Research Article Open Access
Resection of Pancreas and Duodenum During Radical Nephrectomy: Analysis of A Single Center Experience
Juan Chipollini1 and Gaetano Ciancio1,2*
1Department of Urology, University of Miami Miller School of Medicine, Miami, Florida, USA
2Department of Surgery, Division of Transplantation, Miami Transplant Institute, University of Miami Miller School of Medicine, Jackson Memorial Hospital, Miami, Florida, USA
- Corresponding Author:
- Gaetano Ciancio
M.D., University of Miami Miller School of Medicine
Department of Surgery and Urology, University of Miami Miller
School of Medicine, Jackson Memorial Hospital, Miami Fl
Miami Transplant Institute, P.O. Box 012440, Miami, Fl 33101, USA - Tel: (305) 355-5460
Fax: (305) 355-5797
E-mail: gciancio@med.miami.edu
Received date: February 26, 2016; Accepted date: April 25, 2016; Published date: April 29, 2016
Citation: Chipollini J, Ciancio G (2016) Resection of Pancreas and Duodenum During Radical Nephrectomy: Analysis of A Single Center Experience. J Gastrointest Dig Syst 6:420. doi:10.4172/2161- 069X.1000420
Copyright: © 2016 Chipollini J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Introduction: To analyze our experience of radical nephrectomy (RN) with en-bloc resection of neighboring organs for T4 renal cell carcinoma (RCC) based on our experience with multivisceral transplantation techniques.
Materials and Methods: After Institutional Review Board approval, a retrospective review was performed for patients who underwent RN and resection of adjacent organs and with a minimum follow up of 6 months.
Results: Ten patients underwent RN along with resection of pancreas (80%) and 2nd portion of duodenum (30%). Other organs simultaneously removed were inferior vena cava (IVC) (30%), spleen (70%), adrenal (100%), and left colon (10%). Major complications (Clavien-Dindo grade ≥ III) were seen in 6 patients (60%). The overall recurrence rate was 70%.The 2-year overall survival (OS) was 42.9 ± 13% and 2-year disease free survival (DFS) 28.6 ± 4.6%.
Conclusions: Those patients that undergo complete resection of T4 RCC face a high recurrence rate, but some have potential for durable survival response. Multi-institutional studies are needed to determine those patients that may benefit from aggressive surgical interventions, and should be considered for clinical trials of adjuvant targeted therapies.
Keywords
Advanced renal cell carcinoma; Pancreas; Duodenum; Organ resection
Introduction
Approximately one third of RCC cases make their debut in advanced stages of the disease [1,2]. Most of the 14,080 estimated deaths from RCC in 2015 presented with locoregional or widespread disease [3]. While surgical extirpation has been the mainstay for smaller lesions, the role and timing of surgery in cases of aggressive disease with adjacent invasion in the era targeted therapies remains controversial [4].
Resection for locally advanced RCC has increasingly been performed with acceptable morbidity and mortality rates in the last few years [5,6], and there are some series describing such an aggressive approach with modest survival benefit in this group of patients[7,8]. Furthermore, clinical and pathological stage discrepancy is common in surgically treated patients which can have significant impact on clinical outcome [9].
We report our surgical experience of this unique group of patients with confirmed T4 disease invading neighboring organs in which complete removal was achieved by means of RN in conjunction with en-bloc resection of adjacent organs such as pancreas and duodenum.
Materials and Methods
After obtaining Institutional Review Board approval, a retrospective chart review was performed on patients who underwent surgical treatment of locally advanced RCC along with resection of adjacent organs and had a minimum of 6 month follow up. Patients were followed regularly every 3 months with history and physical examinations as well as chest and abdominal imaging every 3-6 months. All relevant data on demographics, clinical presentation, disease characteristics, surgical details, survival outcomes and postoperative complications were collected and analyzed. All observations were censored at the end of the study period (April 2015). OS was measured from the date of surgery to the date of death from any cause; DFS was measured from the date of surgery to the date of locoregional recurrence or distant metastases. Postoperative complications were assessed using the Clavien-Dindo Classification System [10], and defined as occurring within a 30-days period of intervention date. Due to the small sample size, no logistic regression analysis was feasible.
