Review Article Open Access
Endocrine Disrupting Compounds in Lotic Ecosystems: A Review on Its Occurrence, Sources and Effects on Chironomus riparius
Pinheiro C1,2*, Pereira R1,2 and Vieira M1,21Department of Biology, Faculty of Sciences of University of Porto, Rua do Campo Alegre S/N, 4169-007 Porto, Portugal
2CIIMAR–Centro Interdisciplinar de Investigação Marinha e Ambiental, Universidade do Porto, Terminal de Cruzeiros do Porto de Leixões, Av. General Norton de Matos S/N, 4450-208, Matosinhos, Portugal
- *Corresponding Author:
- Pinheiro C
Department of Biology
Faculty of Sciences of University of Porto
Rua do Campo Alegre S/N, 4169-007 Porto, Portugal
Tel: +351 912 546 737
E-mail: cmpinheiro@fc.up.pt
Received date: March 06, 2017; Accepted date: March 21, 2017; Published date: March 28, 2017
Citation: Pinheiro C, Pereira R, Vieira M (2017) Endocrine Disrupting Compounds in Lotic Ecosystems: A Review on Its Occurrence, Sources and Effects onChironomus riparius. Environ Pollut Climate Change 1:117.
Copyright: © 2017 Pinheiro C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Environment Pollution and Climate Change
Abstract
Water plays a vital and irreplaceable role in the entire ecological balance. The increased human population growth, complemented by an intensification of agriculture, industrial development and urbanization, triggered an increase in pressures on water resources and in its consequent degradation, being that rivers one of the most resources intensively used and disturbed by man. Recent research has revealed the existence of hundreds of organic contaminants named “Emerging Organic Contaminants”. Within this vast group of contaminants, exists a class of xenobiotics capable of mimic hormones designated endocrine disrupting compounds. Since information, about this compounds is rare and spread out, the aim of our work is to examine the occurrence and sources of endocrine disrupting compounds and its effects in Chironomus riparius, an important aquatic insect used in ecotoxicological studies.
Keywords
Emerging organic contaminants; Endocrine disrupting compounds; Occurrence; Lotic ecosystems; Chironomus riparius.
Introduction
Rivers are one of the most intensively used and disturbed resources by man [2,3], wherein pollution is a severe worldwide problem that urgently requires the implementation of plans and ideas for routine monitoring. Every day, two million tons of human waste is discharged into water courses, including industrial wastes and other chemicals such as agricultural pesticides and fertilizers [4]. Even though water quality investigations have traditionally focused on nutrients, bacteria, metals and priority pollutants, recent research has revealed the existence of hundreds of new organic contaminants in wastewater and impacted urban surface waters [5,6]. These compounds can be mentioned as “Emerging Organic Contaminants” (EOCs).
Emerging organic contaminants can be defined as naturally occurring, manufactured or manmade chemicals or materials which have now been discovered or are suspected to be present in several environmental compartments and whose toxicity or persistence are likely to alter the metabolism of a living being [7]. That said, Houtman [8] classified EOCs in three categories: (1) compounds newly introduced to the environment; (2) compounds that have only recently been detected in the environment due to improved detection techniques and (3) compounds that have been known for a long time but have only recently been shown to have adverse effects on living beings (e.g. hormones). Nowadays, more than 1000 emerging pollutants, their metabolites and transformation products, are listed as present in Europe’s aquatic environment (http://www.norman-network.net).
Occurrence of EOCs can result from point (mainly urban and industry) and/or diffuse (agriculture) pollution. EOCs from urban or industrial WWTP are directly discharged into rivers where their environmental fate is of concern [9]. Rivers disperse EOCs to other water bodies, including aquifers, estuaries and marine systems. There is also direct discharge of wastewater to aquifers. This technique has been used in countries like Israel, Spain, US, Australia, South Africa and Japan [10-12]. EOCs discharged to groundwater may also occur through on-site (septic) waste treatment systems, threatening the groundwater supplies [13].
The fact that most of this compounds are chemicals that are extrinsic to most of the organisms’ normal metabolism, i.e., xenobiotics, it becomes important to understand the sources, occurrences and effects of the EOCs on behalf of understanding risks and developing monitoring and mitigation policies. Thus, the aim for this review is to examine the: (1) occurrence and sources of endocrine disrupting compounds, xenobiotics capable of mimic hormones, and (2) effects of a variety of endocrine disrupting compounds in Chironomus riparius, an important aquatic insect used in ecotoxicological studies mainly, due to its association with benthic sediments where many pollutants accumulate.
Endocrine disrupting compounds
The United States Environmental Protection Agency [14] has defined environmental “endocrine-disrupting compounds” (EDCs) as exogenous agents that interfere with the ‘‘synthesis, secretion, transport, binding, action, or elimination of natural hormones in the body that are responsible for the maintenance of homeostasis, reproduction, development and/or behaviour’’. However, several opinions as to what defines an EDC can be found in the literature [14]. These contaminants tend to mimic or antagonize the effects of hormones, alter the pattern of synthesis and the metabolism of hormones and modify the hormone receptor levels, thus disrupting the normal functioning of the endocrine system. Their harmful effects are on growth, development and reproduction in certain species and are associated with human disorders like infertility and birth defects [15].
