Research Article Open Access
Impact of Hiatal Hernia on Pediatric Gastroesophageal Reflux Disease
Au Yeung KJ1, Cannon ML2, Arkachaisri T3, Gillespie S4 and Karnsakul W1,4*
1Department of Pediatrics, Division of Pediatric Gastroenterology and Nutrition, Johns Hopkins University School of Medicine, Baltimore, Maryland, USA
2Robert C Byrd Health Sciences Center, West Virginia University School of Medicine, Morgantown, West Virginia, USA
3Department of Paediatric Subspecialties, KK Women's and Children's Hospital, Singapore
4Robert C Byrd Health Sciences Center, West Virginia University School of Medicine, Morgantown, West Virginia, USA
- *Corresponding Author:
- Wikrom Karnsakul, MD
Assistant Professor, Division of Pediatric Gastroenterology and Nutrition
Johns Hopkins University School of Medicine, CMSC-2
600 North Wolfe Street, Baltimore, MD. 21287, USA
Tel: 410-955-8769
Fax: 410-955-1464
E-mail: wkarnsa1@jhmi.edu
Received date: July 27, 2015 Accepted date: August 19, 2015 Published date: August 27, 2015
Citation: Au Yeung KJ, Cannon ML, Arkachaisri T, Gillespie S, Karnsakul W (2015) Impact of Hiatal Hernia on Pediatric Gastroesophageal Reflux Disease. J Gastrointest Dig Sys 5:330. doi:10.4172/2161-069X.1000330
Copyright: © 2015 Yeung KJA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objectives: To identify the frequency of hiatal hernia (HH) and the impact of HH size in children with gastroesophageal reflux disease (GERD). Methods: Outpatient records were retrospectively reviewed in children ages 0-17 years old, diagnosed clinically with GERD. HH was diagnosed based on an upper gastrointestinal (UGI) series and HH body was characterized as either small (<2 cm) or large (≥2 cm). For patients who underwent an upper endoscopy, presence and severity of esophagitis was diagnosed with esophageal biopsies and correlated with size of HH. Results: There were 155 children diagnosed clinically with GERD who underwent an UGI. HH was diagnosed in 80 (51%) children via UGI (mean age 7.5 y, 32 female, 48 male). Small HH was observed in 60 children (75%; mean age 7 y, 25 female, 35 male) and large HH in 20 (25%; mean age 3.2 y, 7 female, 13 male). Erosive esophagitis was uncommon in children with HH. Abdominal pain, chest pain and heartburn were commonly reported chief complaints in children (age >5yr) with a large HH. Children with large HH were more frequently found to fail medications and require Nissen fundoplication than those with a small HH (p=0.032). Conclusions: Children with GERD had high frequency of HH based on UGI. Children with large HH required significantly more anti-reflux surgery than those with small HH. A larger scale longitudinal prospective study is needed to further investigate the impact of HH on GERD complications and the effects of therapy.
Keywords
Hiatal hernia; Gastroesophageal reflux disease; Esophagitis; Children and adolescents; Radiology
Abbreviations
HH: Hiatal Hernia; GERD: Gastroesophageal Reflux Disease; UGI: Upper Gastrointestinal Series; EGD: Esophagogastroduodenoscopy; BMI: Body Mass Index; GEJ: Gastroesophageal junction; GER: Gastroesophageal Reflux; LES: Lower Esophageal Sphincter; LA: Los Angeles classification; SD: Standard deviation; TLESR: Transient Lower Esophageal Sphincter Relaxation; PPI: Proton Pump Inhibitors
Introduction
Although the relationship between hiatal hernia (HH) and gastroesophageal reflux disease (GERD) has long been recognized, there are many unknown aspects to the long-term impact of HH on GERD in children. Inthat early detection and conventional treatment of sliding HH in children resulted in a favorable outcome for children with persistent vomiting [1]. However, about one-third of the children without treatment continued to suffer from GERD after their fourth birthday with a 10% chance of developing an esophageal stricture. HH is defined as an axial displacement of the proximal part of the stomach through the diaphragmatic hiatus. Type I HH is caused by widening of the muscular hiatal tunnel and circumferential laxity of the phrenoesophageal membrane, thus allowing the gastric cardia to herniate above the diaphragm.
