Review Article Open Access
Powder XRD Technique and its Applications in Science and Technology
Ashish Chauhan1* and Priyanka Chauhan21Small and Medium Pharmaceutical Industry Centre, Mohali, Punjab, India
2Shikhar S. Sadan Sr. Secondary School, Dhampur, Bijnor, India
- *Corresponding Author:
- Ashish Chauhan
Small and Medium Pharmaceutical Industry Centre|
Mohali, Punjab, India
Tel: 9464616773
E-mail: aashishchauhan26@gmail.com
Received date: September 19, 2014; Accepted date: October 16 2014; Published date: October 20, 2014
Citation: Chauhan A, Chauhan P (2014) Powder XRD Technique and its Applications in Science and Technology. J Anal Bioanal Tech 5:212 doi: 10.4172/2155-9872.1000212
Copyright:© 2014 Chauhan A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
The review presents a brief overview of advanced X-Ray Powder Diffraction Technique. It is an indispensable method of material investigation, characterization and quality control. It has many salient features and advantages that promote its wide range use. It has numerous applications that cater to industrial and academic research that ultimately enriches the growth and development of Science and Technology.
Keywords
XRD Technique; Diffractometer; Powder X-Ray Diffraction
Introduction
Diffraction occurs when light is scattered by a periodic array with long-range order, producing constructive interference at specific angles. The atoms in a crystal are periodically arranged thus diffract light. The wavelength of X-ray are similar to the distance between atoms, Powder X-ray Diffraction (PXRD) techniques uses this principle to elucidate the crystalline nature of materials. The scattering of X-rays from atoms produce a diffraction pattern that contains information about the atomic arrangement in crystal. Amorphous materials like glass do not have periodic array with long-range order so; they do not produce any significant peak in diffraction pattern [1-3].
Powder X-Ray Diffractometer and Diffractogram
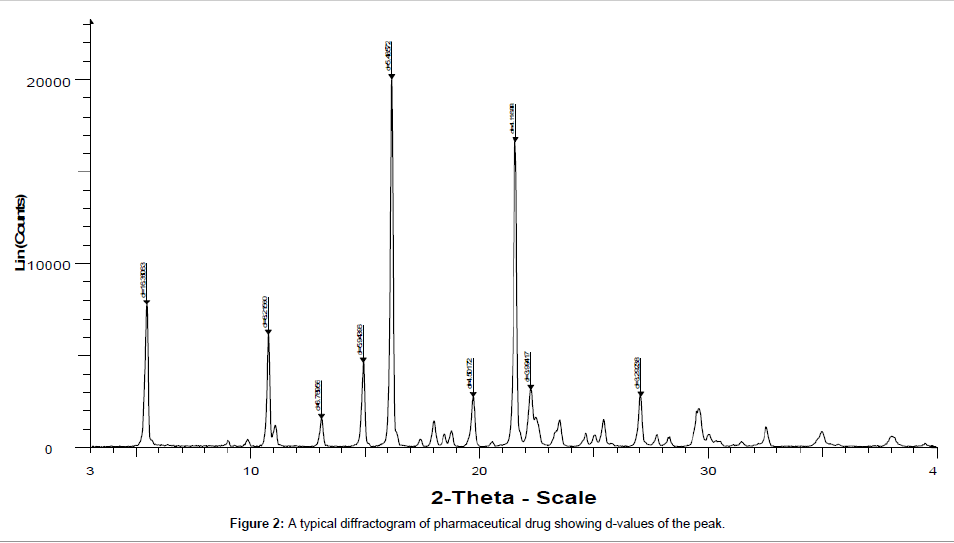
Powder XRD is a compact advanced instrument (Figure 1). It has various salient features and new accessories like variable temperature assembly and humidity chamber that can further expand the horizon of its applications by providing the information on effect of temperature and humidity on the nature of material. When X-ray falls over a crystal, it diffracts in a pattern characteristic to its structure. In powder X-ray diffraction, the diffraction pattern is obtained from a powder of the material, rather than an individual crystal. Powder diffraction is often easier and more convenient than single crystal diffraction as it does not require individual crystals. A diffraction pattern plots intensity against the angle of the detector, 2θ. The result obtained is called diffractogram (Figure 2). In a diffraction pattern, the peak position depends upon the wavelength. Absolute intensity (number of X-rays observed in a given peak) may vary by instrumental and experimental parameters. Diffractometers can be operated both in transmission and in reflection configurations. The reflection one is more common. Interactions between the incident X-ray beam and the sample produce intense reflected X-rays by constructive interference when conditions satisfy Bragg’s Law. This law describes the general relationship between the wavelength of the incident X-rays, the incident angle of the beam and the spacing between the crystal lattice planes of atoms. Constructive interference occurs when the differences in the travel path of the incident X-rays is equal to an integer multiple of the wavelength [1-3].
