Research Article Open Access
Source Rock Evaluation using Total Organic Carbon (TOC) and the Loss- On-Ignition (LOI) Techniques
Al-Selwi A and Joshi M*
Faculty of Chemical and Energy Engineering, Universiti Teknologi Malaysia, UTM, Malaysia
- *Corresponding Author:
- Joshi M
Faculty of Chemical and Energy Engineering
Universiti Teknologi Malaysia, UTM, Malaysia
Tel: +(6)075535559
E-mail: mollyj@petroleum.utm.my
Received date: September 08, 2015; Accepted date: November 17, 2015; Published date: November 21, 2015
Citation: Al-Selwi A, Joshi M (2015) Source Rock Evaluation using Total Organic Carbon (TOC) and the Loss-On-Ignition (LOI) Techniques. Oil Gas Res 1:105. doi:10.4172/2472-0518.1000105
Copyright: © 2015 Al-Selwi A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Oil & Gas Research
Abstract
This paper analyzes the methods used to determine Total Organic Carbon (TOC) content in rock samples. These methods include the High Temperature Catalytic Oxidation HTCO and the Loss-on-ignition (LOI). The study was conducted on shale samples. The main objective is to identify the potential petroleum productivity of these rocks. Two samples were tested. One is presumed to be an organic shale, while the other is a typical ‘inorganic shale” sample. In the Loss-on-ignition method, the effect of sample size and time of exposure on TOC values were also investigated. The results show that Loss-on-ignition yields higher values than the HTCO method. In addition, size and exposure time cause a significant effect on TOC values. Therefore, these two parameters have to be mentioned along with each TOC measurement.
Keywords
Kerogen; Source rocks; Total organic carbon (TOC); Loss-on-ignition (LOI); Total Carbon (TC)
Introduction
In hydrocarbon exploration and petroleum reservoir development, reservoir rock quality and source rock productivity are of utmost importance in oil and gas industry. Quantification of the source rock and an assessment of its potential to generate hydrocarbons is extremely important [1,2]. These two parameters predict the type of hydrocarbon to be expected and its quality. Kerogen type is identified by determining oxygen-carbon and hydrogen-carbon ratios, and the quality is determined by vitrine reflectance values [3,4]. Determination of the Total Organic Carbon is a fundamental part of source rocks evaluation, and it is a very critical determinant of the potential productivity of discovered oil reservoirs and therefore to the subsequent course of action of oil and gas companies [5,6]. Many source rocks also include inorganic sources of carbon such as carbonates and most notably calcite, dolomite, and siderite. These minerals break down at high temperature, generating carbon dioxide and thus their presence must be corrected in order to determine the organic carbon content [2].
There are a number of techniques for the determination of the Total Organic Carbon and the Total Carbon. These techniques are classified as qualitative, semi-qualitative, and quantitative.
For the qualitative technique, two methods are commonly used; one utilizes Nuclear Magnetic Resonance (NMR) spectroscopy and the other method is based on Diffuse Reflectance infrared Fourier Transform (DRIFT) spectroscopy. NMR is a very useful method in organic matter determination and humification procedure of rock samples. In the NMR technique, the organic matter extraction is not required; test is conducted for the whole rock sample. The downside of this method is that it is expensive and takes longer time [7,8]. DRIFT spectroscopy provides a rapid and inexpensive means of differentiating carbon forms in soils and sediments [8]. Both inorganic and organic compounds can be identified using this technique [9]. In his experiments, Rumpel et al., [8] determined the presence of lignite and its contribution to the total organic carbon reading. Quantification of lignite content was carried out by this technique. However, TOC was determined by a dry combustion method [8,9].
Semi-quantitative methods are based upon the unselective removal of all organic material using gravimetric sample weight loss determination. The two basic semi-quantitative techniques are Losson- Ignition (LOI) and Hydrogen Peroxide Digestion method. The hydrogen peroxide (H2O2) digestion method involves the destruction of the organic material in the rock sample by oxidation [10].
