Moving Toward Consensus Terminology in Ophthalmic Practice
Received: 22-Dec-2015 / Accepted Date: 19-Feb-2016 / Published Date: 25-Feb-2016 DOI: 10.4172/2476-2075.1000105
Abstract
The dual purposes of this review are to point out inherent problems in communication due to lack of consensus terminology, and to review preferred ophthalmic terms or to suggest others in order to improve eye care. With advances in informatics and the ongoing evolution of modern health care delivery, a common lexicon will also improve basic research and clinical outcomes. It is hoped that this paper will raise awareness of these issues and open profession-wide discussion with the ultimate goal of improving medical communication for ophthalmic patient care.
Keywords: Communication; Ophthalmic; Standardization; Terminology
6155Introduction
The magnitude of effective communication between medical team members and patients in order to achieve excellence in patientcentered healthcare cannot be understated [1,2]. Improper exchange of clinical findings can lead to medical errors [3], and the profound significance of successful communication has been described as “the most important medical instrument” in the lay press [4]. As for all health care providers, the optometrist-patient relationship is also dependent upon effective communication [5], as is the link between referring optometrists and consulting ophthalmologists. Indiscriminate use of medical terms undermines these aims in several, key ways.
Informatics
Interdisciplinary use of merged biomedical and technological data is growing in usage for the delivery of modern health care. Ophthalmic informatics has flourished to include applications of telemedicine, long-distance consultation, and electronic health records/coding among other applications. The exacting nature of data transmission requires the use of specific terminology for accuracy. An important function of the World Health Organization’s International Classification of Diseases (ICD) is to monitor overall trends of health for clinical and administrative purposes on both local and global scales. This is impossible without a common frame of reference—in this case consistency in human disease diagnosis.
Research
Non-standardized terminology limits the ability of researchers to study specific disease entities, on epidemiological and individual levels. If investigators include or exclude the wrong patients because of variations in diagnosis between examiners, then results become nongeneralizable, and advances in medical care are hindered. As an example, it is instructive to note that three large randomized controlled trials—the Advanced Glaucoma Intervention Study (AGIS), the Collaborative Initial Glaucoma Treatment Study (CIGTS), and the Early Manifest Glaucoma Trial (EMGT)—differed in their criteria to identify progression of visual field loss [6]. Such non-consensus complicates prognostic projections and interdisciplinary care.
Prognosis
Effective communication via patient education regarding ophthalmic disease obviously depends on accurate diagnosis, which in turn, depends on consensus terminology. Inconsistent working definitions among health care providers can lead to different diagnoses and varying prognoses—and, possibly, to unpredictable degrees of patient anxiety.
Accurate prognosis is especially important to patients who demonstrate advancing, primary open-angle glaucoma (what is the rate of progression?), who have developed a retinal detachment (is the macula on or off?), who wish to learn if the fellow eye will develop vision loss (what is the risk of bilateral disease?), or for myriad other ophthalmic findings.
Students without clinical experience are often overwhelmed by the multitude of medical definitions; however, providers with years of clinical experience also realize the limitations of definitions for some medical diagnoses. Careful consideration of the vagaries of “macular degeneration” is a prime example [7].
Referrals
Indiscriminate use of terminology inhibits effective communication between providers involved in the referral process [8]. This is especially important in ophthalmic practice, where optometrists frequently refer patients to ophthalmologists for surgical care. Inappropriate referrals squander provider time and waste financial resources.
All providers have surely been faced with a patient who presents for a second opinion or who provides a definite history of an ophthalmic disease, only to discover at the end of the examination that the patient has nothing of the kind. Was the first doctor charting on the wrong patient—or worse yet, incompetent? Not necessarily. More than likely the two providers merely define clinical entities in different ways.Consider the variability of what diverse providers across many disciplines indicate by a diagnosis of “Pink Eye.”
For all of these reasons, it is inherent that eye doctors be “on the same page” when communicating ophthalmic findings to patients, staff members, and other providers.
Literature Review
The available medical literature is quite limited on the topic of standardization of terminology in ophthalmic care. The most robust attempts involve age-related macular degeneration [7] and uveitis [9], although the extent to which ophthalmic providers follow these recommendations is—as yet—unstudied.