Operative details
Our techniques for en-bloc mobilization of spleen and pancreas, as well as anterior mobilization of the liver in order to facilitate resection of large renal tumors have been previously described [11-13]. For left sided exposure, once the descending colon is adequately mobilized, we dissect the spleen off the diaphragm and mobilize en-bloc with the pancreas toward the midline, thereby exposing the entire upper retroperitoneal space from the diaphragm to the inferior border of the kidney. When caval exploration is necessary, we prefer to fully mobilize the liver as previously described [14-16]. This allows for wide exposure of central retroperitoneal structures such as the abdominal aorta, IVC, root of the mesentery, and portions of the pancreas and duodenum.
Results
Patient characteristics
In total 10 patients underwent RN and resection of neighboring visceral structures as described (Table 1). Median follow-up was 25 months (SD ± 10.8). None of the patients received pre-surgical targeted therapies for neoadjuvant purposes. Except for a single case (10%) in which an incidental mass was found on imaging, majority of patients were symptomatic (90%).
| Parameter | Value |
|---|---|
| No of patients | 10 |
| Male, n (%) | 5 (50%) |
| Age, median (y) ± SD | 53 ± 10.6 |
| Follow up, median (mo) ± SD | 25 ± 10.8 |
| Asymptomatic, n (%) | 1 (10%) |
| Symptomatic, n (%) | 9 (90%) |
| Abdominal pain | 3 (33%) |
| Hematuria | 2 (22%) |
| Fatigue | 2 (22%) |
| GI bleeding | 1 (11%) |
| Collapsed lung | 1 (11%) |
| EBL, median (mL) | 675 (range, 150-2500) |
| Transfusion, median (units) | 2.5 (range, 0-8) |
| Pancreaticoduodenectomy, n (%) | 1 (10%) |
| Partial pancreatectomy, n (%) | 3 (30%) |
| Distal pancreatectomy, n (%) | 4 (40%) |
| Other organs removed, n (%) | |
| Partial duodenum | 2 (20%) |
| Spleen | 7 (70%) |
| Adrenal gland | 10 (100%) |
| Left colon | 1 (10%) |
Table 1: Demographic information and operative details.
Complaints ranged from abdominal pain (33%), hematuria (22%), fatigue (22%), and gastrointestinal bleeding (11%) due to tumor erosion into duodenum. One patient had M1 disease at the time of surgery after bronchoscopy confirmed endobronchial RCC metastases. One patient had equivocal signs of T4 disease on imaging but the majority was confirmed intraoperatively. None of the patients had obstructive jaundice.
Operative outcomes and postoperative complications
At the time of resection, neoplastic extension to the pancreas was seen in 8 patients (80%), mostly coming from left sided tumors (right to left ratio; 1:7). The second portion of duodenum was affected in two right-sided tumors (20%) requiring partial duodenectomy, while 1 case (10%) had both pancreas and duodenum involvement requiring a pancreaticoduodenectomy (i.e., Whipple procedure) (10%). The other pancreatic resections consisted of subtotal pancreatectomy (30%) and distal pancreatectomy (40%) depending if body and tail or tail of the pancreas were involved, respectively. The spleen and the ipsilateral adrenal were also en-bloc excised in 7 (70%) and 10 (100%) cases, respectively. An extended lymphadenectomy completed the procedure in all cases (100%). Intracaval involvement was additionally detected in three of the patients (30%): one case of level II and two cases of level III (20%) thrombi as described per Neves and Zincke [17]. There were no perioperative deaths. Major complications (Clavien-Dindo grade ≥ III) were seen in 6 patients (60%). The most common postoperative complications were intraabdominal collection (30%) and pulmonary complications (20%).
