the location plays a dominant role in forming carbon isotope composition of the above objects. The difference in environmental conditions determines facial isotopic shifts. The analysis of natural isotopic data confirms this conclusion. It allows saying that carbon isotopic difference between oils and organic matter is inherited from the difference between lipid fraction and other part of biomass of “living matter” that was the source of initial biogenic material. It is shown that global photosynthesis performs in global carbon cycle a regulatory role ensuring cycle’s movement to the ecological compensation point. The regulatory role is based on the ability of photosynthesizing organisms to respond to the increased oxygen concentration in the course of evolution by reducing assimilation and increasing photorespiration. The achievement of ecological compensation point means that oxygen atmosphere was stabilized and further accumulation of organic matter in the Earth’s crust stopped.
Keywords
CO2 assimilation; Environmental factors; Facial isotopic shifts; Global photosynthesis; Living matter; Oscillatory pattern; Photorespiration; Sedimentary organic matter
Introduction
To understand the global carbon cycle, it is necessary to find out the meaning of the two terms - “global photosynthesis” and “living matter”. “Living matter” – is a term, introduced by Vernadsky in 1926 [1], which he defined as the total biomass of all living on Earth organisms. Global photosynthesis is the process of synthesis of organic matter from carbon dioxide and water under sunlight, carried by all living on the Earth organisms with special photosynthetic apparatus. Formally, the global equation of photosynthesis can be written as it is written for any photosynthetic organism.
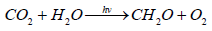
Since the global photosynthesis involves all of the photosynthesizing organisms living on the Earth, then given they constitute the beginning of all food chains, the total biomass of all living organisms, including photosynthesizing biomass and biomass of all consumers in trophic chains, can be regarded as a product of global photosynthesis. According to Vernadsky, it is the “living matter”.
Another product of global photosynthesis is an oxygen molecule in the Earth’s atmosphere. With a good approximation it can be regarded as such if to neglect the quantity of oxygen, which was in atmosphere prior to photosynthesis emergence [2].
What is common and what is the difference between global photosynthesis from regular photosynthesis of individual organism?
The regular photosynthesis of individual organism is described in detail in the literature [3]. What is common and what is the difference between global photosynthesis and regular photosynthesis of individual organism? At first we’ll see the common features. Considering that each photosynthesizing organism has CO2 assimilation and photorespiration, let’s assume that the same ability inherent in global photosynthesis. The first is responsible for the total biomass growth on the Earth, while the second is used in oxidation of part of the assimilated carbon to cover the energy costs of organisms partly to ensure adaptation of them to environmental conditions and to supply other metabolic needs. Part of respiration flux is used for biosynthesis of total biomass.
Assimilatory and photorespiratory metabolic fluxes of global metabolism are in reciprocal relations, i.e., the increase one of them is accompanied by a decrease of the other [4]. The contribution of the fluxes to the total biomass growth depends on CO2/O2 concentration ratio. The increase in CO2 concentration intensifies CO2 assimilation, whereas the increase in O2 concentration is followed by the intensification of photorespiration and by subsequent decrease of total biomass.
The main difference between global photosynthesis and that of photosynthesizing organism is in that it does not account for the changes associated with ontogenesis. It means that global photosynthesis doesn’t depend on time, but depends on the environmental conditions in a given place at a given moment. As compared with photosynthesis of individual organism, global photosynthesis can be imagined as an instant photo taken at the moment. These images, taken in different locations of the Earth at the same time, will be different depending on the conditions of photosynthesis in these locations only.
The control over the ratio of assimilatory and photorespiratory fluxes in a photosynthesizing cell, is carried out, as known, by the key enzyme of photosynthesis is ribulose bisphosphate carboxylase/ oxygenase (Rubisco) having carboxylase and oxygenase function and has a working feedback mechanism [5]. Functions oscillate, switching over depending on the ratio of CO2/O2 in the environment. The switching time determines the duration carboxylase and oxygenase phases [6] and contribution to the biomass of both processes. We accept that the same principle lies in the basis of the synthesis of the total photosynthetic biomass on the Earth.
