Case Report Open Access
Electrophysiological Recognition Of An Atrio-ventricular Mahaim Fibre Pathway.
James P Higham*University of Bristol Medical School, Bristol, England, UK
- *Corresponding Author:
- James P Higham
Medical student, University of Bristol Medical School
Bristol, England, UK
Tel: +44 7852864768
Fax: +44 7852864768
E-mail: jh12417@my.bristol.ac.uk
Received date: July 20, 2016; Accepted date: September 12, 2016; Published date: September 15, 2016
Citation: Higham JP (2016) Electrophysiological Recognition of an Atrio-ventricular Mahaim Fibre Pathway. Arrhythm Open Access 1:118.
Copyright: © 2016 Higham JP. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at
Abstract
Mahaim fibres are cardiac accessory bundles which can cause dangerous tachyarrhythmias. Despite their first description being many y ears ago, their recognition and diagnosis still proves difficult. The electrocardiographic features of Mahaim pathways are subtle and shared with other accessory pathways; as such, an electrophysiology study must be used to diagnose this pathway. Even then, it is often a process of elimination which leads to the eventual diagnosis. Here, I will present the case of a young woman with a peculiar history who was ultimately diagnosed with a Mahaim fibre pathway. Using this case, I will describe the key electrophysiological features and the treatment of the Mahaim fibre pathway.
Introduction
Mahaim fibres (MF) were originally described anatomically in 1938 [1]. They were shown to be conductive accessory fibres arising from either the atrio-ventricular node (AVN) or from the His bundle, with their distal insertion in the right ventricle [2-4]. These pathways were named nodo-ventricular and fasciculo-ventricular, respectively. MF were associated with pre-excitation of the ventricles and dangerous tachyarrhythmias due to retrograde conduction along the AVN-His system and atrial re-entry, making an understanding of their pathophysiology of vital importance. As more sophisticated electrophysiological techniques were developed and data gathered from patients with this condition, there appeared to be a mismatch between the early anatomical studies and case reports. In one case, the AVN and His bundle of a 29 year old male was frozen in a cryoablation procedure with no effect on pre-excitation: the AVN and His bundle pathway was not at all involved in this patient’s condition [5]. Previously, Becker et al. had identified a group of specialised fibres on the lateral tricuspid annulus which mimicked AVN function and projected to the right ventricle, though this did not change the mainstream ideology about MF-type tachycardias at the time [6]. The fibres were found to conduct both anterogradely and retrogradely, explaining how they could lead to dangerous re-entrant arrhythmias. A growing body of evidence suggested that the symptoms of MF were due to these AVN-like fibres (atrio-ventricular MF), with the other classes of MF being far less dangerous. In fact, it is now accepted that fascicluo-ventricular fibres play no role in clinical tachycardias [6]. There is, however, still debate over where exactly these atrioventricular fibres insert; opinion is divided between the His fascicles and ventricular conduction system [7]. This issue is beyond the scope of this case report and only the recognition of these AVN-mimicking fibres will be discussed here.
Case Presentation
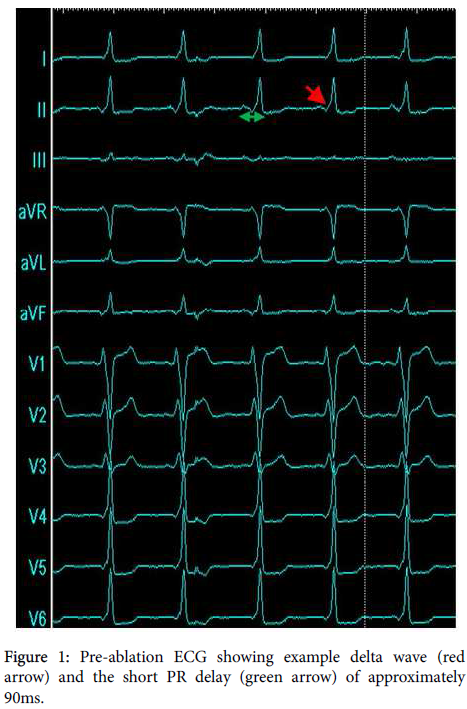
A 37-year-old Caucasian woman had a routine ECG following a three month history of chest pain and dizziness. Her symptoms typically lasted for between three and five minutes, with a rapid onset and offset. There was associated shortness of breath and occasional palpitations but no syncope. Deep breathing alleviated her symptoms. Both her siblings have had myocardial infarcts (MI; aged 29 and 32) and her father died of a MI; she has no other cardiovascular risk factors. Her examination was unremarkable: no added heart sounds were audible and no signs of vascular disease noted. The 12-lead ECG revealed a short PR interval (90 ms), pre-excitation delta waves and large R waves in V1. This patient was referred to a cardiology clinic and went on to have an exercise test. This test found pre-excitation throughout exercise, up to 187 beats per minute (100% of predicted heart rate). Her chest pain and other symptoms subsided soon after the exercise test, but it was agreed that she should have a catheter electrophysiology study and ablation for suspected Wolffe-Parkinson White Syndrome (WPWS). During the procedure, it became clear that her symptoms could not be due to WPWS and the properties of the accessory pathway were consistent with an atrio-ventricular MF. The pathway was located on the lateral tricuspid annulus, but ablation failed to completely block the pathway. However, given that this fibre only conducted anterogradely and blocked at high heart rates, it is deemed safe as it is unlikely to cause an arrhythmia.
