Research Article Open Access
Novel Dual Magnetic Drug Targeting and Hyperthermia Therapy in Hepatocellular Carcinoma with Thermosensitive Polymer-Coated Nanoparticles
Chang PEJ1,6*, Purushotham S2, Rumpel H3, Kee IHC4, Ng RTH4, Chow PKH4-6, Ramanujan RV2 and Tan CK1,6
1Department of Gastroenterology and Hepatology, Singapore General Hospital, Singapore
2School of Materials Science and Engineering, Nanyang Technological University, Singapore
3Department of Diagnostic Radiology, Singapore General Hospital, Singapore
4Department of Experimental Surgery, Singapore General Hospital, Singapore
5Department of General Surgery, Singapore General Hospital, Singapore
6Duke-NUS Graduate Medical School, Singapore
- Corresponding Author:
- Dr Jason Chang Pik Eu
Department of Gastroenterology and Hepatology
Singapore General Hospital, Outram Road, Singapore 169608
Tel: (+65) 62223322
Fax: (+65) 62273623
E-mail: jason.chang@sgh.com.sg
Received Date: May 26, 2014; Accepted Date: July 28, 2014; Published Date: August 05, 2014
Citation: Chang PEJ, Purushotham S, Rumpel H, Kee IHC, Ng RTH, et al. (2014) Novel Dual Magnetic Drug Targeting and Hyperthermia Therapy in Hepatocellular Carcinoma with Thermosensitive Polymer-Coated Nanoparticles. J Gastroint Dig Syst 4:198. doi:10.4172/2161-069X.1000198
Copyright: © 2014 Chang PEJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Background: Magnetic drug targeting (MDT) uses magnetic fields to localize magnetic nanoparticles (MNP) to tumor. Once localized, applying an alternating magnetic field (AMF) to the MNPs generates heat energy. Poly-N-isopropylacrylamide (PNIPA) is a thermosensitive polymer that contracts when heated, releasing any drugs which are bound to it. Utilizing these properties, MNP coated with PNIPA polymer on to which doxorubicin is loaded can be localized to hepatocellular carcinoma (HCC) with an external magnetic field. Application of an AMF will then generate localized magnetic hyperthermia (MH) within the tumor, thus releasing bound doxorubicin. This study explores this novel drug delivery model which can be used for targeted dual therapy (MDT and MH) of HCC.
Aim: To demonstrate that doxorubicin-loaded-PNIPA-coated-MNP can be delivered intra-arterially to target HCC in a rat model and that heat is generated and doxorubicin is released when an AMF is applied.
Methods: Morris hepatoma cells are implanted into the livers of buffalo rats. HCC development is confirmed on MRI using a specially-constructed rat-MRI coil. 0.5 ml of doxorubicin-loaded-PNIPA-coated-MNP solution is injected into the hepatic artery and localization of MNP in HCC is confirmed with MRI. Rats are sacrificed for histology of liver and other organs. AMF is applied to doxorubicin-loaded-PNIPA-coated-MNP solution and the temperature measured to demonstrate local hyperthermia in vitro. The amount of doxorubicin released is measured by spectrophotometry.
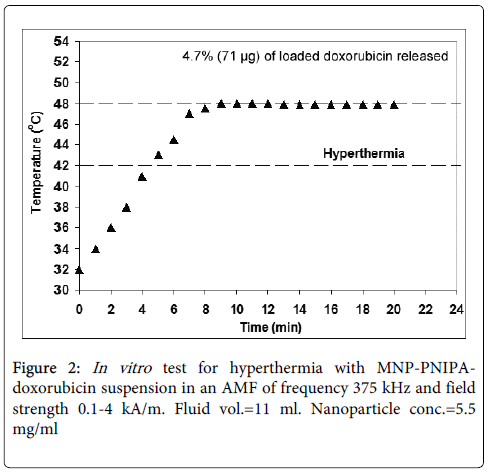
Results: Successful intra-arterial delivery of MNP was confirmed on post-injection MRI. On histology, iron particles were seen in HCC but not in normal liver or other organs. When AMF was applied, temperature of the suspension reached the target temperature of 42°C within 5 minutes and remained within hyperthermia range(42°C-48°C) for 15 minutes. During this period of hyperthermia, 4.7% (~71 μg) of loaded doxorubicin was released.
Conclusions: We have demonstrated in a rat model the feasibility of intra-arterial doxorubicin-loaded-PNIPA-coated-MNP for synergistic dual therapy of HCC using targeted hyperthermia and doxorubicin.