The Advantages of Powder XRD and its Scope
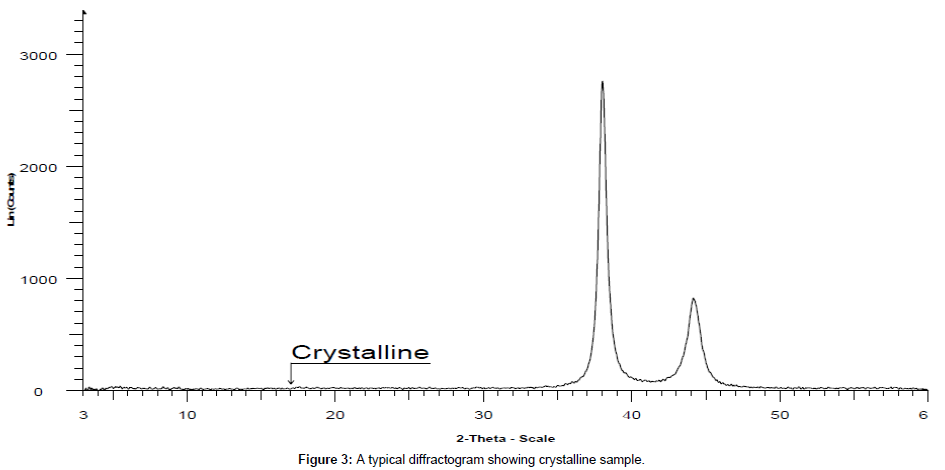
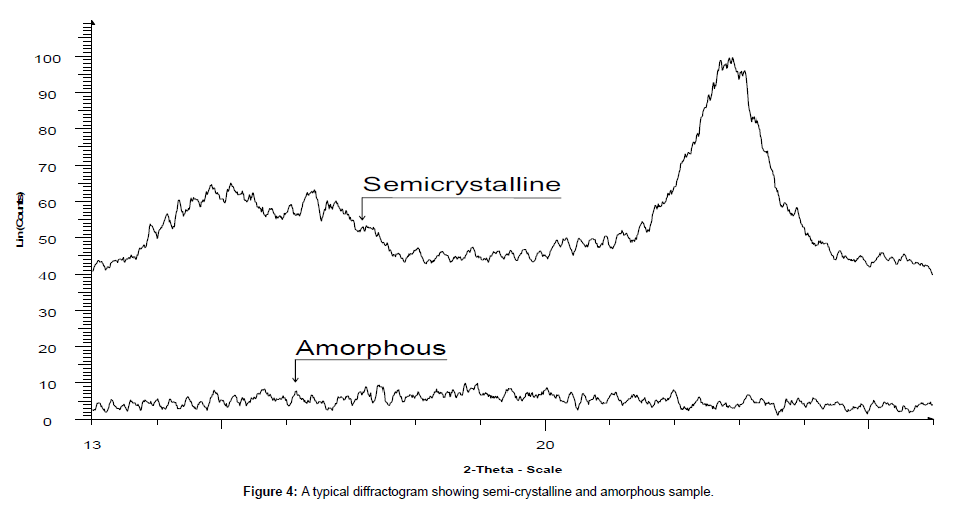
PXRD has numerous advantages like non-destructive nature, high sensitivity, reliability, depth profiling (glancing incident angle), easy sample preparation, system is user friendly, operational procedure is convenient, fast speed, effective resolution, low maintenance cost, proper automation, easy data interpretation that could be used for both qualitative and quantitative analysis, in wide range of applications. However, it has a few disadvantages due to use of harmful radiations moreover there is requirement of standard reference to match for an inference and an expensive instrument. It is used to study the crystalline content, identify the crystalline phases, spacing between lattice planes, scales of existence, preferential order and epitaxial growth of crystallites. Since every material has its unique diffraction patterns so materials and compounds can be identified by using a database of diffraction patterns. The percentage purity of a sample can be accessed by diffraction pattern by considering the proportion and composition of impurities present. PXRD is the basic requisite for differentiating the crystalline sample (Figure 3) from semi-crystalline like in polymer e.g cotton and amorphous materials e.g phenol formaldehyde complex resin (Figure 4). It is the primary tool for solid state characterization.