According to Kresinova et al. [16], the most commonly studied and monitored EDCs are the natural oestrogens such as, oestrone (E1), 17β-estradiol (E2) and estriol (E3), synthetic oestrogens such as, 17α-ethinylestradiol (EE2) and industrial compounds such as bisphenol A (BPA) and nonylphenol (NP).
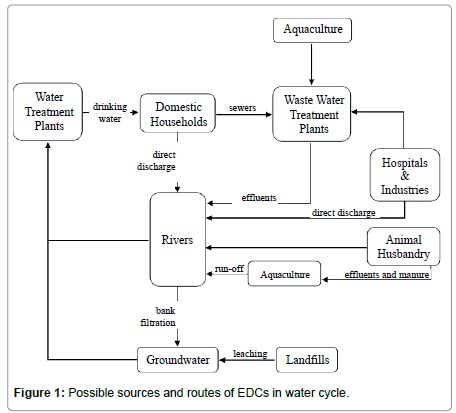
Possible sources and routes of the previously mentioned EDCs, in water cycle, are shown in Figure 1 [17]. It shows that rivers are affected direct or indirectly by five major sources, namely aquaculture, animal husbandry, domestic households, hospitals and industries and landfills.
Agriculture is also an important source to consider, since most of the drugs used in veterinary medicine end up in animal excreta. When manure is used on agricultural fields, the non-metabolized compounds that exist in it (or their biologically active metabolites) may affect the aquatic organisms mainly due to run-offs [18]. Moreover, pesticides are frequently applied to agricultural fields, which, coupled to their moderate persistence in the soil, can result in persistent surface water contamination [19,20].
Rivers are the first media receiving these estrogenic agents mainly arising from the discharge of effluents from WWTPs [21]. Given the important role and ecological services that lotic ecosystems provide to human communities, it is important to monitor the existing levels of EDCs on the resources used to produce drinking and irrigation water, as well as in those used for recreational purposes. Table 1 shows the occurrence and distribution of these six categories of EDCs, that can potentially cause an estrogenic endocrine disruption at very low concentrations, in surface water from ten countries dispersed worldwide [22].
From the collected data, we can argue that the concentrations of E1 in surface water are usually higher than the other natural oestrogens, E2 and E3. This might be due to the higher output rate of E1 from organisms and to the transformation of E2 to E1. Despite this, the synthetic oestrogen, EE2, is present at trace levels of several ng/L in surface water (Table 1). Considering that EE2 is considerably more persistent than E1, E2 and E3 [23], it is necessary to reduce the input of EE2 in surface water through an effective treatment of the effluents by the WWTPs.
Outstandingly, significantly higher levels of BPA and NP, when compared to the steroid oestrogens, were present in most surface water samples (Table 1), probably due to the wide usage of these synthetic chemicals in industries and the defectively treated wastewater. The discharge of domestic and industrial wastewater has been considered as a main cause of the presence of these xenoestrogens in surface water in comparison to other sources [24].
Effects on aquatic invertebrates–Chironomus riparius
Different authors consider that assays utilizing invertebrates offer some advantages over vertebrate models, since their use involves fewer ethical concerns over their welfare, doses are easier to deliver in the aquatic medium and their shorter life-span and inexpensive cultural requirements allow larger sets of data to be collected [25].
However, the scientific knowledge on the invertebrate’s endocrine system is very incomplete for most of the phyla [25] even though invertebrates are key-components of aquatic ecosystems. Moreover, the available information, on the biological effects of EDCs in these organisms, is scattered in comparison with vertebrates [26-28] Therefore, a problem arises since invertebrates are at considerable risk of exposure to these compounds.
Chironomids are a ubiquitous and ecologically diverse family of dipterans. Their ability to tolerate a wide range of conditions, including salinity, temperature, pH, current velocity and reduced levels of dissolved oxygen allows them to have a widespread distribution [29]. Chironomids’ larvae are mostly used in aquatic ecotoxicological studies mainly because of their association with benthic sediments where many contaminants accumulate [29,30].
Chironomus riparius is widely used in environmental toxicology, and has recently been selected as a reference organism for investigations of the potential endocrine disrupting effects of chemicals [31,32].
Bisphenol-A: Bisphenol-A (BPA) is widely used as an intermediate in the production of epoxy and polycarbonate resins, in dental sealants, and in mixtures with other plastic products [33,34]. The high production and widespread use of this industrial compound has been raising concern mainly because it has been shown to have endocrine activity in both vertebrates [35] and invertebrates.
Hahn et al. [36] detected, in a semi-static test system using 1.0, 100 and 3000 μg/L of BPA, an alteration of vitellogenin/vitellin production in males and female. Yolk concentrations in males decreased by 20 to 25% after exposure to BPA in all concentrations, while females were only affected in the highest BPA treatment, where yolk immunoreactivity was reduced by about 10% compared to the control [36].