Keywords
Hepatocellular carcinoma; Superparamagnetic; Iron oxide nanoparticles; Magnetic drug targeting; Magnetic hyperthermia; Thermosensitive polymer; Doxorubicin
Abbreviations
HCC: Hepatocellular Carcinoma; MNP: Magnetic Nano Particles; MDT: Magnetic Drug Targeting; PNIPA: Poly-N-isopropylacrylamide; MH: Magnetic Hyperthermia; AMF: Alternating Magnetic Field
Introduction
The success of chemotherapy in cancer treatment is dependent not only on the efficacy of the drug, but also on adequate delivery to the target tissue. The dose and duration of chemotherapy is often limited by systemic side effects including nausea, anorexia, diarrhea, mucosal ulcerations and myelosuppression. This is especially true of patients with hepatocellular carcinoma (HCC), who often have underlying cirrhosis and hence are often unable to tolerate high doses of chemotherapy. Systemic chemotherapy for advanced HCC has only marginal activity with frequent toxicity and has not shown survival benefit in the past [1]. Despite this, systemic chemotherapy has an important role in the treatment of HCC because a large proportion of patients present at advanced stage of disease, which precludes surgical intervention or local ablative treatment [2]. For this group of patients, systemic chemotherapy offers the only hope of prolonging survival. Recent trials with multi-kinase inhibitors have shown promising survival benefit [3]. However, this new treatment is also associated with systemic side effects such as diarrhea, fatigue and hand-foot skin reaction [4]. Localized targeted delivery of chemotherapy allows for a reduction of systemic side effects as the drug is delivered directly to the tumor, hence limiting exposure to other tissues. This allows the delivery of an adequate concentration of the chemotherapeutic agent directly to the cancer, without being limited by excessive systemic toxicity.
Magnetic drug targeting (MDT) is a novel drug delivery system which has been used in loco-regional cancer treatment [5-7]. The advantage of MDT is the ability to direct the drug-bearing magnetic particles to the tumor by the application of an external magnetic field. In addition, it provides the ability to maintain the particles at the desired location, thus allowing for targeted release of the bound drug. In recent years, there have been important advances in the biomedical applications of nanotechnology [8], including the use of magnetic nanoparticles in MDT. Nanoparticles are less than 100 nanometers in diameter and possess unique properties due to their small size and large surface area to volume ratio. Iron oxide nanoparticles posses low coercivity (a phenomenon known as superparamagnetism) and hence are able to exist as a suspension with minimal aggregation, allowing for intra-arterial or intravenous delivery to the patient. This feature is useful for a biological drug delivery system. By applying an external magnetic field gradient, the ferrimagnetic particles can be directed to the site of the tumor. In addition, when an alternating magnetic field (AMF) is applied, the nanoparticles are able to generate heat, thus providing localized hyperthermia [9-11].
In MDT, the magnetic nanoparticles (MNP) are coated by a polymer to which a cytotoxic drug is bound. This allows for the targeted delivery of the drug to the tumor by the application of an external magnetic field at the site of the tumor. Poly-N-isopropylacrylamide (PNIPA) is a thermo-sensitive polymer with the attractive property of significant volume shrinkage at temperatures above the lower critical solution temperature (LCST) of 34°C [12]. Drugs that are bound to PNIPA are released as the polymer shrinks when heated above the LCST [13]. This provides a system for triggered drug release.
Hyperthermia has been utilized in the treatment of a variety of cancers. Necrosis of tumor cells is achieved when temperatures above 41-42°C are maintained for a sustained period [9]. In the treatment of HCC, radiofrequency ablation (RFA) utilizes hyperthermia. However, RFA is limited by tumor size, positioning of electrodes, difficult accessibility of deep seated tumors, proximity to blood vessels and other organs as well as risk of seeding of tumor along the needle tract. In addition, RFA is unable to differentiate tumor tissue from normal non-tumor tissue and thus inflicts indiscriminate destruction of normal tissues as well as tumor tissue. The heating property of magnetic nanoparticles (magnetic hyperthermia) has been explored as a potential modality of cancer treatment [10,11]. Magnetic hyperthermia offers the additional advantage of targeted delivery of hyperthermia to the tumor site by heating magnetic particles which have been localized to the tumor by a magnetic field.
There is limited experience with MDT and magnetic hyperthermia in the treatment of HCC. While previous studies have explored the efficacy of tumor targeting and localized delivery of cytotoxic drugs in animal models of HCC, these only involved single-modality treatment, i.e. either targeted delivery of chemotherapy [6,7,14] or magnetic hyperthermia [15,16]. In this study, we evaluate the feasibility of simultaneous dual treatment modalities of magnetically targeted delivery of chemotherapy and concomitant hyperthermia treatment using iron oxide nanoparticles coated with thermosensitive PNIPA polymer to which a cytotoxic drug (doxorubicin) is bound.
Aim
Our study is a proof of principle experiment, designed in two parts (an in vivo animal experiment and in vitro analysis of the characteristics of the MNP-PNIPA-doxorubicin suspension) in order to demonstrate the following:
Iron oxide MNP can be delivered intra-arterially and targeted with an external magnetic field to localize to HCC in a rat liver model (in vivo experiment)
When an alternating magnetic field is applied to the MNP solution, heat is generated to a temperature sufficient to effect tumor cell death (in vitro experiment)
Doxorubicin (loaded to PNIPA-coated MNP) can be released in response to an increase in local temperature when an external alternating magnetic field is applied (in vitro experiment).