Adult patients with a large HH are known to be more prone to GERD development than those with a smaller HH [2,3]. It has an estimated prevalence of 10-80%, and usually is asymptomatic [4]. In infants and children, invasive procedures are not routinely used to diagnose GERD, but rather clinical signs and symptoms. Therefore, the incidence of HH could be under-diagnosed since the diagnosis of HH requires radiographic or endoscopic examinations. Moreover, current pediatric data relating the size of HH to its clinical significance is lacking. The aim of this study was to investigate the frequency of HH in children based on upper gastrointestinal (UGI) series and the impact of HH size on GERD.
Methods
Following internal review for feasibility and subsequent study approval, outpatient records were reviewed of children 0-17 years old who were diagnosed with GERD and also had an UGI from May 2004 to February 2008 at West Virginia University School of Medicine. GERD was diagnosed based on one or a combination of factors including clinical history, upper endoscopy [(esophagogastroduodenoscopy (EGD)] with biopsies, and an UGI series. Patients with eosinophilic esophagitis, food allergy, inflammatory bowel disease, post-Nissen fundoplication, gastrointestinal obstruction, and chronic or intermittent steroid use were excluded from the study. The following information was collected on each patient: age, gender, body mass index (BMI), neurodevelopmental status, and clinical symptoms at presentation. Clinical symptoms collected included abdominal pain, vomiting, extraesophageal symptoms such as cough, hoarseness, recurrent pneumonia, and asthma, chest pain, heartburn, dysphagia, food refusal, choking, and irritability. Data was also collected regarding history of medical and surgical interventions, responses to therapy, UGI findings specific for HH size, and endoscopic and pathologic esophageal biopsy reports. Medical anti-reflux therapy included use of histamine-2 receptor antagonists, lansoprazole, and/or omeprazole to suppress acid production, and metoclopramide as a prokinetic drug. A failure of medical anti-reflux therapy is defined as persistent symptoms of GERD or a presence of aspiration pneumonia and erosive esophagitis while on treatment longer than 3 months.
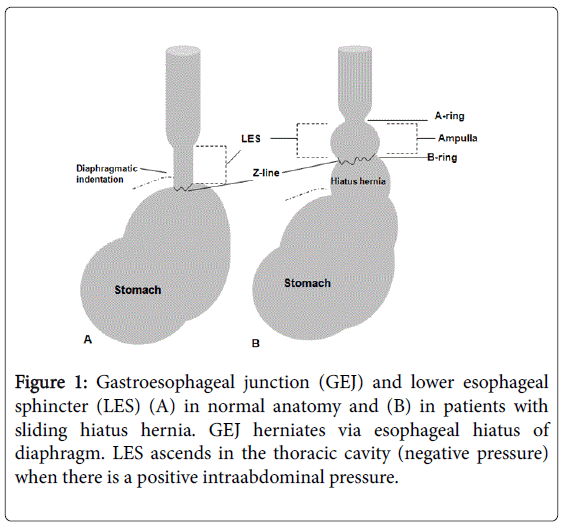
An UGI series was performed by a pediatric radiologist observing standard radiation safety protocol. Infants were fed with dilute barium utilizing a nipple and bottle, whereas older children were given the appropriate amount of barium to drink utilizing a cup and straw. If an enteric tube were needed to administer barium, it was removed at the end of the examination to assess for HH and reflux. The esophagus and gastroesophageal junction (GEJ) were observed during swallowing. The patient was evaluated for HH, gastric emptying, bowel rotation and for position of the ligament of Treitz, and mucosal abnormalities. Gastroesophageal reflux (GER) was evaluated with change in patient position, but without application of manual abdominal pressure. HH was diagnosed when the esophageal vestibule or the upper part of the gastric folds reached 2 cm or more above the diaphragmatic indentation (the so-called B-ring or Schatzki ring) during quiet respiration (Figure 1). The A-ring is the superior-most aspect of the lower esophageal sphincter (LES). A small HH was diagnosed by the presence of an HH body of less than 2.0 cm in length, and a large HH was identified if the HH body was 2.0 cm or greater in length (Figure 1).