Watts et al. [37] demonstrated that BPA could impact the number of emergences and the percentage of adult emergence. These effects occurred mainly in Chironomus riparius second generation, where the emergence of male and female adults was significantly delayed at concentrations ranging from 78 ng/L to 750 μg/L. Two years later Watts et al. [38] exposed Chironomus riparius larvae to sub-lethal concentrations of BPA (10 ng/L–1.0 mg/L). Molting was delayed and larval wet weight significantly reduced at the highest treatment concentration (1.0 mg/L).
| EDCs | E1(ng/L) | E2(ng/L) | E3(ng/L) | EE2(ng/L) | BPA(ng/L) | NP(ng/L) |
|---|---|---|---|---|---|---|
| AUSa | 0.55–20.91 | 0.39–3.77 | n.d.–1.9 | n.d.–0.52 | 4–59 | 287–2058 |
| BRAa,e | n.d.–39 | n.d.–14.8 | n.d.–2.3 | n.d.–25 | 25-84 | n.d. |
| CHNa,c,d | n.d.–17.8 | n.d.–0.42 | n.d.–0.37 | n.d.–38.1 | n.d.–3336.7 | 0.35–865 |
| ESPa | n.d.–17 | n.d. | n.d. | n.d. | n.d.–126 | 96–1483 |
| FRAa | 0.8–3.9 | 0.8–3.6 | 0.6–3.1 | 0.6–3.5 | 2–175 | 78–467 |
| ITAa | n.d.–10 | n.d. | – | n.d. | n.d.–140 | 130 - 580 |
| JPNa | 17.1–107.6 | 2.6–14.7 | n.d. | n.d. | 16.5–150.2 | 51.6–147 |
| MEXa | n.d. | n.d. | – | n.d. | 7 | 89–655 |
| MYSb | n.d. | 0–0.004 | 0–0.002 | 0–0.02 | – | – |
| NLDa | n.d.–7.2 | n.d.–1.0 | – | n.d.–0.4 | n.d. - 1000 | n.d.–4100 |
| PRTg | n.d.–26.9 | n.d.–11.5 | – | n.d. | n.d.–98.4 | – |
| TURf | n.d.–6.04 | n.d.–10.2 | n.d.–16.0 | n.d.–14.0 | – | – |
| USAa, h | 1.12–12.9 | n.d.–8.8 | n.d.–3.3 | n.d. | 1.37–57.14 | 18–690 |
E1–oestrone; E2–17β-estradiol; E3–estriol, EE2–17α-ethinylestradiol, BPA–bisphenol Aand NP–nonylphenol |
||||||
Table 1 : Occurrence and distribution of six EDCs in surface water all over the world.(a Zhang et al. [21], b,c Wang et al. [72], d Zhang et al. [16], e,f Aydin and Talinli [68]; Campanha et al. [69], Praveena et al. [70], g Rocha et al. [71], h Wang et al. [73], n.d.: not detected, –: not available
Planelló et al. [39] showed for the first time that BPA has a direct interaction, at a molecular level, with the insect endocrine system. Furthermore, they found that BPA acted similarly between vertebrates and invertebrates, i.e., BPA modulated the expression of the ecdysone receptor gene (EcR) in Chironomus riparius larvae, in a comparable way to that seen for the oestrogen receptor and other steroid receptors well characterized in vertebrates. Although more intensive studies are needed, the authors claim that EcR may be a useful tool for the screening of environmental xenoestrogens in insects. Moreover, the absence of a significant effect on ribosomal production suggests that BPA, for the times and dose tested in their study, did not affect the basic cell metabolism. In contrast, it was found that BPA can increase HSP70 gene expression, which is commonly considered to be an indication of cellular stress [39,40].
Park and Choi [41] and Martinez-Paz et al. [42], confirmed the genotoxicity potential of BPA in Chironomus riparius by using the comet assay, despite using different concentrations, 5–500 μg/L and 0.5–3 mg/L, respectively.
The study presented by Martinez-Paz et al. [42] is one of the first reports that support the activation of DNA repairing mechanisms under prolonged exposure to BPA genotoxicity, i.e., they showed that the extent of DNA damage at the lower concentration decreased with exposure time, possibly due to DNA repair activity.
Butyl benzyl phthalate: Butyl benzyl phthalate (BBP) is an important industrial chemical mainly used as a plasticizer in the production of vinyl tiles [43]. Because BBP is a phthalate, which softens and increases the flexibility of plastics without binding chemically to them, it tends to leach into the surrounding environment, becoming a ubiquitous pollutant and, consequently, entering the food chain [43].
Herrero et al. [44] tested short-term effects (24 h), long-term effects (48 h) and delayed toxicity of BBP in Chironomus riparius. Their results showed that BBP triggered a clear dose-dependent effect on the transcriptional levels of hsp70, hsp40, and hsp27 after 24 h exposures, but in different ways. On one hand, the highest concentrations produced a significant overexpression of hsp70 and hsp27 genes. On the other hand, the gene coding for the 40 kDa protein was inhibited even at the lowest concentrations. Prolonging BBP exposures to 48 h caused widespread inhibition of all the genes studied except for hsc70. In contrast, hsp70, hsp40 and hsp10 inducible genes tended towards significant overexpression after the toxin removal, in the delayed toxicity experiments [44]. Other studies have already confirmed BBP’s ability to affect the expression of heat-shock genes [40,45].
Exposure to BBP for 24 h caused a similar dose-dependent overexpression of EcR and ERR at higher concentrations. Their responses were also similar in longer experiments, with a moderate decrease for all concentrations after 48 h exposure and a marked upregulation in the delayed toxicity tests [44]. Planelló et al. [45] had already shown a significant overexpression of EcR in these insects due to BBP.