Methods
Synthesis of magnetic nanoparticles (MNP)
Iron oxide MNP was synthesized by thermal decomposition of an iron-oleate precursor in an organic solvent at elevated temperature. The synthesis route is described in detail elsewhere [17] and was slightly modified for our work with the inclusion of an additional processing step. The organic residues from the synthesized magnetic nanoparticles were removed by heating at 400°C for 2 hours. During this process, the synthesized magnetite (Fe3O4) was converted to maghemite (γ-Fe2O3), a more stable phase in the nanometer size range. The MNP were washed multiple times with acetone, ethanol and distilled water and dried under vacuum at room temperature for 72 hours.
Coating of MNP with PNIPA
Hydrogel coating of poly-n-isopropylacrylaimde (PNIPA) onto the MNP was achieved by the surface free-radical polymerization method [12]. All the steps were carried out in a nitrogen environment. In brief, a suspension of 410 mg MNP, 600 mg N-isoproplyacrylamide (NIPA), 5.1 mg N,N-methylene(bis)acrylamide and 7.2 µl N,N,N’,N”-tetramethylenediamine in 40 ml of deoxygenated ultra-pure water was prepared under ultrasonication. After the suspension was formed, 100 µl of ammonium persulfate was added and the mixture was ultrasonicated for 2 hours. The PNIPA-coated MNP were removed by centrifugation, washed with water multiple times and dried under vacuum.
Loading of MNP-PNIPA particles with doxorubicin
Doxorubicin solutions of water and phosphate buffer saline (PBS) were prepared by dissolving known quantities of the drug in the relevant solvent over a wide range of concentrations and reference plots prepared by measuring the absorbance at a wavelength of 482 nm in a Shimadzu UV1700 UV-visible spectrophotometer. From the absorbance versus concentration plot, the concentrations of doxorubicin solutions could be determined by UV-visible spectroscopy.
Hydrogel-encapsulated MNP were ultrasonically dispersed in a doxorubicin hydrochloride-water solution of known concentration, and incubated for 16 hours. The particles were separated by centrifugation for 10 min at 8000 rpm and dried under vacuum. The concentration of the supernatant was measured by UV-visible spectroscopy and the drug loading of the particles was calculated.
Characterization of MNP
Electron microscopy
Electron microscopy of the particles was carried out on a JEOL 2010F transmission electron microscope. Particle size was measured from transmission electron micrographs using ImageJ image processing software.
X-ray diffraction
X-ray diffraction studies were performed on vacuum dried MNP powders using a Shimadzu 6000 X-ray diffractometer. Phase identification was done by matching peak positions and relative intensities to reference patterns using diffraction pattern analysis software.
Thermo-gravimetric analysis
A TA instruments TGA Q500 was used to measure the quantity of polymer coated onto the particles via thermo-gravimetric analysis. The samples were heated to 600°C at a rate of 20°C/min and weight loss measured during the process.
Vibrating sample magnetometry
Magnetic properties of the MNP powders were evaluated using a LakeShore 7404 vibrating sample magnetometer at room temperature, in an applied field range of 0 to 10 kG.
Optimum concentration of MNP suspension for MRI visualization
Preliminary studies were performed to determine the optimum concentration of MNP suspension for MRI visualization. A concentration of 0.36 mg/ml was found to be ideal to facilitate distinct visualization of the MNP using a conventional MRI scanner. At this concentration, the MNP were identified by areas of decreased intensity due to heavy T2*shortening on the T1-weighted gradient-echo-sequences.
In vitro experiment
Generation of heat and doxorubicin release in an alternating magnetic field
Sixty one milligrams of MNP-PNIPA-doxorubicin suspension were dispersed in 11 ml of phosphate buffered saline (pH 7.4) in a glass bottle and placed inside a water-cooled 5-loop copper induction coil energized by an alternating current generator (Inductelec, UK) at a fixed operating frequency of 375 kHz. An alternating magnetic field (AMF) of 0.1-4 kA/m was generated by the passage of the alternating current through the induction coil. Ceramic wool thermal insulation was used around the bottles to minimize heat loss. The temperature of the MNP-PNIPA-doxorubicin suspension in response to the AMF was measured using a fiber optic thermometry unit (Luxtron MD600) connected to a laptop computer. The temperature was stabilized within the hyperthermia range (between 42°C and 48°C) by manually varying the strength of the AMF as necessary. After 15 minutes of exposure to hyperthermia temperature, the AMF was turned off and the particles were separated by centrifugation. The concentration of doxorubicin released in response to the temperature of the suspension was measured as described earlier.
In vivo animal experiment
Adult male Buffalo rats weighing between 250-300 g were procured from Charles River Laboratories (Australia) and housed in the Department of Experimental Surgery, Singapore General Hospital. All animal studies were conducted under approved guidelines set forth by the Institutional Animal Care and Use Committee of the hospital, which is an International Association for Assessment and Accreditation of Laboratory
Animal Care accredited centre. The rats were fed ad libitum and housed in individual ventilated cages under the standard housing conditions. The animals were kept for 4 weeks prior to MRI scan. Morris Hepatoma 7777 cells (American Tissue Culture Corporation, Rockville, MD) were used for direct subcapsular implantation into the rat liver to induce HCC growth.