The samples subjected for analysis are generally in the form of finely divided powders and diffraction can be obtained from flat surfaces made coplanar to the holder. The sample for analysis could be of vast array, including inorganic complexes, organic compounds, fiber, polymers, metals, composites, metallurgical samples, pharmaceuticals, earth sciences, microelectronics and nanotechnology. PXRD can also be wisely used to study and explore the pseudo crystalline structure of mesoporous materials and colloidal materials provided that the length scales are in the correct regime and proportion. It is frequently used for the analysis of Asbestos, catalyst, ceramics, chemicals, clays & minerals, cement, composite, corrosion products, fly-ash, environmental studies, semiconductors, textiles, plastics, nanomaterials, pharmaceuticals, metals, alloys etc. [1,3].
Applications of Powder X-Ray Diffraction in Science and Technology
Polymorph study
PXRD is helpful in identification and characterization of polymorph, monitoring the stability, method development and validation for identification and quantification of drugs in Pharmaceutical Industries. X-rays are partially scattered by atoms when they strike the surface of a crystal. The part of the X-ray that is not scattered simply passes through the next layer of atoms, where again part of the X-ray is scattered and part of it passes through to the next layer. This causes an overall diffraction pattern, similar to how a grating diffracts a beam of light. In order for an X-ray to diffract, the sample must be crystalline and the spacing between atom layers must be close to the radiation wavelength. If beams diffracted by two different layers are in phase, constructive interference occurs and the diffraction pattern shows a peak. However, if they are out of phase, destructive interference occurs appear and no peak is observed. Diffraction peaks only occur if it follows Bragg’s Law. Since, a highly regular structure is needed for diffraction to occur, only crystalline solids diffract, the PXRD of amorphous materials do not depict any significant peak in diffraction pattern [2-6].
Variable Temperature and Relative Humidity study by PXRD
Solid phase transitions such as polymorph inter-conversions are routinely examined by X-ray diffractometer using variable temperature sample stages (VT-XRD). Both subambient and elevated temperature stages are available that can help to study the sample behavior at variable temperature conditions. VT-XRD helps to directly identify the crystalline phase as a function of temperature. XRPD is also commonly used to investigate the structure of variable hydrates that are crystalline and contain nonstoichiometric water within channels in the crystal lattice. The amount of water present in a variable hydrate occurs as a function of the change in relative humidity (RH) in the environment of the sample. The peak positions in the diffractogram refers to dimensions of the unit cell so a change in the size of the unit cell due to the presence of water can be screened by comparison of XRPD patterns under different RH environments [4-9].