The effects of BBP on the transcriptional activity of GAPDH, CYP4G and GPx as well as variations in GST enzyme activity were also studied by Herrero et al. [44]. GAPDH transcriptional levels suffered no noteworthy changes, except for a significant overexpression at the highest BBP concentration after 24 h and a slight widespread repression after 48 h. No effects were detected in either GYP4G or GPx gene neither in 24 h acute exposures or delayed toxicity tests, except for a significant increase of the CYP4G level after removing 1 μg/L BBP. GST activity was significantly reduced, even at the lowest dose, in the first 24 h in the presence of BBP. This effect was emphasized after 48 h [44]. Moreover, BBP affects the levels of ribosomal transcription. According to Planelló et al. [45], a decrease in the levels of immature rRNA was caused by exposure to BBP at the higher concentrations tested, from 1 mg/L and above.
Nonylphenol: Nonylphenols (NP) are products of the degradation of nonylphenol polyethoxylates, which are widely used as surfactants with commercial, household, industrial and institutional applications. The discharge of effluents from sewage treatment plants represents the major source of nonylphenol in the environment. Here, nonylphenol can accumulate in different environmental partitions such as, river sediments and biota, acting in a more lipophilic and toxic way than their parent compounds [46,47].
In laboratory bioassays, Meregalli et al. [48] investigated mouthpart deformities of Chironomus riparius when larvae were exposed to 4NP (10, 50 and 100 μg/L). Survival of the larvae was not affected by the tested concentrations, but the frequency of mentum deformities increased significantly with 4NP at 50 and 100 μg/L.
The process of vitellogenesis is under hormonal control and the hormones involved are ecdysteroids and juvenile hormones, which in the adult insect, do not trigger molting processes but play a new role in gonadal maturation [49]. Hahn et al. [36] determined in a semistatic test system using 4NP (1.9 μg/L–2.0 mg/l) an alteration of vitellogenin/ vitellin production in males. The results showed a significant reduction in males’ yolk immunoreactivity at 1.9 and 30 μg/L and an increase at 2.0 mg/L.
Hagger et al. [50] suggested that environmental chemicals, that affect reproductive processes, do so, partly, through DNA damage pathways. Among the available genotoxicity indicator tests, the Comet assay has recently attracted much attention. The Comet assay, also called the single-cell gel electrophoresis (SCGE) assay, primarily measures DNA strand breakage in single cells. DNA strand breaks are potential pre-mutagenic lesions and are sensitive markers of genotoxic damage [51]. Both, Park and Choi [41] and Martinez-Paz et al. [42] found, in their studies, that DNA breakage increased in a dose-dependent manner under short NP treatments (24 h). Martinez-Paz et al. [42] also revealed that genetic damage significantly decreased after four days of exposure, suggesting the activation of repairing mechanisms under prolonged exposures in this species.
In a more recent work, Martinez-Paz et al. [52], studied the Hsp27 gene as a sensitive marker in response to exposure to chemicals in benthic invertebrates, concluding that nonylphenol did not alter the Hsp27 gene after the exposures assayed, in Chironomus riparius. Morales et al. [40] also studied the transcriptional regulation of an Hsp gene, Hsp70. Here, they demonstrate 4-nonylphenol produced a significant increase in Hsp70 mRNA levels early after exposure. However, none of the experimental treatments assayed caused a significant alteration in the expression level of the Hsc70 gene. Hsps are suitable as an early warning bioindicator of environmental hazard, because of their sensitivity to even minor changes in cellular homeostasis and their conservation along the evolutionary scale. Among Hsps, the Hsp70 family represents one of the most highly conserved proteins identified to date, and has constitutive as well as regulated members in all the organisms examined [53].
Nair and Choi [54] studied the effect of nonylphenol on the modulation of EcR mRNA, by assessing Chironomus riparius ecdysone receptor (CrEcR) mRNA expression. They found that the mRNA expression level of CrEcR was significantly up-regulated on exposure at 50 μg/L. Thus, stating its oestrogenic effects as an endocrine disruptor, within environmentally relevant concentrations.
Pentachlorophenol: Pentachlorophenol (PCP) was once one of the most widely used biocides, mainly as a wood preservative, but also for the formulation of fungicidal and insecticidal solutions and for incorporation into other pesticide products [55]. Nowadays, PCP is a restricted use pesticide and is no longer available to the public due to its carcinogenic and endocrine disrupting effects [55,56]. Morales et al. [40] studied the effect of PCP in the expression of heat-shock genes, concluding that, at a concentration of 1 μM, no significant effects were observed in the expression of HSP70 and HSC70. Its genotoxicity potential was explored by Martinez-Paz et al. [42], which results showed for the first time that this compound is genotoxic to the aquatic insect Chironomus riparius.
Tebufenozide: Tebufenozide is a diacylhydrazine moult-inducing insecticide that has been developed for the control of larval lepidopteran pests in agriculture including forest and fruit-crops [57]. This compound is a non-steroidal ecdysone agonist that mimics natural moulting hormones, which contain, mainly, 20-OH ecdysone in larval insect. Insecticidal activity of Tebufenozide is shown by inducing premature and incomplete larval moult [57].
According to research, Hahn et al. [58] was the first work to report No-observed-effect concentration (NOEC) and lowest-observedeffect concentration (LOEC) values of 13.2 and 17.2 μg/L, respectively, for Chironomus riparius during a 24 day toxicity test after static contamination of first instar larvae with tebufenozide. A semistatic exposure of fourth-instar larvae was also performed, revealing a lower susceptibility of elder larvae (NOEC 30 μg/L, LOEC 60 μg/L and LC50 81.94 μg/L). In the semistatic exposure, pupa mortality was twice as high in males as in females during the 100 μg/L treatment. This sexrelated difference probably resulted from the endocrine activity of Tebufenozide. One year later, Hahn et al. [36], tested vitellogenesis as a marker for possible effects of endocrine-disrupting agents using concentrations that are environmentally relevant. Tebufenozide did not affect the yolk protein content after a semistatic exposure of 10 μg/L but it reduced the yolk protein content of males at 80 μg/L.