The first stage of the study was a baseline MRI of the rat liver using a purpose-built Helmotz coil in a 3 Tesla MRI machine with a gradient strength of 40 mT/m (Siemens Allegra, Erlangen, Germany). T1-weighted images were taken by means of a 3D (RF spoiled) gradient-echo sequences with field of view (FOV) 80 mm, slice thickness of 1 mm, matrix 256x192. The sequence parameters were TR/TE 6.0/3.5. As image acquisition lasts multiple free breathing cycles, a simple random 16 averaging procedure was performed, minimizing the motion artifacts. The rats were sedated with a cocktail mixture of ketamine (50 mg/kg) and diazepam (5 mg/kg) intraperitoneally during the MRI procedure to reduce movement artifacts. In the second stage, the anaesthetized rats underwent laparotomy for hepatic subcapsular implantation of Morris hepatoma cells. Post-laparotomy analgesia was provided via acetaminophen 1-2 mg/ml in the drinking water. The diet, activity, weight and well-being of the rats were monitored post-implantation. At 2 weeks post-implantation, the rats were subjected to a repeat MRI to confirm and assess for development of HCC. If the HCC was more than 2 cm, the rats progressed to the next stage of the experiment. If the HCC was too small, an additional 2-4 weeks were allowed for adequate tumor growth before the MRI was repeated. This served as the baseline pre-treatment MRI scan.
In the third stage, the laparotomy wound was re-opened for selective cannulation of the hepatic artery and injection of MNP-PNIPA-doxorubicin solution under a directed external magnetic field. Cannulation was performed under the operating microscope using a 1 ml syringe with a 21-gauge needle bent at 60 degrees. 0.5 ml of MNP-PNIPA-doxorubicin suspension at a concentration of 0.36 mg/ml was delivered directly into the hepatic artery. A magnet wrapped in sterile gauze was placed at the liver surface directly over the tumor site to localize the MNP-PNIPA-doxorubicin particles. Immediately after delivery of the MNP solution, an MRI was performed to document localization within the HCC.
In the final stage of the in vivo experiment, the rats were euthanized using intraperitoneal phenobarbitone at 80 mg/kg. The animal was then laparotomized and sections of the liver (tumor and normal liver) and other intra-abdominal organs (spleen, kidney and stomach) were harvested to determine the histological distribution of the MNP particles.
Results
In vitro experiments
Characteristics of iron-oxide MNP
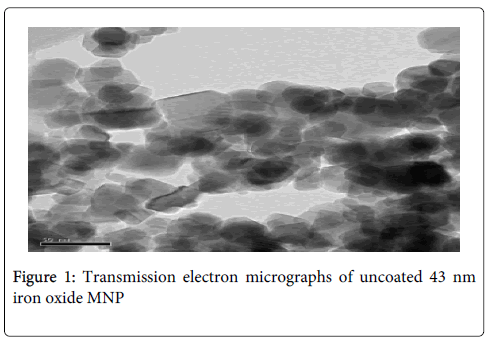
After synthesis and heat treatment, the iron oxide MNP were confirmed to be maghemite (γ-Fe2O3) by pattern matching of the X-ray diffraction pattern to reference Joint Committee on Powder Diffraction Standards (JCPDS) files. The average size of these irregularly shaped nanoparticles before the polymer coating was 43 nm with a size range of 34 nm to 57 nm as measured from the transmission electron micrograph (Figure 1).Thermo-gravimetric analysis revealed that after PNIPA coating, the polymer content of the MNP was 6% of the total weight of the particles. From UV-visible spectrophotometry of the doxorubicin-loading solution, it was calculated that the doxorubicin content of these polymer coated magnetic nanoparticles was 2.5 % of their total weight. The saturation magnetization of the MNP remained high and was not significantly affected by the polymer coating. This aids guidance of the MNP to the target tumor by an external magnetic field gradient.
Hyperthermia and doxorubicin release in response to alternating magnetic field
When exposed to the AMF, the temperature of the MNP-PNIPA-doxorubicin suspension rose steadily in less than 5 minutes to a temperature >42°C (Figure 2). It then remained in the hyperthermia range (42°C-48°C) for 15 minutes while maintaining a plateau temperature close to 48°C. After this heating and cooling cycle, 4.7% (equivalent to 71 μg) of loaded doxorubicin was released. This in vitro experiment thus demonstrated that the MNP suspension was capable of generating hyperthermia in the range required for tumor cell death when an AMF was applied. Furthermore, the ability to control the temperature in the range between 42°C and 48°C would avoid damage to healthy tissue if used in vivo. We have also demonstrated the ability of the MNP-PNIPA-doxorubicin suspension to release doxorubicin when the temperature is raised beyond the LCST.
