Case Report Open Access
Intranasal Diprospan Injection for Chronic Rhinosinusitis Treatment-Two Case Reports
Anna Yu Poghosyan1*, Armen L Zargaryan1 and Levon R Galstyan21Department of Maxillofacial and ENT Surgery “Heratsy” No 1 University Hospital, 60 Abovyan Str, Yerevan 0025, Armenia
2Department of Maxillofacial Surgery in “Shengavit” Medical Center, 9 Manadtyan Str, Yerevan 0006, Armenia
- *Corresponding Author:
- Anna Yu. Poghosyan
Head of ENT and Maxillofacial Surgery
Department in “Heratsy” â�?�?1 Hospital 60 Abovyan Str
Yerevan, 0025, Armenia
Tel: +37491474169
E-mail: anna.yu.poghosyan@gmail.com
Received Date: April 02, 2016; Accepted Date: April 14, 2016; Published Date: April 20, 2016
Citation: Poghosyan AY, Zargaryan AL, Galstyan LR (2016) Intranasal Diprospan Injection for Chronic Rhinosinusitis Treatment: Two Case Reports. J Orthop Oncol 2:109. doi:10.4172/2472-016X.1000109
Copyright: © 2016 Poghosyan AY, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Orthopedic Oncology
Abstract
Chronic rhinosinusitis with nasal polyposis (CRSwNP) affects 0.5–4% of the world population and is present in ∼20% of patients with CRS. There are no definitive guidelines for the treatment of CRS largely due to the lack of consensus on the etiology of the disease. The management of chronic rhinosinusitis with nasal polyps, aimed at improving clinical symptoms, includes both surgical and medical treatments, but there is no universally accepted management protocol. Two case reports of CRSw NP treatment by intranasal Diprospan injection are presents. In both cases significant clinical and radiological improvement was observed without any side effects.
Keywords
Chronic rhinosinusitis; Nasal polyposis; Diprospan injection
Abbreviations
CRS: Chronic Rhinosinusitis; CRSwNP: Chronic Rhinosinusitis with Nasal Polyposis; INCSs: Intranasal Corticosteroids; CT: Computed Tomography
Introduction
Chronic rhino sinusitis with nasal polyposis (CRSwNP) affects 0.5–4% of the world population and is present in ∼20% of patients with CRS [1-6]. Rhinosinusitis is defined as a sudden onset of two or more symptoms, one of which should be either nasal blockage or nasal discharge (anterior or posterior nasal drip). Other symptoms are facial pain or pressure, and impairment or loss of smell [7-9]. It is also important to gauge the severity of the patient’s condition as well as the impact on the patient’s quality of life. The pathogenesis of CRS remains controversial. Multifactorial factors altering the host-environment interaction such as bacteria, fungi, viruses, allergens, or environmental toxins may trigger the inflammatory process [10]. That is why there are no definitive guidelines for the treatment of CRS largely due to the lack of consensus on the etiology of the disease [2]. The management of chronic rhino sinusitis with nasal polyps, aimed at improving these symptoms, includes both surgical and medical treatments, but there is no universally accepted management protocol [11]. Topical treatment with intranasal corticosteroids (INCSs) has been widely used to control disease symptoms in patients with CRSwNP. INCSs can be classified as modern (mometasone, fluticasone, and ciclesonide) versus first-generation corticosteroids (budesonide, beclomethasone, betamethasone, triamcinolone, and dexamethasone). Effectiveness of INCSs depends on type, dose, delivery method (i.e., nasal spray, drops, direct irrigation of the sinuses, catheters, and atomizer), and length of the treatment. However, the constant use of nasal sprays or drops causes the patient lots of inconveniences and impacts the quality of life, especially for people living active lifestyle.
The purpose of this study is to present two case reports of CRSwNP treatment by intranasal Diprospan injection.
Case Report 1
A 62-year old female patient was referred to the department of ENT and Maxillofacial Surgery of “Heratsy” â�?�?1 University Hospital on September 2015 with complaint on persistent headache, facial pain, nasal congestion, rhino rhea with clear mucous discharge, difficulty breathing through nose, loss of smell, dyspnoea, general malaise, cough and lower extremities edema.