Each photosynthesizing organism has a so-called CO2 compensation point, which corresponds to the state when the quantity of carbon assimilated in photosynthesis is equal to the quantity of carbon oxidized in the photorespiration. The state below the compensation point makes the physical existence of the organism is impossible, while the excess of the assimilated carbon over the oxidized means the growth of the biomass. Global photosynthesis has the same feature. The analog of CO2 compensation point in global photosynthesis termed as ecological compensation point. It corresponds to the state when the total amount of the assimilated carbon (total photosynthesizing biomass) becomes equal to the amount of organic material returned back completely to the oxidized inorganic form. Above this point the excess of carbon assimilated in photosynthesis turns into organic matter deposited in the Earth crust. With oxygen growth in the course of photosynthesis evolution the carbon cycle system spontaneously strives to ecological compensation point. On achieving this point the system goes into stationary state. Oxygen and carbon dioxide begin to oscillate around the steady meaning. It means that in the course of evolution oxygen reached its maximal meaning, while carbon dioxide its minimal meaning [7]. Thus, the oxygen and carbon dioxide concentrations in the environment, as well as the ecological compensation point are determined by the features of photosynthesis itself [8,9].
Carbon isotope fractionation in global photosynthesis
Following the adopted logic we assume that CO2 assimilation and photorespiration in global photosynthesis are followed by carbon isotope fractionation like photosynthesis of an individual organism. CO2 assimilation results in enrichment of total biomass in 12C isotope whereas photorespiration leads to accumulation in biomass of 13C.
Let’s see what affects carbon isotope composition of “living matter” on the Earth. The “living matter”, as said, consists of photosynthesizing and heterotrophic parts. The latter includes biomass of all organisms forming trophic chains. Since the photosynthesizing biomass is a source of the whole “living matter”, it can be regarded as a product of photosynthesis. If so, in the equation of global photosynthesis CН2O will denote the “living matter”.
From numerous isotopic studies of heterotrophic assimilation it was found that this assimilation isn’t accompanied with noticeable carbon isotope fractionation [10,11] and isotope composition of consumer’s biomass is determined by carbon isotope effect at the stage of photosynthesizing assimilation. It allows concluding that the “living matter” should be of the same isotope composition as its photosynthesizing part.
Besides carbon isotope fractionation associated with photosynthesis, in cells of any “living matter”, regardless of whether it is heterotrophic or photosynthetic, the intracellular carbon isotope fractionation takes place. This leads to a natural redistribution of carbon isotopes between fractions. The lipid fraction of biomass turns to be 3-5%o “lighter” than other factions (lignin, protein and carbohydrate) [12]. This is a result of a specific way of biosynthesis of lipid components in a cell that occurs through structural C2-fragments and is accompanied by a carbon isotope effect [13,14].
The enrichment in a “light” isotope begins to occur immediately after the death of the organisms in the course of sedimentation in water column and in burial of biogenic material in the sediment. The most frequent case is the burial in a subaqueous environment, which is considered below. Microbial and chemical oxidation of biogenic material commences already in the water column and in the sediment. Of the major biochemical fractions the lipid one is the most stable to oxidation. That’s why the initially small lipid fraction in the “living matter” begins to increase with oxidation development. This is clearly manifested in the shift of the isotopic composition of organic matter relative to the source of carbon of biogenic material. At all stages of sedimentation and digenesis there was a steady enrichment of organic matter in 12C, which indicates an increase in the relative proportion of total carbon of the lipid component [15,16]. But it doesn’t mean structural and chemical stability of the lipid fraction components.
Depending on carbon isotope composition of sedimentary organic matter two main factors determining carbon isotope composition: Sedimentary organic matter is an intermediate product on the way of biogenic material transformation to the final oxidized inorganic carbon forms in global carbon cycle [17]. As we use carbon isotopic technique to study carbon turnover, carbon isotope composition of sedimentary organic matter is of special interest. Carbon isotope composition of sedimentary organic matter is determined by two reasons:
1) The difference in photosynthesis conditions leading to different values of carbon isotope effects. This in turn determines different carbon isotope composition of “living matter” at different locations;
2) The uneven oxidation of the fractions in different locations leads to a change relative to the initial ratio of the fractions and to the emergence of additional isotopic shifts. As a result, the variance in the carbon isotope composition between organic matter and initial biogenic material emerges.