Figure 1: Gastroesophageal junction (GEJ) and lower esophageal sphincter (LES) (A) in normal anatomy and (B) in patients with sliding hiatus hernia. GEJ herniates via esophageal hiatus of diaphragm. LES ascends in the thoracic cavity (negative pressure) when there is a positive intraabdominal pressure.
A routine EGD was performed in patients under general anesthesia by endoscopy using a flexible endoscope (Pentax-EG 1840, 2540, or 2731, depending upon patient age). Reflux esophagitis was graded using the criteria of the Los Angeles (LA) classification [5]. Anti-reflux medications were not generally discontinued prior to the EGD. The presence of HH was recorded during an EGD when gastric folds were assessed as extending 2.0 cm or more above the diaphragmatic indentation during quiet respiration and air insufflation into the stomach; however, a close retrograde view to measure the size of the HH with the distended stomach was not performed. Two biopsy specimens were routinely obtained from the distal esophagus and midesophagus. Histological assessment included: evaluation for basal zone hyperplasia, papillary length, dilation of intraepithelial blood vessels and semi-quantitative cellular infiltration by lymphocytes, neutrophils, and eosinophils. These are standardized parameters for the diagnosis of reflux esophagitis. Barrett's esophagus was defined as intestinal metaplasia on esophageal biopsies. Severe GERD complications were collected and defined by erosive esophagitis, iron deficiency anemia, stricture, Barrett’s esophagus and adenocarcinoma.
Statistics
Statistical analysis was performed with SPSS v. 15 (SPSS, Inc, Chicago IL). The mean and standard deviation (SD) were used to describe demographic data. The chi-square test or Fisher's exact test were used for differences between the large and small HH groups. A pvalue < 0.05 was considered to be significant.
Results
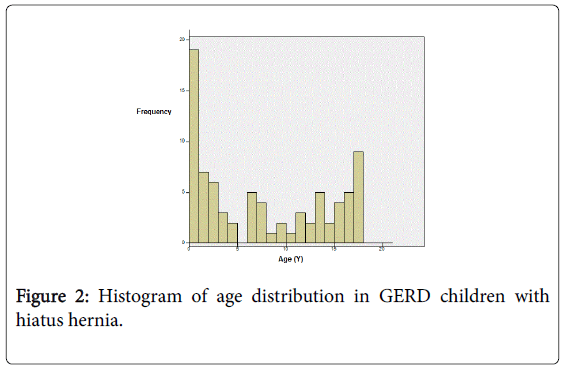
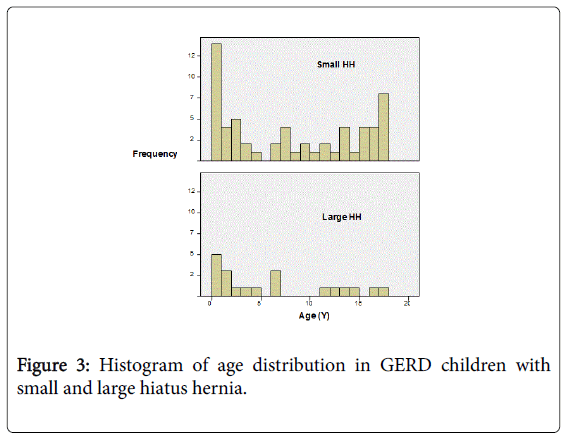
UGI results: We reviewed 155 charts of children who were diagnosed with GERD and also had an UGI during May 2004 to February 2008. HH was detected in 80/155 children (51.6%; mean age 7.5 ± 6.48 y, 32 female, 48 male). Small HH was observed in 60 children (75%; mean age 7 y, 25 female), and large HH was observed in 20 children (25%; mean age 3.2 y, 7 female). Among the group diagnosed with HH, a bimodal distribution was observed, with the higher peak occurring in early infancy (Figures 2 and 3). Clinical symptoms at presentation included vomiting, extra-esophageal symptoms (cough, hoarseness, recurrent pneumonia, and asthma), dysphagia, food refusal, choking, and irritability. The chief complaints and presenting symptoms among children older than 5 years were abdominal pain, chest pain and heartburn (no difference among HH sizes).
