Research Article Open Access
GC-MS Fingerprinting of Fatty Acids of Freshwater Mollusc Lamellidens Marginalis using Different Columns, TR-Waxms and TR-FAME
Arabinda Mahanty1, Smriti Ranjan Maji2, Satabdi Ganguly1 and Bimal Prasanna Mohanty1*
1ICAR - Central Inland Fisheries Research Institute, Fishery Resource and Environmental Management Division, Barrackpore, Kolkata, India
2Bose Institute (Centenary Building), Central Instrument Facility (P. D. Lab), P 1/12 C.I.T, Scheme VIIM, Kolkata, India
- *Corresponding Author:
- Bimal Prasanna Mohanty
ICAR - Central Inland Fisheries Research Institute
Fishery Resource and Environmental Management Division
Barrackpore, Kolkata, India
Tel: +919230618153
E-mail: bimalmohanty12@rediffmail.com
Received Date: February 10, 2015; Accepted Date: April 06, 2015; Published Date: April 09, 2015
Citation: Mahanty A, Ranjan Maji S, Ganguly S, Mohanty BP (2015) GC-MS Fingerprinting of Fatty Acids of Freshwater Mollusc Lamellidens Marginalis using Different Columns, TR-Waxms and TR-FAME. J Anal Bioanal Tech 6:238. doi: 10.4172/2155-9872.1000238
Copyright: ©2015 Mahanty A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
GC-MS is an important instrument in lipid profiling and lipid biomarker identification. The choice of columns and detectors is important in analysis and identification of the biomolecules. In the present study, GC-MS fingerprinting of fatty acids (FAs) in different tissues of freshwater mollusc Lamellidens marginalis was carried out in GC-MS with two different columns, TR-FAME and TR-WaxMS, to examine if choice of columns and detectors influence the identifications of the lipids. These sentinel organisms are used in aquatic pollution monitoring studies as they accumulate toxic environmental contaminants to levels well above those present in the surrounding environment thus providing information on the spatio-temporal pollution trends. FAs identified in different tissues include the saturated FAs palmitic acid (C16:0), margaric acid (C17:0), stearic acid (C18:0), monounsaturated FAs (MUFAs) oleic acid (C18:1), eicosenoic acid (C20:1) and polyunsaturated fatty acid (PUFAs) linoleic acid (C18:2). More numbers of FAs were detected in TR-FAME column than TR-WaxMS column. Several non-fatty acid compounds like butylated hydroxyl toluene (BHT) and dibutyl phthalate were also identified in the FAME (fatty acid methyl ester) preparation using NIST (MS) library. It was interesting to note that the FA peaks (6/14) were outnumbered by the non-fatty acid peaks (8/14). Such compounds could be taken as unidentified FAs unless powerful detectors like MS are available and can lead to erroneous inferences. The study showed that the choice of columns and detectors influenced the identification of the compounds. The information generated on fatty acid profiles of this bivalve could serve as baseline data for this species in searching for lipid biomarkers for aquatic pollution monitoring studies.
Keywords
Lipid biomarkers; Fatty acid; GC-MS fingerprinting; Lamellidens marginalis ; Pollution monitoring
Introduction
The freshwater molluscs form an important part of the aquatic ecosystems. These sentinel organisms are used in aquatic pollution monitoring programs as they accumulate toxic environmental contaminants to levels well above those present in the surrounding environment thus providing information on the spatio-temporal pollution trends [1,2]. Lamellidens marginalis is a bivalve mollusc predominantly distributed in the freshwater riverine environment. This sedentary and benthic organism is used for monitoring pesticides, artificial color pigments and environmental contaminants in aquatic ecosystems in the Indian subcontinent [2-4].
Lipids are a diverse group of molecules which are hydrophobic or amphiphilic in nature, the lipid structure and their properties enable these compounds to fulfill many biological functions from energy storage through membrane structure to signaling intermediates [5]. Among the different groups of lipid molecules, Fatty acids (FAs) are important biomolecules that regulate key metabolic pathways and play major role in health and diseases. These molecules play leading role in the tissue construction and maintenance of all healthy cells. They serve as the cell’s guard, operating the sodium-potassium pump that regulates the opening and the closing of the metabolic pathways. As the FA composition of an organism changes with its age, size, environmental conditions etc. these could be used as potential biomarkers for a vast array of clinical conditions [6-10].