To assess the influence of these two reasons, let’s see first the scale of natural variations of carbon isotope composition of photosynthesizing organisms at different environmental conditions. Different natural parameters effect on carbon isotope composition of biomass. They are concentration of CO2 and O2 in the environment, pH values of the water in aquatic environment, salinity, light intensity, temperature, water availability, mixing and aeration, temperature and other parameters derived from the above [17-19]. The scale of isotopic variations covers range up to 20%o. After the burial of biogenic material the isotopic differences associated with uneven fractions oxidation begin to show. But their value, as said before, can’t exceed 5%o, i.e., maximal isotopic differences between fractions observed in living organisms. The case corresponds to the full oxidation of all fraction excepting lipid one. Evidently, this case is very unlikely. It allows concluding that photosynthesis conditions play a dominant role. Another conclusion is in that carbon isotope composition of sedimentary organic matter mainly depends on environmental parameters in photosynthesis.
Environmental factors exerting an impact on carbon isotope composition of sedimentary organic matter. Mechanism of formation of carbon isotope composition of “living matter” and sedimentary organic matter: As it follows from the above, isotopic differences of sedimentary organic matter should reflect isotopic differences of the “living matter” in the relevant period. From the above the environmental factors exerting an impact on photosynthesizing organisms have the same effect on the carbon isotope composition of organic matter.
Below we show the examples of impact of different environmental parameters on carbon isotope composition of photosynthetic biomass. Natural observations and in vitro experiments showed that the carbon isotopic variations depending on CO2 concentrations in the environment may achieve 25%o [20]. It was also found [21] that the 12C enrichment of biomass turned out to be much less than the carbon isotope effects of RuBP carboxylation on the enzymes isolated from the biomass of these organisms. The effects were about 60- 65%o. Such a great difference was found later to be mainly a result of photorespiration [22].
Environmental factors have different effects on carbon isotope composition of biomass. Among them the variations of CO2 concentration in the environment exerts the strongest effect. High CO2 concentrations results in 12C enrichment of biomass in the in vitro experiments [11,20,23]. Similarly pH manifests itself in aquatic environment. The low pH values, corresponding to high CO2 concentrations, provided 12C accumulation in biomass of marine alga Cyclotella, whereas high pH values resulted in abrupt enrichment in 13C [24,25]. In nature one can see the same picture [17].
The effect of environmental oxygen concentration on carbon isotope composition of biomass was firstly considered to be insignificant [26,27], since the role of photorespiration was underestimated and the reciprocal relation of assimilation and photorespiration was unknown. Later the role of carbon isotope effect of photorespiration was recognized as important [22,28] and some researchers have indicated the role of oxygen concentration on photorespiratory function of Rubisco [23,29]. It was shown that low O2 content caused the observed enrichment of organic matter 12C [30].
Numerous data showed that the environmental parameters, directly or indirectly affecting the CO2 uptake by photosynthesizing cells or facilitating CO2 availability, result in 12C enrichment of biomass. The parameters are: water salinity [31], water availability [32], temperature [27], cell density per volume unit [11], mixing and aeration of water [33,34]. But total effect of these parameters as a rule doesn’t exceed several per mille.
Light intensity exerts an impact on carbon isotope composition as well [35]. The isotopic shifts under low intensity have multidirectional character, but at high intensity isotopic shifts are always followed by 13C enrichment of biomass up to 5%o [36]. In nature so-called midday 13C enrichment of biomass is also associated with maximum light intensity [37]. The exposed leaves of plants reveal the accumulation of 13C as compared with shadowed leaves [38]. The effect of light intensity on carbon isotope composition of biomass can be explained by its impact on carboxylase/oxygenase activity of Rubisco. Under intense illumination CO2 the supply of Rubisco becomes insufficient to provide effective work of Calvin cycle. In this case photosynthesizing organisms use instead of CO2 another substrate O2, involving photorespiratory pathway [39]. It is followed by 13C enrichment of biomass.