Screening the Crystal structure and Lattice parameters using PXRD
While it will always be advantageous to solve the crystal structure using single-crystal diffraction, it is not always possible as some crystalline substances do not possess the necessary size and quality. Traditionally, structural analyses using powder diffraction data are conducted using patterns obtained from synchrotron sources. When a complete structural solution is not possible from XRPD, it may still be possible to use a high quality XRPD pattern to obtain useful information such as the unit cell parameters (indexing) and the crystalline space group.
Crystal structure determination from powder diffraction data is sometimes tedious due to the overlap of reflections in a powder experiment. A number of methods are used for structural determination like simulated annealing and charge flipping. The crystal structures of known materials can be evaluated as a function of temperature or pressure, using the Rietveld method. It is full pattern analysis technique. It is used to determine unknown structures from powder data. A number of programs can be used for structure determination such as TOPAS.
X-ray diffraction provides ample information about the lattice parameters. The position of a diffraction peak is determined by the size and shape of unit cell of the crystalline phase. Peak represents a lattice plane and therefore can be characterized by Miller index. If the symmetry is high as in case of cubic or hexagonal, it is not difficult to identify the peak index for an unknown phase. This is very useful in solid-state chemistry to identifying new materials. Once a pattern gets indexed, it serves as reference for new entities [10-17].
Use of PXRD in Pharmaceutical industry
XRD is the key technique for drug analysis. It serves a major role in all stages of drug development, testing and production. It is an essential part of analytical research and development, quality control of the active ingredients, excipients and final products Minor changes may cause major batch-to-batch inconsistency that may cause critical problems and lead to problems in the manufacturing of the pharmaceutical dosage form, the quality of the formulation, the bioavailability and drug stability.
Many new drug moiety exhibit different forms (polymorphs or solvates) that vary in their physical properties. Differences in these forms can affect the quality or efficacy of the new drug. It helps in elucidation of the relevant polymorphic and pseudo-polymorphic forms in pharmaceutical development. The materials ranging from Active Pharmaceutical Ingredients to finished dosage forms have to be properly screened by XRD as it ultimately cause changes in diffraction patter and affects the bio-availability [8,9,18].
Use of PXRD in Nano and Material science
The term particle size and crystallite size refer to two distinct properties in a material. Particles comprise of several small crystallite. Crystallite size is the fundamental property of materials. Properties of nanomaterials depend on crystals size and not particle size. PXRD can measure millions of crystals and accurately determine the size distribution of nanomaterials. It can be widely used for studying the nature of polymers and composites in Material Science [19,20].
PXRD use in Forensic studies
PXRD is useful in trace analysis. It can be used to contact traces of paint flakes, hair, glass fragments, stains of any description and loose powdered materials. Identification and comparison of trace quantities of material can help in the conviction of a suspect of his involvement in a crime [1,3].
Geological applications of PXRD
PXRD is the key tool in mineral exploration. The advent of PXRD has revolutionized the geological sciences to a great extent. It is not easy to precede the characterization and identification of the samples without powder XRD. Every mineral is defined by a characteristic crystal structure that gives a unique X-ray diffraction pattern, allowing rapid identification of minerals present within a rock and soil sample. The PXRD data can be fruitful to determine the proportion of the different minerals present in a given sample [3,6].
Microelectronics industry using PXRD
Microelectronics industry uses silicon and gallium arsenide substrates in integrated circuit production; there is a need to fully characterize these materials by incorporating the information’s obtained by PXRD. XRD topography detects and forms the image of the defects within a crystal [1,3,6].
Use of PXRD in glass industry
Glasses are amorphous in nature. The X-ray pattern of amorphous compounds does not contain any significant peak. However, there are many uses of PXRD in the glass industry. It includes identification of crystalline particles that creates faults in bulk quantity of glass, measurements of crystalline coatings for texture, crystallite size and crystallinity [10,12,13,17].