Small versus large HH: Age and gender showed no difference between groups. Eleven children (6 with small HH, 5 with large HH) had delayed development and there was no statistically significant difference between the two groups (p=0.08). In 49 HH patients who had BMI documented in the chart, 23 were diagnosed with obesity with no statistical difference between small and large HH.
EGD results: Only 45 of the HH children underwent an EGD (34 small HH and 11 large HH on UGI). Only one patient who had a large HH on the UGI series also had HH identified during the EGD. The other 44 had no landmarks documented in the chart to indicate HH was evident endoscopically. Mild esophagitis (Grade A) was diagnosed more frequently than erosive esophagitis (Grade B to D) in each group of HH size (Table 1); however, there was no statistical difference between small and large HH patient groups to associate the severity of esophagitis with the size of HH (Table 1). Iron deficiency anemia was diagnosed in one of the patients who had a large HH and erosive esophagitis. Barrett's esophagus did not occur in any of the children with HH. However, one child without HH had Barrett’s esophagus whose follow-up biopsies were negative for intestinal metaplasia after completing 3 month anti-reflux medical treatment.
| Size of hernia | Small HH | Large HH | p value |
|---|---|---|---|
| Number of Patient | 60 | 20 | |
| Age (Y) | |||
| 0-5 | 24 (30%) | 11 (55%) | 0.484 |
| 06-Nov | 14 (17%) | 4 (20%) | |
| Dec-17 | 22 (27.5%) | 5 (25%) | |
| Male | 35 (58%) | 13 (65%) | |
| Obesity (>85 %le BMI) n=49 | 19 (31%) | 4 (2.0%) | 0.724 |
| Delayed development n=11 | 6 (7.5%) | 5 (6%) | 0.12 |
| abdominal pain | 23 (38%) | 5 (25%) | 0.417 |
| vomiting | 40 (67%) | 13 (68%) | 0.891 |
| cough | 1 (1.6%) | 5 (25%) | 1 |
| hoarseness | 6 (10%) | 6 (30%) | 0.673 |
| recurrent pneumonia | 1 (2%) | 20 (5%) | 0.44 |
| asthma | 15 (24%) | 6 (30%) | 1 |
| supraesophageal | 0.645 | ||
| symptoms | 17 (28%) | 6 (30%) | 0.887 |
| chest pain/heartburn | 13 (21%) | 9 (45%) | 1 |
| dysphagia/food refusal/choking | 7 (11%) | 2 (10%) | 1 |
| irritability | 0 | 8 (40%) | 0.25 |
| Mild esophagitis (n= 40/45) | 31 | 9 | 0.582 |
| Erosive esophagitis | 3 | 2 | 0.582 |
| (n=5/45) | |||
| Iron deficiency anemia | 0 | 1 | |
| Acid suppression therapy | 51 (85%) | 16 (80%) | 0.727 |
| Prokinetic | 29 (48.3%) | 10 (50%) | 1 |
| Nissen Fundoplication | 2 (3%) | 4 (20%) | 0.032 |
| Family history of HH | 10 (1.6%) | 3 (15%) | 0.705 |
| Family history of GERD | 22 (36%) | 6 (30%) | 0.69 |
Table 1: Demographic and clinical presentation data on 80 children with HH.
