Research Article Open Access
Safety of Blood Transfusion: An Egyptian Study
| Dalia Omran1*, Eiman A Hussein2 and Mohamad Nagib3 | ||
| 1 Department of Tropical Medicine, Cairo University, Egypt | ||
| 2 Departments of Clinical Pathology and Hepatology, Cairo University, Egypt | ||
| 3 Department of Internal Medicine, Cairo University, Cairo, Egypt | ||
| Corresponding Author : | Dalia Omran Faculty of Medicine Department of Tropical Medicine Cairo University, Nasr City, Egypt Tel: +(202)01000087802 Fax: +(202)23649281 E-mail: daliaomran2007@yahoo.com |
|
| Received December 04, 2013; Accepted December 13, 2013; Published December 17, 2013 | ||
| Citation: Omran D, Hussein EA, Nagib M (2013) Safety of Blood Transfusion: An Egyptian Study. J Infect Dis Ther 1:124. doi: 10.4172/2332-0877.1000124 | ||
| Copyright: © 2013 Omran D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | ||
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
||
Visit for more related articles at Journal of Infectious Diseases & Therapy
Abstract
Introduction: Blood safety presents a serious challenge in Egypt, having the highest prevalence of hepatitis C virus (HCV).
Objectives: To evaluate the effectiveness of blood donor recruitment strategies, the seroprevalence of positive infectious markers among volunteer donors (VD) and family replacement donors (RD) at a University Hospital Blood Bank was studied.
Methodology: Donors’ records covering the period between 2006 and 2012 were reviewed. All donations had been tested using the following screening tests ; Ortho for HCV antibody (enzyme-linked immunosorbent assay test system Version 3.0, Ortho Clinical Diagnostics, Inc., Raritan, NJ) and Siemens (Siemens Enzygnost assay, Healthcare Diagnostic Products, Marburg, Germany) for HBsAg (Enzygnost HBsAg 6.0), human immune deficiency virus (HIV)-1 and HIV-2 antibodyies (Enzygnost HIV integral II), and syphilis antibodies. Reactive samples were not confirmed with additional tests.
Results: Of 308,762 donors, 63.4% were VD. The overall prevalence of HCV antibodies, HBsAg, human immunodeficiency virus (HIV) and syphilis antibodies were 4.3%, 1.22%, 0.07%, and 0.13%, respectively. After stratification for age and gender, all tested markers (except HIV) were significantly higher among RD.A significant decline in HCV seropositivity was observed in RD and VD from 8.9% and 4.2% in 2007/2008 to 3.8% and 1.5% in 2011/2012, respectively. A significant decrease in HBsAg was demonstrated in VD from 1.2% in 2007/2008 to 0.53% in 2011/2012. During the period 2007- 2011, an increasing trend for HIV and syphilis was noted.
Conclusion: The decreasing trend in HCV antibodies and HBsAg could be related to the strict donor selection criteria.