While a comprehensive appraisal of ophthalmic terminology is beyond the scope of this paper, reminders of more common medical and ophthalmic terms can be presented as a segue to more in-depth discussion and evaluation. Basic definitions of all of the following terms can be readily reviewed at various on-line medical dictionaries such as: The Free Dictionary (http://medicaldictionary. thefreedictionary.com), Medline Plus (http:// www.nlm.nih.gov/medlineplus/mplusdictionary.html), and Dictionary.com (http://dictionary.reference.com/medical).
Terms For Consideration
While the terms and phrases that follow are by no means intended to form an inclusive list, variable usage with respect to the following words is commonplace in clinical practice and these inconsistencies are likely well-known to most ophthalmic providers.
General medical terms
Etiology (literally “giving a reason for”) is synonymous with causation (an event that produces an effect), but neither of these terms equates to an association (non-causal relationship). Unilateral optic disk edema is associated with many conditions, but its direct causation remains obscure.
Adverse drug reaction is an injury caused by taking a medication; whereas a side effect is an outcome secondary to the one intended, and could be advantageous or detrimental. Sildenafil citrate was initially studied as therapy for hypertension and angina pectoris (ineffective for both), but offers a classic example of a secondary side effect becoming the primary indication for use.
As is well-known, “-itis” is a suffix denoting inflammation; however, this designation is misleading for conditions such as retinitis pigmentosa [10] (gradual, non-acute retinal disease), asteroid hyalitis [11] (the unchanging presence of refractile, vitreal bodies), and optic neuritis [12] (typically applied to a
, not an inflammatory, process). “Optic neuritis” is probably best referred to descriptively as “disk edema,” and “asteroid hyalosis” now seems to be the preferred term for the common vitreous finding; however, it seems unlikely that the long-ingrained misnomer “retinitis pigmentosa” will soon disappear from ophthalmic or lay usage.
Cranial nerve palsy, paralysis, paresis are variably defined and often used synonymously—creating great ambiguity in clinic and for research [13]. The confusion surrounding these neurological terms arises for eye doctors as cranial nerve dysfunction can be partial or complete, temporary or permanent, localized or generalized, depending on the degree of functio laesa at a given time of clinical examination (sometimes a prognostic dilemma). Currently there is no consensus regarding these terms—perhaps a challenge for neurologists to address in the years to come.
Non-specific medical classification schemes
Grading scales are frequently used in medicine to classify stages of health and disease, and the oldest of these—the American Society of Anesthesiologists Physical Status (ASA PS) classification system from 1963—remains a valuable tool for surgical patients to this day [14]. Unfortunately, ophthalmic classification schemes have not been validated over time or to this degree. Eye providers are familiar with subjective determinations of “mild/moderate/severe” yet these judgments remain arbitrary and may vary between clinicians. “Moderate” phacomorphic glaucoma as described one provider may be interpreted as “mild” by another. “Visually-significant” cataract proves an equally-nebulous descriptor.
1+/2+/3+/4+ determinations are attempts to standardize physiological evaluation, yet—with the exception of uveitis9—exact correlations of various ophthalmic findings to numeric grading scales have not been established. Creation of continuous scales can also lead to improved research methodology.
Like clinically-significant macular edema (CSME) in diabetic eye disease, perhaps ophthalmic researchers must define all ocular disease entities in “clinically-significant” terms in order to establish a basis for more exact terminology and to replace the widespread variable usage of the schemes described above. Creation of treatment criteria for CSME and proliferative diabetic retinopathy helped remove some subjective determinations regarding photocoagulation therapy; however, this concept has not yet extended to other retinal treatments or other ophthalmic sub-specialties.
Ophthalmic terms
Which leads to a discussion of specific ophthalmic diagnoses: it may be conjectured that the most reliable and repeatable classification systems rely upon standardized definitions of diseases processes. In the absence of validated grading schemes to further classify certain diseases, there are several ophthalmic entities worthy of review or in need of consensus definition. These are generally presented in an anterior to posterior fashion with respect to ocular anatomy.
RAPD=Relative Afferent Pupillary Defect. Omission of the “R” (in favor of “APD”) neglects the critical distinction that makes the acronym accurate. The phrase only makes anatomical sense if comparing direct to consensual responses in the same eye – i.e. the direct response is judged relative to the consensual response to light stimulus (assuming the patient has two contralateral, afferent optic nerve pathways). Long-gone is the non-descriptive phrase, Marcus- Gunn pupil.