She had a history of radical bilateral Coldwell-Luc operation 7 years ago, which was carried out on the occasion of chronic sinusitis with polyposis. Acute cerebrovascular accident in 2010, hypertension, asthma and cardio-pulmonary insufficiency were noted by the patient.
Anterior rhinoscopy showed large polyps, which grow down to nasal cavities external opening. Nasal mucosa was bluish with clear mucus.
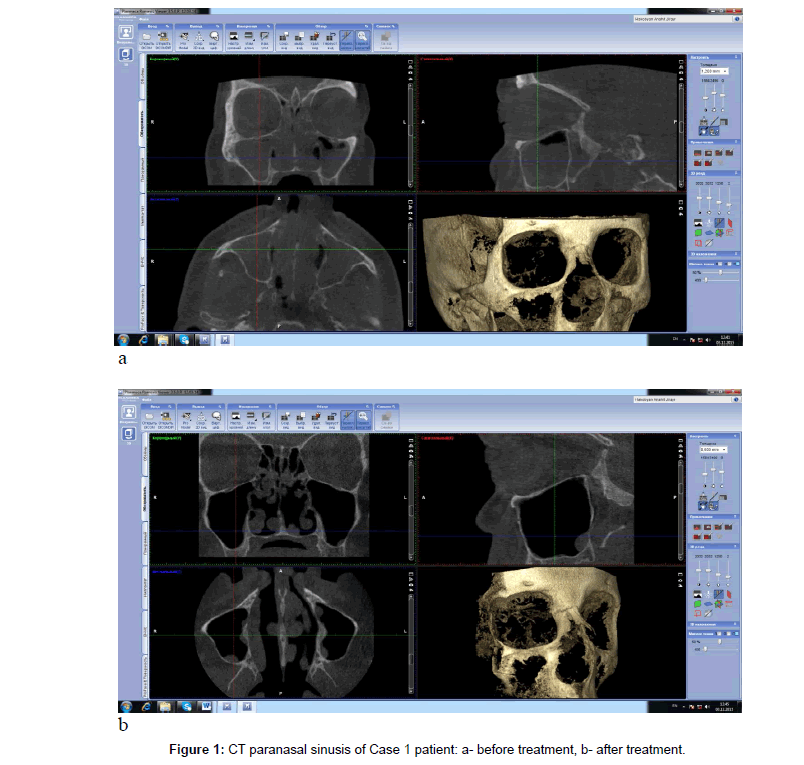
Coronal CT scan showed appearance of sinonasal polyposis with polypoid soft tissue masses within nasal cavity and paranasal sinusis. Opacification of all paranasal sinusis with the nasal cavity filled with polyps was observed (Figure 1a).
As a treatment of choice medical therapy was carried out. Injection of 1 ml Diprospan solution was done in the lateral wall of nasal cavity. Intranasal insulin needle prick conducted at a depth of 3-4 mm from the external nasal opening in the region of mucodermal fold. Aspiration probe was necessary before injection.
On control examination after one week patient noted, that had less facial pain, headache and rhinorrhea. Anterior rhinoscopy showed a decrease in polyps’ sizes. The second injection with the same technique was done.
The next examination was done in a month. She already did not have any complains and noted, that she could smell. The last injection was done and the patient was appointed for clinical and CT scan examination in two months.
On her last examination she did not have any complains. She noted excellent breathing, smelling, absence of rhinorrhea, coughing and even lower extremities edema.
Coronal and axial CT scan showed pneumatization of all sinusis and several residual small soft tissue masses in the nasal cavity (Figure 1b).
Case Report 2
A 28-year old male was referred to the department of ENT and Maxillofacial Surgery of “Heratsy’’ â�?�?1 University Hospital on August 2015 with complaint on persistent headache, nasal congestion, rhino rhea with anterior and posterior discharge, inability to nasal breathing and loss of smell. He gave a history of disease first manifestations in 2012.
Anterior rhinoscopy reveals pale, grey polypoid masses arising middle meatus and prolapsing into the nasal cavity.
