Research Article Open Access
Impact of Diesel-butanol Blends on Performance and Emission of Diesel Engine
Swamy RL1, Chandrashekar TK2, Banapurmath NR3* and Khandal SV4
1Department of Mechanical Engineering, B.G.S Institute of Technology, BG Nagar, India
2Department of Mechanical Engineering, Mangalore Institute of Technology and Engineering, Moodbidri, India
3Department of Mechanical Engineering, B.V.B. College of Engineering and Technology, Hubli, India
4Department of Mechanical Engineering, B.V.B. College of Engineering and Technology, Hubli, India
- *Corresponding Author:
- Banapurmath NR
Department of Mechanical Engineering
B.V.B. College of Engineering and Technology, Hubli, India
Tel: +91 9880726748
E-mail: nr_banapurmath@rediffmail.com
Received date: September 03, 2015 Accepted date: October 13, 2015 Published date: October 17, 2015
Citation: Swamy RL, Chandrashekar TK, Banapurmath NR, Khandal SV (2015) Impact of Diesel-butanol Blends on Performance and Emission of Diesel Engine. Oil Gas Res 1:101. doi: 10.4172/2472-0518.1000101
Copyright: © 2015 Swamy RL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Oil & Gas Research
Abstract
The aim of lowering the pollutants and to enhance the performance of diesel engines has intensified research in diesel engines. The goal of this study was to assess combustion, performance and emission characteristics of diesel engine using diesel-oxygenate blends. In this direction, experimental investigations were carried out on a single cylinder four stroke direct injection water cooled diesel engine using butanol blended fuels in different volume ratios with diesel fuel. The butanol had no solubility or stability problems when blended with diesel fuel. As there was not phase separation in the blends, no additive was added. The experimental investigation was done with four different blends of butanol on volume basis [B0 (0% Butanol and 100% Diesel), B5 (5% Butanol and 95% Diesel), B10 (10% Butanol and 90% Diesel), B15 (15% Butanol and 85% Diesel) and B20 (20% Butanol and 80% Diesel)] to study the impact of using butanol -diesel blends on diesel engine performance, combustion and emissions. The outcome indicates that the brake thermal efficiency increased with an increase in butanol contents in the blended fuels at overall operating conditions. At higher loads, reduced CO emission levels were observed for blends of butanol. HC emissions increased for all blends of butanol compared with diesel fuel due to high fuel consumption and high latent heat of vaporization which lowers cylinder temperatures and causes the emission of unburned hydrocarbons at lower load. NOx emissions with diesel–butanol blends were found to be comparable with neat diesel at low loads due to lower calorific value and high latent heat of vaporization of butanol results in reduced flame temperature and slightly higher NOx at high loads with increased butanol percentage in the blend compared to neat diesel. Butanol showed lowest smoke opacity at high engine loads compared to diesel fuel operation.
Keywords
Engine power; Oxygenates; Synthetic fuel; Butanol; Optimization; Stability; Blending; Miscibility; Higher alcohol
Introduction
Diesel engines are the most popular well known efficient prime mover among the internal combustion engines because of their simple, robust construction coupled with high thermal efficiency and specific power output with better fuel economy, much longer life span and reliability which results in their wide spread use in transportation, thermal power generation and many more industrial and agricultural applications [1,2]. In spite of many advantages, the diesel engine is inherently dirty and is the most significant contributor of various air polluting exhaust gases such as particulate matter (PM), oxides of nitrogen (NOx), carbon monoxide (CO) and other harmful compounds which contribute to serious public health problems.
In view of increased concerns regarding the effects of diesel engine particulate and NOx emissions on human health and the environment and more stringent government regulation on exhaust emissions, reducing the NOx and particulate emission from diesel engines is one of the most significant challenges. A lot of research work has taken up in this direction to develop after treatment and in-cylinder control techniques by modifying fuels to mitigate the tailpipe NOx emission and formation in the cylinder respectively. It is difficult by using the traditional way to simultaneously reduce NOx and PM in diesel engine owing to the existing of trade-off curve between NOx and PM [3-5]. The rapid depletion, uneven distribution of petroleum fuels, their ever increasing costs and great concern over pollution led to search for an alternative fuel to replace conventional fuels. The most promising alternative possibility to clear this critical issue is to use the oxygenated fuels either in pure form or blended with diesel to provide sufficient oxygen and promote combustion and reducing PM emission and possibly decreasing NOx emission.