Discussion
Electrocardiographic changes are usually subtle in patients with atrio-ventricular Mahaim fibres (AVMF). Pre-excitation, due to there being a shorter distance between the sino-atrial node (SAN) and the MF than there is between the SAN and AVN, gives rise to short PR intervals and a delta wave preceding the R wave (Figure 1). The R wave transition between leads V1 - V6 can be delayed, and large R waves seen in V1. A pattern resembling left bundle branch block can sometimes be seen due to the right ventricle being stimulated before the left, though this is a variable and transient feature.
An AVMF will only be diagnosed during a catheter electrophysiology study, because the surface ECG features of this pathway are shared with many others. For a diagnosis to be made, the properties of the pathway must be deduced. This includes the conductive properties – for example, the direction of conduction – and the pharmacological properties.
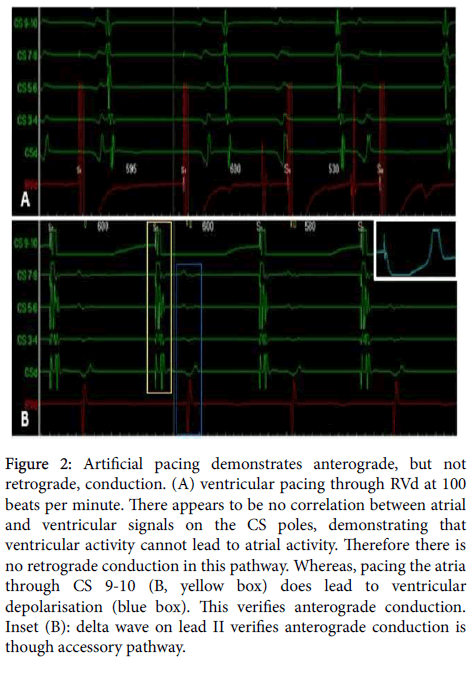
In this case, the right ventricle was first artificially paced at 100 beats/minute by the RVd catheter. As can be seen in Figure 2A, stimulation of the right ventricle does not lead to capturing and depolarisation of the atria; rather, there appears to be a dissociation between atrial and ventricular events on the CS poles. This demonstrates that there is no retrograde conduction in this pathway, ruling out such things as WPWS. Contrarily, pacing the CS 9-10 pole (i.e. pacing the atria) resulted in ventricular capture, verifying anterograde conduction (Figure 2B). Additionally, a delta wave can be seen in lead II when pacing from CS 9-10, revealing that this anterograde conduction is via the accessory pathway (Figure 3B, top-right inset, initial spike is a pacing artefact). AVMF pathways which conduct exclusively in the anterograde direction are not considered to be particularly dangerous, as there is low risk of re-entrant arrhythmias. Further, when the right atrium was paced, rather than CS9-10, a left bundle branch block-like morphology was seen (data not shown), confirming that this pathway conducted from the right atrium to right ventricle and, eventually, round to the left ventricle. At higher pacing frequencies, this accessory pathway blocked and normal ECG morphology resumed (data not shown): this pathway is, therefore, unlikely to lead to supra-ventricular tachycardia. To further validate this pathway’s safety, a post-ablation exercise stress test should be carried out.