In vivo animal experiment
Successful intra-arterial delivery of MNP solution
Various concentrations of MNP suspension and different methods of delivery were evaluated in order to determine the most efficacious model. We developed a technique of bending the 21-gauge needle at a 60 degree angle to stabilize the needle within the small lumen of the rat hepatic artery so that the entire volume of MNP solution could be delivered into the hepatic artery at a constant rate.
Localization of MNP particles to HCC
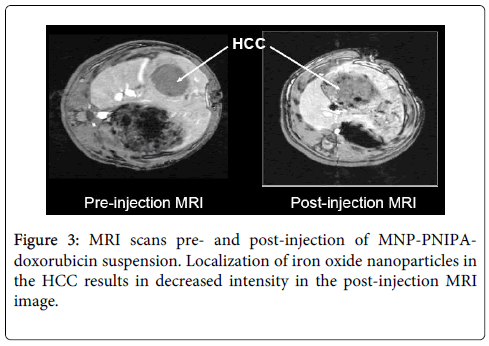
Following intra-arterial delivery of the MNP-PNIPA-doxorubicin suspension, the MNP particles successfully localized to the HCC. This was confirmed by the post-MNP injection MRI scans (Figure 3). In the post-MNP injection MRI, the localized uptake of MNP by the tumor was evidenced by the decreased intensity of the HCC. The intensity of the MNP is decreased because of the high concentration used (T2*effect). In contrast, the rest of the non-tumor liver and other intra-abdominal organs did not show decreased intensity, providing radiological evidence of localization to the HCC and not elsewhere within the abdomen.
Histological study of necropsy specimens
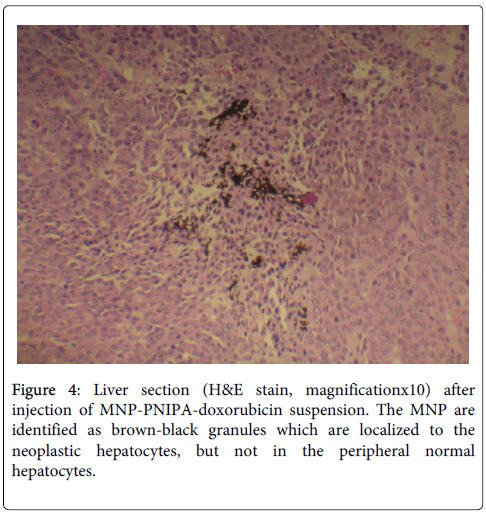
Sections from the tumor-bearing and normal parts of the liver, kidney, spleen and stomach were fixed in paraffin and stained with hematoxylin and eosin (H&E) to determine the presence of iron particles in the various viscera. In the liver sections, the MNP had agglomerated to become visible under light microscopy, and were identified as brown-black granules. They were localized to the tumor and were not found in the normal (non-tumor) liver tissue (Figure 4). Examination of the other viscera found no traces of MNP collection under light microscopy. Sections of the distal hepatic artery showed minimal deposition of MNP along the walls of the vessel. This provides histological evidence of successful delivery and targeting of the MNP particles to the tumor by the external magnetic field, with no evident spillover to neighboring organs.
Discussion
In this study, we investigated the feasibility of combining dual modalities of anti-neoplastic treatment (magnetic targeted delivery of cytotoxic agent and magnetic hyperthermia) using MNP in the treatment of a rat model of HCC. We have demonstrated successful intra-arterial delivery of MNP-PNIPA-doxorubicin suspension and localization to intra-hepatic tumor based on MRI imaging and histology. We have also demonstrated generation of in vitro hyperthermia and controlled drug release from the MNP-PNIPA-doxorubicin suspension when exposed to an AMF. This explores the unique concept of utilizing MNP complexed with a thermosensitive polymer for the dual purpose of targeted chemotherapy and concurrent thermotherapy in HCC.
The use of MNP for targeted delivery of chemotherapy has been studied in many different cancers [5-7]. The success of MDT depends on several factors: (a) the magnetic particles should be of an optimal size to allow efficient delivery to the tumor and uptake by tumor cells and not precipitate in the vessels prior to reaching the tumor (b) the method of delivery of the MNP-drug complex should have good access to the tumor and avoid excessive clearance by the reticulo-endothelial system (first-pass effect) (c) the magnetic field should be of sufficient strength to be able to attract the magnetic particles to the tumor and prevent dispersion outside the intended field (d) there should be an efficient method of confirming localization of the magnetic particles within the tumor (e) the chemotherapeutic drug should be effectively released at the site of the tumor at a concentration sufficient to cause tumor cell death without excessive toxicity to surrounding healthy tissue (f) it should cause minimal local and systemic toxicity and should be safe for clinical use [5].