Dilation of conjunctival vasculature can be referred to as “injection” (older meaning for “congestion”) or “hyperemia” (also indicating increased blood flow to a part of the body). However, insofar as most medical disciplines use the term “injection” in reference to the process of introducing a liquid into the body—therefore indicating the use of needles—perhaps “hyperemia” is less likely to be misunderstood.
“Pink eye” is a descriptive, but non-specific expression for conjunctival hyperemia. Unfortunately, this phrase typically implies contagious “infection” to the general population. Ophthalmic providers understand that conjunctivitis is a generic, inflammation (i.e.an “-itis”) caused by a variety of antecedents: allergic, chemical, environmental, infectious, immune-mediated, mechanical, thermal or toxic. While this term is still ubiquitous on well-known websites (Centers for Disease Control, National Eye Institute, Mayo Clinic,among others [15-17], the confusion and anxiety its use creates for patients suggests that it is perhaps time to abandon it in favor of more etiological descriptors for conjunctivitis.
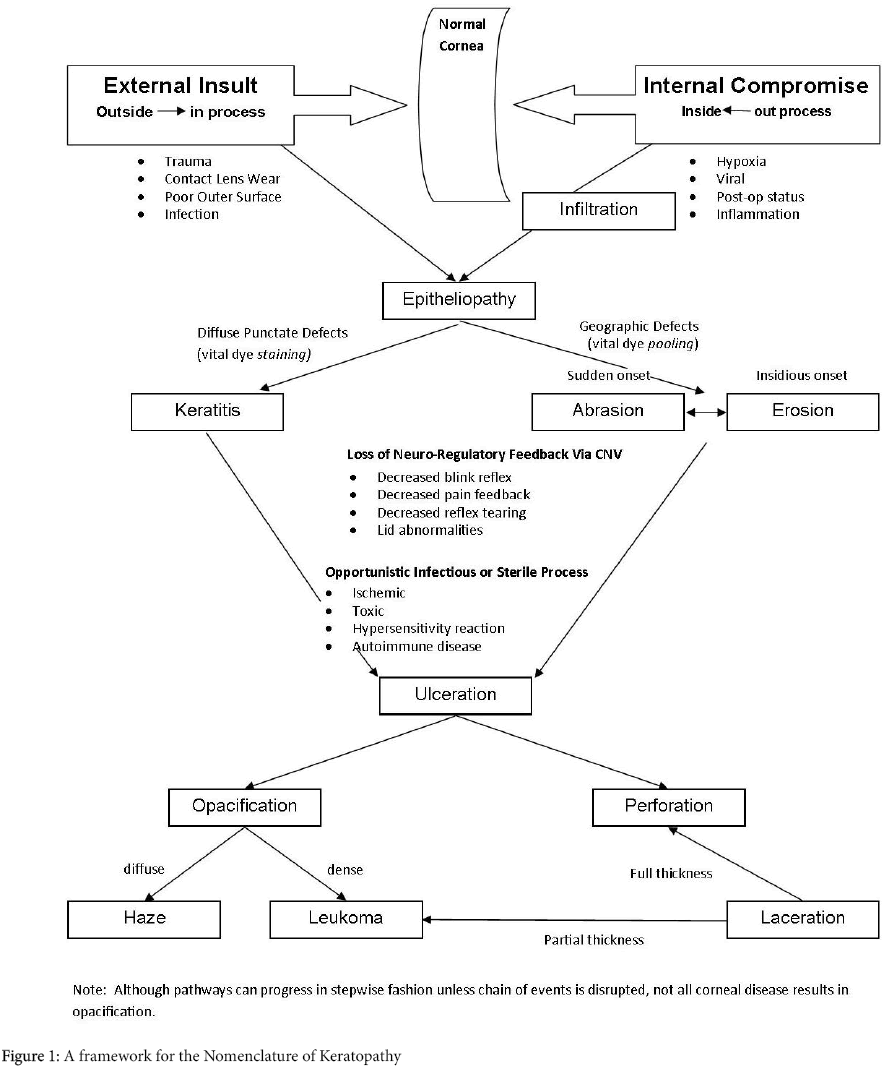
Although less important to patients, haphazard use of the terminology of keratopathy is a well-known problem for eye doctors familiar with the literature of the cornea. The diagnostic dilemmas involving keratitis, ulcer, infiltrate, abrasion, erosion and the like not only bedevil patient care, but are quite common in publications of corneal research, hindering clear communication of findings for providers. Perhaps a consideration of this vocabulary is best appreciated within a context of disease progression. Because of so much ambiguity regarding terminology for the cornea, a new framework for keratopathy is presented in Figure 1.