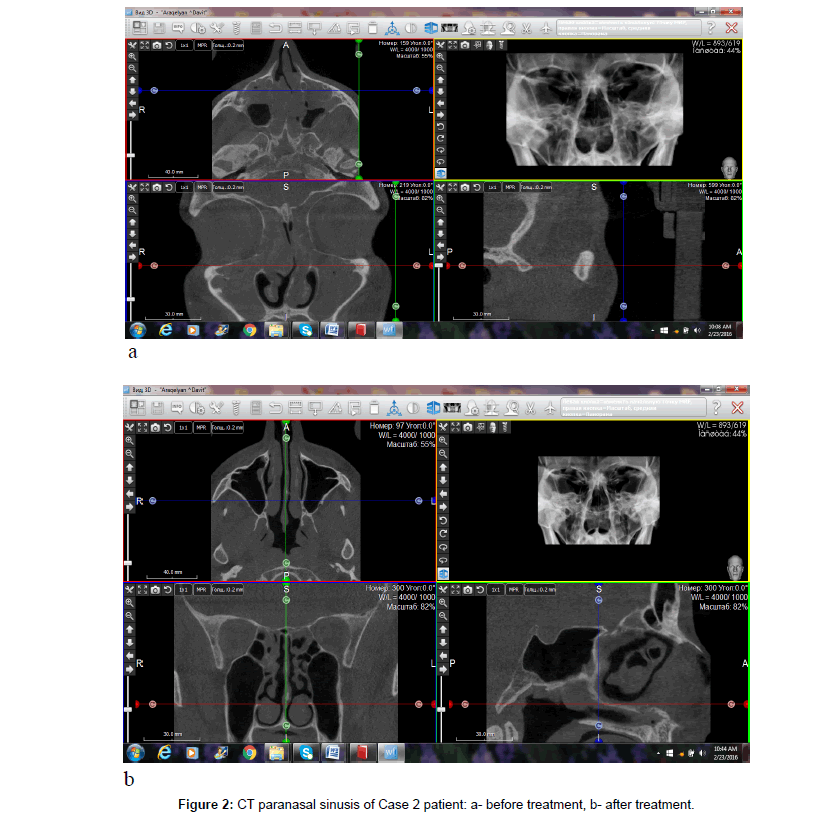
Coronal and axial CT scan showed appearance of sinonasal polyposis with polypoid soft tissue masses within nasal cavity and paranasal sinusis. Opacification of all paranasal sinusis with the nasal cavity filled with polyps and nasal septum deviation was observed (Figure 2a).
Medical therapy was carried out. Injection of 1 ml Diprospan solution was carried out by the scheme presented above (first day, one week, and one month).
On control examination after one week the patient noted, that had less headaches and rhinorrhea. Anterior rhinoscopy showed a decrease in polyps’ sizes. After one month he already did not any complains and noted good smelling. Three months after the last injection the patient was appointed for control clinical and radiological examination.
He did not have any complains, noted excellent breathing, smelling and absence of rhinorrhea.
Coronal and axial CT scan showed pneumatization of all sinusis and several residual small soft tissue masses in the right ethmoidal sinus (Figure 2b).
Discussion
CRS is a common health problem which significantly affects quality of life [6,7,10].
CRS, with or without nasal polyps in adults is defined as inflammation of the nose and the paranasal sinuses characterized by two or more symptoms, one of which should be either nasal blockage/ obstruction/congestion or nasal discharge (anterior/posterior nasal drip) ± facial pain/pressure ± reduction or loss of smell for ≥ 12 weeks [10,12]. In addition to history, physical signs are important criteria in the diagnosis of CRS. This should be supported by demonstrable disease with endoscopic signs of nasal polyps, and/or mucopurulent discharge primarily from middle meatus and/or edema/mucosal obstruction primarily in middle meatus [10].