Quantitative methods include wet chemistry and dry chemistry techniques. Wet chemistry method is done by sample extraction and sample quantitation. Sample extraction employs the quick organic matter dichromate oxidation. Walkley-Black method is probably the best known of the rapid dichromate oxidation procedure [11]. Equation 1 shows Walkley-Black method reaction:
2Cr2O7 2- + 3CO + 16H+ = 4Cr3+ + 3CO2 + 8H2O (1)
Sample quantitation is conducted by manual titration, automated titration using potentiometric determination, calorimetry, gravimetric determination, or volumetric/manometric measurement [10].
On the other hand, dry chemistry technique is based on combusting the sample at high temperatures and turning it into CO2. This CO2 is pushed to detectors and quantified as Total Organic Carbon. The detection/quantification is conducted by titrimetric, gravimetric, manometric, spectrophotometric, or gas chromatographic techniques [12,13].
Loss On Ignition (LOI) is a common and widely used method to estimate the organic and carbonate content of sediments [14-17]. It is the quickest and cheapest among all the methods employed for determining carbon content [14]. In a first reaction, organic matter is oxidised at 500-550°C to carbon dioxide and ash. In a second reaction, carbon dioxide is evolved from carbonate at 900-1000°C, leaving oxide. LOI provides a fast and inexpensive means of determining carbonate and organic contents of clay-poor calcareous sediments and rocks with precision and accuracy comparable to other, more sophisticated geochemical methods.
In this study, Total Organic Carbon for carbonaceous shale and inorganic shale samples was determined using the Dry Chemistry Technique also known as High Temperature Catalytic Oxidation (HTCO) using Shimadzu TOC-VCSH® analyzer and the Loss-onignition (LOI) technique.
The samples were collected from two different locations. The sample size of both shale samples was around 500 grams. The organic shale was collected from the coastal area of Terengganu (eastern coast of Malaysia), close to an oil field site. The inorganic shale sample was collected from Johor Bahru, Malaysia.
Methodology
Sample preparation
Two types of shale samples were prepared, organic shale and inorganic shale. The sample surfaces were cleaned to remove any outer contaminations, such as twigs, roots, stems, wood chips, branches, etc. The larger particles are relatively inert chemically therefore their removal may lead to a more accurate assessment of the samples [10].
The two samples were, then, crushed using two-stage mechanical crushing machine and turned into powder. The size of the powdered particles was less than 500 μm. The main downside of this method is its instability due to accumulation of residues that are non-volatile in the tube of combustion. Therefore homogenized sample size should be used to avoid errors [10]. The samples were sealed and kept away from moisture and contamination.
In the Loss-on-Ignition method, after crushing, the samples were oven-dried overnight at 105ºC to remove the water content and weighed. Alumina crucibles were dried as well, and weighed with the samples prior to installing in the muffle furnace. Sample oven-drying temperature that is used to determine the moisture in solid combustion residue samples must be between 104 -110°C [18]. For loss-on-ignition analysis, samples should be dried for 12 hours at 105°C (using a Termarks oven) to determine both the water content (or loss-ondrying) and dry sediment percentage [18]. Oven-drying constitutes a potential source for error with this method due to the loss of volatile organic compounds. However, these losses are generally small (<1% of the TOC content in a non-contaminated soil or sediment) [10] (Figure 1).
Experimental procedures
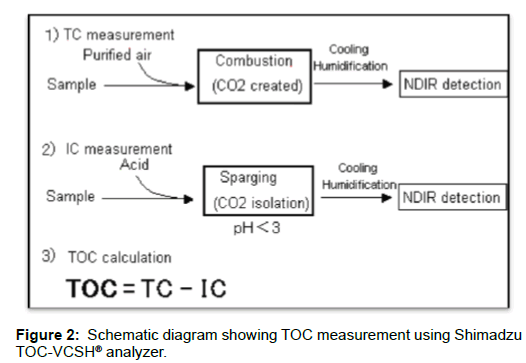
For the HTCO method, Shimadzu TOC-VCSH® analyzer was used. The 680°C combustion catalytic oxidation method brings about complete combustion of samples by heating them to 680°C in an oxygen-rich environment inside TC combustion tubes filled with a platinum catalyst. Since this procedure works on the simple principle of oxidation through heating and combustion, pretreatment and posttreatment using oxidizing agents are unnecessary, which enhance operability. The carbon dioxide generated by oxidation is detected using an infrared gas analyzer (NDIR).