Therapy: Prokinetic therapy (metoclopramide) was prescribed in 29 children (48%) with small HH and 10 children (50%) with large HH; antacid therapy was utilized in 51 children (80%) with small HH and 16 children (80%) with large HH. When medical therapy failed, a Nissen fundoplication was performed, significantly improving reflux symptoms in 4 children with large HH and 2 children with small HH (p=0.032) as described. Three of these children (1 large HH, 2 small HH) had a delayed developmental history (p=0.031) as described in Figure 3. Besides HH size and developmental delay, other parameters such as age, gender, clinical symptoms, histological findings of the esophagus, and medical therapies showed no statistical significant difference for the requirement of Nissen fundoplication.
Discussion
Although, the UGI series is neither sensitive nor specific for the diagnosis of GER, it is a common investigative tool used in children with persistent vomiting to detect anatomic abnormalities such as HH. Based on UGI, we documented the frequency of HH in a group of children diagnosed with GERD to be 51.6%, small HH being more frequent than large. We could not confirm this same rate of HH with EGD, but were able to show through biopsies that mild esophagitis occurred for a large portion of patients with HH without a difference between size of HH. We did not find a significant association between having a large or small HH and various patient characteristics such as developmental delay, obesity, age or gender. However, this data characterized a specific pediatric population with large HH and/or developmental delay who are more likely to fail medical therapy and ultimately be referred for Nissen fundoplication to definitively treat GERD.
Understanding anatomy and physiology of the LES and HH
The LES, a functional barrier with no anatomical landmarks, is an intraluminal zone with a basal pressure greater than that of the stomach and esophagus, approximately half of which lies intraabdominally in adults. Manometric study has shown that the length of LES increases with age, measuring 3mm in neonates, 0.5-0.75 cm in infants under 3 months of age, 1 cm in older infants less than a year old, 1-3 cm in older children, and 3-5 cm in adults [6].
The anatomy consists of the intrinsic part of the LES which is formed by the intrinsic muscles of the distal esophagus and the gastric cardia. It is augmented externally by the crural diaphragm, the angle of His which is the valve-like intra-abdominal portion (approximately 2 cm) of the esophagus, and the phreno-esophageal ligament, which inserts circumferentially into the esophageal musculature close to the squamo-columnar junction. HH is characterized by the herniation of parts of the abdominal contents through the esophageal hiatus of the diaphragm [7,8]. The most common type of HHs is characterized by a widening of the muscular hiatal diaphragmatic opening with laxity of the phreno-esophageal membrane, allowing some of the gastric cardia to herniate upwards (Figure 3).
Mechanisms responsible for GER in individuals with HH include not only transient LES relaxations (TLESRs), but also swallowassociated reflux and GER associated with straining, more than those without HH [9,10]. During a swallow, the esophageal body is shortened by the longitudinal esophageal muscle contractions, reducing LES length, which makes HH even more pronounced. Understanding physiologic changes of the LES and HH during swallowing helps explain the discrepancy in diagnosing small HH on UGI as compared to EGD, as was documented in this study.
Methods to diagnose HH in children
HH is primarily diagnosed by UGI series or by EGD. In adult patients, literature has suggested guidelines during EGD to inspect for HH utilizing proper air insufflation prior to gastric intubation and during intra-gastric retroflection [4]. It is unknown, however, whether diagnostic guidelines for HH and measuring size of HH during endoscopy should be different for infants and children. In this study, only one radiologist performed the UGI series with technique as described above. In comparison, there has been at least one other investigation of HH in children based on criteria of at least three gastric folds above the diaphragm seen on UGI [11,12]. Such extensive criteria for classifying HH may under-diagnose a small HH. Finally, the other difference in diagnosing HH size by UGI versus EGD is influenced by deep sedation or anesthesia required for pediatric endoscopy. The dynamic phenomenon of HH induced by swallowing, respiration, and valsalva during an UGI is reduced with anesthesia [4]. Thus, we surmise that the lower frequency of HH detected during an EGD compared to UGI in our series was due possibly to the differences in technique between endoscopy and radiology to identify particularly smaller-sized HH. Scarpato et al reported the prevalence of HH approximately 20.7% in 111 children who had EGD for evaluating GI symptoms. 13 However, they believed that HH could be overestimated via EGD given the lower esophageal sphincter relaxation under conscious or deep sedation [13,14]. Wu et al reported ratio of reflux episodes detected by pH-probe to that by impedance probes >1 was more frequently observed in children with than without with the sensitivity and specificity of 93.8 and 79.6% respectively [15].