Figure 2: Artificial pacing demonstrates anterograde, but not retrograde, conduction. (A) ventricular pacing through RVd at 100 beats per minute. There appears to be no correlation between atrial and ventricular signals on the CS poles, demonstrating that ventricular activity cannot lead to atrial activity. Therefore there is no retrograde conduction in this pathway. Whereas, pacing the atria through CS 9-10 (B, yellow box) does lead to ventricular depolarisation (blue box). This verifies anterograde conduction. Inset (B): delta wave on lead II verifies anterograde conduction is though accessory pathway.
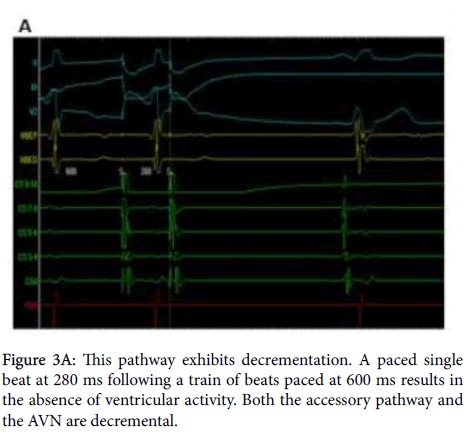
It has been previously documented that AVMF exhibit decrementation and are sensitive to adenosine - which are both properties of the AVN, too. Decrementation - a characteristic in which an increase in atrial pacing frequency leads to a prolonged atrioventricular delay - was demonstrated by pacing the CS 9-10 pole with an inter-stimulus interval of 600 ms, followed by a single beat paced after 280 ms. Pacing is then ceased; this protocol is known as “paced single beat”. Subsequent to the more rapid atrial stimulation, there was no ventricular activity recorded, demonstrating the signal did not conduct through the AVN or the accessory pathway (Figure 3A). After a period of approximately 900 ms, a cardiogenic atrial depolarisation followed by ventricular depolarisation is noted, showing the SAN has begun to pace the heart again at roughly 60 beats per minute. Had this pathway been - for example - WPW, ventricular activity would have been recorded due to conduction down the non-decrementing WPW pathway (Bundle of Kent).
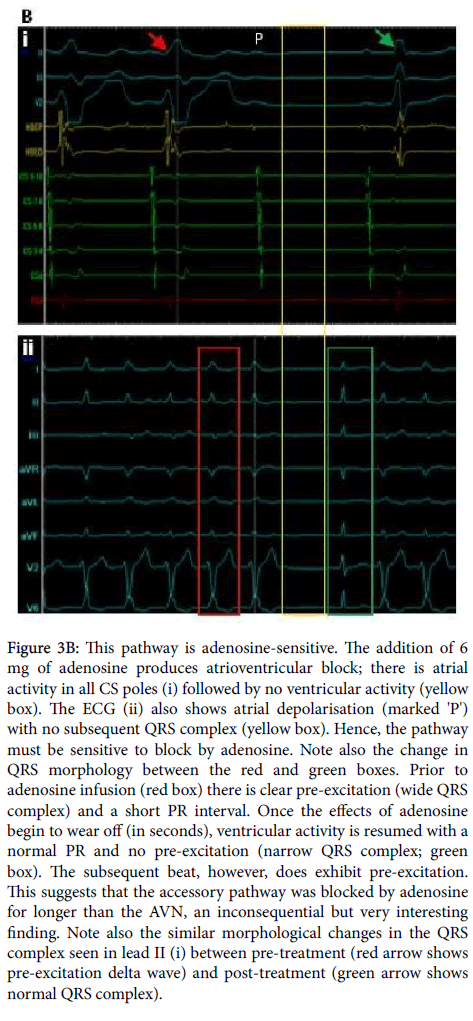
Adenosine is a natural metabolite and a drug used in the treatment of AVN re-entrant arrhythmias and some supra-ventricular tachyarrhythmias [8,9]. This drug is a negative dromotrope, mainly working by L-type calcium channel inhibition, to decrease AVN conduction and, therefore, increases atrio-ventricular delay. Adenosine can block decremental and non-decremental pathways [10,11], but for a diagnosis to be made it is vital to know the adenosine sensitivity status of the pathway in question. In the case presented here, the heart was not artificially paced as before, and 6 mg of adenosine was administered. Shown in Figure 3B is the response to adenosine: lead II shows a P wave with no subsequent QRS complex and the CS poles and RVd show no ventricular activity following atrial depolarisation. Once again, both the AVN and the accessory pathway have been blocked. Taken together, the properties so far are highly suggestive of an AVMF pathway, as they align with very few other known accessory pathways and these properties are well documented for Mahaim pathways.