Anterior rhinoscopy is a noninvasive means of viewing the nasal mucosa [13]. However, it is difficult to view beyond the anterior portion of the nasal passages, even after administration of topical decongestants. Nasal endoscopy, although more invasive, is a preferable method for obtaining a magnified view of the nasal mucosa, turbinates, and interior of the nasal airway in the preoperative and post-surgical patient. Endoscopy aids in assessing the integrity of the mucosa, as well as directly seeing mucosal changes, polyps, crusting, and/or discharge. Cultures can also be obtained endoscopically [13]. Discolored nasal discharge, polyps, or polypoid swelling seen with anterior rhinoscopy or endoscopy, and edema or erythema of the middle meatus or ethmoid bulla seen on endoscopy, are consistent with CRS [2,14]. In Case 1 patient polyps are closed both nasal passages and endoscopicaly was possible to view only 1,5 cm of the nasal passage depth. Radiologic imaging is necessary to view the sinuses and to make or confirm the diagnosis of CRS [2,5]. Plain film X-rays have not been proven to be useful in CRS. Computed tomography (CT) scanning is the imaging method of choice. Direct coronal CT affords an excellent view of the bony structures and mucosal lining. These coronal CT scans are indicated in patients with recurrent sinusitis or CRS in order to properly consider treatment options and sequence the steps in the evaluation. In surgical candidates, CT scanning clearly defines the surgical anatomy and the extent of the disease process [13]. Mucosal thickening, bony changes, or air-fluid levels seen on CT are consistent with CRS [2]. There are instances when lateral or axial imaging is required to determine the extent of the disease process. Magnetic resonance imaging (MRI) is not recommended for diagnosis of CRS due to its lack of specificity [2].
Surgery has long been a treatment of choice for persistent CRS, and with the advent of endoscopy, most surgeries are now minimally invasive. Although studies have shown positive outcomes from surgery, with subjective improvement ranging from 70% to 98% of patients, surgery does not necessarily cure the disease and should be considered as an adjunct to medical therapy. However, when it comes to CRS with sinonasal polyposis, postoperative recurrence often occurred, even when aeration is improved [15-17]. And medical treatment is often still required after surgery [6,18]. For Case 1 patient the recurrent was observed after Coldwell Luk and ethmoidal polypectomy operation and it is necessary to note her asthmatic component.
Randomized controlled trials compared the efficacy between surgery and medication in CRS. Ragab et al. [19] showed no difference in total symptom scores in the medication group (erythromycin plus nasal steroid plus nasal douche) and surgical group (endoscopic sinus surgery plus nasal steroid plus nasal douche). The Cochrane review suggested that functional endoscopic sinus surgery has not been demonstrated to confer additional benefits to those obtained by medical treatment [20]. Piromchai et al. [10] recommend surgical intervention only when there is no response to maximal medical treatment.
Topical treatment with intranasal corticosteroids (INCSs) has been widely used to control disease symptoms in patients with CRSwNP [16,21-23]. The aim of corticosteroid therapy in CRS is to reduce inflammation via directly reducing eosinophil viability and activation [24,25]. In addition, an indirect effect can reduce the secretion of chemotactic cytokines from the nasal mucosa and polyp’s epithelial cells [26].
In a recently published systematic review looking at the effect of INCSs, a substantial positive effect on patients’ symptoms was found [27]. INCS treatment favors the direct drug delivery to diseased mucosa and has the potential for delivering higher local drug concentrations, minimizing systemic absorption, and systemic side effects [3,4]. Effectiveness of INCSs depends on type, dose, delivery method (i.e., nasal spray, drops, direct irrigation of the sinuses, catheters, and atomizer), and length of treatment [6,28,29]. INCSs can be classified as modern (mometasone, fluticasone, and ciclesonide) versus first-generation corticosteroids (budesonide, beclomethasone, betamethasone, triamcinolone, and dexamethasone). The delivery method of topical steroids is an imperative factor. Classification of delivery methods can be divided by site (nose or paranasal sinus), volume, and pressure. The delivery methods to the nasal site include drops, sprays, and nebulizers. However, simply applying topical steroid through the nostrils does not imply delivery of the drug into the sinus. To deliver topical medicine into the sinuses, an appropriate access and delivery is required [29]. Paranasal sinus delivery requires devices cannulated through the nose [10]. In our cases the Diprospan intranasal intradermal injection was used for GRSwNP treatment. Diprospan is the brand of betamethasone dipropionate and betamethasone sodium phosphate. It is indicated for intramuscular, intra-articular, peri-articular, intradermal, intralesional and soft tissue injection. Each ml of Diprospan Suspension contains 5 mg of betamethasone as dipropionate and 2 mg of betamethasone as sodium phosphate in a sterile buffered vehicle. It provides a combination of highly soluble and very slightly soluble esters of betamethasone that produce anti-inflammatory, antirheumatic and anti-allergic effects. Prompt therapeutic activity is achieved by the soluble ester, betamethasone sodium phosphate, which is quickly absorbed after injection. Sustained activity is provided by betamethasone dipropionate, which is only slightly soluble and becomes a repository for slow absorption, thereby controlling symptoms over a prolonged period. The small crystal size of betamethasone dipropionate permits the use of a fine gauge needle (up to 26 g) for intradermal and intralesional administration.