Does HH size impact GERD complications?
Based on the fact that patients with large HH were more likely to fail medical management and require Nissen fundoplication, there was a significant association between the size of HH and GERD. Studies investigating HH in adults that included endoscopic evaluation have shown an association with size of HH and GERD severity [16]. The increasing size of HH has been associated with greater esophageal acid exposure, more prolonged episodes of reflux, and longer acid clearance times in a study of nine controls and 38 patients with GERD [17]. Furthermore, HH is known to cause impaired esophageal acid clearance through a mechanism of decreased amplitude of esophageal peristalsis [18]. It has been suggested that increased acid exposure time in patients with HH is explained by a reservoir effect in HH, from which acid can reflux to the esophagus during swallowing and LES relaxation [19,20].
Therapeutic effect of medication management for HH
Gorenstein et al. observed significant failure rates of medical therapy in children with HH, both with and without neurological disorders [12]. The authors, however, did not clarify whether medical treatment with proton pump inhibitor (PPI) was used prior to surgery. We evaluated a subset of patients who received an EGD in our study to compare size of HH to biopsy findings and found no statistical differences between HH groups. Mild esophagitis was the most common finding in both groups. Likewise, there was no difference between HH groups for complications from GERD diagnosed by biopsy such as erosive esophagitis, stricture, and Barrett’s esophagus. Possibly the low rate of complications was explained by the PPI use providing protective mucosal effect, but there was no control for being on antacid therapy.
We discovered that patients with a large HH received statistically significant more Nissen fundoplication surgeries (p=0.032). Large HH is often associated with severe GERD symptoms and a poorer response to treatment. In our study, in addition to having a large HH, delayed neuro-development seemed to have a higher likelihood of getting a Nissen fundoplication as compared to those with a small HH or normal neurological development.
One limitation of this study is that it lacks objective criteria for the diagnosis of GERD such as with pH/impedance probe studies. Not all the children had upper endoscopy to confirm the gross endoscopic findings of HHs and the degree of GERD histopathology as routinely performed. However, objective data was evaluated through a subset of 45 patients who had an EGD and compared biopsy results between the patients who had evidence of large versus small HH seen on UGI. Furthermore, diagnosis of HH on the UGI was consistent among patients because objective criteria were used by a single pediatric radiologist who performed the test on all the patients. Children less than 5 years old seemed to be the majority in both small and large HH. Although there have been no data on the cutoff length to differentiate size of HH in young children, LES length are usually around 2-3 cm in this age group [6]. We might underestimate the size of HH in young infants. Another potential bias with the study population is that clinical practice only warrants an UGI series in GERD patients with more severe and persistent symptoms. No longitudinal records of GERD symptoms was available. Last but not least, it is possible that Nissen fundoplication was suggested under the bias of the fact that some of those children were developmentally delayed or had large HHs.
Conclusion
Knowledge about the role of the HH and HH size in pediatric GERD has still been limited due to a lack of a uniform diagnostic criteria and a gold standard diagnostic test and a larger population study. Better diagnostic techniques for HH may include the utilization of UGI, EGD, and esophageal pH probe and impedance study and/or esophageal motility test. Further prospective study at different age groups will improve understanding the impact of HH on the development, progression, and treatment of GERD in children.
References
- CARRE IJ (1960) Postural treatment of children with a partial thoracic stomach ('hiatus hernia'). Arch Dis Child 35: 569-580.
- Ott DJ, Glauser SJ, Ledbetter MS, Chen MY, Koufman JA, et al. (1995) Association of hiatal hernia and gastroesophageal reflux: correlation between presence and size of hiatal hernia and 24-hour pH monitoring of the esophagus. AJR Am J Roentgenol 165: 557-559.