Oxygenated fuels are the attractive class of synthetic fuels in which Oxygen atoms are chemically bound within the fuel structure. This Oxygen bond in the oxygenated fuel is energetic and provides a chemical energy that result in no loss of efficiency during combustion. The optimization of oxygenated fuels, to be used either as, neat fuel or as an additive, offers significant potential for reduction in particulate emission. Oxygenated fuels used either as alcohols or ethers. Lower alcohols viz. Ethanol and Methanol have been widely used as alternate fuels, but they show solubility problems in diesel fuel. Moreover, percentages of ethanol above 10% blended with diesel fuel usually reduce engine power and increase brake specific fuel consumption (BSFC). Butanol has higher heating value and cetane number, less volatility and it is less hydrophilic than ethanol. Also, butanol is less polar than shorter alcohols which favor miscibility with diesel fuels. All these properties are closer to diesel fuel which makes butanol an alternative to ethanol as fuel (in blend with diesel) in compression ignition engines. During this experimentation, butanol tested with diesel in blended form to investigate the performance, combustion and emissions of a diesel engine to provide additional data in this field of research. Butanol, a 4-carbon alcohol (butyl alcohol), is generally used as a transportation fuel. Butanol is commonly produced using fossil fuels, but it can also be produced from biomass through the fermentation of the feed stocks such as corn, sugar beets, and other types of biomass generally called as biobutanol. Biobutanol is considered a renewable fuel, in addition, butanol has higher heat of vaporization, oxygen content and flammability temperature and therefore has a positive influence on engine performance and emission characteristics of CI engine.
Literature Review
Several researchers have conducted experimental investigation on the diesel engine fueled with diesel blended fuels. Some of them are briefly highlighted in the following section [6] compared the shortterm performance of a direct injection diesel engine fuelled with different 1-butanol/diesel blends (from 10% to 30% of 1-butanol by volume) and 1-pentanol/diesel fuel blends (from 10% to 25% of 1-pentanol by volume), without any modifications of the engine. Experimental results showed a slight engine power loss and an increase in brake thermal efficiency when the engine was fuelled with higher alcohols blends instead of straight diesel fuel and a diesel engine, without any modifications, can run successfully on a blend up to 30% 1-butanol/70% diesel fuel or 25% 1-pentanol/75% diesel fuel. Butanol blends exhibit slightly better BSFC behavior than pentanol blends and neat diesel fuel. Octavio [7] showed that slight increase in NOx and THC emissions, while reduced CO emissions were found with these alcohol blended fuels. The lower values of smoke opacity and particle concentration with alcohol blends indicate that the presence of oxygen in the fuel is the main factor that led to the lower soot generation. Octavio [8] identified that alcohol blends tested at ‘cold’ start produced combustion instabilities with higher concentrations of NOx, THC and CO compared with diesel fuel used as reference. Under warm conditions, the effect of oxygen content of the alcohol blends tested was more important than the effect of the differences in cetane number and enthalpy of vaporization. Gaseous emissions from ethanol–diesel blends were slightly lower than those derived from butanol–diesel blends. Merola [3] carried out experimental investigation with neat diesel fuel and blends of 80% diesel-20% n-butanol by volume in compression ignition engines equipped with a common rail injection system. The test results indicate that BU20 reducing engine out emissions of NOx and smoke without significant penalties on engine performance. The neat diesel (BU00) gave the best smoke-NOx tradeoff result at injection pressures higher than 120 MPa. Rahman [9] studied the influence of injection timing on engine performance and exhaust emissions with diesel, biodiesel, alcohol and other alternative fuels. Results shown that incase of diesel fuel, advancement in injection timing results in lower carbon monoxide (CO) and hydrocarbon (HC) emission along with increased nitrogen oxides (NOx) emission. Advance injection timing increases brake thermal efficiency (BTE) and decreases brake specific fuel consumption (BSFC). Biodiesel–diesel blends produce more HC and CO emission, but reduce NOx emission when injection timing is retarded. Advancement in injection timing results in higher exhaust gas temperature with increase of biodiesel percentage in the blends. Wang [10] and Banapurmath [11] observed lower particulate matter (PM) emission with