The size of the magnetic particles is one of the most important factors in the design of a successful MDT system. For in vivo biomedical applications, MNP must be made of a non-toxic and non-immunogenic material, with particle sizes small enough to remain within the circulation after injection and to pass through the capillary systems without causing vessel embolism. They must also have a high magnetization so that their movement in the blood can be directed and so that they can be immobilized in the targeted pathologic tissue by an external magnetic field [18]. Early trials in MDT using magnetic albumin microspheres caused embolisation of vessels, preventing effective delivery of the drug to the tumor [7]. The use of ferrofluids bound to anhydroglucose polymers provided a more stable drug delivery system, partly due to the smaller size of the MNP [6]. The first clinical experiment of MDT in humans was performed using nanoparticles of 100 nm in size [19]. This study demonstrated that MDT using nanoparticles was well tolerated with low toxicity to the patients. However, there was limited anti-tumor activity, possibly due to limitations imposed by the size of the magnetic particles used. More recent studies in MDT have used smaller nanoparticles (10-20 nm) to demonstrate successful tumor targeting in animal models [20,21]. We studied the tumor-targeting and heating characteristics of a range of MNP sizes. The 43 nm MNP (size range 34-57 nm) respond readily to a magnetic field and are were capable of excellent heating power output when exposed to alternating magnetic field [22]. The ability to produce MNP of various sizes allowed us to modify the characteristics of the MNP suspensions in order to maximize efficacy of drug-binding, targeted drug release and hyperthermia by modifying the production parameters as required. This aspect of our study is an advantage over other studies which have used commercially available MNP solutions with fixed parameters.
In our in vivo experiments, we studied the feasibility of delivery of the MNP-PNIPA-doxorubicin complex solution into rat livers with HCC via direct injection into the hepatic artery. We have chosen to study this model as we envisage a potential transition to clinical application in the treatment of HCC in humans, where the drug-bound MNP suspension can be delivered into the hepatic artery by a catheter introduced via the femoral artery, similar to the method used for conventional transarterial hepatic chemoembolization of HCC. Several studies of MDT in animal models have used intravenous delivery of the magnetic fluids [23] or direct injection of the magnetic material into the tumor [24]. Compared to intravenous delivery, intra-arterial administration has the advantage of avoiding the first pass effect and hence allows for delivery of a greater concentration of drug to the tumor with lower toxicity. Direct intra-tumoral injection is invasive and involves a risk of tumor seeding along the needle tract as well as bleeding from the highly vascular HCC. We have found that delivery of 0.5 ml of the MNP-PNIPA-doxorubicin suspension into the distal hepatic artery resulted in adequate uptake of magnetic particles into the tumor. Necropsy studies demonstrated minimal deposition of particles along the wall of the vessel and no significant spillover to neighboring organs, supporting the feasibility of intra-arterial delivery of drug-bound MNP for future HCC treatment.
Doxorubicin was used in this study because it is the most established chemotherapy drug in HCC. This system offers the flexibility of complexing the polymer with other chemotherapeutic agents as well. Hence, future studies exploring the efficacy of this technique might involve the usage of alternative drugs, including sorafenib which has recently been shown to provide survival benefit in HCC. This may theoretically provide a reduction in systemic toxicity with possible improvement in efficacy.
A unique aspect of our study is the use of MNP to simultaneously execute two modalities of cancer therapy–targeted chemotherapy and magnetic hyperthermia. The feasibility of MDT has been established in both animal and human studies and has been proven to be safe with little toxicity. However, although studies in animal models have shown successful tumor regression [5,6], a phase I trial in patients with advanced solid tumor showed only slight tumor reduction in 2 out of 14 patients [19]. Hence although the idea of MDT is theoretically attractive, it has yet to demonstrate clinical efficacy. MNP have also been used in the treatment of cancer by magnetic fluid hyperthermia (MFH). MFH involves the localized deposition of magnetic particles within the tumor, which are then heated by the application of an external AMF. Previous studies have established the feasibility of MFH to generate temperatures above 42°C, the temperature at which cell necrosis occurs [25]. A pilot study utilizing MFH for treatment of recurrent prostate cancer has demonstrated the clinical feasibility of this approach [26]. The combination of MDT and MFH in a single delivery system has the potential synergistic anti-tumor effect. While this concept has been studied for the treatment of malignant melanoma [27], to date there have been no studies of such dual therapy for HCC.
In the in vitro aspect of our study, we have demonstrated that when an appropriate AMF was applied to a suspension of doxorubicin-loaded PNIPA-MNP, the nanoparticles generated sufficient heat to raise the temperature of the surrounding medium to the range required for tumor cell kill (42-48°C). We were able to maintain this temperature by controlling the strength and duration of the magnetic field. This is important to ensure that the heat produced is not excessive, thus avoiding thermal injury to surrounding tissues and excessive discomfort to the patient [28,29]. In vitro studies exploring the potential cytotoxicity of PNIPA-MNP have demonstrated that concentrations of 0.5 mg/ml of PNIPA-grafted nanoparticles had low cytotoxicity after 72 hours of incubation [30]. Even at higher concentrations of 25-1000 mg/ml, no significant cytotoxic or genotoxic response was observed in human cell lines exposed to PNIPA nanoparticles [31].