- Patti MG, Goldberg HI, Arcerito M, Bortolasi L, Tong J, et al. (1996) Hiatal hernia size affects lower esophageal sphincter function, esophageal acid exposure, and the degree of mucosal injury. Am J Surg 171: 182-186.
- Gordon C, Kang JY, Neild PJ, Maxwell JD (2004) The role of the hiatus hernia in gastro-oesophageal reflux disease. Aliment PharmacolTher 20: 719-732.
- Sugiura T, Iwakiri K, Kotoyori M, Kobayashi M (2001) Relationship between severity of reflux esophagitis according to the Los Angeles classification and esophageal motility. J Gastroenterol 36: 226-230.
- Semeniuk J, Kaczmarski M, Uścinowicz M (2008) Manometric study of lower esophageal sphincter in children with primary acid gastroesophageal reflux and acid gastroesophageal reflux secondary to food allergy. Adv Med Sci 53: 283-292.
- Dodds WJ (1977) 1976 Walter B. Cannon Lecture: current concepts of esophageal motor function: clinical implications for radiology. AJR Am J Roentgenol 128: 549-561.
- Mittal RK (1997) Hiatal hernia: myth or reality? Am J Med 103: 33S-39S.
- Sloan S, Rademaker AW, Kahrilas PJ (1992) Determinants of gastroesophageal junction incompetence: hiatal hernia, lower esophageal sphincter, or both? Ann Intern Med 117: 977-982.
- van Herwaarden MA, Samsom M, Smout AJ (2000) Excess gastroesophageal reflux in patients with hiatus hernia is caused by mechanisms other than transient LES relaxations. Gastroenterology 119: 1439-1446.
- van Herwaarden MA, Samsom M, Smout AJ (2004) The role of hiatus hernia in gastro-oesophageal reflux disease. Eur J GastroenterolHepatol 16: 831-835.
- Gorenstein A, Cohen AJ, Cordova Z, Witzling M, Krutman B, et al. (2001) Hiatal hernia in pediatric gastroesophageal reflux. J PediatrGastroenterolNutr 33: 554-557.
- Scarpato E, D'Armiento M, Martinelli M, Mancusi V, Campione S, et al. (2014) Impact of hiatal hernia on pediatric dyspeptic symptoms. J PediatrGastroenterolNutr 59: 795-798.
- Pensabene L, Miele E, Del Giudice E, Strisciuglio C, Staiano A (2008) Mechanisms of gastroesophageal reflux in children with sequelae of birth asphyxia. Brain Dev 30: 563-571.
- Wu JF, Hsu WC, Tseng PH, Wang HP, Hsu HY, et al. (2013) Combined multichannel intraluminal impedance and pH monitoring assists the diagnosis of sliding hiatal hernia in children with gastroesophageal reflux disease. J Gastroenterol 48: 1242-1248.
- Berstad A, Weberg R, Frøyshov Larsen I, Hoel B, Hauer-Jensen M (1986) Relationship of hiatus hernia to reflux oesophagitis. A prospective study of coincidence, using endoscopy. Scand J Gastroenterol 21: 55-58.
- Kasapidis P, Vassilakis JS, Tzovaras G, Chrysos E, Xynos E (1995) Effect of hiatal hernia on esophageal manometry and pH-metry in gastroesophageal reflux disease. Dig Dis Sci 40: 2724-2730.
- Jones MP, Sloan SS, Jovanovic B, Kahrilas PJ (2002) Impaired egress rather than increased access: an important independent predictor of erosive oesophagitis. NeurogastroenterolMotil 14: 625-631.
- Mittal RK, Lange RC, McCallum RW (1987) Identification and mechanism of delayed esophageal acid clearance in subjects with hiatus hernia. Gastroenterology 92: 130-135.
- Sloan S, Kahrilas PJ (1991) Impairment of esophageal emptying with hiatal hernia. Gastroenterology 100: 596-605.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 19403
- [From(publication date):
October-2015 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 14621
- PDF downloads : 4782