Histological details and survival outcomes
Histological examination revealed clear cell RCC in 9 (90%) of the specimens and chromophobe type in another (10%). The median tumor size was 15.1 cm (SD ± 3.92). There were no positive margins in all pancreatectomy specimens. There were 2 positive renal vein margins. A total of 43 lymph nodes were assessed and 1 showed involvement. All tumors were T4 as per American Joint Committee on Cancer (AJCC) 2010 TNM staging system [18]. The 2-year OS was 42.9 ± 13% and 2-year DFS 28.6 ± 4.6% (Table 2).
| Variable | Value |
|---|---|
| AJCC pT4, n (%) | 10 (100) |
| RCC conventional type, n (%) | 9 (90) |
| Tumor grade, median ± SD | 4 ± 0.85 |
| Longest diameter of tumor, median ± SD (cm) | 15.1 ± 3.92 |
| Positive margins, n (%) | 2 (20) |
| Lymph node metastases, n (%) | 1 (2.3) |
| Median survival from time of resection ± SE (months) | 16.5 ± 10. |
| 2-yr OS ± SE (%) | 42.9 ± 13 |
| 2-yr DFS ± SE (%) | 28.6 ± 4.6 |
Table 2: Pathological characteristics and survival outcomes.
Discussion
With the widespread use of computer tomography, the classic triad of flank pain, hematuria, and flank mass is infrequently seen today and only a third of RCC cases present with advanced disease. Adjacent visceral invasion is rare in RCC, likely due to the protective nature of the anterior and posterior renal fasciae [19]; In cases where RCC has spread to other organs, aggressive surgical extirpation such as upfront nephrectomy with or without metastasectomy has been advocated as the standard of care for those fit to withstand aggressive surgical interventions [20,21].
The current literature is nonetheless scarce when it comes to describing nephrectomy and concomitant resection of other organs. In one series, colon, pancreas, and diaphragm were the most commonly resected organs in patients (n = 12) with T4 disease. Although 10 patients developed recurrence at a median time of 2.3 months, 5 (42%) patients were still alive at the time of analysis [7]. In another study of 26 patients at the Memorial Sloan Kettering Cancer Center (MSKCC), pancreas (23%) and bowel (23%) resections were among the more commonly resected organs, along with liver (39%), IVC (35%), vertebral body (31%), spleen (12%), and psoas muscle (7%); with 4 (15%) of those patients still alive at follow up [8].
In our cohort, median time to recurrence was 6.5 months for those that eventually died vs 20 months for patients currently alive (p = 0.00018). While several risks scores and nomograms are available for localized RCC [1,22,23], there are no validated algorithms that differentiate patients with locally advanced RCC that may benefit from surgical intervention vs those who are destined to progress rapidly. In the setting of metastatic RCC (mRCC), clinical factors have already been evaluated as prognostic tools.
Motzer and associates evaluated pretreatment clinical features in 670 patients enrolled in Phase II and Phase III clinical trials at MSKCC in order to create a multivariate model that can predict survival. Prognostic factors associated with shorter survival were low Karnofsky performance status (KPS) (<80%), lactate dehydrogenase level (LDH) >1.5 x the upper limit of normal , hemoglobin level < the lower limit of normal, high corrected serum calcium level (>10 mg/dL), and absence of nephrectomy. The median time to death in patients with 0 risk factors (favorable risk), 1 or 2 risk factors (intermediate risk), and ≥3 risk factors (poor risk) were 20 months, 10 months, and 4 months, respectively [24].
The MSKCC model was later modified in 2002 and was restricted to 400 patients who received IFN-α. Five variables were used as risk factors for short survival: low KPS, high LDH, low serum hemoglobin, high corrected serum calcium, and time from initial RCC diagnosis to start of IFN-α therapy of less than one year. The median time to death for patients deemed favorable, intermediate, and poor risk was 30 months, 14 months, and 5 month, respectively [25].