Figure 3b: This pathway is adenosine-sensitive. The addition of 6 mg of adenosine produces atrioventricular block; there is atrial activity in all CS poles (i) followed by no ventricular activity (yellow box). The ECG (ii) also shows atrial depolarisation (marked 'P') with no subsequent QRS complex (yellow box). Hence, the pathway must be sensitive to block by adenosine. Note also the change in QRS morphology between the red and green boxes. Prior to adenosine infusion (red box) there is clear pre-excitation (wide QRS complex) and a short PR interval. Once the effects of adenosine begin to wear off (in seconds), ventricular activity is resumed with a normal PR and no pre-excitation (narrow QRS complex; green box). The subsequent beat, however, does exhibit pre-excitation. This suggests that the accessory pathway was blocked by adenosine for longer than the AVN, an inconsequential but very interesting finding. Note also the similar morphological changes in the QRS complex seen in lead II (i) between pre-treatment (red arrow shows pre-excitation delta wave) and post-treatment (green arrow shows normal QRS complex).
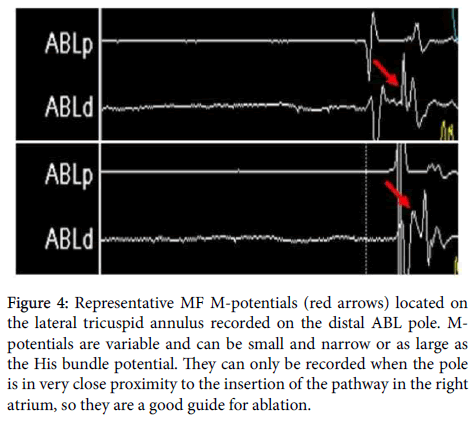
Once suspicions of an AVMF have been raised, one can go on to confirm this by searching for the fibre itself using the ablation catheter (ABL) to record signals around the tricuspid annulus [12]. If nothing can be found, other MF pathways, or even other types of accessory pathways, must be considered. The MF has a characteristic electrical signal, called the “M-potential”, which is indicative of the location of the fibre and commonly used to direct ablation. Other techniques, like electroanatomic mapping of the right atrium, can be of use in more difficult cases, and there are numerous ways of locating the MF, though these are often more time-consuming than simply locating the Mpotential. Here, M-potentials were recorded from the distal ABL (ABLd) pole on the lateral tricuspid annulus (Figure 4). With a site for the MF confirmed, radiofrequency ablation can be carried to destroy the pathway, isolating the atria and ventricles, other than the AVN pathway.
Figure 4: Representative MF M-potentials (red arrows) located on the lateral tricuspid annulus recorded on the distal ABL pole. M-potentials are variable and can be small and narrow or as large as the His bundle potential. They can only be recorded when the pole is in very close proximity to the insertion of the pathway in the right atrium, so they are a good guide for ablation.
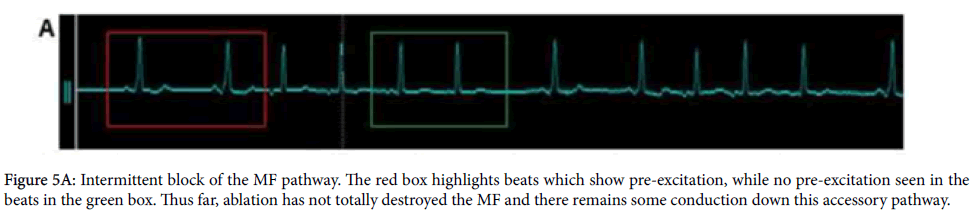
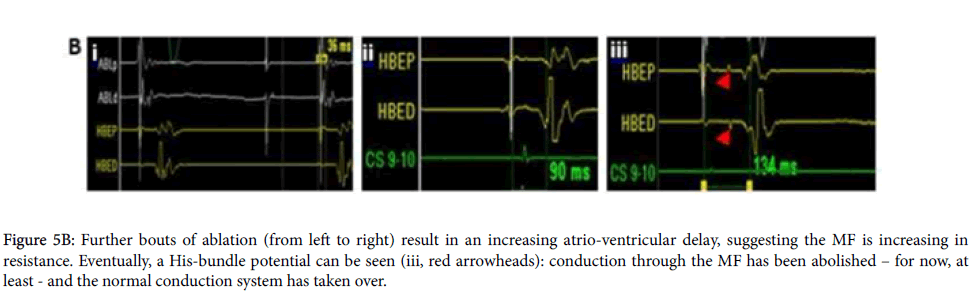
Ablation at the site of the M-potential initially resulted in an intermittent block of the MF (Figure 5A), suggesting incomplete electrical isolation. Continued ablation prolonged the atrio-ventricular conduction interval – measured in the His bundle poles - from 36 ms to 134 ms (Figures 5Bi and iii) and abolished pre-excitation (Figure 5A). Further, the His bundle poles detected a complete atrial, His bundle and ventricular signal for the first time in the procedure (Figure 5B): conduction was now exclusively by the AVN-His bundle system. Unfortunately, continued monitoring revealed only an intermittent block of the pathway had been achieved, and some conductive fibres remained. Consequently, this patient’s PR interval is variable and there is transient pre-excitation. This is not deemed dangerous, however, because this AVMF conducts only in the anterograde direction and blocks at high heart rates, and so is unlikely to precipitate an arrhythmia.