Crystallinity study by PXRD
The XRD analysis of crystalline compounds gives a diffraction pattern consisting of a well-defined, narrow, sharp and significant peak while amorphous materials do not give clear peaks rather the pattern has noise signals, smeared peak or it can have some short order bumps. Many polymers depict semi-crystalline behavior and produce halo pattern. Powder XRD can be used to determine the crystallinity by comparing the integrated intensity of the background pattern to that of the sharp peaks. Different scientists have reported variable methods to find out the percentage crystallinity and crystallinity index. Ashish Chauhan and Balbir Kaith have widely used it for polymer characterization [21-24].
Studying phase transitions by PXRD
Under certain conditions, such as 0°C for water at 1 atm, a new arrangement of atoms or molecules may become stable, leading to a phase transition. At this point new diffraction peaks will appear or old ones disappear according to the symmetry of the new phase. If the material melts to an isotropic liquid, all sharp lines will disappear and be replaced by a broad amorphous pattern. If the transition produces another crystalline phase, one set of lines will suddenly be replaced by another set. In some cases however lines will split or collapse, e.g. if the material undergoes a continuous, second order phase transition. In such cases the symmetry may change because the existing structure is distorted rather than replaced by a completely different one. For example, the diffraction peaks for the lattice planes (100) and (001) can be found at two different values of q for a tetragonal phase, but if the symmetry becomes cubic the two peaks may coincide [16,17].
Size and strain broadening
Peak size broadening in PXRD pattern gives many informations. There are many factors that cause peak broadening such as instrumental factors, the presence of defects, differences in strain, size of the crystallites. It is easy to calculate the effects of size and strain. Where size broadening is independent of q (K=1/d), strain broadening increases with increasing q-values. In most cases there will be both size and strain broadening. It is possible to separate these by combining the two equations in what is known as the Hall-Williamson method:
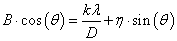
Thus, when we plot 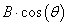 vs.
vs. 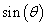 we get a straight line with slope η and intercept
we get a straight line with slope η and intercept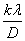 The value of η is the strain in the crystallites, the value of D represents the size of the crystallites. The constant k is typically close to unity and ranges from 0.8 to 1.39 [13,16,17].
The value of η is the strain in the crystallites, the value of D represents the size of the crystallites. The constant k is typically close to unity and ranges from 0.8 to 1.39 [13,16,17].
Conclusion
Advances in PXRD instrumentation and software have improved the efficacy of pharmaceutical drugs, efficiency of solid state characterization and rapid analysis of composites and materials. Variable temperature and humidity techniques are especially powerful in understanding structural changes of pharmaceutical drugs to enhance the stability and screen the drug substances under variable conditions. Ongoing developments involving structure determination directly from PXRD are encouraging to determine the crystal structures of new synthesized compounds and in pharmaceuticals. XRD in association with DSC, TGA, DTA, FTIR techniques can solve numerous problems encountered in industries for pharmaceutical formulation and pharmaceutic developments. The excellence of this technique is catering to academic research and commercial use in industries. Powder X-Ray Diffraction technique is a key analytical technique to serve the growth and development of Science and Technology.
Acknowledgements
I am grateful to God for blessing me assets of my life in the guise of parents. I thank my mother, Smt. Madhvi Chauhan and father, Dr Kripal Singh Chauhan for all their efforts in my favor and precious guidance.
References
- Sharma BK (2000) Instrumental Methods of Chemical Analysis. Krishna Prakashan Media, Meerut, India, 514.
- Dann SE (2002) Reactions and Characterization of Solids. Royal Society of Chemistry, USA, 10.
- Skoog DA, Holler FJ, Crouch SR (2007) Principles of Instrumental Analysis. Sixth Edition, Thomson Brooks, USA.
- Kishi A (2011) Detailed observations of dynamic changes such as phase transitions, melting and crystallization using an XRD-DSC with a high-speed, high-sensitivity two dimensional PILATUS detector. Rigaku J 27: 9-14.
- Borghettia GS, Carinia JP, Honoratob SB, Ayalab AP, Moreirac JCF, et al. (2012) Physicochemical properties and thermal stability of quercetin hydrates in the solid state. Thermochim Acta 539: 109-114.