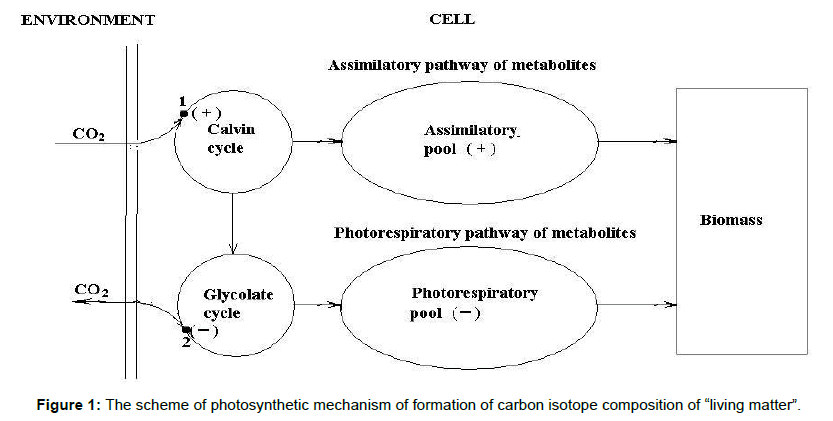
Figure 1 illustrates the principal scheme, explaining the mechanism of influence of different environmental factors on carbon isotope composition of living matter and, hence, sedimentary organic matter. It illustrates the impact of CO2/O2 concentration ratio in the environment. In the point 1, RuBP acceptor fixes CO2 and carbon isotope fractionation occurs resulting in 12C enrichment (+) of the assimilated carbon. In the point 2 glycine decarboxylation occurs followed by carbon isotope fractionation resulting in 13C enrichment (-) of the assimilated carbon. Switching over the enzyme from assimilatory function to photorespiratory occurs by RuBP carboxylase/oxygenase.
The mechanism takes into account that global photosynthesis fractionates carbon isotopes like a photosynthesizing cell, including isotope fractionation in assimilation and photorespiration. Note that these processes are in reciprocal relations and oscillate.
The key element of this mechanism is Rubisco engine, switching over assimilation to photorespiration function and back. In assimilatory phase the mechanism provides 12C accumulation in biomass (Figure 1). In this phase the total biomass on the Earth and the amount of sedimentary carbon in the Earth’s crust increases. In photorespiratory phase the mechanism provides 13C accumulation in biomass (Figure 1). Total biomass on the Earth and the amount of sedimentary carbon in the Earth’s crust decreases.
From the Figure 1 it becomes clear why the environmental parameter CO2/O2 ratio exerts the stronger effect on carbon isotope composition of biomass as compared with others. It is the result of the fact that it is connected directly with Rubisco engine. In contrast, the relatively small isotopic variations due to other parameters are explained by indirect impact of them. In general, the relatively small scale of isotopic variations observed in photosynthesis is due to a strong coordination of different photosynthetic processes in a cell to ensure optimal conditions for Calvin cycle functioning.
It was shown [3,40] that the coordination includes energy (ATP) and reducing equivalents (NADPH) formation coupled with electron transport chain associated with photosystem II. Thus, various environmental parameters determine CO2/O2 ratio inside a cell. The latter controls carboxylase/oxygenase activity of Rubisco and determine assimilation and photorespiration and, hence, carbon isotope composition of biomass.
Global photosynthesis in space and time
Global photosynthesis is manifested in space and in time in the form of facial isotopic shifts and temporal isotopic shifts of sedimentary organic matter correspondingly [41]. The difference between these two terms is the same as the difference between a photograph and a video. Facial isotopic shifts reflect the conditions of photosynthesis at this time in this location. That is why we attribute them to organic matter in rocks of the same age. As shown before, the main environmental parameter, exerting an impact on carbon isotope composition of organic matter, is the CO2/O2 concentration ratio in the surrounding.
It
is known that in the course of photosynthesis evolution the CO2/ O2 ratio on the Earth permanently changed from maximal value at the photosynthesis origin to minimal value at the ecological compensation point [7]. The changes of the ratio in the course of evolution were a saw tooth with gradual decrease of the average value to the ecological compensation point. It means that if to compare samples of organic matter separated by large time intervals they would correspond to different CO2/O2 ratios and have different values of carbon isotope composition.Facial isotope differences: Following the above definition we assert that a set of environmental parameters makes within a cell a certain ratios of assimilatory and photorespiratory fluxes, which form carbon isotope composition of “living matter” and further of sedimentary organic matter. It was confirmed by many researchers, who disclosed distinctive links of organic matter with the assumed zones of organisms’ habitats differing in CO2/O2 ratios and corresponding to marine, fresh water, terrigenous and salt marshes’ conditions [42-44]. They evidence that the initial isotopic discrepancies are remained, despite of transformations, and inherited at different stages of organic matter transformations [16,44]
The correspondence of zones with different CO2/O2 ratios to isotopic differences of carbon isotope composition can be traced not only for organic matter but for oils as well. As said before, oils origin is associated with the lipid fraction of the “living matter” which is enriched in 12C relative to other parts of biomass. If compared the difference between carbon isotope composition of “living matter“ and its lipid fraction with the corresponding difference in the carbon isotope composition of organic matter and genetically related oil, it is easy to see that both values are very close. It allows concluding that the latter difference is inherited from the “living matter“. From this fact it follows that no noticeable carbon isotope fractionation occurs in oil generation and the role of kinetic isotope effect of C-C and C-H bonds cleavage in oil formation is strongly overestimated [45,46].