Figure 5B: Further bouts of ablation (from left to right) result in an increasing atrio-ventricular delay, suggesting the MF is increasing in resistance. Eventually, a His-bundle potential can be seen (iii, red arrowheads): conduction through the MF has been abolished – for now, at least - and the normal conduction system has taken over.
Conclusion
AVMF fibres are rarely diagnosed in the first instance using a surface ECG, due to many accessory pathways giving similar morphologies. Here, an electrophysiology study revealed an anterograde, decrementing, adenosine-sensitive pathway; all features at odds with the initial diagnosis of WPWS. Further investigation revealed M-potentials on the lateral tricuspid annulus, characteristic of an AVMF. Ablations lead to an intermittent block of the accessory pathway, which is safe because there is no retrograde conduction – unlikely to precipitate a re-entrant arrhythmia – and the pathway blocks at high heart rates, so a supra-ventricular tachycardia is also unlikely. This case not only demonstrates the properties of this rare accessory fibre, but also places emphasis on the techniques and logic required to diagnose such a pathway. Finally, it gives an insight into how it is decided whether or not a pathway presents any danger to the patient based on its electrophysiology.
References
- Ellenbogen KA, Ramirez NM, Packer DL, O'Callaghan WG, Greer GS, et al. (1986) Accessory nodoventricular (Mahaim) fibers: a clinical review. Pacing Clin Electrophysio l9: 868-884.
- Mahaim I (1947) Kent's fibers and the A-V paraspecific conduction through the upper connections of the bundle of His-Tawara. Am Heart J 33: 651-653.
- Neuss H, Thormann J (1977) Functional properties of mahaimfibers. KlinWochenschr 55: 1031-1037.
- Gallagher JJ, Smith WM, Kasell JH, Benson DW, Sterba R, et al. (1981) Role of Mahaimfibers in cardiac arrhythmias in man. Circulation 64: 176-189.
- Klein GJ, Guiraudon GM, Kerr CR, Sharma AD, Yee R, et al. (1988) "Nodoventricular" accessory pathway: evidence for a distinct accessory atrioventricular pathway with atrioventricular node-like properties. J Am Coll Cardiol 11: 1035-1040.
- Sternick EB (2003) Mahaim fibre tachycardia: recognition and management. Indian Pacing Electrophysiol J 3: 47-59.
- Tchou P, Lehmann MH, Jazayeri M, Akhtar M (1988) Atriofascicular connection or a nodoventricular Mahaimfiber? Electrophysiologic elucidation of the pathway and associated reentrant circuit. Circulation 77: 837-848.
- Schuller JL, Varosy PD, Nguyen DT (2013) Wide complex tachycardia and adenosine. JAMA Intern Med 173: 1644-1646.
- Lerman BB (2015) Ventricular tachycardia: mechanistic insights derived from adenosine. Circ Arrhythm Electrophysiol 8: 483-491.
- Ellenbogen KA, R ogers R, Old W (1989) Pharmacological characterization of conduction over a Mahaimfiber: evidence for adenosine sensitive conduction. Pacing Clin Electrophysiol 12: 1396-1404.
- Betts TR (2006) Mahaim tachycardia and intravenous adenosine. Heart 92: 1408.
- Bohora S, Dora SK, Namboodiri N, Valaparambil A, Tharakan J (2008) Electrophysiology study and radiofrequency catheter ablation of atriofascicular tracts with decremental properties (Mahaim fibre) at the tricuspid annulus. Europace 10: 1428-1433.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 12829
- [From(publication date):
September-2016 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 11930
- PDF downloads : 899