- Vogt FG, Williams GR (2010) Advanced approaches to effective solid-state analysis: X-ray diffraction, vibrational spectroscopy, and solid-state NMR. Am Pharm Rev 13: 58-65.
- Suzuki T, Araki T, Kitaoka H, Teradab K (2012) Characterization of nonstoichiometric hydration and the Dehydration Behavior of Sitafloxacin Hydrate. Chem Pharm Bull (Tokyo) 60: 45-55.
- Vogt FG, Dell’Orco PC, Diederich AM, Su Q, Wood JL, et al. (2006) A study of variable hydration states in topotecan hydrochloride. J Pharm Biomed Anal 40: 1080-1088.
- Teng J, Bates S, Engers DA, Leach K, Schields P, et al. (2010) Effect of water vapor sorption on local structure of poly(vinylpyrrolidone). J Pharm Sci 99: 3815-3825.
- Hušak M, Jegorov A, Brus J, van Beek W, Pattison P, et al. (2008) Metergoline II: structure solution from powder diffraction data with preferred orientation and from microcrystal. Struct Chem 19: 517-525.
- Fraundorf P, Shuhan Lin (2004) Spiral powder overlays. Microscopy and Microanalysis. 10: 1356-1357.
- Cullity BD (1978) Elements of X-ray Diffraction. Addison Wesley, USA.
- Liss KD, Bartels A, Schreyer A, Clemens H (2003) High-Energy X-Rays: A tool for Advanced Bulk Investigations in Materials Science and Physics. Textures and Microstructures 35: 219-252.
- Chien WM, Chandra D, Franklin J, Rawn CJ, Helmy AK (2005) X-Ray diffractormetry studies and lattice parameter calculation on KNO3-NH4NO3 solid solutions, International Centre for Diffraction Data. Advances in X-Ray Analysis 48: 39-44.
- Li P, Chu Y, Wang L, Wenslow RM Jr, Yu K, et al. (2014) Structure Determination of the theophylline-nicotinamide cocrystal: a combined powder XRD, 1D solid state NMR and theoretical calculation study. Crys Eng Comm 16: 3141-3147.
- Skakle J (2005) Application of X-Ray powder diffraction in Material Chemistry. Chem Recources 5: 252-262.
- Bearden JA (1967) X-Ray Wavelengths. Reviews of Modern Physics 39: 78-124.
- Chyall LJ (2012) Current Applications of X-Ray Powder Diffraction in the Pharmaceutical Industry. Chyall Pharma, US.
- Chauhan A, Kaith B (2012) X-Ray Powder Diffraction Studies to Evaluate the Transition in Graft-copolymers Procured from Roselle fiber. Journal of Natural Fiber 9: 87-97.
- Chauhan A, Kaith B (2011) X-Ray Diffraction Studies and Assessment of Roselle Graft-copolymers. Malaysian Polymer Journal 6: 155-164.
- Chauhan A, Singh B (2011) X-Ray Powder Diffraction Studies to Accredit the Morphological Transformations in Hibiscus sabdariffaGraft-copolymers. International Journal of Polymer Analysis and Characterization 16: 319-328.
- Chauhan A, Kaith B (2013) XRD and Physico-chemical Evaluation of Hibiscus sabdariffa cellulose-Butyl acrylate-co-vinyl monomer graft. American Journal of Biochemistry and Molecular Biology 3: 61-70.
- Chauhan A, Kaith B (2012) XRD Elaborate the Metamorphosis in Graft Copolymers. J Anal Bioanal Tech 3: 1-6.
- Singha AS, Kaith BS, Chauhan A (2006) X-Ray Diffraction Studies and Thermogravimetric / Differential Thermal Analysis of Graft Co-polymers of Methylacrylate onto Hibiscus sabdariffa Fiber. J Polym Mat 23: 349-356.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 90113
- [From(publication date):
November-2014 - Aug 25, 2025] - Breakdown by view type
- HTML page views : 82673
- PDF downloads : 7440