Infiltration: Typically a gradual process of abnormal accumulation of a substance in body cells, tissues or organs. In the cornea, primarily white blood cell accumulation—can be asymptomatic in early stages.
Keratitis: Inflammation of the cornea. Essentially indicated by vital dye staining, although some presentations may be asymptomatic.
Abrasion: A sudden wound caused by rubbing or scraping of (i.e. abrading) the surface. Often self-limited and resolves completely without residua.
Erosion: The gradual wearing away of a surface. In the cornea, caused by a loose attachment of the epithelium to its underlying basement membrane; may eventually heal without long-term sequelae.
Ulceration: The process of lesion formation resulting from a break in the surface of a tissue with accompanying disintegration of underlying material and eventual necrosis of sub-surface tissue (i.e. loss of tissue). Historically described as always resulting in cicatrix formation [18-20], in the cornea, this indicates loss of tissue to at least stromal depth. Given the problems describing keratopathy, likely need to label in accordance with this older definition.
Opacification: The process of making something non-transparent or impenetrable to light.
Haze: Diffuse aggregation of particles. In the cornea, a type of opacification that results in a clouding of vision that makes objects appear indistinct.
Leukoma: A dense, white opacity of the cornea.
Laceration: A rough, jagged wound produced by tearing a body tissue. Can leave an opacity if extending into the corneal stroma.
Perforation: The process of passing through a body or structure. With respect to the cornea, a full-thickness event, as indicated by a positive Seidel Sign.
It may be useful to consider that a bacterial keratitis can lead to ulceration, but not every corneal disease involving bacteria forms an “ulcer” with necrosis and loss of tissue to the stromal level (and therefore;“opacity”). In fact a true corneal ulcer would be unexpected early in a keratopathy.
It must be conceded that “Recurrent Corneal Erosion Syndrome” might be a misnomer for two reasons: a “syndrome” involves a constellation of findings, and the process is probably more accurately termed a “recurrent corneal abrasion” due to non-adhesion of epithelial cells to the underlying basement membrane—an outside-in mechanism versus an inside-out process.
Dendritic keratitis may be the most accurate descriptor for the branching corneal epithelium staining pattern commonly observed in Herpes Simplex Virus (HSV) keratopathy, as resolution of the dendritiform lesion does not always result in corneal opacification, as implied by the term “ulcer.”
Finer distinctions of these and other corneal processes should greatly help facilitate reporting of keratopathy.
At this time, glaucoma is widely recognized as an optic neuropathy with associated visual field loss [21]. Removal of intraocular pressure (IOP) from the definition of primary open-angle glaucoma has also retired the misleading terms “normal tension glaucoma” and “ocular hypertension” (within the context of OAG) [22], and as such tonometry can no longer be referred to as a “glaucoma test.”
Although use of the term “glaucoma” is too ambiguous by itself to be clinically useful, further classification of primary and secondary forms of open- and closed-angle optic neuropathies has yet to reach consensus levels. By way of example, the European Glaucoma Society [23] and the World Glaucoma Association [24] do not yet agree on taxonomies for childhood glaucoma. Terminology regarding adult, secondary forms of glaucoma remains equally ambiguous. Use of the designator “glaucoma suspect” remains vague and ill-defined at this time. It is hoped that variations in current nomenclatures will be resolved to the benefit of patients and clinicians alike.
Perhaps no ophthalmic term is more concerning to patients than cataract. Even in this day of extremely-successful cataract surgery, this diagnosis continues to have a strong emotional effect on patients. Nearly all adults have heard of “cataract” and patients seem to dread it. Furthermore, after this word has been dropped—even in passing— there is no amount of explanation to remove it from a patient’s mind.