the increase of oxygenate content in the blends, and ethanol addition into diesel fuel increases HC, CO, NOx and NO emissions and decreases particle number concentration. Increased brake thermal efficiency has been reported with diesel/biodiesel-ethanol blends operation. Gnanamoorthi [12] showed that increased peak in-cylinder pressure and higher heat release occurs closer to TDC for ethanol blend and it increases with increase in ethanol in the blend. There was CO and HC emissions and found that which is closer than those with base fuel up to 30% ethanol, but much higher CO and HC emission with 40% and 50% ethanol and there is a slight decrease in NOx emission. Ahmet [13] reported high engine power for DE10 at an engine speed of 3000 rpm and 45oCA advanced injection angle. Further, the maximum engine torque has been reported for DE10 at 1400 rpm and injection advance of 25oCA. The minimum brake specific fuel consumption (BSFC) was obtained for DE20 blends in 1200 rpm and injection advance of 35oCA. Baskar [4] conducted experiments on diesel engine under different load conditions using various fuel combinations. They showed both Diphenyl ether and Diethylene glycol dimethyl ether blends substantially lowers the exhaust gas opacity. The maximum reduction 60% has been reported with Diphenyl ether 15 and Diethylene glycol dimethyl ether15 blends as compared to base reference diesel fuel. In addition, oxygenated diesel blends have shown significant reduction in CO and HC emissions with only a slight penalty of NOx emissions. Oxygen enrichment of the conventional fuel is not accompanied by a sharp increase of the in-cylinder NO concentration due to decrease of the local temperature as a result of the lower fuel heating value. Oxygenated diesel fuels produce a favorable shift in PM/NOx trade-off. Nurun [14] have reported that the combustion with oxygenated fuels were much faster than that of conventional diesel fuel. This was mainly due to the oxygen content in the fuel molecular structure and the low volatility of the oxygenated fuels. The lower volatile oxygenated fuel evaporated earlier and very good air-fuel mixing was achieved during combustion which eventually resulted in lower exhaust emissions. Miyamoto [15] investigated the combustion and emissions of diesel engine operated on different types of oxygenates in a single cylinder, four stroke cycle, DI diesel engine. They reported significant improvements in smoke, particulate matter, NOX, HC, engine noise and thermal efficiency with the oxygenated fuels. Gonzalez [16] focused study on selecting most promising oxygenate compounds as blending components in diesel fuel foe an advanced engine testing with an objective of reducing particulate emissions in the exhaust. The results have shown that PM emission reductions were proportional to the oxygen content of the fuel. Both Tripropylene Glycol Mono-Methyl Ether and Dibutyl Maleate oxygenated test fuels contained 7% wt. oxygen and their total PM emissions were found to be similar. David [17] evaluated the performance of reference base diesel and five blends containing oxygenates. The experimental results have shown that oxygenated fuels reduced PM and NOx under some operating conditions, but produced little effect on either HC or CO emissions. Aliphatic oxygenates at 6% wt. Oxygen in the reference fuel reduced PM emissions by 15-27%. Kannan [5] have shown that the combustion characteristics with ethanol addition improved in-cylinder peak pressure, cumulative heat release (CHR), rate of heat release (ROHR), and in-cylinder peak temperature and combustion duration. Regarding emission characteristics, the experimental results showed significant reduction in smoke, carbon monoxide (CO) and total hydrocarbon (THC) emissions with extended oxygen mass percentage in the fuel at higher engine loads. However, an increased oxide of nitrogen (NOx) emissions has been reported at high loads although the common tradeoff between smoke and NOx was found to be more prominent for the oxygenated fuels. Gvidonas [18] observed lower NOx levels with ethanol blended fuels and HC emissions for richer combustible mixtures, whereas the influence of a higher ethanol mass content on CO emissions and smoke opacity depends on the air–fuel ratio and engine speed. Dulari [19] carried out experimental investigations in a single cylinder, four stroke, air cooled direct injection (DI) diesel engine, fueled with bioethanol, adopting the fumigation technique. The results indicated that, the bioethanol fumigation showed an overall longer ignition delay of 2-3oCA for all the flow rates in comparison with diesel, at full load. The maximum brake specific nitric oxide (BSNO) and smoke emissions were found to be lower, by about 24.2% and 25% in the bioethanol fumigation compared to that of diesel operation