| Keywords | |
| Blood transfusion; Safety; Screening; Infections | |
| Introduction | |
| Although transfusion of human blood can be life saving, it is not without risks. Transfusion-transmitted infections (TTI) can be transmitted through blood transfusions [1]. The TTI problem is directly proportional to the prevalence of infections among blood donors [2]. Hepatitis B virus (HBV), hepatitis C virus (HCV), Human immune deficiency virus (HIV) and syphilis are the most important lethal agents in TTIs. The incidence rates of these infections are hard to calculate due to the asymptomatic and the latent nature of these diseases [3]. | |
| Blood safety presents a serious challenge in Egypt, having the highest recorded prevalence of HCV antibodies in the world. Prevalence of HCV was reported to be 13.9% among healthy populations. Adults have higher HCV prevalence (15.7%) than children (4.0%). Geographically, HCV prevailed more in the Nile delta (15.8%) than in Upper Egypt (9.02%) [4]. | |
| The route of transmission of hepatitis C virus is still controversial and complicated. Transmission of blood or blood products leads to infection in the majority of cases. While the majority of intravenous drug users become infected by repetitive exposure to shared contaminated injection equipments, Needle-stick injuries in health-care settings represent another high risk category. HCV is less commonly transmitted through; having sex with an infected person, being born to an HCV-infected mother, or sharing of personal items contaminated with infectious blood [5]. | |
| Given the constrained economy which limits the implementation of sensitive screening techniques as nucleic acid amplification technology (NAT), proper recruitment of blood donors becomes of paramount importance. | |
| In an attempt to evaluate the effectiveness of blood donor recruitment strategies, the seroprevalence of positive infectious markers among volunteer donors (VD) and family replacement donors (RD) at Cairo University Hospital Blood Bank was studied. | |
| Methodology | |
| This study was a hospital based retrospective one. The data was collected from the blood bank of Cairo University hospital. Cairo University hospital comprises more than 5200 beds representing the largest medical institute in the Middle East and probably one of the largest centres in the world. The study protocol was approved by the institutional committee for the protection of human subjects and conformed to the guidelines of the 1975 Declaration of Helsinki. | |
| Donors’ records covering the period from October 2006 to September 2012 were reviewed. All donations had been tested using the following screening tests; Ortho for HCV antibody (enzymelinked immunosorbent assay test system Version 3.0, Ortho Clinical Diagnostics, Inc., Raritan, NJ) and Siemens (Siemens Enzygnost assay, Healthcare Diagnostic Products, Marburg, Germany) for HBsAg (Enzygnost HBsAg 6.0), human immune deficiency virus (HIV)-1 and HIV-2 antibodies (Enzygnost HIV integral II), and syphilis antibodies. Reactive samples were not confirmed with additional tests. | |
| The age of VD and RD was determined for the period of one year (October 2011-September 2012) and compared. | |
| Statistical Analysis | |
| Data was statistically analyzed with SPSS version 17.0 for windows 7. P value less than 0.05 was considered statistically significant. | |
| Results | |
| Of 308,762 donors, 63.4% were VD with male predominance (256,272) representing 83% of the total donors. | |
| The prevalence of positive infectious markers among RD and VD during the period of the study is presented in Table 1. | |
| After stratification for age and gender, The total prevalence rates of HBV, HCV and syphilis markers for 6-year study period among RD was significantly higher as compared to VD (p=0.03, p<0.001 and p+0.02, respectively). | |
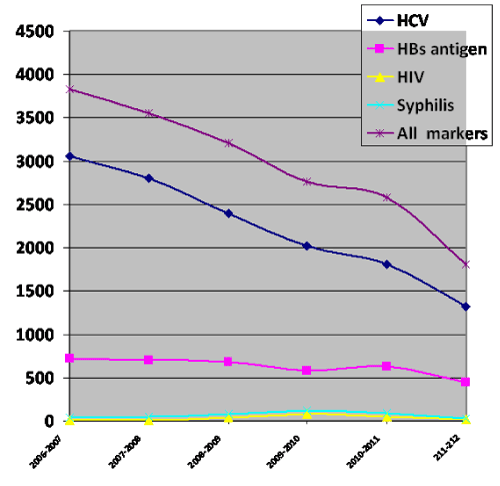
| A consistent significant steady decline in the seropositivity for HCV antibodies was observed in both RD and VD from 8.9% and 4.2% in 2007/2008 to 3.8% and 1.5%% in 2011/2012, respectively (P=0.0001). A steady significant decrease in HBs antigen was demonstrated in VD from 1.2% in 2007/2008 to 0.53% in 2011/2012 (P=0.004). | |
| A significant decline of HBs antigen was observed in RD, only during 2011-212, when compared to the previous years (P=0.02). | |