Gas Chromatography-Mass Spectrometry (GC-MS) is a sensitive analytical technique that is used in a wide range of applications such as environment monitoring, flavor and fragrance analysis [11], pesticide analysis, metabolite analysis [12], forensic and criminal cases etc [13]. It is considered as the method of choice for detection of volatile compounds due to its high sensitivity over other analytical techniques like Liquid Chromatography-Mass Spectrometry (LC-MS) [14]. With the selection of suitable column, a wide range of compounds such as eicoscanoids, essential oils, FAs, wax, esters, perfumes, terpenes can be analyzed in GC-MS [14]. GC-MS combines the separation capability of GC with fragments identification capability of MS. Thus the results obtained in GC-MS are much more confirmatory compared to GC.
The present study was undertaken with the objective of generating information on the lipids especially FAs composition of the foot, gill and mantle tissues of Lamellidens marginalis which could be used as the baseline data in lipid biomarker discovery for aquatic ecosystem health monitoring.
Materials and Methods
Collection of bivalve molluscs
Live bivalve molluscs were collected from river Ganga at Ballykhal, Kolkata (22°39'17"N 88°20'51"E) and foot, gill and mantle tissues were collected and stored at -40°C for further experiments.
Lipid Extraction and preparation of Fatty Acid Methyl Esters
Lipid extraction was carried out as per Folch et al. [15]. In brief, frozen tissues were homogenized (using a motor pestle) in the organic solvent mixture (chloroform–methanol, 2:1), keeping the solvent/ tissue ratio 20:1, and washed by centrifugation. Washing was repeated five times with fresh solvent mixture. The chloroform fractions, enriched with lipids, were collected, pooled, and dried in a rotary evaporator. The dried lipids were weighed, dissolved in chloroform, and stored in small amber glass laboratory bottles at −20°C. Fatty acid methyl esters (FAME) were prepared from the extracted fat as per Matecalfe, Schmitz, and Petha [16]. The FAMEs were dissolved in hexane for further analysis in GC-MS.
Fatty Acid Analysis by GC-MS using TR-WaxMS Column
The Gas Chromatography- Mass Spectrometry (GC-MS) analyses of the FAs were carried out using a GC (Trace GC Ultra, Thermo Fisher Scientific India Pvt. Ltd.) equipped with a capillary column (TR-WaxMS, 30 m × 0.25 mm [ID] × 0.25 μm film thickness) and a MS (POLARISQ, Thermo Fisher Scientific India Pvt. Ltd.) attached to it. For separation of FAs, the oven temperature programme was set as stated; 1 min initial hold at 50°C, temperature raised from 50-150°C at the rate of 20°C per min followed by a hold of 15 min at 150°C, temperature raised from 150-240°C at the rate of 20°C per min and a final hold of 2 min at 240°C. Helium was used as a carrier gas with column flow rate of 1.0 ml per min. The MS conditions were as follows; ionization voltage 70 eV, Mass range of 40-500 and the scan time equal to the GC run time. The individual constituents showed by GC were identified and quantified by comparing the retention times and peak areas to those of standards (ME-14-KT and ME-19-KT, SUPELCO Analytical) and by using the NIST Library (version 2.0, 2008).
Fatty Acid Analysis by GC-MS using TR-FAME Column
The FAs were also analyzed using a GC-MS (Trace GC Ultra-ITQ 900, Thermo Scientific) equipped with a capillary column (TR-FAME, 30m × 0.25 mm [ID] × 0.25 μm film thickness). The oven programming and all other MS parameters were kept same as described under FA analysis by GC-MS using TR-WaxMS column.