at full load. Subramanian [20] carried out the experimental investigations to assess the effect of using diethyl ether to improve performance and reduce emissions of a DI diesel engine running on water- diesel emulsion. It was found that use of neat water diesel emulsion significantly lowered NOx and smoke levels as compared to neat diesel operation. It also increases the brake thermal efficiency at high outputs. However, there is a rise in HC, CO emissions and ignition delay. Nagdeote [21] carried out experimental investigation to evaluate the effects of using diethyl ether and ethanol as additives to biodiesel / diesel blends on the performance and emissions of a direct injection diesel engine. The test fuels were denoted as DI ( 100% diesel), BD (20% biodiesel and 80% diesel in vol.), BDET (15% biodiesel, 80% diesel, and 5% diethyl ether in vol.) and BDE (15% biodiesel, 80% diesel and 5% ethanol, in vol.) respectively. The result shows that, compared with BD, slightly lower brake specific fuel consumption (BSFC) for BDET was observed. Drastic reduction in smoke is observed with BDET and BDE at higher engine loads. BDET reflects better engine performance and combustion characteristics than BDE and BD. Sulakshana [22] carried out the experimentation to study the effects of blends DB10, DBE10, DBE20 i.e. diesel 90% -10 % biodiesel , diesel 80% -10 % biodiesel - 10% ethanol , diesel 70% - 10% biodiesel - 20% ethanol for same diesel engine. The results showed maximum BP for blended fuels of DB10, DBE10, DBE20 dropped by 3.65%, 6.17% and 7.58% respectively compared with neat diesel fuel. Maximum BSFC for blend DB10, DBE10, DBE20 increased by 7.69%, 7.82%, and 33.62% respectively compared with neat diesel fuel. Blend DBE10 has optimum viscosity and calorific value which gives slightly less brake torque and brake power. Diesel-ethanol-biodiesel blends showed reduced NOx, PM, Smoke with slight increment in HC emissions while keeping CO emissions at same level compared with diesel fuel. Finally by considering all parameters DBE10 was best alternative fuel. SayiLikhitha [23] investigated the effect of blending of Diethyl ether (DEE) with diesel at various proportions (5%, 7.5% and 10%) on the performance of diesel engine. The experimental results indicated that with the increase in the concentration of DEE to diesel increases the brake thermal efficiency, mechanical efficiency and decreases the specific fuel consumption.
Experimental Setup
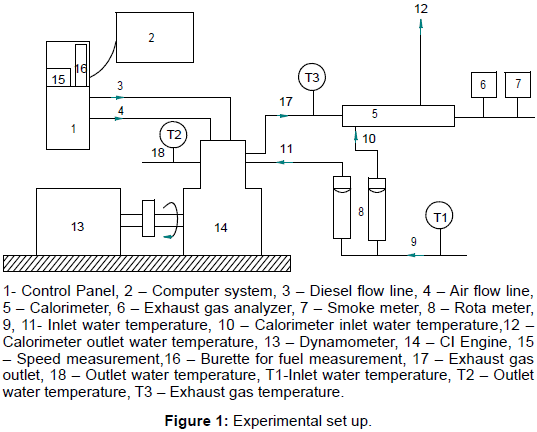
Experiments were conducted on a Kirloskar TV1 type, four stroke, single cylinder, water-cooled diesel engine test rig fueled diesel. Figure 1 shows the line diagram of the test rig used. Eddy current dynamometer was used for loading the engine. The fuel flow rate was measured on the volumetric basis using a burette and stopwatch. The engine was operated at a rated constant speed of 1500 rev/min. The emission characteristics were measured by using HARTRIDGE smoke meter and five gas analyzer during the steady state operation. Experimentation is carried out with the hemispherical shaped combustion chamber provided in the existing engine at an injection pressure of 205 bar with an injector of 3 holes of ɸ 0.3 mm at an injection timing of 23o bTDC. Finally the results obtained were analyzed. Properties of the fuels used viz. diesel and butanol are shown in Table 1. The specification of the compression ignition (CI) engine is given in Table 2.
| Sl No | Property | Diesel | Butanol |
|---|---|---|---|
| 1 | Molecular Weight | 181 g/mol | 74.1216 g/mol |
| 2 | Molecular Formula | C13H24 | C4H9OH |
| 3 | Appearance | Light yellow liquid | Colorless liquid |
| 4 | Density | 850 kg/m3 | 809.8 kg/m3 |
| 5 | Boiling point | 266°C | 117.6°C |
| 6 | Flash point | 85°C | 37°C |
| 7 | Volatility | Volatile | Volatile |
| 8 | Kinematic viscosity at 40°C | 3.05 cSt | 2.22 cSt |
| 9 | Auto-ignition temperature | 316°C | 343°C |
| 10 | Heating value | 43000 kJ/kg | 37000 kJ/kg |
| 11 | Surface tension at 20°C | 0.023 N/m | 0.024 N/m |
| 12 | Latent heat of vaporization | 250 kJ/kg | 550 kJ/kg |
| 13 | Flammability | Flammable | Flammable |
| 14 | Cetane number | 45-55 | 17 |
| 16 | Carbon content (% weight) | 84-87 | 64.82 |
| 17 | Hydrogen content (% weight) | 13.87 | 13.60 |
| 18 | Oxygen content (% weight) | 0 | 21.59 |
| 19 | Stoichiometric air-fuel ratio | 15 | 11.19 |
Table 1: Properties of the fuels used.
| Sl No | Parameters | Specification |
|---|---|---|
| 1 | Type of engine | Kirloskar make Single cylinder four stroke direct injection diesel engine |
| 2 | Nozzle opening pressure | 200 to 205 bar |
| 3 | Rated power | 5.2 KW (7 HP) @1500 RPM |
| 4 | Cylinder diameter (Bore) | 87.5 mm |
| 5 | Stroke length | 110 mm |
| 6 | Compression ratio | 17.5 : 1 |
Table 2: Specifications of the engine.