Within this context, perhaps it is useful to remember that a cataract is an “an opacification [see definition above] of the lens that leads to measurably decreased visual acuity and/or some functional disability as perceived by the patient” [25]. This is not equivalent to normal, senescent yellowing (nuclear sclerosis) of the lens – thus “NS” is not the same as “NSC.” Premature use of the word cataract creates unneeded anxiety on the part of the patients and can result in extra visits and consultation time on the part of the provider.
Posterior capsule opacification (PCO) is another ophthalmic misnomer in that the posterior capsule is not directly involved in the opacification process, yet the phrase is widely used in clinical care and unlikely to be rebranded. Posterior capsular fibrosis is one of two types of PCO—the other being Elschnig’s pearls [26].
Swelling of the optic disk is, in fact, most literally described as “papilledema.” Unfortunately, this term has been characterized differently by neurology to indicate optic disk edema that is caused by increased intracranial pressure. It is this usage that has caused confusion for eye doctors. Except for atypical cases, increased intracranial pressure typically results in bilateral disk edema due to the intracranial anatomy. Associated terms like papillopathy, papillitis, and optic neuritis further confuse this topic—perhaps “disk edema” with a specification of unilateral or bilateral is the most precise label for this finding, in that this phrase does not imply etiology or a pathological mechanism.
The definition of amaurosis fugax largely depends on the specialty of the provider making the diagnosis. A non-specific description of symptoms (and not pathognomonic for any specific clinical finding), the phrase “amaurosis fugax” best describes a spectrum of fleeting visual obscurations involving light and dark phenomena. This author has suggested an alternative nomenclature to encompass this spectrum of events [27].
Uveitis terminology has been classified by the Standardization of Uveitis Nomenclature (SUN) Working Group, with anatomic, chronicity, grading of cells and flare, and measures of activity clearly delineated [9]. It is hoped that this important work is embraced by eye doctors in the years to come.
Light sensitivity and photophobia are poorly defined and variably used in the medical literature. Inconsistencies extend into ophthalmic practice (as well as in the lay press and advertising) and documentation among providers varies widely. *Light sensitivity of the eyes remains ill-defined, but is essentially subjective discomfort caused by exposure to light. This is in contrast to photophobia, which may be better considered as "a pathological intolerance of light,” as might be encountered in ocular albinism or with chronic anterior uveitis. An abnormal process, "pathological" implies a disease process, an objective finding. *Light sensitivity of the eyes should not be confused with the term photosensitivity, which is usually defined as an abnormal dermatological reaction to ultraviolet light (usually following exposure to certain drugs or to other sensitizing chemicals), resulting in accelerated burning and blistering of the skin.
Light sensitivity is quite common, largely relieved by tinted spectacle lenses, and accounts for the vast majority of light-related complaints offered by ophthalmic patients. By contrast, photophobia is (fortunately) rare, requires medical treatment of the underlying cause (if possible), and may be present under otherwise “normal” light exposure. These distinctions are especially important with regard to legal determinations regarding vehicle window tinting, disability claims, or other medicolegal cases.
The terminology for diabetes classification changed in 2003, favoring etiologic (“type 1” and “type 2”) rather than pharmacologic distinctions [28]. Gone are the Type I/Type II, IDDM/NIDDM, dietcontrolled and insulin-dependent designators. Dichotomous grading of diabetic retinopathy now favors “non-proliferative” versus “proliferative” distinctions [29] (“background diabetic retinopathy” has fallen out of vogue); yet mild/moderate/severe modifiers are still variably used in the context of non-proliferative diabetic retinopathy.
Clinically-Significant Macular Edema (CSME) is a specific type of Diabetic Macular Edema (DME), and was defined in the Early- Treatment Diabetic Retinopathy Study to set criteria for focal laser photocoagulation treatment. However, in more recent years, the marketing of intravitreal injections has resulted in less use of CSME in favor of the more generic DME. It is hoped that retinologists will once again define DME with respect to treatment algorithms in order to help facilitate the ophthalmic referral process and ongoing patient care, although this does not appear to be under discussion at the present time.