Results and Discussion
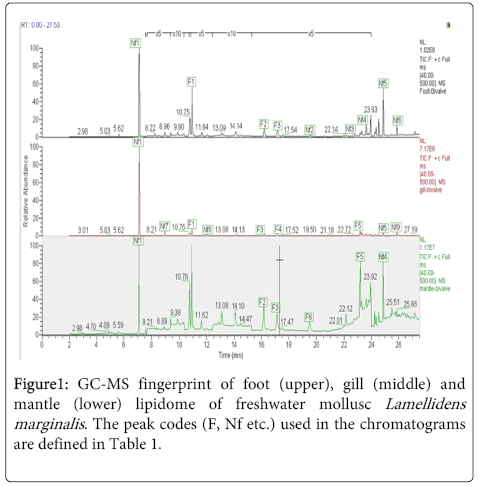
GC-MS is a powerful tool that is being increasingly used in biomarker discovery. It can be used for analysis of wide range of biological compounds including FAs, essential oils, eicoscanoids, wax, esters by the selection of suitable columns. As many of the FAs are non-volatile compounds, these needs to be derivatized into their methyl ester forms for analysis in GC-MS. A number of columns like TR-FAME, TR-WaxMS, Trace TR-1 are recommended for analysis of esters [17]. In the present study, FA composition of three different tissues of L. marginalis has been analyzed using two different columns; TR-FAME and TR-WaxMS. The compounds detected in different tissues are summarized in Table 1 and Figure 1.
| Sl.No. | Compounds identified : Fatty acid /fatty acid derivative (F)/ Non- fatty acid peaks (Nf) | Foot | Gill | Mantle | M/Z Value | GC-MS fingerprint Peak code No. ** |
|---|---|---|---|---|---|---|
| Fatty acid / fatty acid derivative (F) | ||||||
| Saturated fatty acids (in mg/g tissue) | ||||||
| 1. | C 16 : 0 ( Palmitic acid ) | 3.9 | 1.2 | 3.0* | 143,74 | F1 |
| 2. | C 17 : 0 (Margaric acid ) | 0.78 | - | 1.06* | 199, 255 | F2 |
| 3. | C 18 : 0 (Stearic acid) | 0.65 | 0.2* | 0.8* | 87, 298 | F3 |
| Unsaturated fatty acids (in mg/g tissue) | ||||||
| 4. | C 18 : 1 ( Oleic acid ) | - | 3.34 | - | 55, 222 | F4 |
| 5. | C 20 : 1 (Eicosenoic acid) | - | 1.68 | 3.6* | 292, 324 | F5 |
| 6. | C 18 : 2 (Linoleic acid) | - | - | 2.7 | 67, 262 | F6 |
| B. Non-fatty acid (Nf) peaks (in % total area) (Compounds identified through NIST library search ) | ||||||
| 7. | Butylated hydroxyl toluene ( BHT ) | 39.7* | 73.5* | 11.4* | 205, 220 | Nf1 |
| 8. | Methyl-9-cis-11-trans-octadecadienoate | 0.38 | - | - | 81, 95 | Nf2 |
| 9. | 3,5-di-tert Butyl-4-hydroxybenzaldehyde | 0.3 | - | - | 191, 219 | Nf3 |
| 10. | Dibutyl phthalate | 3.25* | - | 16.52* | 149, 150 | Nf4 |
| Dibutyl phthalate | 28.5* | - | 4.73* | 149, 223 | Nf5 | |
| 11. | Phenol 4,4’-thiobis(methylene)bis(2,6-bis)(1,1-dimethylethyl) | 6.25* | - | - | 203, 219 | Nf6 |
| 12. | Furan 2,5 dihydro 2,2 dimethyl 5-(1-methyl ethyl) -3-(1-methy ethyl) | - | 1.09* | - | 137, 165 | Nf7 |
| 13. | Phlorobutyrophenone | - | 0.46 | - | 153, 196 | Nf8 |
| 14. | Phenol-2,6-bis(1,1-dimethyl propyl)-4-methyl | - | 7.74* | - | 219, 258 | Nf9 |
* Detected both in TR-FAME and TR-WaxMS column; other compounds were detected only with TR-FAME column. ** For identifying the peak in the GC-MS fingerprint refer codes in Figure 1.
Table 1: peak codes of chromatograms.
Fatty acids
Several FAs as well as non fatty acid components in different concentrations were detected in the foot, gill and mantle tissues of the bivalve analyzed by GC-MS using both TR-WaxMS and TR-FAME column.
In foot, steric acid (C18) was the only FA detected when the FAMEs were analyzed using the TR-WaxMS column while palmitic acid (C16:0), margaric acid (C17:0) and stearic acid (C18:0) were detected when the TR-FAME column was used.