It follows from this standpoint that the observed 12C enrichment of oils is a result of initial enrichment of lipid fraction due to intracellular carbon isotope fractionation [15,47]. One more important point should be explained. It is the cause of the dependence of carbon isotope composition of fractions and metabolites of “living matter” on isotope effect of photosynthesis. The thing is that carbon isotope effect of photosynthesis arises at the entry into any photosynthesizing cell. It means that all intracellular isotope effects that appear later and result in carbon isotope heterogeneity of biomass, including isotopic shifts its fractions and metabolites, should be summarized with photosynthesis effect and hence should reflect photosynthesis conditions. Thus the facial isotopic differences reflect a variety of photosynthesis conditions on the Earth at the same time.
Temporal isotope differences: If two samples of organic matter relating to the rocks of different ages and this time interval comprise one or more orogenic cycles, it is necessary to take into account the change in CO2/O2 ratio arising due to photosynthesis evolution. Naturally, it is more correct to compare temporal isotope differences for the samples of the same faces.
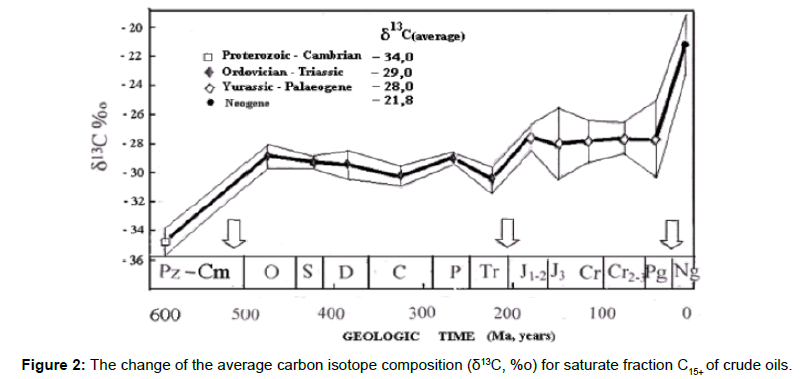
Data of Hayes et al. [48] disclose distinctive 13C enrichment of sedimentary marine organic matter relating to different intervals of geological time. They studied carbon isotope discrimination (the difference in carbon isotope composition of organic matter and carbonates, ε=δ13Corg C - δ13CcarbC):
| Neoproterozoic | from 800 to 750 Ma | e< -32% |
| from 685 to 625 Ma | -32<e< -28% | |
| Phanerozoic | less than 625 Ma | -28<e< -22% |
Distinct reduction of carbon isotope discrimination with time was found.
The same regularity one can find by studying carbon isotope composition of oils and their fractions in time comprising Neoproterozoic and Phanerozoic [49]. The study disclosed a similar change of carbon isotope composition of oil fractions C15+ as was seen before for organic matter. Carbon fractions become “heavier” with time (isotope discrimination become less). It should be emphasized that oil, despite to organic matter, reflects the average trend of carbon isotope composition in time, since during migration from source rock to reservoir, oil absorbs hydrocarbons from different sources. But in each case the general trend to 13C enrichment remains.
Figure 2 illustrates four steps of 13C enrichment of the fraction C15+ of oils occurring in Phanerozoic. Vertical bars are standard deviations, which increase with decreasing age. Arrows indicate Cambrian- Ordovician, Triassic-Jurassic, and Paleogene-Neogene boundaries where 13C enrichment occurs.
The 13C enrichment is associated with a 4-fold increase in oxygen content in the atmosphere in the course of 4 orogenic cycles [49].