In presented cases we have got clinical and radiological proved treatment of III and IV stages of CRSwNP by intranasal Diprospan injection. No adverse effects were reported by the patients in reported cases.
Conclusion
In reported cases intranasal subdermal Diprospan injection represents as a safe therapy in a primary and postoperative management of CRSwNP. Intranasal Diprospan injection showed significant improvement in patients’ symptoms, clinical and radiological imagine.
References
- Ah-See KW, Evans AS (2007) Sinusitis and its management. BMJ 334: 358-361.
- Benninger MS, Ferguson BJ, Hadley JA, Hamilos DL, Jacobs M, et al. (2003) Adult chronic rhinosinusitis: definitions, diagnosis, epidemiology, and pathophysiology. Otolaryngol Head Neck Surg 129: S1-32.
- Demoly P (2008) Safety of intranasal corticosteroids in acute rhinosinusitis. Am J Otolaryngol 29: 403-413.
- Derendorf H, Meltzer EO (2008) Molecular and clinical pharmacology of intranasal corticosteroids: clinical and therapeutic implications. Allergy 63: 1292-1300.
- Emanuel IA, Shah SB (2000) Chronic rhinosinusitis: allergy and sinus computed tomography relationships. Otolaryngol Head Neck Surg 123: 687-691.
- Fandino M, Macdonald KI, Lee J, Witterick IJ (2013) The use of postoperative topical corticosteroids in chronic rhinosinusitis with nasal polyps: a systematic review and meta-analysis. Am J Rhinol Allergy 27: e146-157.
- Hamilos DL (2000) Chronic sinusitis. J Allergy Clin Immunol 106: 213-227.
- Hartog B, van Benthem PP, Prins LC, Hordijk GJ (1997) Efficacy of sinus irrigation versus sinus irrigation followed by functional endoscopic sinus surgery. Ann Otol Rhinol Laryngol 106: 759-766.
- Hoffmans R, Schermer T, van Weel C, Fokkens W (2011) Management of rhinosinusitis in Dutch general practice. Prim Care Respir J 20: 64-70.
- Piromchai P, Kasemsiri P, Laohasiriwong S, Thanaviratananich S (2013) Chronic rhinosinusitis and emerging treatment options. Int J Gen Med 6: 453-464.
- Passali D, Bernstein JM, Passali FM, Damiani V, Passàli GC, et al. (2003) Treatment of recurrent chronic hyperplastic sinusitis with nasal polyposis. Arch Otolaryngol Head Neck Surg 129: 656-659.
- Ragab SM, Lund VJ, Scadding G (2004) Evaluation of the medical and surgical treatment of chronic rhinosinusitis: a prospective, randomised, controlled trial. Laryngoscope 114: 923-930.
- Kalish L, Snidvongs K, Sivasubramaniam R, Cope D, Harvey RJ (2012) Topical steroids for nasal polyps. Cochrane Database Syst Rev 12: CD006549.
- Johansson L, Holmberg K, Melen I, Stierna P, Bende M (2002) Sensitivity of a new grading system for studying nasal polyps with the potential to detect early changes in polyp size after treatment with a topical corticosteroid (budesonide). Acta Otolaryngol 122: 49-53.
- Kang IG, Yoon BK, Jung JH, Cha HE, Kim ST (2008) The effect of high-dose topical corticosteroid therapy on prevention of recurrent nasal polyps after revision endoscopic sinus surgery. Am J Rhinol 22: 497-501.
- Khalil HS, Nunez DA (2006) Functional endoscopic sinus surgery for chronic rhinosinusitis. Cochrane Database Syst Rev : CD004458.