Reproduction is also an endpoint, as suggested by the OECD guideline [59]. This is important, considering the role that reproduction plays as the main process linking the individual to the population. Tassou and Schulz [60] were the first to demonstrate that the exposure to environmentally relevant sub-lethal concentrations of Tebufenozide affected developmental and reproductive processes of Chironomus riparius. Their results indicated a reduction in reproduction and emphasised the importance of considering reproduction as an endpoint for the detection of EDCs.
Tributyltin: Tributyltin (TBT) belongs to the organotin compounds or stannanes, used for disinfection, antifouling and preservation in industrial processes. Due to its toxic, persistent, bioaccumulative and endocrine disruptive activity, TBT is included in the EU list of priority compounds in water [61].
A laboratory study of Hahn and Schulz [62] resulted in sex-specific effects of TBT on molting hormone biosynthesis and imaginal disc development of Chironomus riparius. Ecdysteroid synthesis decreased significantly in female larvae at all concentrations (50, 500 and 5000 ng TBT as Sn/l), whereas a significant increase of biosynthesis rate occurred in male larvae in the 500 ng/l treatment. In vivo experiments with development of the genital imaginal disc within a 48 h exposure period revealed a significantly slower development in female larvae and a significantly faster development in male larvae at all concentrations tested (10, 50, 200 and 1000 ng TBT as Sn/l).
In the same year, Martinez-Paz et al. [42] and Morales et al. [63] demonstrated that TBT had the highest values of DNA breakage at the lowest concentration (0.1 ng/L) when compared to other organic pollutants. Morales et al. [63] also showed that TBT can induce a significant overexpression of the EcR gene, the ultraspiracle (usp) gene and the ecdysone-inducible E74 gene, all key ecdysone-responsive genes, as well as of the oestrogen-related receptor gene (ERR), i.e., TBT it is capable of activate hormonal nuclear receptor and early-responsive genes as within only 24 h of exposure at environmentally relevant concentrations.
Triclosan: Triclosan (TCS), a halogenated phenol, is a non-ionic, broad spectrum antimicrobial. The hormonal activity of TCS has been demonstrated by its capability of modulating thyroid hormone-related genes and anuran development [64] and it has endocrine disruptive effects in fishes [65].
Martinez-Paz et al. [42] provided the first evidence of TCS’ potential genotoxic damage because of DNA fragmentation in freshwater invertebrates, through comet assay on Chironomus riparius. TCS also increases the expression of hsp27 gene at high concentrations of 1000 μg/L in this species [52].
17α-ethinylestradiol: 17α-ethinylestradiol (EE2) is a synthetic steroid that has become one of the most commonly used active ingredients for oral birth-control contraception [65].
In a two-generation experiment using Chironomus riparius and EE2, Watts et al. [37] could demonstrate that emergence times and the percentage of adult emergence were affected. These effects were mainly associated with the second generation of test animals. At very low concentrations (1.0 ng/L) of EE2, both the first and second generation of adults emerged significantly earlier than control animals.
Watts et al. [38] exposed Chironomus riparius larvae to sub lethal concentrations of EE2 (10 ng/l–1.0 mg/L). Molting was delayed and larval wet weight significantly reduced at the highest treatment concentration (1.0 mg/L). However, in contrast to Meregalli and Ollevier [66] deformities in the mentum of mouthparts were observed at low exposure concentrations (10 ng/L–10 μg/L).
Conclusion
The aim of this review was to help to understand how the large occurrence and the vast sources of EDC’s could affect the species Chironomus riparius by analysing different endpoints. Since this species plays an important role in the food chains of aquatic communities, representing a major link between producers and secondary consumers [67], the study of the effects of these xenobiotics can bring some evidences about the populations’ and communities’ future.
Although Chironomus riparius provides an excellent model to address some fundamental questions regarding the endocrine disrupting compounds’ effects in aquatic communities, a scarce number of research papers have been published about the potential endocrine disruption in this insect since the beginning of the century. During our initial research, only eight papers, on the effects of endocrine disrupting compounds on Chironomus riparius, were found, representing, therefore, less than 1% of those listed in the NORMAN network database.
Most of the EDCs studied did cause an effect at ecological relevant concentrations to C. riparius and some had effects on the following generations. These bioaccumulated EDCs inherited from the mother not only influence the morphological and physiological development of the offspring but also the offspring’s’ reproductive behavior as adults and, in consequence, this adult behavior can have further consequences on the sexual development of their own young [68].
Understanding the response to EDCs means identifying the action mechanism in the endocrine system of these organisms and relating them to the alterations observed in the endpoints studied. Therefore, future research on endocrine effects in aquatic insects, using a variety of different endpoints at different life stages of a species, and validation of hazard/risk assessment procedures could enable better future protection of these ecologically essential invertebrates.