Choroidal Neovascularization (CNV) is the final common pathway by which anatomical breaks in Bruch’s Membrane typically result in chorioretinal disease, typicallly with profound visual consequence. Choroidal Neovascular Membrane (CNVM), Subretinal Neovascularization (SRNV), and Subretinal Neovascular Membrane (SRNVM) are terms analogous to CNV and inconsistently used in the ophthalmic literature. A simple search of PubMed citations by keywords reveals that CNV is the most commonly used of these four terms, and thus, perhaps the most widely recognized.
A similar search of PubMed citations reveals epiretinal membrane (ERM) to be the most frequently used term to describe fibrocellular proliferation along the internal limiting membrane of the retina. Thus, “macular pucker” and other, related terms appear to have fallen out of favor, with ERM currently the favored term—and acronym—for this common retinal finding.
“Macular Degeneration” is generically used for a litany of retinal conditions affecting the macula in a wide age-range of patients; however, this should come as no surprise as there are no widelyaccepted, germane definitions [7]. For maculopathy involving drusen formation in persons older than 55 years of age, age-related macular degeneration is now the consensus term with AMD as the preferred acronym (as opposed to ARM or ARMD). Additionally, the terms “wet” and “dry” AMD are no longer recommended due to the lack of specificity across the continuum of AMD findings. Early or intermediate AMD, neovascular AMD and GA are now suggested to describe age-related macular changes, that is, for persons older than 55 years of age [7]. Thus, a diagnosis of AMD in younger patients is inconsistent with these recommendations and should be reconsidered.
A retinal break is a full-thickness retinal defect and includes holes, tears and dialyses.[30] A break can lead to a detachment (separation of neurosensory retina from underlying retinal pigment epithelium—the de facto “basement membrane” for the neurosensory retina), but not vice versa. Thus a written description for the peripheral retina stating “no holes, breaks or tears” is doubly redundant.
Misnomers for ophthalmic medications
Pharmaceutical agents that lower IOP are frequently referred to as “anti-glaucoma” agents; however, this misnomer is a holdover from the days when IOP was the sole diagnostic criterion for glaucoma. Currently, there is no proven treatment for the characteristic optic neuropathy that is recognized as the final common pathway for glaucoma, and clinicians do not “treat” glaucoma [31]. Rather ophthalmic providers manipulate the only known modifiable risk factor for glaucoma, IOP. It is, therefore, most accurate to describe this therapy as “ocular hypotensive.”
The term “antibiotic” is frequently used interchangeably with antibacterial, but it is worth remembering that a medication that is “against life” (literal definition) also includes the pharmaceutical classifications of antifungal, antiparasitie, antiprotozoan and antiviral agents. Using the more specific terms can prevent misunderstandings with providers or patients—especially in veritable or recalcitrant cases of ocular infection.
Conclusion
The information presented in this paper is intended to bring awareness to the need for consistent use of standardized ophthalmic terminology. The use of uniform nomenclature for ophthalmic diseases will further medical research, facilitate the use of informatics, provide more accurate medical prognosis, and improve communication between providers and their patients. A common frame of reference terminology will improve clinical care, which is the ultimate goal of all health care providers.
Disclosures
The views expressed in this article are those of the author and do not necessarily represent the positions of the Department of Veterans Affairs.
The author reports no proprietary or commercial conflicts of interest for any product mentioned or concept discussed in this article.
Acknowledgement
The author would like to thank Antonia Varner for the figure design.
References
- Scotten M, Manos EL, Malicoat A, Paolo AM (2015) Minding the gap: Interprofessional communication during inpatient and post discharge chasm care.Patient EducCouns 98: 895-900.
- Luetsch K, Rowett D (2015) Interprofessional communication training:benefits to practicing pharmacists.Int J Clin Pharm 37: 857-864.
- O’Daniel M, Rosenstein AH (2008) Professional Communication and Team Collaboration.Patient Safety and Quality:An Evidence-Based Handbook for Nurses.
- Chase D (2012) Communication is the Most Important Medical Instrument.Tech Crunch.
- Brandenburg R, Pesudovs K (2014) Teaching communication skills:an Australian optometry program’s new course. Optometric Education 40:19-27.
- Heijl A, Bengtsson B, Chauhan BC, Lieberman MF, Cunliffe I, et al. (2008) A comparison of visual field progression criteria of 3 major glaucoma trials in Early Manifest Glaucoma Trial patients.Ophthalmology 115:1557–1565.