Regulatory role of global photosynthesis
Global photosynthesis performs in global carbon cycle a regulatory role ensuring cycle’s movement to the ecological compensation point. The regulatory role is based on the ability of any photosynthesizing organisms to respond to the increased oxygen concentration by reducing assimilation and increasing photorespiration. The interaction of CO2 assimilation and photorespiration occurs by negative feedback mechanism [4]. Any increase in oxygen concentration causes an intensification of photorespiration which is followed by enrichment of biomass in 13C. The similar changes in global photosynthesis are associated with corresponding 13C enrichment of sedimentary organic matter. The formation of the mechanism of adaptation which emerged with advent of photosynthesis to resist oxygen action (the mechanism of photorespiration) later was transformed into mechanism of adaptation to all stressors.
How long oxygen content in the atmosphere could increase? The analysis allows concluding [7] that the rise of oxygen could last as long as the amount of the reduced carbon derived in photosynthesis doesn’t become equal the amount of carbon return back into oxidized inorganic form. This state is called the ecological compensation point, when the system achieves steady state. In case of deviation from this state under the action of any reason, the system spontaneously returns back to the initial state. Miocene was likely the time when the ecological compensation point was achieved. Two facts give indirect arguments in favor of this conclusion. First is the emergence of C4 plants having a new mechanism of CO2-assimilation. The second is the last wave of oil generation indicating the formation of rocks rich in organic matter. Both arguments evidence about low environmental concentration of CO2 and high concentration of O2 and indicate the end of orogenic cycle. Further any signs of orogenic cycles were not detected. The longterm orogenic cycles were completely replaced by short-term climatic oscillations. It was refection of the fact that the equilibrated system became sensitive to collisions of separate lithospheric plates [7].
Conclusions
Global photosynthesis has all the features typical to the normal photosynthesis of individual organism of C3 type, excepting ontogenetic features. They include: the existence of reciprocal processes assimilation and photorespiration, the possession of the key enzyme Rubisco having carboxylase/oxygenase activity, the existence of oscillatory mechanism switching over assimilation to photorespiration and back, carbon isotope fractionation in CO2 assimilation and photorespiration with opposite signs of isotope effects and some others.
Taking into account these features of global photosynthesis, the mechanism of formation of carbon isotope composition of “living matter” and of sedimentary organic matter in the frames of global carbon cycle model is suggested. The main photosynthetic enzyme Rubisco, having carboxylase/oxygenase activity, plays the key role in this mechanism.
The analysis of the natural carbon isotope data in conjunction with the mechanism of global carbon cycle functioning shows that environmental conditions of photosynthesis play a dominant role in formation of carbon isotope composition of “living matter” and sedimentary organic matter as well. Among them the content of CO2 and O2 in the environment are the most important. Many others environmental parameters (pH, salinity, mixing, water availability, etc.) exert an impact via their influence on the CO2/O2 ratio inside a cell. The role of the intracellular carbon isotope fractionation in formation of carbon isotope composition of the total carbon of “living matter” and sedimentary organic matter is insignificant.
Facial isotopic shifts of sedimentary organic matter are determined by effect of photosynthesis depending on environmental conditions in habitats. The fractions of sedimentary organic matter undergo the same isotopic shifts as organic matter itself. The isotopic difference between oils and organic matter of the source rocks is inherited from the difference between lipid fraction and other part of biomass of the assumed “living matter” in the related habitat. The isotopic difference between lipid fraction and other part of biomass is a result of intracellular carbon isotope fractionation in cells of “living matter”.
The temporal isotopic differences of sedimentary organic matter are due to the change in CO2/O2 ratio in the environment arising owing to evolution of photosynthesis during the period separating sampling of organic matter. Of course, samples should be taken from the same facies.
It is shown that global photosynthesis performs in global carbon cycle a regulatory role ensuring cycle’s movement to the ecological compensation point. The regulatory role is based on the ability of photosynthesizing organisms to respond to the increased oxygen concentration in the course of evolution by reducing assimilation and increasing photorespiration. The achievement of ecological compensation point means that oxygen atmosphere was stabilized and further accumulation of organic matter in the Earth’s crust stopped.
References
- Vernadsky VI (1986) The Biosphere. 1926 ed. Complete Annotated Edition: Copernicus Books.
- Rutten MG (1971) The Origin of life by natural causes.
- Edwards G, Walker DA (1983) C3, C4: Mechanisms, and cellular and environmental regulation of photosynthesis. Univ of California Press.
