Melioidosis Acquired by Traveler to Thailand: A Case Report
Received: 15-Jun-2018 / Accepted Date: 18-Jul-2018 / Published Date: 20-Jul-2018 DOI: 10.4172/2332-0877.1000372
Keywords: Melioidosis; Burkholderia pseudomallei; Traveller; Case report
Introduction
Melioidosis is a rare infection caused by Burkholderia pseudomallei (B. pseudomallei ), which with clinical manifestations ranging from skin abscess to overwhelming sepsis and death. B. pseudomallei is a gram-negative, saprophytic bacillus found in soil and water, and it is endemic in Southeast Asia, Northern Australia and other tropical regions [1-3]. B. pseudomallei is also a major cause of communityacquired septicaemia and pneumonia in adults in the Asia-pacific region, particularly in northeast Thailand [3,4].
In China, the first case of melioidosis was reported in 1990 [5]. Since then several sporadic and epidemic cases had been reported in several tropical provinces, including Hainan, Guangdong, and Guangxi [6,7]. However, the report of melioidosis in Chongqing, China is unknown, here we present a case of melioidosis with prominent tissue necrosis and urinary tract infection in a man in Chongqing.
Case Presentation
In May 2017, a 55-year-old man was admitted to hospital because of erosions and ulcers on right leg with purulent discharge and painful sensation for a one-month history. The erythematous skin changes initially occurred from the anterior tibia and extended up to left leg, which gradually enlarged to erosions and ulcers, and with urinary tract infection recently. No further complaints were elicited during a systematic enquiry, specifically no chills, night sweats, or weight loss. There was no history of trauma caused by soil or plants. The patient had received a diagnosis of type two diabetes mellitus and he took metformin (500 mg 2 times/d) before admission. He didn't smoke or drink alcohol. He was born in Chongqing, China, and recently had travelled to Thailand on April 2017. One month after arriving home, the patient noted swelling and pain in both legs, with a 15-day history of fever, productive cough with white sputum, then developed a sweating, frequent urination, urgency, dysuria.
On admission to the hospital, his temperature was 38.8°C, a heart rate of 109 beats per minute, a blood pressure level of 142/94 mmHg. Laboratory investigations revealed a leukocyte count of 12.3 × 109 cells/L with markedly elevated 91.0% neutrophils. The C-reactive protein level was greater than 240.4 mg/L. The liver functions stated significantly elevated levels of aspartate aminotransferase (4941.1 U/L, normal range: 15.0-40.0 U/L), alanine aminotransferase (3194.3 U/L, normal range: 9.0-50.0 U/L). The inflammatory markers showed mildly increased procalcitonin (0.93 ng/mL, normal range: 0-0.25 ng/ mL). In addition, the glycated haemoglobin A1c blood glucose test result was 11.2%, which is consistent with poorly controlled diabetes, and the results of an HIV test and malaria screen were both negative. Blood and urine were drawn for culturing, and a course of empirical treatment was begun. Subsequently, the aerobic bottles of two sets of blood cultures were positive for gram-negative bacilli, and the same result was also obtained in urine culture. After culturing for 24 h, yielded an oxidase-positive, gram-negative bacillus that grew rapidly on blood agar as a grey colony with a metallic sheen (Supplementary Figure 1). The organisms were initially identified as B. pseudomallei by VITEK-2 Compact GN card (bio number 0003451513500211). The antimicrobial drug therapy was changed to co-amoxiclav, and two days later, when the identification of the organisms was confirmed by 16S rDNA gene sequencing analysis, the patient’s treatment was switched to intravenous meropenem. During the 7-day treatment course, the patient was transferred to the Department of Infectious Disease, the First Affiliated Hospital of Chongqing Medical University for treatment. Subsequently, the patient’s urinary tract infection and fever resolved, and after 15 days of receiving meropenem, his general condition improved. Unfortunately, the patient didn't attend his scheduled outpatient appointment after discharge.
Since VITEK-2 Compact (GN Card) (bioMerieux, France) is prone to produce false positive or false negative results [7], 16S rDNA gene sequencing analysis is required to confirm the identification of B. pseudomallei . And then the isolates were sent to a biosafety level two plus Mycobacteriology Laboratory for further analysis. Moreover, PCR amplification, we used the two primers F (5’- GATCATGGCTCAGATTGAAC-3’) and R (5’- CTACGGTTACCTTGTTACGA-3’) that could specifically detect the B. pseudomallei isolates [8,9], produced the predicted 1397bp band for B. pseudomallei isolates (Supplementary Figure 2). The PCR product was sequenced by BGI (Beijing, China). The isolate showed a 99% similar to B. pseudomallei (GenBank accession nos. CP004379.1 and CP004380.1). Based on these data, we concluded that this infection case was caused by B. pseudomallei .