- Ferris FL 3rd, Wilkinson CP, Bird A,Chakravarthy U,Chew E, et al. (2013) Clinical classification of age-related macular degeneration. Ophthalmology 120:844-851.
- Low vision referrals are too low, study says.Review of Optometry.[Cited on December 15, 2014.]Available from:http:// www.reviewofoptometry.com/content/d/news_review/i/3117/c/52140/.
- Jabs DA, Nussenblatt RB, Rosenbaum JT (2005)Standardization of uveitis nomenclature for reporting clinical data. Results of the first international workshop.Am J Ophthalmol140:509-516.
- Dunbar Roy AB (1895)Retinitis pigmentosa:Report of case.Annals of Ophthalmology and Otology 4:24-28.
- March W, Shoch D, O'Grady R (1974) Composition of asteroid bodies. See comment in PubMed Commons below Invest Ophthalmol 13: 701-705.
- Skorin Jr L (2008)Chapter 22:Neuro-ophthalmic disorders.In:Clinical Ocular Pharmacology.Barlett JD, Jaanus SD, eds, 5th ed.Butterworth-Heinemann:St Louis.
- Linder TE, Abdelkafy W, Cavero-Vanek S (2010) The management of peripheral facial nerve palsy: "paresis" versus "paralysis" and sources of ambiguity in study designs. Otol Neurotol 31: 319-327.
- Hackett NJ, De Oliveira GS, Jain UK, Kim JY (2015) ASA class is a reliable independent predictor of medical complications and mortality following surgery. Int J Surg 18: 184-190.
- Centers for Disease Control.Definition of pink eye.[Cited on May 5, 2015.]Available from:http://www.cdc.gov/features/conjunctivitis/.
- National Eye Institute.Facts about the cornea and corneal disease.[Cited on May 2013.]Available from:https://nei.nih.gov/health/cornealdisease.
- Mayo Clinic.Definition of pink eye.[Cited on July 16, 2015.]Available from:http://www.mayoclinic.org/diseases-conditions/pink-eye/basics/definition/con-20022732.
- Herzig AJ (1908)A short resume of the common conjunctival and corneal diseases.NY Med J.87:1193-2000.
- Verhoff FH (1917) The treatment of hypopyon keratitis.JAMA 68:1964-1969.
- Schoch LE (1918) Minor eye injuries and the workmen’s compensation law.Pennsylvania Med J 21:278-281.
- Quigley HA (2005) New paradigms in the mechanisms and management of glaucoma. Eye (Lond) 19: 1241-1248.
- Sommer A (2011) Ocular hypertension and normal-tension glaucoma: time for banishment and burial. Arch Ophthalmol 129: 785-787.
- European Glaucoma Society (2014)Terminology and Guidelines for Glaucoma.
- World Glaucoma Association (2013)Childhood Glaucoma:Consensus Series 9.Amsterdam:Kugler Publications.
- American Optometric Association Consensus Panel on Care of the Adult Patient with Cataract.Optometric Clinical Practice Guideline Care of the Adult Patient with Cataract.
- Apple DJ, Solomon KD, Tetz MR, Assia EI, Holland EY, et al. (1992) Posterior capsule opacification. SurvOphthalmol 37: 73-116.
- Varner P (2015)Redefining amaurosis fugax.African Vision and Eye Health 74: 9
- Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (2003) Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 26 Suppl 1: S5-20.
- Vander JF (2012) Chapter 3:Diabetic Retinopathy.In:Color Atlas and Synopsis of Clinical Ophthalmology, 2nd ed.Lippincott Williams & Wilkins.
- Kaiser PK, Friedman NJ, Pineda R II (2014)Chapter 10:Retina and Choroid.In:The Massachusetts Eye and Ear Infirmary Illustrated Manual of Ophthalmology, 4th ed.
- Ritch R (1994) Exfoliation syndrome-the most common identifiable cause of open-angle glaucoma. J Glaucoma 3: 176-177.
Citation: Varner P (2016) Moving Toward Consensus Terminology in Ophthalmic Practice. Optom open access 1: 105. DOI: 10.4172/2476-2075.1000105
Copyright: © 2016 Varner P, This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use,distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 12047
- [From(publication date): 3-2016 - Aug 16, 2025]
- Breakdown by view type
- HTML page views: 11098
- PDF downloads: 949