- Andre MJ (2011) Modelling 18O2 and 16O2 unidirectional fluxes in plant: II. Analysis of Rubisco evolution. BioSystems 103: 251-264.
- Igamberdiev AU, Roussel MR (2013) Feedforward non-Michaelis mechanism for CO2 uptake by Rubisco: Contribution of carbonic anhydrases and photorespiration to optimization of photosynthetic carbon assimilation. BioSystems 107: 158-166.
- Dubinsky AY, Ivlev AA (2011) Computational analysis of the possibility of the oscillatory dynamics in the processes of CO2 assimilation and photorespiration. Biosystems 103: 285-290.
- Ivlev AA (2015) Global redox cycle of biospheric carbon: interaction of photosynthesis and earth crust procsess BioSystems 137: 1-11.
- Igamberdiev AU, Lea PJ (2006) Land plants equilibrate O2 and CO2 concentrations in the atmosphere Photosynth Res 87: 77-194.
- Jahren AH, Arens NC, Harbeson SA (2008) Prediction of atmospheric d13CO2 using fossil plant tissues. Rev Geophys 46: 1-12.
- Parker PL (1964) The biogeochemistry of the stable isotopes of carbon in a marine bay Geochim et Cosmochim Acta 28: 1155-1164.
- Sackett WM, Eckelmann WR, Bender ML, Be AW (1965) Temperature dependence of carbon isotope composition in marine plankton and sediments. Science 148: 235-237.
- Ivlev AA (2001) Carbon isotope effect (13C/13C) in biological systems. Separation Science and Technology 36: 1819-1914.
- Monson KD, Hayes JM (1980) Biosynthetic control of the natural abundance of carbon 13 at specific positions within fatty acids in Escherichia coli. J Biol Chem 255: 11435-11441.
- Ivlev AA (2012) Oscillatory nature of metabolism and carbon isotope distribution in photosynthesizing cells. Photosynthesis-fundamental aspects. Intech Publishers. Croatia 341-366.
- Galimov EM (1968) Geochemistry of stable carbon isotopes.
- Ivlev AA (2016) Global carbon cycle and organic matter accumulation in the Earth crust. Geoinfor Goestat: An Overview 2016 4: 1.
- Degens ET (1969) Biogeochemistry of stable carbon isotopes. In Organic Geochemistry. Springer Berlin Heidelberg 7: 207-226.
- Ivlev AA 2004. Contribution of photorespiration to changes of carbon isotope characteristics in plants affected by stress factors. Russ J Plant Physiol 51: 271-280
- Rundel PW, Ehleringer JR, Nagy KA (1989) Stable isotopes in ecological research. Springer-Verlag. Ecological studies 68: 525.
- Pardue IW, Scalan RS, Van Baalen C, Parker PL (1976) Maximum carbon isotope fractionation in photosynthesis by blue-green and green algae. Geochim et Cosmochim Acta 40: 309-312.
- Estep MLF, Tabita FR, Parker PL, Baalen CV (1978) Carbon isotope fractionation by ribulose-1,5,-biphosphate carboxylase from various organisms. Plant Physiol 61: 680-687.
- Ivlev AA (1993) On the flow of “light” and “heavy” carbon in the coupling of assimilation and photorespiration. Fiziologia rastenii 40: 872-880.
- Williams DG, Gempko V, Frevolini A, Leavitt SW, Wall GW, et al. (2001) Carbon isotope discrimination by Sorghum bicolor under СО2 enrichment and drought. New Phytologist 150: 285-293.
- Benedict CR, Scott JR (1976) Photosynthetic carbon metabolism of a marine grass. Plant Physiol 57: 876-880.
- Benedict CR, Wong WW, Wong JH (1980) Fractionation of the stable isotopes of inorganic carbon by seagrasses. Plant Physiol 63: 512-517.
- Smith BN (1976) Evolution of C4 photosynthesis in response to changes in carbon and oxygen concentrations in the atmosphere through time. Biosystems 8: 24-32.
- Troughton JH (1980) 13C as a carbon indicator of carboxylation reactions. In: Encyclopedia of Plant Physiology. Heidelberg. Springer-Verlag 6: 185-190.
- Igamberdiev AU, Ivlev AA, Bykova NV, Threlkeld C, Lea PJ, Gardestrom P (2001) Decarboxylation of glycine contributes to carbon isotope fractionation in photosynthetic organisms. Photosynthesis Research 67: 177-184.