Results and Discussions
This section explains the performance, emission and combustion characteristics of the diesel and oxygenated blended fuel butanol in different proportions.
Performance characteristics
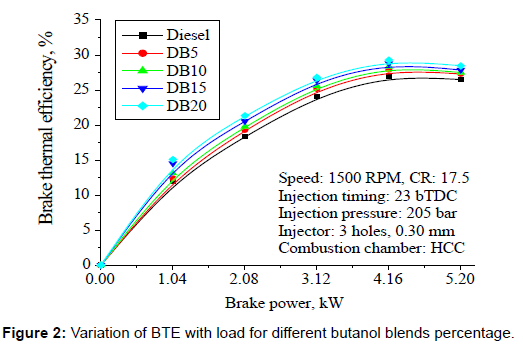
Brake thermal efficiency: Figure 2 shows the variation of BTE at different loads for different blends of butanol. BTE increases with an increase in load for all blends. Higher the percentage of butanol in the mixture, improvement in the brake thermal efficiency can be observed compare to neat diesel fuel. This is due to better combustion because of the presence of oxygen, which involves higher combustion efficiency. Butanol minimizes the interfacial tension between two or more interacting immiscible liquids helped the better atomization of fuel, which improves the combustion of diesel. With butanol- diesel fuel blend operation, the high latent heat of evaporation of butanol which produce more cooling effect that results in low exhaust gas temperature which tends to lesser the heat loss through exhaust and hence higher brake thermal efficiency can be obtained. Furthermore, butanol has a lower flame temperature than neat diesel fuels thereby limiting the heat losses in the cylinder, which further enhance the BTE. In addition, the longer ignition delay due to lower cetane number of butanol involves a rapid rate of released energy which reduces the heat loss from the engine because there is not enough time for this heat to leave the cylinder through heat transfer to the coolant.
Combustion characteristics
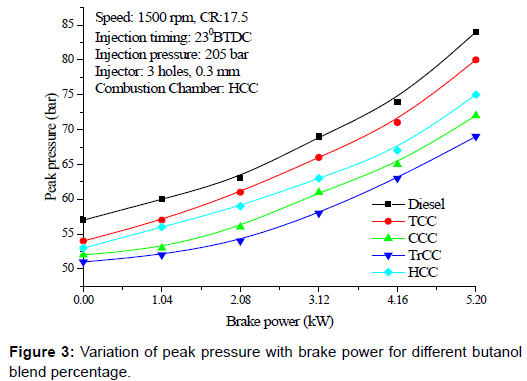
Peak pressure: Figure 3 shows the variation of peak pressure with brake power for diesel butanol blends which is higher than that of the neat diesel. Auto ignition temperature of the fuel is a predominant element which causes variations in the shape of the curve in the pressure angle diagram of engine. Due to combustion of diesel with lower self -ignition temperature, peak pressure is obtained initially, and then a depression is formed due to continuous heat absorption by butanol due to its high latent heat for vaporization. Due to latent heat of evaporation of the butanol higher than that of the diesel and stabilization of the mixture, when auto ignition condition of butanol which is slightly higher than the diesel is reached in the cylinder, combustion takes place and hence sudden rise of temperature and pressure is observed. With the increase of butanol, ignition retards and combustion duration shortens, which contributes to rapid butanol combustion.
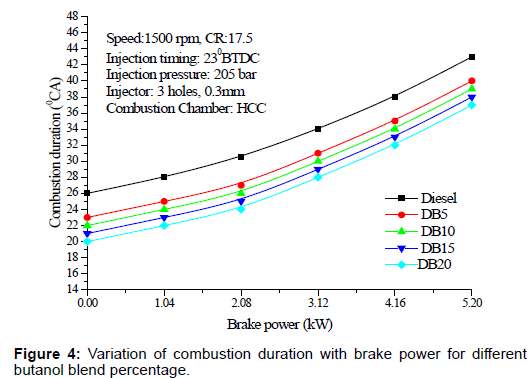
Combustion duration: Figure 4 shows the variation of combustion duration with brake power for different butanol blend percentage with diesel. Combustion duration increased with load and butanol blend percentage. The trend could be due to the higher auto ignition temperature and higher latent heat of evaporation of butanol compared to diesel.