| During the period between 2007- 2011, increasing trends for HIV and syphilis reactivity rates were noted among both RD and VD (Table 1). | |
| The overall prevalences of anti HCV antibodies, HBs antigen, HIV antibody and syphilis antibody were 4.3%, 1.22%, 0.07%, and 0.13%, respectively (Figure 1). | |
| Discussion | |
| In our study, the overall seroprevalence of HBsAg was 1.22% (1.7%/1.1% in RD/VD respectively), The prevalence of HBsAg reactivity using ELISA technique in an Egyptian study was 1.30% in the period 2000-2005 [6] and was 2.13% in the preceding 5 years (1995- 2000) [7]. The prevalence of HBsAg in a study done among student voluntary blood donors in Mansoura University in the academic year 2002-2003 was 4.3% [8]. Those students were probably not vaccinated against HBV; at the time of their birth, the vaccine was not included in the Expanded Programme of Immunization in Egypt. | |
| The decrease in HBV prevalence suggests an effec¬tive screening of blood and blood products. Despite the decline in the prevalence of HBsAg among Egyptian poly¬transfused patients over the past 20 years, it is still high when compared to other parts of the world. This could be attributed to the high infection rates as Egypt has intermediate levels of HBV infection (2–7% chronic infec¬tion) [9]. In Egypt HBV transmission is apparently a mixture of horizontal and perinatal transmission. However, the majority of HBV infection is acquired by the former route [10]. | |
| Screening in most Egyptian blood banks is performed for HBsAg only but not for core antibodies or ALT. El-Za¬yadi et al. [11] called for implementing the anti-HBc test as a routine assay. Altindi�?�? et al. [12] recommended screening of blood units by sensitive PCR-based methods even if they were negative for HBsAg, to unreveal possible occult HBV infections. This can prevent or at least decrease the transmission risk of HBV infection, which is still an important health problem. A recent Egyptian study showed that occult hepatitis B infection detected by HBV-DNA testing constituted 32.4% of polytransfused children [13]. | |
| The prevalence of HCV in Egypt in 1997 was 69% among polytransfused children and 55% among blood donors [14]. In the period between 2000- 2005, The overall average prevalence of HCV antibodies among Egyptian blood donors was 4.04% when estimated in the period between 2000-2005 [6] compared to 6.88% in the preceding 5 years (1995-2000) [7]. The prevalence of HCV seropositivity among blood donors in Minya Governorate (Upper Egypt) in the period between 2000-2008 was estimated to be 9.02%. In Mansora University, 2.7% of the student blood donors were positive for HCV [8]. | |
| In the present study, the overall prevalence of HCV was 4.3% (6.2%/ 3.3% among RD/VD respectively). A consistent steady decline in the seropositivity for HCV antibody was observed in both RD and VD from 8.9% and 4% in 2006/2007 to 3.8% and 1.5% in 2011/2012, respectively (P=0.0001). Similarly, a recent Egyptian study revealed that the prevalence of HBsAg and anti-HCV antibodies was 1.4% and 3.5%, respectively [15]. | |
| The significant decreasing trends in HCV antibodies (P=0.0001) in the present study could be related to the strict donor selection criteria (predonation screening for risk factors of viral hepatitis). Given the low predictive value of the single-positive HCV-ELISA, performance of repeated ELISA tests and PCR-based methods is widely recommended. | |
| It had been reported that the overall prevalence of HIV-1 infection among the Egyptians was 0.18% (54/29261); of which 4.8% (28/582) were blood and factor VIII recipients, 0.15% (3/1961) drug addicts, 0.18% (3/1650) fever of unknown origin patients, 0.23% (6/2602) sexually transmitted disease patients, 1.9% (5/269) HIV-1 contacts, 0.07% (7/9778) international travelers, and 0.02% (2/12070) blood/ product donors [16]. | |
| The World Health Organization estimates a rate of 0.15% among the total Egyptian population. In 2000, only 0.04% of 16 559 voluntarily-tested Egyptians were positive for HIV [17]. A quarter of the reported HIV cases in Egypt were infected iatrogenically, especially in haemodialysis centers [18]. | |