Fortunately, advances in the understanding of molecular and genetic contributors of RCC have led to the emergence of newer targeted therapeutic agents for its treatment, as well as an increased interest in finding new molecular biomarkers for prognostic and therapeutic purposes.
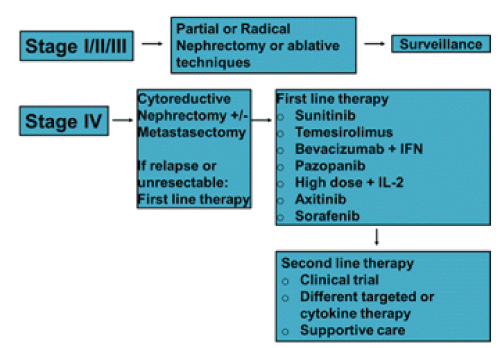
The treatment of advanced RCC has evolved significantly following the identification of the von Hippel-Lindau gene and the subsequent development of antiangiogenic therapies. There are currently 8 US Food and Drug Administration approved agents available for the treatment of mRCC.
Five of these agents target either the vascular endothelial growth factor or its receptors, two inhibit activity of the mammalian target of rapamycin, and one is a recombinant form of the endogenous cytokine IL-2 (Figure 1).
Due to this better understanding of RCC tumorigenesis, targeted molecular therapies have emerged as equally effective alternatives to cytokines, and have shown a survival benefit when combined with surgery in m RCC [26]. Nevertheless, the role of targeted agents for nonmetastatic high risk disease is still controversial today. Several phase 3 trials of adjuvant treatments with sunitinib, sorafenib, pazopanib, axitinib, and everolimus are currently ongoing. Thus far, one study (NCT00326898) has shown no differences in DFS or OS in sunitinib and sorafenib treated patients vs placebo at interim analysis, although there was significant discontinuation rate due to low tolerability [27].
At last follow-up there are currently 5 patients still alive. One patient was able to live for a little over 8 years before death. Despite the majority of patients having negative surgical margins, there was an overall 70% recurrence rate among the cohort which was comparable to the MD Anderson (83%) and MSKCC (76%) experiences for T4 patients. A contemporary, multi-institutional cohort is much needed to validate risk factors that can further improve prognostication, risk stratification, and survival outcomes for patients with advanced RCC. The retrospective nature and small number of patients are acknowledged limitations of this study. Even for large tertiary referral centers, this patient population with locally advanced RCC and adjacent organ involvement can be slow to accrue and enrollment in ongoing adjuvant therapy clinical trials should be encouraged and offered to all eligible patients.
Conclusion
We present a series of patients with locally advanced RCC with involvement of adjacent viscera. Although these patients have a great risk for recurrence, there is potential for durable response. Results of prospective randomized trials are eagerly awaited regarding the use of adjuvant targeted therapies for these high risk patients.
References
- Frank I (2002) An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. J Urol 168: 2395-2400.
- Lam JS, Belldegrun AS, Pantuck AJ (2006) Long-term outcomes of the surgical management of renal cell carcinoma. World J Urol 24: 255-266.
- Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65: 5-29.
- Ljungberg B, Bensalah K, Canfield S, Dabestani S, Hofmann F, et al. (2015) EAU guidelines on renal cell carcinoma: 2014 update. EurUrol 67: 913-924.
- Sperti C, Moletta L, Patanè G (2014) Metastatic tumors to the pancreas: The role of surgery. World J GastrointestOncol 6: 381-392.
- Santoni M, Conti A, Partelli S, Porta C, Sternberg CN, et al. (2015) Surgical resection does not improve survival in patients with renal metastases to the pancreas in the era of tyrosine kinase inhibitors. Ann SurgOncol 22: 2094-2100.
- Margulis V (2007)Renal cell carcinoma clinically involving adjacent organs: experience with aggressive surgical management. Cancer 109: 2025-2030.