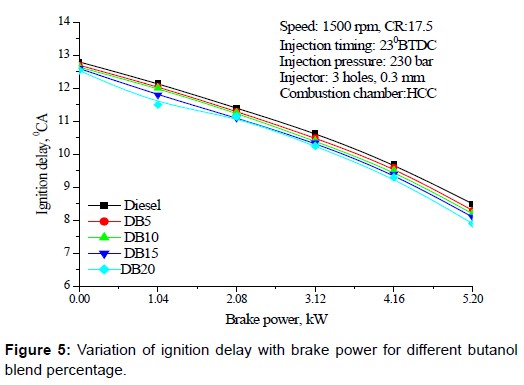
Ignition delay: Figure 5 shows the variation of ignition delay with brake power for different butanol blend percentage with diesel. Ignition delay decreased with load and increased with butanol blend percentage. The trend could be due to the higher auto ignition temperature and higher latent heat of evaporation of butanol compared to diesel.
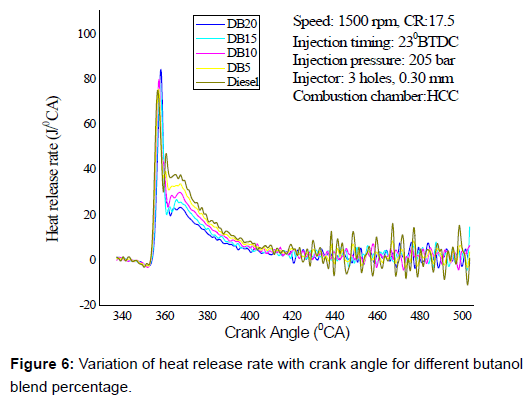
Heat release rate (HRR): Figure 6 shows the variation of heat release rate with crank angle for diesel and different diesel butanol blends at 100% loading conditions which occur closer to TDC. The peak heat release rate increases with an increase in butanol concentration in the diesel. The butanol, as an oxygenator improves combustion for all fuel blends and faster laminar flame speed of butanol that results in sudden heat release rate than that of diesel. The heat release curve has slightly negative depression during the ignition delay period. This is due to loss of heat from the cylinder during fuel vaporization phase. During the premixed combustion, higher heat is released with butanol blend than diesel due to higher ignition delay, which may be due to higher latent heat of evaporation and lower cetane number of butanol in the blend.
Emission characteristics
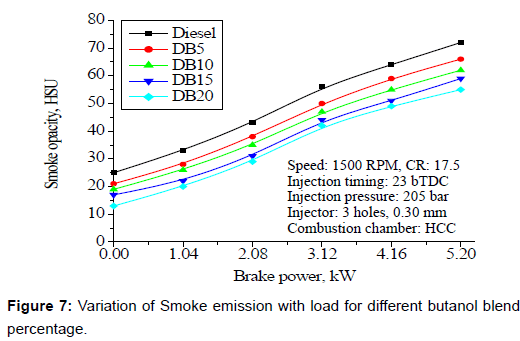
Smoke emissions: Figure 7 illustrates the smoke opacity with engine loads for different butanol blend percentage. Smoke opacity is higher at low loads, which may be due to short combustion cycle at high speed, long delay period and shortage of oxygen which may be due to improper mixing or usage of rich fuel. Smoke opacity is higher with butanol blends at lower loads than diesel. This is because of poor evaporation rate of blended fuel due to high latent heat of evaporation of butanol. At high loads, the flame temperature is high, which results in low smoke opacity with butanol blends than the base fuel. Additionally, high volatility of butanol has a remarkable effect on the reduction of smoke opacity, especially at high engine loads.
HC emissions: HC is partially burned and unburned fuel emission. Figure 8 shows the variation of Total Hydrocarbon with load for different percentage of butanol blends. At lower loads, THC emissions increased for all blends compared with diesel fuel due to fact that low cetane number of blends that promotes quenching effect, to be the main cause for the increase of THC emissions. Higher fuel consumption and high latent heat of vaporization which lowers cylinder temperatures which causes the emission of unburned hydrocarbons. At higher load, no noticeable change in THC emissions for the blends and remains almost same as that of diesel fuel.