A key feature in our study design that enables combined delivery of MDT and MFH is the use of MNP that are coated by a thermosensitive polymer, PNIPA. Heating of the MNP-PNIPA-doxorubicin complex causes conformational change of the PNIPA polymer resulting in release of bound doxorubicin, hence the concentration of doxorubicin released is expected to be temperature-dependent. Therefore, doxorubicin release may be controlled by varying the temperature of the MNP-PNIPA-doxorubicin solution, which is in turn controlled by the varying the AMF. The AMF can thus act as a “therapeutic switch” –once the MNP-PNIPA-doxorubicin particles are in place within the tumor, application of the AMF can activate hyperthermia as well as release of doxorubicin within the target tissue, providing a system of controlled synergistic tumor destruction.
Another unique application of this system is the feasibility of using MRI technology for both diagnostic and therapeutic purposes. We envisage a “one-stop” dual modality therapy whereby after infusion of MNP-PNIPA-doxorubicin solution, MRI is used to visualize localization of the magnetic nanoparticles within the tumor and then a magnetic field applicator is used to generate an AMF to effect hyperthermia and cytotoxic drug release. A magnetic field applicator has been developed for human use (MFH300, MagForce® Nanotechnologies GmbH, Berlin) that is able to generate the magnetic fields necessary to affect hyperthermia treatment in human clinical trials [26].
Our concept of simultaneous dual modality therapy for HCC is plausible and attractive. Related experiments performed by other researches have provided ancillary information that supports the feasibility of this technique [19,24,27]. However, further studies are required to provide in-depth characterization and refinement of this technique before progressing to human trials. We have successfully provided proof of principle of the possibility of intra-arterial delivery of MNP-PNIPA-doxorubicin complex solution and localization by magnetic field to intra-hepatic HCC in a rat model [32]. In our in vitro studies, we have demonstrated the heating and drug-release characteristics of the MNP-PNIPA-doxorubicin suspension. Hence, we have shown in principle, the feasibility of the concept of dual modality therapy with MDT and magnetic hyperthermia for HCC using iron oxide nanoparticle technology. However, our proof-of-principle study was not designed to demonstrate tumor regression in vivo. This important aspect needs to be addressed by further studies. Other issues that need to be addressed in future studies include the effect of tumor size, vascular pattern and position on MNP delivery and targeting, the optimal concentration of doxorubicin required for adequate tumor control, as well as the optimal method of generating an external magnetic field of adequate strength and field size to ensure optimal targeting of MNP to the tumor. The immediate next stage of our experiments is in vivo confirmation of our in vitro observations in a large animal model, with the aim of demonstrating tumor regression. At present, the early success of this novel dual modality therapy technique holds great promise for the future development of targeted synergistic treatments for HCC.
References
- Bruix J, Sherman M; Practice Guidelines Committee, American Association for the Study of Liver Diseases (2005) Management of hepatocellular carcinoma.Hepatology 42: 1208-1236.
- Okada S (1998) Chemotherapy in hepatocellular carcinoma.Hepatogastroenterology 45 Suppl 3: 1259-1263.
- Llovet J, Ricci S, Mazzaferro V (2007) Sorafenib improves survival in advanced hepatocellular carcinoma: Results of a phase III randomized placebo-controlled trial (SHARP trial). Journal of Clinical Oncology.
- Lang L (2008) FDA approves sorafenib for patients with inoperable liver cancer.Gastroenterology 134: 379.
- Alexiou C, Arnold W, Klein RJ, Parak FG, Hulin P, et al.(2000)Locoregional cancer treatment with magnetic drug targeting. Cancer Research 60:6641-6648.
- Lübbe AS, Bergemann C, Huhnt W, Fricke T, Riess H, et al. (1996) Preclinical experiences with magnetic drug targeting: tolerance and efficacy.Cancer Res 56: 4694-4701.
- Widder KJ, Morris RM, Poore GA, Howards DP, Senyei AE (1983) Selective targeting of magnetic albumin microspheres containing-dose doxorubicin: total remission in Yoshida sarcoma-bearing rats. Eur J Cancer ClinOncol 19:135-139.
- Pope-Harman A, Cheng MM, Robertson F, Sakamoto J, Ferrari M (2007) Biomedical nanotechnology for cancer.Med Clin North Am 91: 899-927.
- Wust P, Hildebrandt B, Sreenivasa G, Rau B, Gellermann J, et al. (2002) Hyperthermia in combined treatment of cancer.Lancet Oncol 3: 487-497.
- Moroz P, Jones SK, Gray BN (2002) Magnetically mediated hyperthermia: current status and future directions.Int J Hyperthermia 18: 267-284.
- Jordan A, Scholz R, Wust P, Fahling H, Felix R (1999) Magnetic fluid hyperthermia (MFH): Cancer treatment with AC magnetic field induced excitation of biocompatible superparamagnetic nanoparticles. Journal of Magnetism and Magnetic Materials 201:413-419.
- Ang KL (2007)Venkatraman, S., and Ramanujan, R. V. Magnetic PNIPA hydrogels for hyperthermia applications in cancer therapy. Materials Science and Engineering: C, 27: 347-351.