- Karellas ME, Jang TL, Kagiwada MA, Kinnaman MD, Jarnagin WR, et al. (2009) Advanced-stage renal cell carcinoma treated by radical nephrectomy and adjacent organ or structure resection. BJU Int 103: 160-164.
- Svatek RS, Lotan Y, Herman MP, Duchene DA, Sagalowsky AI, et al. (2006) The influence of clinical and pathological stage discrepancy on cancer specific survival in patients treated for renal cell carcinoma. J Urol 176: 1321-1325.
- Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, et al. (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250: 187-196.
- Ciancio G (2009) En bloc mobilization of the pancreas and spleen to facilitate resection of large tumors, primarily renal and adrenal, in the left upper quadrant of the abdomen: techniques derived from multivisceral transplantation. EurUrol 55: 1106-1111.
- Ciancio G, Gonzalez J, Shirodkar SP, Angulo JC, Soloway MS (2011) Liver transplantation techniques for the surgical management of renal cell carcinoma with tumor thrombus in the inferior vena cava: step-by-step description. EurUrol 59: 401-406.
- CiancioG,Livingstone AS, Soloway M (2007) Surgical management of renal cell carcinoma with tumor thrombus in the renal and inferior vena cava: the University of Miami experience in using liver transplantation techniques. EurUrol 51: 988-994.
- Ciancio G, Vaidya A, Soloway M (2003) Early ligation of the renal artery using the posterior approach: a basic surgical concept reinforced during resection of large hypervascular renal cell carcinoma with or without inferior vena cava thrombus. BJU Int 92: 488-489.
- Ciancio G, Vaidya A, Savoie M, Soloway M (2002) Management of renal cell carcinoma with level III thrombus in the inferior vena cava. J Urol 168: 1374-1377.
- Ciancio G, Hawke C, Soloway M (2000) The use of liver transplant techniques to aid in the surgical management of urological tumors. J Urol 164: 665-672.
- Neves RJ, Zincke H (1987) Surgical treatment of renal cancer with vena cava extension. Br J Urol 59: 390-395.
- Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann SurgOncol 17: 1471-1474.
- Raptopoulos V, Lei QF, Touliopoulos P, Vrachliotis TG, Marks SC Jr (1995) Why perirenal disease does not extend into the pelvis: the importance of closure of the cone of the renal fasciae. AJR Am J Roentgenol 164: 1179-1184.
- Russo P, Synder M, Vickers A, Kondagunta V, Motzer R (2007) Cytoreductive nephrectomy and nephrectomy/complete metastasectomy for metastatic renal cancer. ScientificWorldJournal 7: 768-778.
- Bhat S (2010) Role of surgery in advanced/metastatic renal cell carcinoma. Indian J Urol 26: 167-176.
- Sorbellini M, Kattan MW, Snyder ME, Reuter V, Motzer R, et al. (2005) A postoperative prognostic nomogram predicting recurrence for patients with conventional clear cell renal cell carcinoma. J Urol 173: 48-51.
- Karakiewicz PI (2007) Multi-institutional validation of a new renal cancer-specific survival nomogram. J ClinOncol 25: 1316-1322.
- Motzer RJ, Bacik J, Mazumdar M (2004) Prognostic factors for survival of patients with stage IV renal cell carcinoma: memorial sloan-kettering cancer center experience. Clin Cancer Res, 10: 6302S-6303S.
- Motzer RJ, Bacik J, Murphy BA, Russo P, Mazumdar M (2002) Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J ClinOncol 20: 289-296.
- Flanigan RC, Salmon SE, Blumenstein BA, Bearman SI, Roy V, et al. (2001) Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med 345: 1655-1659.
- Haas NBMJ, Uzzo RG (2015) Initial results from ASSURE (E2805): Adjuvant sorafenib or sunitinib for unfavorable renal carcinoma, an ECOG-ACRIN-led, NCTN phase III trial in J ClinOncol.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 11477
- [From(publication date):
April-2016 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 10498
- PDF downloads : 979