FA profile of gill showed presence of palmitic acid (C16:0), margaric acid (C17:0) and stearic acid (C18:0) both in TR-FAME and TR-WaxMS column and additionally oleic acid (C18:1) and eicosenoic acid (C20:1) were also detected when the FAMEs were analyzed using the TR-FAME column.
Mantle lipidome showed the presence of palmitic acid, margaric acid, stearic acid in both TR-WaxMS and TR-FAME column whereas additionally eicosenoic acid and linoleic acid (C18:2) were detected only in TR-FAME.
Among the saturated FAs palmitic acid (C16:0) and stearic acid (C18:0) were detected in all the three tissues analyzed while margaric acid (C17:0) was detected only in foot and mantle. Unsaturated FA oleic acid (C18:1) was only found in the gill and linoleic acid (C18:2) was present only in mantle. Eicosenoic acid (C20:1) was detected in both gill and mantle.
It was observed that more number of FAs were detected using TRFAME column than TR-WaxMS column. TR-WaxMS column is recommended for analysis of esters, alcohols, essential oils etc. whereas TR-FAME column provides excellent separation of cis/trans isomers and FAMEs with various degrees of unsaturation. The detection of higher number of FA by the application of TR-FAME can thus be attributed to its unique design for providing greater separation of FAMEs [17].
Palmitic acid is the most common FA found in animals, plants and microbes. Palmitic acid is the first FA produced during FA synthesis and the precursor to longer FAs [18]. It is amongst the most abundant saturated FA present in many animal tissues [10] and in the present study, palmitic acid was found to be the most abundant FA in all the three tissues of the bivalve L. marginalis .
Margaric acid is a saturated FA which occurs in trace amount in milk fat of ruminants and in very minute quantities in animal and plant fats [19]. However, it was found to be a major constituent of the foot and mantle lipidome of the bivalve.
Stearic acid is among the long chain FA which plays crucial role in lipogenesis through a desaturation process [20,21]. Stearic acid is one of the most common saturated FAs found in nature following palmitic acid and it was found be one of the FA which was found in all the three tissues studied.
Non-fatty acids
Butylated hydroxyl toluene (BHT) and dibutyl phthalate (DBP) were the primary non-fatty acid components. BHT was detected in all the three tissues analyzed while dibutyl phthalate was detected only in foot and mantle. Other non-fatty acid components like.methyl-9- cis-11-trans-octadecadienoate, 3,5-di-tert-butyl-4- hydroxybenzaldehyde,furan 2,5-dihydro 2,2-dimethyl 5-(1-methyl ethyl)-3-(1-methylethyl) were also found in different tissues.
BHT is widely used as a food preservative (E321) for fats. It preserves fats (stops them going rancid) because it is an antioxidant. BHT acts as viral membrane disrupting agent can also be used as a preventative, when there is a risk of viral infection. BHT is used as an antioxidant for minimizing oxidation of FA during the process of fat extraction. Therefore, presence of this compound in all the three samples is quite obvious.
DBP was found to be the most abundant non-fatty acid component next to BHT. It is used mainly in nail products as a solvent for dyes and as a plasticizer that prevents nail polishes from becoming brittle. Phthalates are also used as fragrance ingredients in many other cosmetics, but consumers won't find these listed on the label [22]. The sampling site Kolkata is a major city in the country and is surrounded by large number of industries which uses pthalate like substances. Therefore, presence of these substances in the tissue lipid extract could be attributed to leaching of phthalate like substances from industries and their subsequent bioaccumulation in bivalve tissues.
The FA profile of freshwater mollusc Lamellidens marginalis was generated in the present study which would serve as baseline information for studies on the pollution induced lipid biomarkers. This study also showed that a number of compounds which are soluble in organic solvents also get extracted during the lipid extraction process and remain as unidentified FAs (lipids) unless a powerful detector like MS is used to detect them, which is important from quality control (QC) point of view. The present study clearly showed that the results of lipid analysis would vary with the column used. Although the TR-WaxMS column is recommended for analysis of esters, lower numbers of FAMEs were detected in comparison to the TR-FAME which emphasizes the selection of appropriate column for sensitive experiments especially biomarker discovery. Besides, the bivalve and gastropods are used as food components in different parts of the world. The FA composition data generated for L. marginalis could serve for comparing its food value with other bivalves and other food organisms also.