- Klouda L, Mikos AG (2008) Thermoresponsive hydrogels in biomedical applications.Eur J Pharm Biopharm 68: 34-45.
- Wang JM, Xiao BL, Zheng JW, Chen HB, Zou SQ (2007). Effect of targeted magnetic nanoparticles containing 5-FU on expression of bcl-2, bax and caspase 3 in nude mice with transplanted human liver cancer. World J Gastroenterol 13: 3171-3175.
- Jones SK, Winter JG, Gray BN (2002) Treatment of experimental rabbit liver tumours by selectively targeted hyperthermia.Int J Hyperthermia 18: 117-128.
- Muraoka A, Takeda S, Matsui M, Shimizu T, Tohnai I, et al. (2004) Experimental study of a novel thermotherapy for hepatocellular carcinoma using a magnesium ferrite complex powder that produces heat under a magnetic field. Hepatogastroenterology 51: 1662-1666.
- Park J, An K, Hwang Y, Park JG, Noh HJ, et al. (2004) Ultra-large-scale syntheses of monodispersenanocrystals.Nat Mater 3: 891-895.
- Tartaj P, Morales MP, Veintemillas-VerdaguerS, Gonzalez-Carreno T, Serna CJ (2003) The preparation of magnetic nanoparticles for applications in biomedicine. J Phys D: ApplPhys36.
- Lubbe AS, Bergemann C, Reiss H, Schriever F, Reichardt P, et al. (1996) Clinical experiences with magnetic drug targeting: a Phase I study with 4’-epirubicin in 14 patients with advanced solid tumors. Cancer Res 56:4686-4693.
- Liang S, Wang Y, Yu J, Zhang C, Xia J, et al. (2007) Surface modified superparamagnetic iron oxide nanoparticles: as a new carrier for bio-magnetically targeted therapy.J Mater Sci Mater Med 18: 2297-2302.
- Xu H, Song T, Bao X, Hu L(2005) Site-directed research of magnetic nanoparticles in magnetic drug targeting. Journal of Magnetism and Magnetic Materials 293:514-519.
- Hergt R, Dutz S, Muller R, Zeisberger M (2006) Magnetic particle hyperthermia: nanoparticles magnetism and materials development for cancer therapy. Journal of Physics: Condensed Matter 19: 2919-2934.
- Chen J, Wu H, Han D, Xie C (2006) Using anti-VEGF McAb and magnetic nanoparticles as double-targeting vector for the radioimmunotherapy of liver cancer.Cancer Lett 231: 169-175.
- Yan S, Zhang D, Gu N, Zheng J, Ding A, et al. (2005) Therapeutic effect of Fe2O3 nanoparticles combined with magnetic fluid hyperthermia on cultured liver cancer cells and xenograft liver cancers. Journal of Nanoscience and Nanotechnology 5: 1-8.
- Moroz P, Jones SK, Gray BN (2002) Tumor response to arterial embolization hyperthermia and direct injection hyperthermia in a rabbit liver tumor model.J SurgOncol 80: 149-156.
- Johannsen M, Gneveckow U, Eckelt L, Feussner A, Waldofner N, et al. (2005) Clinical hyperthermia of prostate cancer using magnetic nanoparticles: Presentation of a new interstitial technique. Int J Hyperthermia 21: 637-647.
- Ito A, Fujioka M, Yoshida T, Wakamatsu K, Ito S, et al. (2007) 4-S-Cysteaminylphenol-loaded magnetite cationic liposomes for combination therapy of hyperthermia with chemotherapy against malignant melanoma. Cancer Sci 98: 424-430.
- Johannsen M, Gneveckow U, Taymoorian K, Thiesen B, Waldorfner N, et al. (2007) Morbidity and quality of life during thermotherapy using magnetic nanoparticles in locally recurrent prostate cancer: results of a prospective phase I trial. International Journal of Hyperthermia 23: 315-323.
- Wust P, Gneveckow U, Johannsen M, Bohmer D, Henkel T, et al. (2006) Magnetic nanoparticles for interstitial thermotherapy – feasibility, tolerance and achieved temperatures. International Journal of Hyperthermia 22: 673-685.
- Lien YH, Wu TM, Wu JH, Liao JW (2011) Cytotoxicity and drug release behavior of PNIPAM grafted on silica-coated oxide nanoparticles. J Nanopart Res 13: 5065-5075.
- Naha PC, Bhattacharya K, Tenuta T, Dawson KA, Lynch I, Gracia A, et al. (2010) Intracellular localization, geno- and cytotoxis response of polyN-isopropylacrylamide (PNIPAM) nanoparticles to human keratinocyte (HaCaT) and colon cells (SW 480). ToxicolLett 198: 134-143
- Purushotham S, Chang PE, Rumpel H, Kee IH, Ng RT, et al. (2009) Thermoresponsive core-shell magnetic nanoparticles for combined modalities of cancer therapy.Nanotechnology 20: 305101.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 16091
- [From(publication date):
August-2014 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 11361
- PDF downloads : 4730