| In a study done by NCI, the incidence of HIV antibody reactivity during the period extending from 2000 to 2005 was found to be 0.0% [6] compared to two cases 5 years before [7]. In the current study, the overall prevalence of HIV antibody was 0.07% (0.07%/0.07% for RD/ VD, respectively). A rising trend was observed in both RD and VD from 0.02% in 2006/2007 to 0.1% in 2010/2011. This rising burden of HIV among blood donors (who represent the healthy sector of the community) emphasizes a clear need for an integrated approach to the emerging threat of HIV. It is important, not neglect the high prevalence of HIV in neighboring African countries [19-23]. | |
| The overall prevalence of syphilis antibodies was 0.13% (0.2% / 0.1% for RD/VD, respectively). Increasing trends for syphilis reactivity rates were noted among both RD and VD (0.1% / 0.05%, respectively in 2006/2007 to 0.23%/ 0.13%, respectively in 2010/2011). | |
| In our study, the mean age of HCV positive donors was 36.8 ±9.6, while the mean age for HBsAg positive donors was 28 ± 7.1 years. In a previous study conducted to evaluate the incidence of infectious markers among donors during the Egyptian revolution, age was the most consistently associated factor with HCV prevalence. Prevalence of HCV increased with age from 0.5% in those below 30 years to a peak of 4.2% in the age group 40 to 53 years. Data of the same study also demonstrated a high incidence of HBsAg among adolescents and young adults below30 years of age (76.2%) [24]. HBsAg seroclearance with age had been documented as the main reason for decreasing HBsAg incidence with increasing age [25]. | |
| The main limitation of this study was that; it was conducted among donors from blood bank of one university hospital, the results may not be generalizable to all blood banks in Egypt | |
| Only a national epidemiologic study can provide definitive informations about the overall prevalence of the studied infectious diseases in Egypt. Lacking such a national sample survey, our study represents a useful compromise. | |
| Conclusion, the decreasing trends in HCV antibodies and HBsAg in VD over the last 6 years is a promising sign, and could be related to the strict donor selection criteria. It is important to implement sensitive screening techniques as nucleic acid amplification technology (NAT) and advocate more strategies on community education programs (more public health campaigns) to raise awareness about risky behaviors to limit further spread of HCV in Egypt. | |
| Acknowledgements | |
| We acknowledge workers in the blood bank for helping us in collecting the donors’ data. | |
References
- World Health Organization (WHO) 2010 Blood Safety Unit: Aide-Memoire for national blood programmes.
- Jain R, Jose B, Coshic P, Agarwal R, Deorari AK (2008) Blood and blood component therapy in neonates. Indian J Pediatr 75: 489-495.
- Bhawani Y, Rao PR, Sudhakar V (2010) Seroprevalence of transfusion transmissible infections among blood donors in a tertiary care hospital of Andhra Pradesh. Biology and Medicine 2: 45-48.
- Lehman EM, Wilson ML (2009) Epidemic hepatitis C virus infection in Egypt: estimates of past incidence and future morbidity and mortality. J Viral Hepat 16: 650-658.
- Tibbs CJ (1995) Methods of transmission of hepatitis C. J Viral Hepat 2: 113-119.
- Eissa SA, Abdel Meguid LM, Ebeid SM, Abou Elfetouh RM, Abdel Moneim GM (2007) National Cancer Institute Experience in Healthy Egyptian Blood Donors as Regards Blood Group Frequencies and Seroprevalence of Hepatitis B Virus, Hepatitis C Virus & HIV: 10 Year Evaluation. J Egypt Natl Canc Inst 19: 71-76
- Soheir AE, Lobna MA, Samir ME, Sherief AAN, Mahmud DF (2002) Prevalence of anti-HIV, HBs-Ag and anti-HCV reactivity in different categories of Egyptian blood donors: Experience of the National Cancer Institute in the last 5 years. J Egypt Nat Cancer Inst 14: 217-221.
- El-Gilany AH, El-Fedawy S (2006) Bloodborne infections among student voluntary blood donors in Mansoura University, Egypt. East Mediterr Health J 12: 742-748.
- El Gawhary S, Omar N, Abdel Rahman L, Mahmoud M (2009) Hepatic viruses screening in multitransfused Egyptian thalassemia patients. Jounal of Arab Children 20: 193-202.
- El-Zayadi A (2007) Hepatitis B virus infection: The Egyptian situation. Arab J Gastroenterol 8: 94-98.
- El-Zayadi AR, Ibrahim EH, Badran HM, Saeid A, Moneib NA, et al. (2008) Anti-HBc screening in Egyptian blood donors reduces the risk of hepatitis B virus transmission. Transfus Med 18: 55-61.