Acknowledgement
This article is a result of the project INNOVMAR - Innovation and Sustainability in the Management and Exploitation of Marine Resources (reference NORTE-01- 0145-FEDER-000035, within RL ECOSERVICES), supported by NORTE 2020, under the PORTUGAL 2020 Partnership Agreement, through the ERDF.
References
- Rodrigues A, Martins G, Ribeiro D, Nogueira R, Monteiro P et al. (2006) Modelação da qualidade da água do rio Ferreira: Avaliação preliminar de riscos ambientais. 8° Congresso da Água, Figueira da Foz, 13-17 de Março de 2006, Associação Portuguesa de Recursos Hídricos, p: 11.
- Matono P, Ilhéu M, Sousa L, Bernardo JM, Formigo N, et al. (2006) Aplicação da Directiva Quadro da Água. Tipos de rios portugueses com base na fauna picola. 8º Congresso da Água, APRH.
- Oliveira JM, Santos JM, Teixeira A, Ferreira MT, Pinheiro PJ, et al. (2007) Projecto Aquariport: Programa Nacional de monitorização de recursos piscícolas e de avaliação da qualidade ecológica de rios. DGRF, Loa, p: 96.
- UN WWAP (2003) Water for people, water for life: 3rd World Water Forum in Kyoto, Japan.
- Richardson SD (2003) Water analysis: Emerging contaminants and current issues. Analytical Chemistry 75: 2831-2857.
- Shannon MA, Bohn PW, Elimelech M, Georgiadis JG, Marinas BJ, et al. (2008) Science and technology for water purification in the coming decades. Nature 452: 301-310.
- Sauvé S, Desrosiers M (2014) A review of what is an emerging contaminant. Chem Cent J 8.
- Houtman CJ (2010) Emerging contaminants in surface waters and their relevance for the production of drinking water in Europe. JIES 7: 271-295.
- Bradley PM, Barber LB, Kolpin DW, McMahon PB, Chapelle FH (2008) Potential for 4-n-nonylphenol biodegradation in stream sediments. Environmental Toxicology and Chemistry 27: 260-265.
- Asano T (2002) Water from (waste)water – the dependable water resource. Water Sci Tech 45: 24.
- Asano T, Cotruvo J (2004) Groundwater recharge with reclaimed municipal wastewater: health and regulatory considerations. Water Research 38: 1941-1951.
- Voudouris K (2011) Artificial recharge via boreholes using treated wastewater: Possibilities and prospects. Water 3: 964-975.
- Dougherty JA, Swarzenski PW, Dinicola RS, Reinhard M (2010) Occurrence of herbicides and pharmaceutical and personal care products in surface water and groundwater around Liberty Bay, Puget Sound, Washington. J Environ Qual 39: 1173-1180.
- USEPA (1997) Endocrine Disruptor Screening and Testing Advisory Committee (EDSTAC) Final Report.
- AWWA Research Foundation (AwwaRF) (2005) Featured topics, EDC, PhACs, PCPs.
- Kresinova Z, Svobodova K, Cajthaml T (2009) Microbial degradation of endocrine disruptors. Chemicke Listy 103: 200-207.
- Zhang ZF, Ren NQ, Kannan K, Nan J, Liu LY, et al. (2014) Occurrence of endocrine-disrupting phenols and estrogens in water and sediment of the Songhua River, North-eastern China. Arch Environ Contam Toxicol 66: 361-369.
- Petrovic M, Eljarrat E, Lopez de Alda MJ, Barceló D (2004) Endocrine disrupting compounds and other emerging contaminants in the environment: A survey on new monitoring strategies and occurrence data. Anal Bioanal Chem 378: 549–562.
- Claver A, Ormad P, Rodriguez L, Ovelleiro JL (2006) Study of the presence of pesticides in surface waters in the Ebro river basin (Spain). Chemosphere 64: 1437-1443.
- Pedersen JA, Yeager MA, Suffet IH (2006) Organophosphorus insecticides in agricultural and residential runoff: Field observations and implications for total maximum daily load development. Environ Sci Technol 40: 2120-2127.
- Caupos E, Mazellier P, CroueJP (2011) Photodegradation of estrone enhanced by dissolved organic matter under simulated sunlight. Water Res 45: 3341-3350.
- Zhang C, Li Y, Wang C, Niu L, Cai W (2016) Occurrence of endocrine disrupting compounds in aqueous environment and their bacterial degradation: A review. Critical Reviews in Environ Sci Technol 46: 1-59.
- Khan U, Nicell JA (2014) Contraceptive options and their associated estrogenic environmental loads: Relationships and trade-offs. Plos ONE 9.
- Luo YL, Guo WS, Ngo HH, Nghiem LD, Hai FI, et al. (2014) A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci Total Environ 473: 619-641.
- Kortenkamp A, Martin O, Faust M, Evans R, McKinlay R, et al. (2011) State of the art assessment of endocrine disruptors. Final Report.
- Crain DA, Eriksen M, Iguchi T, Jobling S, Laufer H, et al. (2007) An ecological assessment of bisphenol-A: Evidence from comparative biology. Reprod Toxicol 24: 225-239.
- Scott AP (2013) Do mollusks use vertebrate sex steroids as reproductive hormones? II. Critical review of the evidence that steroids have biological effects. Steroids 78: 268-281.
- Canesi L, Fabbri E (2015) Environmental effects of BPA: Focus on aquatic species. Dose-Response 13.