Acknowledgements
This work was supported by ICAR under CIFRI core Project No. FREM/ER/12/03/05. The authors are thankful to Director, CIFRI for the facilities and encouragement. SRM is thankful to Director, Bose Institute; Prof. P. C. Sen and Prof. S. K. Dasgupta, Bose Institute, Kolkata for the facilities and encouragement. The authors are thankful to Shri Sk Rabiul, Senior Lab Technician for the technical assistance.
References
- Porte C, Sole M, Borghi V, Martinez M, Chamorro J, et al. (2001) Chemical, biochemical and cellular responses in the digestive gland of the mussel Mytilusgalloprovincialis from the Spanish Mediterranean coast. Biomarkers 6: 335-350.
- Mohanty BP, Gupta SK, Yadav L, Panwar RS (2004) Tissue specific protein profiles of two closely related freshwater molluscs. NatlAcadSciLett 28: 205-208.
- Phadnis SD, Chandagade CA, Jadhav VV, Raut PD (2013) Impact of colour pigments on biochemical parameters of bivalve, Lamellidensmarginalis. J Environ Biol 34: 267-271.
- Kumar S, Pandey RK, Das S, Das VK (2013) Temperature dependent mortality and behavioral changes in a freshwater mussel Lamellidensmarginalis to dimethoate exposure. J Environ Biol 34: 165-170.
- Meikle P, Barlow C, Weir J (2009) Lipidomics and lipid biomarker discovery. AustBiochem 40: 12-16.
- Frank R, Hargreaves R (2003) Clinical biomarkers in drug discovery and development. Nat Rev Drug Discov 2: 566-580.
- Yu LR (2011) Pharmacoproteomics and toxicoproteomics: the field of dreams. J Proteomics 74: 2549-2553.
- Piotrowska-Seget Z, Mrozik A (2003) Signature lipid biomarker (SLB) analysis in determining changes in community structure of soil microorganisms. Polish J Environment Study 12: 669-675.
- Hu C, van der Heijden R, Wang M, van der Greef J, Hankemeier T, et al. (2009) Analytical strategies in lipidomics and applications in disease biomarker discovery. J Chromatogr B AnalytTechnol Biomed Life Sci 877: 2836-2846.
- Mohanty BP, Bhattacharjee S, Paria P, Mahanty A, Sharma AP (2013) Lipid biomarkers of lens aging. ApplBiochemBiotechnol 169: 192-200.
- Paranthaman R, Praveenkumar P, Kumaravel S (2012) GC-MS analysis of phytochemicals and simultaneous determination of flavonoids in Amaranthuscaudatus (Sirukeerai) by RP-HPLC. J Anal Bioanal Tech 3: 147.
- Reade S, Mayor A, Aggio R, Khalid T, Pritchard DM, et al. (2014) Optimisation of sample preparation for direct SPME-GC-MS analysis of murine and human faecal volatile organic compounds for metabolomic Studies. J Anal Bioanal Tech 5: 2.
- Chauhan A, Goyal MK, Chauhan P (2014) GC-MS technique and its applications in science and technology. J Anal Bioanal Tech 5: 222.
- Agilent Technologogies (2007) Considerations for selecting GC/MS or LC/MS for metabolomics.
- Folch J, Lees M, Sloane-Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biological Chemistry 226: 497-509.
- Metcalfe LD, Schmitz AA, Petha JR (1966) Rapid preparation of fatty acid esters from lipids for gas chromatography analysis. Analytical Chemistry 38: 514-517.
- Thermo Electron Corporation (2005) Columns and consumables for GC/MS.
- Nelson D, Cox MM (2013) Lehningers Principles of Biochemistry. WH Freeman, USA.
- COOKE NJ, HANSEN RP, SHORLAND FB (1957) Occurrence in butterfat of n-heptadecanoic acid (margaric acid). Nature 179: 98.
- Sampath H, Miyazaki M, Dobrzyn A, Ntambi JM (2007) Stearoyl-CoA desaturase-1 mediates the pro-lipogenic effects of dietary saturated fat. J BiolChem 282: 2483-2493.
- Legrand P, Rioux V (2010) The complex and important cellular and metabolic functions of saturated fatty acids. Lipids 45: 941-946.
- USEPA Dibutylpthalate (2013) United States Environment Protection Agency.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 16439
- [From(publication date):
April-2015 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 11706
- PDF downloads : 4733