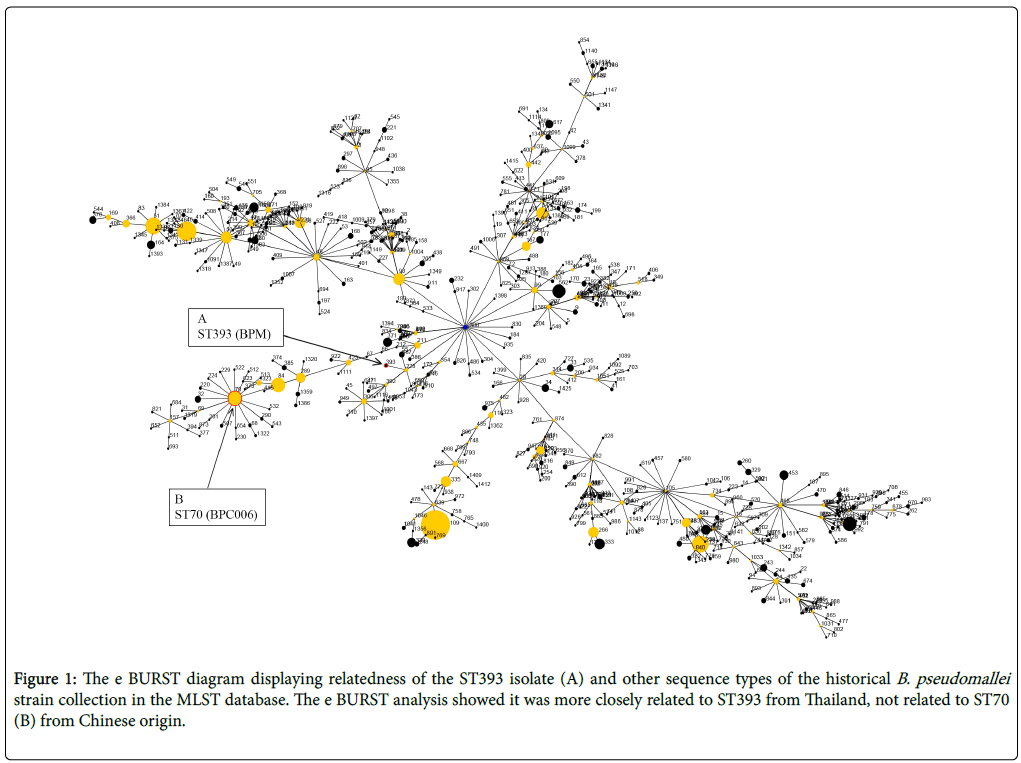
Melioidosis is rare in most parts of China, there were only 0-3 cases reported annually, and most of them occurred in travellers returning from disease-endemic regions [8,9]. In this case, we cannot be certain that whether this melioidosis acquired by the traveller to Thailand, the disease-endemic country in the Asia-pacific region. To our best knowledge, no documented cases of melioidosis acquired in Chongqing had been reported, so the multilocus sequence typing (MLST) was used to study the molecular epidemiology of B. pseudomallei , and the MLST was performed by using PCR amplification conditions and primers as previously described [10], generated an allelic profile (Supplementary Figure 2) that was compared with those of strains of B. pseudomallei , B. thailandensis , and Mallei stored in the database (http://bpseudomallei.mlst.net). The result showed that the novel sequence type (ST) was identified as ST393 (BPM), which is a strain of Thailand origin. However, it had previously been reported that the B. pseudomallei ST70 (BPC006) was the predominantly isolate of Chinese origin, which was considered as China's standard strain [11]. And an evolutionary analysis of ST393 with burst analysis software (http://eburst.mlst.net) suggested that it was not related to ST70 (BPC006) (Figure 1).
Figure 1: The e BURST diagram displaying relatedness of the ST393 isolate (A) and other sequence types of the historical B. pseudomallei strain collection in the MLST database. The e BURST analysis showed it was more closely related to ST393 from Thailand, not related to ST70 (B) from Chinese origin.
Discussion
Clinical diagnosis of melioidosis in non-endemic regions is challenging because signs of the disease are non-specific and similar to those of more common diseases, such as tuberculosis [12]. In addition, laboratory diagnosis is also challenging. In this case, B. pseudomallei grew readily in culture. However, the VITEK-2 Compact didn't provide definitive species identification; we recheck the identification of B. pseudomallei by 16S rDNA gene sequencing analysis. What's more, cultures and analysis of B. pseudomallei isolates were both manipulated in biosafety level two plus laboratory.
Melioidosis is endemic to the Pacific region and Southeast Asia [13,14]. Most cases reported in other non-endemic regions are imported [15]. In Thailand, where the patient had travelled, the number of cases increased from 11.5/100,000 inhabitants in 1997 to 21.3/100,000 in 2006 [16]. The main routes of transmission are flooding, a risk factor for melioidosis, as for this case-patient.