- Anderson RL (1980) Chironomidae toxicity tests — biological background and procedures. In: Buikema AL, Cairns J (Eds.), Aquatic Invertebrate Bioassays. American Society for Testing and Materials (ASTM), p: 209.
- Ibrahim H, Kheir R, Helmi S, Lewis J, Crane M (1998) Effects of organophosphorus, carbamate, pyrethroid and organochlorine pesticides and a heavy metal on survival and cholinesterase activity of Chironomus riparius meigen. Bull Environ Contam Toxicol 60: 448-455.
- OECD (2006) Series on testing and assessment. Detailed review paper on aquatic arthropods in life cycle toxicity test with an emphasis on developmental, reproductive and endocrine disruptive effects, Number 55.
- Taenzler V, Bruns E, Dorgerloh M, Pfeifle V, Weltje L (2007) Chironomids: Suitable test organisms for risk assessment investigations on the potential endocrine disrupting properties of pesticides. Ecotoxicology 16: 221-230.
- Staples CA, Dorn PB, Klecka GM, O'Block ST, Harris LR (1998) A review of the environmental fate, effects, and exposures of bisphenol A. Chemosphere 36: 2149-2173
- Welshons WV, Nagel SC, Saal FSv (2006) Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 147: S56-S69.
- Sohoni P, Tyler CR, Hurd K, Caunter J, Hetheridge M, et al. (2001) Reproductive effects of long-term exposure to bisphenol a in the fathead minnow (Pimephales promelas). Environ Sci Technol 35: 2917-2925.
- Hahn T, Schenk K, Schulz R (2002) Environmental chemicals with known endocrine potential affect yolk protein content in the aquatic insect Chironomus riparius. Environ Pollut 120: 525-528.
- Watts MM, Pascoe D, Carroll K (2001) Chronic exposure to 17 alpha-ethinylestradiol and bisphenol A-effects on development and reproduction in the freshwater invertebrate Chironomus riparius (Diptera: Chironomidae). Aquat Toxicol 55: 113-124.
- Watts MM, Pascoe D, Carroll K (2003) Exposure to 17 alpha-ethinylestradiol and bisphenol A-effects on larval moulting and mouthpart structure of Chironomus riparius. Ecotoxicol Environ Saf 54: 207-215.
- Planello R, Martinez-Guitarte JL, Morcillo G (2008) The endocrine disruptor bisphenol A increases the expression of HSP70 and ecdysone receptor genes in the aquatic larvae of Chironomus riparius. Chemosphere 71: 1870-1876.
- Morales M, Planello R, Martinez-Paz P, Herrero O, Cortes E, et al. (2011) Characterization of Hsp70 gene in Chironomus riparius: Expression in response to endocrine disrupting pollutants as a marker of ecotoxicological stress. Comparative Biochemistry and Physiology C-Toxicology and Pharmacology 153: 150-158.
- Park S-Y, Choi J (2009) Genotoxic effects of nonylphenol and bisphenol A exposure in aquatic biomonitoring species: Freshwater crustacean, Daphnia magna and aquatic midge, Chironomus riparius. Bull Environ Contam Toxicol 83: 463-468.
- Martinez-Paz P, Morales M, Martinez-Guitarte JL, Morcillo G (2013) Genotoxic effects of environmental endocrine disruptors on the aquatic insect Chironomus riparius evaluated using the comet assay. Mutat Res-Gen Tox En 758: 41-47.
- CERHR (2003) NTP-CERHR monograph on the potential human reproductive and Developmental Effects of Butyl Benzyl Phthalate (BBP). National Institutes of Health, Bethesda.
- Herrero O, Planello R, Morcillo G (2015) The plasticizer benzyl butyl phthalate (BBP) alters the ecdysone hormone pathway, the cellular response to stress, the energy metabolism and several detoxication mechanisms in Chironomus riparius larvae. Chemosphere 128: 266-277.
- Herrero O, Planello R, Morcillo G (2015) The plasticizer benzyl butyl phthalate (BBP) alters the ecdysone hormone pathway, the cellular response to stress, the energy metabolism and several detoxication mechanisms in Chironomus riparius larvae. Chemosphere 128: 266-277.
- Planello R, Herrero O, Luis Martinez-Guitarte J, Morcillo G (2011) Comparative effects of butyl benzyl phthalate (BBP) and di(2-ethylhexyl) phthalate (DEHP) on the aquatic larvae of Chironomus riparius based on gene expression assays related to the endocrine system, the stress response and ribosomes. Aquat Toxicol 105: 62-70.
- Baldwin WS, Graham SE, Shea D, LeBlanc GA (1998) Altered metabolic elimination of testosterone and associated toxicity following exposure of Daphnia magna to nonylphenol polyethoxylate. Ecotoxicol Environ Saf 39: 104-111.
- Soares A, Guieysse B, Jefferson B, Cartmell E, Lester JN (2008) Nonylphenol in the environment: A critical review on occurrence, fate, toxicity and treatment in wastewaters. Environ Int 34: 1033-1049.
- Meregalli G, Ollevier F (2001) Exposure of Chironomus riparius larvae to 17 alpha-ethynylestradiol: Effects on survival and mouthpart deformities. Sci Total Environ 269: 157-161.
- Gade G, Hoffmann KH, Spring JH (1997) Hormonal regulation in insects: Facts, gaps and future directions. Physiol Rev 77: 963-1032.