- Marple BF, Stankiewicz JA, Baroody FM, Chow JM, Conley DB, et al. (2009) Diagnosis and management of chronic rhinosinusitis in adults. Postgrad Med 121: 121-139.
- Meltzer EO, Hamilos DL, Hadley JA, Lanza DC, Marple BF, et al. (2004) Rhinosinusitis: establishing definitions for clinical research and patient care. J Allergy Clin Immunol 114: 155-212.
- Rimmer J, Fokkens W, Chong LY, Hopkins C (2014) Surgical versus medical interventions for chronic rhinosinusitis with nasal polyps. Cochrane Database Syst Rev 12: CD006991.
- Rudmik L, Schlosser RJ, Smith TL, Soler ZM (2012) Impact of topical nasal steroid therapy on symptoms of nasal polyposis: a meta-analysis. Laryngoscope 122: 1431-1437.
- Slavin RG, Spector SL, Bernstein IL, Kaliner MA, Kennedy DW, et al. (2005) The diagnosis and management of sinusitis: a practice parameter update. J Allergy Clin Immunol 116: S13-47.
- Small CB, Hernandez J, Reyes A, Schenkel E, Damiano A, et al. (2005) Efficacy and safety of mometasone furoate nasal spray in nasal polyposis. J Allergy Clin Immunol 116: 1275-1281.
- Snidvongs K, Kalish L, Sacks R, Sivasubramaniam R, Cope D, et al. (2013) Sinus surgery and delivery method influence the effectiveness of topical corticosteroids for chronic rhinosinusitis: systematic review and meta-analysis. Am J Rhinol Allergy 27: 221-233.
- Stjarne P, Blomgren K, Cayé-Thomasen P, Salo S, Søderstrøm T (2006) The efficacy and safety of once-daily mometasone furoate nasal spray in nasal polyposis: a randomized, double-blind, placebo-controlled study. Acta Otolaryngol 126: 606-612.
- Suh JD, Kennedy DW (2011) Treatment options for chronic rhinosinusitis. Proc Am Thorac Soc 8: 132-140.
- Vento SI, Ertama LO, Hytönen ML, Wolff CH, Malmberg CH (2000) Nasal polyposis: clinical course during 20 years. Ann Allergy Asthma Immunol 85: 209-214.
- Witterick IJ, Kolenda J (2004) Surgical management of chronic rhinosinusitis. Immunol Allergy Clin North Am 24: 119-134.
- Xaubet A, Mullol J, López E, Roca-Ferrer J, Rozman M, et al. (1994) Comparison of the role of nasal polyp and normal nasal mucosal epithelial cells on in vitro eosinophil survival. Mediation by GM-CSF and inhibition by dexamethasone. Clin Exp Allergy 24: 307-317.
- Xaubet A, Mullol J, Roca-Ferrer J, Pujols L, Fuentes M, et al. (2001) Effect of budesonide and nedocromil sodium on IL-6 and IL-8 release from human nasal mucosa and polyp epithelial cells. Respir Med 95: 408-414.
Relevant Topics
- 3D Printing in Limb-Sparing Surgery
- Adamantinoma
- Aneurysmal Bone Cysts
- Chondrosarcoma
- Chordomas
- Cryosurgery
- Enchondroma
- Ewing’s Sarcoma
- Fibrous Dysplasia
- Giant Cell Tumor of Bone
- Immunotherapy for Osteosarcoma
- Liquid Biopsy in Orthopedic Oncology
- Malignant Osteoid
- Metastatic Bone Cancer
- Molecular Profiling of Bone Tumors
- Multilobular Tumour of Bone
- Orthopaedic Oncology
- Osteocartilaginous Exostosis
- Osteochondrodysplasia
- Osteoma
- Osteonecrosis
- Osteosarcoma
- Primary Bone Tumors
- Sarcoma
- Secondary Bone Tumours
- Targeted Therapy in Bone Sarcomas
- Tumours of Bone
Recommended Journals
Article Tools
Article Usage
- Total views: 16307
- [From(publication date):
June-2016 - Aug 19, 2025] - Breakdown by view type
- HTML page views : 15323
- PDF downloads : 984