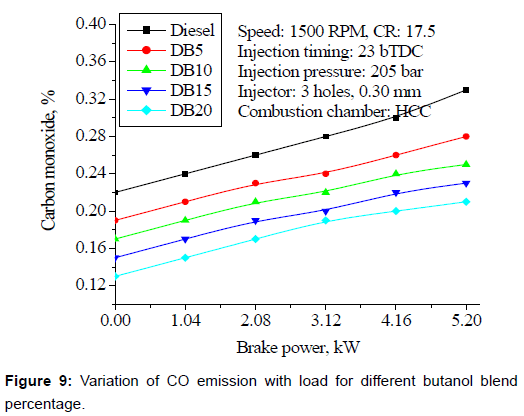
CO emissions: Figure 9 shows the variation of CO with load for different percentage of butanol blends. CO emissions are higher at lower loads. This is due to fact that its latent heat of evaporation is slightly higher than that of diesel; as result there is not enough vaporization and hence very less time to burn fuel completely that results in considerable increase in CO emissions. At higher loads, enough time available for combustion to occur, better mixing and inbuilt fuel oxygen that results in complete combustion and hence slightly reduced the CO emissions, for blends at high load.
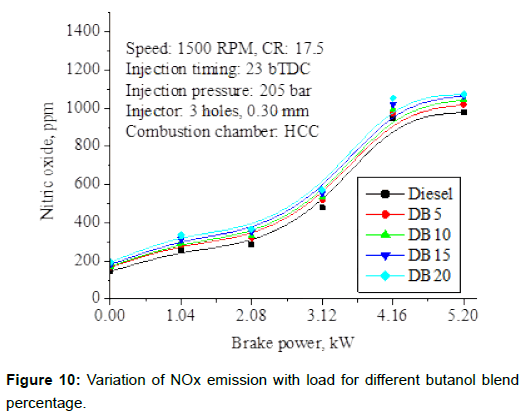
NOx emissions: Figure 10 shows the variations of NOx emissions with engine loads for different butanol blend percentage. The rate of formation of NOx is primarily a function of flame temperature, the residence time of nitrogen at that temperature, and the availability of oxygen in the combustion chamber. It is observed from the figure that NOx emissions with diesel –butanol blends were found to be comparable with neat diesel at low loads due to lower calorific value and high latent heat of vaporization of butanol results in reduced flame temperature. At higher loads, due to increased quantity of fuel injection, the combustion temperature and oxygen availability is more; slightly higher NOx with increased butanol percentage in the blend compared to neat diesel.
Conclusions
The existing single cylinder CI engine was suitably modified to operate on diesel and butyl alcohol blends at near diesel operating conditions. From the experimental study, the effect of blend ratios on the performance of diesel engine operated with diesel and butyl alcohol, the following conclusions were drawn for different loading conditions.
• For diesel and alcohol fuelled blends in diesel engine, the BTE showed increasing trend with increased blend ratio of alcohol in diesel up to 20%.
• However blends beyond 20% were not considered due to reduced engine power and increased brake specific fuel consumption because of lower calorific value of the butanol. Compared to neat diesel, butyl alcohol-diesel blended fuels showed improved performance in terms of increased BTE.
• HC, CO and smoke emissions reduced with increased alcohol concentration in diesel fuel while they increased with increased loading conditions However NOx emissions increased with increased butanol content in diesel fuel.
• Ignition delay, combustion duration, peak pressure and heat release rates increased with increased alcohol content in the diesel fuel.
On the whole it can be concluded that the modified single cylinder engine operated with alcoholic-diesel blended fuels worked satisfactorily with improved engine performance compared to neat diesel fuel operation. This experimental work showed the capability of alcoholic fuels which are renewable energy sources to replace diesel partially and is the need of hour. Finally from the experimental study it can be said that butanol can be a good option for reducing fossil fuel usage in diesel engines and pave the way for the energy security of the country due to escalating fossil fuel prices with dependency on such fuels.
References
- Swamy RL, Chandrashekar TK, Banapurmath NR, Nashipudi P (2014) Effect of Injection Timing, Combustion Chamber Shapes and Nozzle Geometry on the Diesel Engine Performance. Universal Journal of Petroleum Sciences 2: 74-95.
- Swamy RL, Chandrashekar TK (2013) Potentiality of Dibutyl Ether on Diesel Engine Performance and Emissions. International Journal of Engineering Research & Technology 2.
- Merola SS, Tornatore C, Iannuzzi SE, Marchitto L, Valentino G (2014) Combustion process investigation in a high speed diesel engine fuelled with n-butanol diesel blend by conventional methods and optical diagnostics. Renewable Energy 64: 225-237.
- Baskar P, Nanthagopal K, Elango T(2011) The effect of two oxygenates on diesel engine emissions. ARPNJournal of Engineering and Applied Sciences 6.