- Hagger JA, Depledge MH, Oehlmann J, Jobling S, Galloway TS (2006) Is there a causal association between genotoxicity and the imposex effect? Environ Health Perspect 114: 20-26.
- Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low-levels of DNA damage in individual cells. Exp Cell Res 175: 184-191.
- Martinez-Paz P, Morales M, Martin R, Luis Martinez-Guitarte J, Morcillo G (2014) Characterization of the small heat shock protein Hsp27 gene in Chironomus riparius (Diptera) and its expression profile in response to temperature changes and xenobiotic exposures. Cell Stress Chaperones 19: 529-540.
- Mayer MP, Bukau B (2005) Hsp70 chaperones: Cellular functions and molecular mechanism. Cell Mol Life Sci 62: 670-684.
- Agency for Toxic Substances and Disease Registry (ATSDR) (1999). Toxicological profile for pentachlorophenol (Update) (Draft). Public Health Service, U.S. Department of Health and Human Services, Atlanta, GA.
- Nair PMG, Choi J (2012) Modulation in the mRNA expression of ecdysone receptor gene in aquatic midge, Chironomus riparius upon exposure to nonylphenol and silver nanoparticles. Environ Toxicol Pharmacol 33: 98-106.
- Orton F, Lutz I, Kloas W, Routledge EJ (2009) Endocrine disrupting effects of herbicides and pentachlorophenol: In vitro and in vivo evidence. Environ Sci Technol 43: 2144-2150.
- Wing KD, Slawecki RA, Carlson GR (1988) Rh-5849, A nonsteroidal ecdysone agonist - Effects on larval lepidoptera. Science 241: 470-472.
- OECD (2004) OECD guideline for the testing of chemicals, sediment water Chironomid toxicity test using spiked water, OECD 219, adopted 13 April 2004.
- Hahn T, Liess M, Schulz R (2001) Effects of the hormone mimetic insecticide tebufenozide on Chironomus riparius larvae in two different exposure setups. Ecotoxicol Environ Saf 49: 171-178.
- Tassou KT, Schulz R (2013) Low field-relevant tebufenozide concentrations affect reproduction in Chironomus riparius (Diptera: Chironomidae) in a long-term toxicity test. Environ Sci Pollut Res 20: 3735-3742.
- Antizar-Ladislao B (2008) Environmental levels, toxicity and human exposure to tributyltin (TBT)-contaminated marine environment. A review. Environ Int 34: 292-308.
- Hahn T, Schulz R (2002) Ecdysteroid synthesis and imaginal disc development in the midge Chironomus riparius as biomarkers for endocrine effects of tributyltin. Environ Toxicol Chem 21: 1052-1057.
- Morales M, Martinez-Paz P, Ozaez I, Luis Martinez-Guitarte J, Morcillo G (2013) DNA damage and transcriptional changes induced by tributyltin (TBT) after short in vivo exposures of Chironomus riparius (Diptera) larvae. Comp Biochem Physiol C Toxicol Pharmacol 158: 57-63.
- Veldhoen N, Skirrow RC, Osachoff H, Wigmore H, Clapson DJ, et al. (2006) The bactericidal agent triclosan modulates thyroid hormone-associated gene expression and disrupts post-embryonic anuran development. Aquat Toxicol 80: 217-227
- Arcand-Hoy LD, Nimrod AC, Benson WH (1998) Endocrine-modulating substances in the environment estrogenic effects of pharmaceutical products. Int J Toxicol 17: 139-158.
- Tokeshi M (1995) Production ecology. In: P.D. Armitage, P.S. Cranston and L.C. V. Pinder (eds.), The Chironomidae: biology and ecology of non-biting midges. Chapman & Hall, London, p: 584.
- Meregalli G, Pluymers L, Ollevier F (2001) Induction of mouthpart deformities in Chironomus riparius larvae exposed to 4-n-nonylphenol. Environ Pollut 111: 241-246
- Crews D, Willingham E, Skipper JK (2000) Endocrine disruptors: Present issues, future directions. Q Rev Biol 75: 243-260.
- Aydin E, Talinli I (2013) Analysis, occurrence and fate of commonly used pharmaceuticals and hormones in the Buyukcekmece Watershed, Turkey. Chemosphere 90: 2004-2012.
- Campanha MB, Awan AT, de Sousa DNR, Grosseli GM, Mozeto AA, et al. (2015) A 3-year study on occurrence of emerging contaminants in an urban stream of So Paulo State of Southeast Brazil. Environ Sci Pollut Res 22: 7936-7947.
- Praveena SM, Lui TS, Hamin N, Raza S, Aris AZ (2016) Occurrence of selected estrogenic compounds and estrogenic activity in surface water and sediment of Langat River (Malaysia). Environ Monit Assess188: 142.
- Rocha S, Domingues VF, Pinho C, Fernandes VC, Delerue-Matos C, et al. (2013) Occurrence of Bisphenol A, Estrone, 17 beta-Estradiol and 17 alpha-Ethinylestradiol in Portuguese rivers. Bull Environ Contam Toxicol 90: 73-78.
- Wang DP, Luo ZX, Zhang X, Lin LF, Du MM, et al. (2015) Occurrence, distribution and risk assessment of estrogenic compounds for three source water types in Ningbo City, China. Environ Earth Sci 74: 5961-5969.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 3517
- [From(publication date):
April-2017 - Aug 25, 2025] - Breakdown by view type
- HTML page views : 2555
- PDF downloads : 962