- Osmond CB, Bjorkman O (1975) Pathways of CO2 fixation in the CAM-plants Kalanchoe daigremontiana II. Effects of O2 and CO2 concentration in light and dark CO2 fixation. J Plant Physiol 2: 155-162.
- Sanchez A, Lopez-Ortiz BE, Aguiniga-Garcia S, Balart E (2013) Distribution and composition of organic matter in sediments of oxygen minimum zone of the Northeastern Mexican Pacific: paleoceanographic implication. Journal of Iberian Geology 39: 111-120.
- Neales TF, Frazer MS, Roksandic Z (1983) Carbon isotope composition of halophyte Dishyma clavellattum (Aizoaceae) as affected by salinity. Aust J Plant Physiol 10: 537-544.
- Wise RR, Frederick JR, Alm DM, Kramer DM, Hesketh JD, Crofts AR, Ort DR (1990) Investigation of the limitations of photosynthesis induced by leaf water deficit in field grown sunflower (Helianthus annuus L.). Plant Cell Environment 13: 923-931
- Abelson PH, Hoering TC (1960) Carbon isotope fractionation in the system CO2 (gas)-CO2 (aqua)-HCO3 (aqua). Ann Rep Dir Geophys Lab 59: 158-161.
- Abelson PH, Hoering TC (1961) Carbon isotope fractionation in formation of amino acids by photosynthetic organisms. Proc Nat Acad Sci USA 47: 623-629.
- O’Leary MH (1988) Carbon isotopes in photosynthesis. Bioscience 38: 325-336.
- Park R, Epstein S (1960) Carbon isotope fractionation during photosynthesis. Geochim et Cosmochim Acta 21: 110-119.
- Muraoka H, Tang Y, Terashima I, Koizumi H, Washitami I (2000) Contributions of diffusional limitation, photoinhibition and photorespiration to mid-day depression of photosynthesis in Arisaema heterophyllum in natural high light. Plant Cell Environment 23: 235-250.
- Borland AM, Griffiths H, Broadmeadow MS, Fordham MC, Maxwell C (1994) Carbon-isotope composition of biochemical fractions and the regulation of carbon balance in leaves of the C3-crassulacean acid metabolism intermediate Clusia minor L. growing in Trinidad. Plant Physiology 106: 493-501.
- Roussel MR, Ivlev AA, Igamberdiev AU (2007) Oscillations of the internal CO2 concentration in tobacco leaves transferred to low CO2. Journal of Plant Physiology 164:1188-1196.
- Roussel MR, Igamberdiev AU (2011) Dynamics and mechanisms of oscillatory photosynthesis. Biosystems 103: 230-238.
- Ivlev AA (2017) On the physical sense of isotope composition of carbonate and organic carbon in sedimentary rocks and its possible use in stratigraphy. Int J Earth Environ Sci N 5 (in press)
- Waseda A, Nishita H (1998) Geochemical characteristics of terrigenous and marine sourced oils in Hokkaido. Japan Org Geochem 28: 27-41.
- Sackett W (1963) Isotopic carbon composition of marine sediments. Trans Amer Geophys Union 44: 182-189.
- Meyers P (1994) Preservation of elemental and isotopic identification of sedimentary organic matter. Chem Geology 114: 289-302.
- Johns WD (1979) Clay mineral catalysis and petroleum generation. Ann Rev Earth Planet Sci 7: 183-198.
- Peters KE, Walters CC, Moldowan JM (2007) The Biomarker Guide. V.1 Biomarkers and isotopes in the environment and human history. Cambridge University Press.
- Yeh HW, Epstein S (1981) Hydrogen and carbon isotopes of petroleum and related organic matter. Geochim et Cosmochim Acta 45: 753-762.
- Hayes JM, Strauss H, Kaufman AJ (1999) The abundance of 13C in marine organic matter and isotopic fractionation in the global biogeochemical cycle of carbon during the past 800 Ma. Chem Geol 161: 103-125.
- Andrusevich VE, Engel MH, Zumberge JE, Brothers LA (1998) Secular, episodic changes in stable carbon isotope composition of crude oils. Chemical Geology 152: 59-72.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 4623
- [From(publication date):
September-2017 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 3648
- PDF downloads : 975