Melioidosis may affect healthy persons, but melioidosis infection is closely related to underlying diseases, especially related to diabetes [12]. Diabetic patients are prone to concurrent infections due to malnutrition, hypoalbuminemia, and blood glucose fluctuations that can easily damage defence mechanisms and reduce immune function. In a case-control study in northern Australia, diabetes was associated with an adjusted odds ratio of 13.1 [17]. With the prevalence of diabetes projected to increase constantly in many developing countries and the increased international travel [18], the global reach of melioidosis may expand. To sum up, the immunocompromised travellers might be first sentinels for ongoing endemic diseases. When the travellers return with uncommon diseases, physicians should check for underlying diseases such as melioidosis.
Conclusions
Given the frequency of travel between China and other countries, the existence of imported cases from endemic countries until now is striking, and physicians should be aware of its varied manifestations. In particular, melioidosis should be considered when diabetic patients returning from endemic countries, even without risk factors predisposing to severe disease. And this study finally indicates that this melioidosis acquired by the traveller to Thailand.
References
- Peto L, Nadjm B, Horby P, Ngan TT, van Doorn R, et al. (2014) The bacterial aetiology of adult community-acquired pneumonia in Asia: a systematic review. Trans R Soc Trop Med Hyg 108: 326-337.
- Kingsley PV, Leader M, Nagodawithana NS, Tipre M, Sathiakumar N (2016) Melioidosis in Malaysia: A Review of Case Reports. PLoS Negl Trop Dis 10: e0005182.
- Perumal SR, Stiles BG, Sethi G, LHK L (2017) Melioidosis: Clinical impact and public health threat in the tropics. PLoS Negl Trop Dis 11: e0004738.
- Suntornsut P, Wongsuwan N, Malasit M, Kitphati R, Michie S, et al. (2016) Recommended Interventions to Prevent Melioidosis in Northeast Thailand: A Focus Group Study Using the Behaviour Change Wheel. PLoS Negl Trop Dis 10: e0004823.
- Fang Y, Chen H, Zhu X, Mao X (2016) Fatal Melioidosis in a Newborn from Hainan, China. Am J Trop Med Hyg 95: 444-446.
- Tan G, Li J, Chen L, Chen X, Zhang S (2015) Prostatic melioidosis rarely reported in China: two cases report and literatures review. Int J Clin Exp Med 8: 21830-21832.
- Kong Z, Fang Y, Zhang M, Hong J, Tan Z, et al. (2016) Melioidosis acquired by a traveler from Papua New Guinea. Travel Med Infect Dis 14: 267-270.
- Chang K, Luo J, Xu H, Li M, Zhang F (2017) Human Infection with Burkholderia thailandensis, China, 2013. Emerg Infect Dis 23: 1416-1418.
- Ngamdee W, Tandhavanant S, Wikraiphat C, Reamtong O, Wuthiekanun V, et al. (2015) Competition between Burkholderia pseudomallei and B. thailandensis. BMC Microbiol 15: 56.
- Fang Y, Huang Y, Li Q, Chen H, Yao Z, et al. (2012) First genome sequence of a Burkholderia pseudomallei Isolate in China, strain BPC006, obtained from a melioidosis patient in Hainan. J Bacteriol 194: 6604-6605.
- Salam AP, Khan N, Malnick H, Kenna DT, Dance DA, et al. (2011) Melioidosis acquired by traveler to Nigeria. Emerg Infect Dis 17: 1296-1298.
- Cheng AC, Currie BJ (2005) Melioidosis: epidemiology, pathophysiology, and management. Clin Microbiol Rev 18: 383-416.
- Wiersinga WJ, Virk HS, Torres AG, Currie BJ, Peacock SJ, et al. (2018) Melioidosis. Nat Rev Dis Primers. 4: 17108.
- Schultze D, Müller B, Bruderer T, Dollenmaier G, Riehm JM, et al. (2012) A traveller presenting with severe melioidosis complicated by a pericardial effusion: a case report. BMC Infect Dis 12: 242.
- Teparrukkul P, Nilsakul J, Dunachie S, Limmathurotsakul D (2017) Clinical Epidemiology of Septic Arthritis Caused by Burkholderia pseudomallei and Other Bacterial Pathogens in Northeast Thailand. Am J Trop Med Hyg 97: 1695-1701.
- Currie BJ, Jacups SP, Cheng AC, Fisher DA, Anstey NM, et al. (2004) Melioidosis epidemiology and risk factors from a prospective whole-population study in northern Australia. Trop Med Int Health 9: 1167-1174.
- Hall V, Thomsen RW, Henriksen O, Lohse N (2011) Diabetes in Sub Saharan Africa 1999-2011: epidemiology and public health implications. A systematic review. BMC Public Health 11: 564.
Citation: Li J, Hu W, Rao C, Zhang F, Li M, et al. (2018) Melioidosis Acquired by Traveler to Thailand: A Case Report. J Infect Dis Ther 6: 72. DOI: 10.4172/2332-0877.1000372
Copyright: © 2018 Li J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 5603
- [From(publication date): 0-2018 - Dec 23, 2025]
- Breakdown by view type
- HTML page views: 4593
- PDF downloads: 1010