- Kannan D, Senthil PK, Nurun Nabi MD, Hustad JE, Løvås T (2012) Theoretical and experimental investigation of diesel engine performance, combustion and emissions analysis fuelled with the blends of ethanol, diesel and jatropha methyl ester. Energy Conversion and Management53: 322-331.
- Arnal JM, Dorado MP (2012) A comparison of performance of higher alcohols/diesel fuel blends in a diesel engine. Applied Energy 95: 267-275.
- Armas O, García-Contreras R, Ramos A (2014) Pollutant emissions from engine starting with ethanol and butanol diesel blends. Fuel Processing Technology 122: 64-71.
- Armas O, García-Contreras R, Ramos A (2012) Pollutant emissions from engine starting with ethanol and butanol diesel blends. Fuel Processing Technology 100: 63-72.
- Ashrafur Rahman SM, Masjuki HH, Kalam MA, Sanjid A, Abedin MJ (2014) Assessment of emission and performance of compression ignition engine with varying injection timing. Renewable and Sustainable Energy Reviews 35: 221-230.
- Wang X, Cheung CS, Di Y, Huang Z (2012) Diesel engine gaseous and particle emissions fueled with diesel–oxygenate blends. Fuel 94: 317-323.
- Banapurmath NR, Tewari PG (2010) Performance, combustion, and emissions characteristics of a single-cylinder compression ignition engine operated on ethanol–biodiesel blended fuels. Journal of power and Energy 224: 533-543.
- Gnanamoorthi V, Devaradjane G (2013) Effect of Diesel-Ethanol Blends on Performance, Combustion and Exhaust Emission of a Diesel Engine. International Journal of CurrentEngineering and Technology 3.
- Murcak A, Hasimoglu C, Çevik I, Karabektas M, Ergen G (2013) Effects of ethanol–diesel blends to performance of a DI diesel engine for different injection timings. Fuel 109: 582-587.
- Nabi MN, Kannan D, Einar Hustad J, Mustafizur Rahman M (2009) Role of oxygenated fuel to reduce diesel emissions: A Review. InternationalConference on Mechanical Engineering 26-28.
- Miyamoto N, Ogawa H, Nabi Nurun MD, Obata K, Teruyoshi A (1998) Smokeless, Low NOx, high thermal efficiency and low noise diesel Combustion with oxygenated agents as main fuel. Society of Automotive Engineers.
- Gonzalez AD, Piel W, Asmus T, Clark W, Garbak J (2001) Oxygenates screening for advanced Petroleum-based Diesel Fuels: Part 2. The Effect of Oxygenate Blending Compounds on Exhaust Emissions. Society of Automotive Engineers.
- Hilden DL, Eckstrom JC, Wolf LR (2001) The Emissions Performance of Oxygenated Diesel Fuels in a Prototype DI Diesel Engine. Society of Automotive Engineers.
- Labeckas G, Slavinskas S, Mazeika M (2014) The effect of ethanol–diesel–biodiesel blends on combustion, performance and emissions of a direct injection diesel engine. Energy Conversion and Management 79: 698-720.
- Hansdah D, Murugan S (2014) Bioethanol fumigation in a DI diesel engine. Fuel 130: 324-333.
- Subramanian KA, Ramesh A (2002) Use of Diethyl Ether Along with Water-Diesel Emulsion in a Di Diesel Engine. Society of Automotive Engineers.
- Nagdeote DD, Deshmukh MM (2012) Experimental Study of Diethyl Ether and Ethanol Additives with Biodiesel-Diesel Blended Fuel Engine. International Journal of Emerging Technology and Advanced Engineering 2.
- Deshpande SS, Channapattana SV, Pawar AA (2015) Experimental Evaluation of Diesel Engine Performance and Emissions Using Diesel/Biodiesel/Ethanol Blend Fuel. International Journal of Emerging Technology and Advanced Engineering 5.
- Likhitha SS, Durga Prasad B, Vikram Kumar CHR (2014) Investigation on the Effect of Diethyl Ether Additive on the Performance of Variable Compression Ratio Diesel Engine. International Journal of Engineering Research 3: 11-15.
Relevant Topics
Recommended Journals
- Oil & Gas Research Journal
- Renewable Energy and Applications Journal
- Oceanography Journal
- Industrial Pollution Control Journal
- Coastal Zone Management Journal
- Climatology & Weather Forecasting Journal
- Geoinformatics & Geostatistics Journal
- Engineering and Technology Journal
- Petroleum & Environmental Biotechnology Journal
- Polymer Sciences Journal
Article Tools
Article Usage
- Total views: 19645
- [From(publication date):
December-2015 - Sep 01, 2025] - Breakdown by view type
- HTML page views : 14454
- PDF downloads : 5191