- Altindi�?�? M, Uslan I, Cetinkaya Z, Yüksel S, Ciftçi IH, et al. (2007) [Investigation of hemodialysis patients in terms of the presence of occult hepatitis B]. Mikrobiyol Bul 41: 227-233.
- Shaker O, Ahmed A, Abdel Satar I, El Ahl H, Shousha W, et al. (2012) Occult hepatitis B in Egyptian thalassemic children. J Infect Dev Ctries 6: 340-346.
- EL-Alfy M, EL-Sayed M (1997) Prevalence of hepatitis C among thalassemic children. Egytian J Haematology 22: 311-325.
- Khattab MA, Eslam M, Sharwae MA, Hamdy L (2010) Seroprevalence of hepatitis C and B among blood donors in Egypt: Minya Governorate, 2000-2008. Am J Infect Control 38: 640-641.
- Watts DM, Constantine NT, Sheba MF, Kamal M, Callahan JD, et al. (1993) Prevalence of HIV infection and AIDS in Egypt over four years of surveillance (1986-1990). J Trop Med Hyg 96: 113-117.
- (2002) Epidemiological fact sheets on HIV/AIDS and sexually transmitted infections, 2002 update: Egypt. Geneva, Joint United Nations Programme on HIV/AIDS/United Nations Children’s Fund/World Health Organization.
- (2003) Initiative for HIV voluntary counseling and testing. Cairo, Ministry of Health and Population.
- El Sayed NM, Gomatos PJ, Beck-Sagué CM, Dietrich U, von Briesen H, et al. (2000) Epidemic transmission of human immunodeficiency virus in renal dialysis centers in Egypt. J Infect Dis 181: 91-97.
- Jayaraman S, Chalabi Z, Perel P, Guerriero C, Roberts I (2010) The risk of transfusion-transmitted infections in sub-Saharan Africa. Transfusion 50: 433-442.
- Candotti D, Mundy C, Kadewele G, Nkhoma W, Bates I (2010) et al. Serological and molecular screening for viruses in blood donors from Ntcheu, Malawi: high prevalence of HIV-1 subtype C and of markers of hepatitis B and C viruses. J Med Virol 65: 1-5
- Kania D, Sangaré L, Sakandé J, Koanda A, Nébié YK et al. (2009) A new strategy to improve the cost effectiveness of human immunodeficiency virus, hepatitis B virus, hepatitis C virus, and syphilis testing of blood donations in sub-Saharan Africa: a pilot study in Burkina Faso. Transfusion 49: 2237-2240.
- Diarra A, Kouriba B, Baby M, Murphy E, Lefrere JJ (2009) HIV, HCV, HBV and syphilis rate of positive donations among blood donations in Mali: lower rates among volunteer blood donors. Transfus Clin Biol 16: 444-447.
- Hussein E, Teruya J (2012) Evaluation of blood supply operation and infectious disease markers in blood donors during the Egyptian revolution. Transfusion 52: 2321-2328.
- Tai DI, Tsay PK, Chen WT, Chu CM, Liaw YF (2010) Relative roles of HBsAg seroclearance and mortality in the decline of HBsAg prevalence with increasing age. Am J Gastroenterol 105: 1102-1109.
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
| Figure 1 |
Relevant Topics
- Advanced Therapies
- Chicken Pox
- Ciprofloxacin
- Colon Infection
- Conjunctivitis
- Herpes Virus
- HIV and AIDS Research
- Human Papilloma Virus
- Infection
- Infection in Blood
- Infections Prevention
- Infectious Diseases in Children
- Influenza
- Liver Diseases
- Respiratory Tract Infections
- T Cell Lymphomatic Virus
- Treatment for Infectious Diseases
- Viral Encephalitis
- Yeast Infection
Recommended Journals
Article Tools
Article Usage
- Total views: 17137
- [From(publication date):
February-2014 - Aug 17, 2025] - Breakdown by view type
- HTML page views : 12331
- PDF downloads : 4806