The primary interferent in the determination of TOC is the presence of inorganic carbonates (such as minerals calcite and dolomite) which if left untreated will be destroyed along with the organic matter and in the process additional CO2 will be evolved and measured.
HCl (2N) was added to the samples in a syringe followed by sparging to remove inorganic carbon. 30 μL of sample was injected onto a platinum catalyst at 680°C in an oxygen rich atmosphere. The oxidizable content in the sample was turned into gas. The concentration of vaporized CO2 was determined with an infrared gas analyzer (NDIR) detector at 680°C. At the end, CO2 concentration was converted into weight percent [19]. Figure 2 shows TOC measurement using the 680°C combustion catalytic oxidation method and the NDIR method.
By sparging the samples to which a small amount of acid has been added, the inorganic carbon in the sample is converted to carbon dioxide. This carbon dioxide is removed, and the TOC is obtained by measuring the TC in the treated sample. When the carbon dioxide from the IC is removed, POC (purgeable organic carbon) may also be lost. Accordingly, the TOC obtained with this method may be referred to as NPOC. Shimadzu analyser provides option to measure this POC, which is typically lost, enabling more complete TOC measurement.
For The Loss-on-ignition method, a muffle furnace was used to heat the samples to a 550°C for Organic Carbon and 950°C for Total Carbon. Besides estimating TOC, the tests were conducted to understand the effect of samples size, temperature level, and exposure-to-heat time on the total weight loss of the samples, and therefore to the accuracy of total organic carbon (TOC). The sample sizes chosen for the test were 1.5 g and 5 g; the exposure time was set at 1, 2.5, and 4 hours. The variation in sample size and the exposure time was applied for the two types of shales. The effect of these factors was, then, studied and reported. The carbon emitted between 105 and 550ºC was interpreted as the content of organic carbon of the samples, while the carbon evolved between 550 and 925ºC is interpreted as the carbonate content [20,21]. The organic matter which resulted from sample heating to 550°C was, then, divided by a factor of 1.8 to be converted to total organic carbon (TOC). Equation 2 is used to calculate TOC at 550°C [18]:
LOI550 = ((DW105–DW550)/DW105)*100 (2)
To investigate the effect of sample size, the exposure time, and the temperature were fixed at 4 hours and 8°C/min., respectively (Figures 3 and 4).
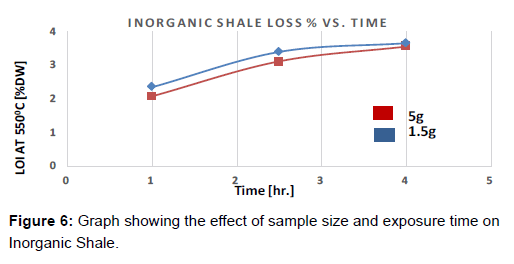
Exposure time is believed to have an effect on the sample weight loss upon ignition. The two samples were prepared to be tested at different exposures (4 hrs and 1 hr) while fixing the temperature level, heating rate, and the sample size (Figures 5 and 6).
Results and Discussion
Direct method (HTCO)
The determination of Total Carbon (TC), Inorganic Carbon (IC), and Total Organic Carbon (TOC) using HTCO analyzer was conducted in two runs for both the samples. “Organic shale” was tested first for TC, IC, and TOC by injecting 30 μl of the sample into the slider using the automated injection and for the inorganic shale results, 70 μl of the sample were injected using the automatic slider into the analyzer. The total carbon, TC was found to be 11.23% and 0.88% for organic shale and the inorganic shale, respectively (Table 1).
| Parameter | Value % (Organic shale) | Value% (Inorganic shale) |
|---|---|---|
| TC | 11.23 | 0.88 |
| IC | .02 | .022 |
| TOC | 11.21 | 0.86 |
Table 1: Summary of TC, IC, and TOC of Organic and Inorganic Shale.
Loss-on-ignition method
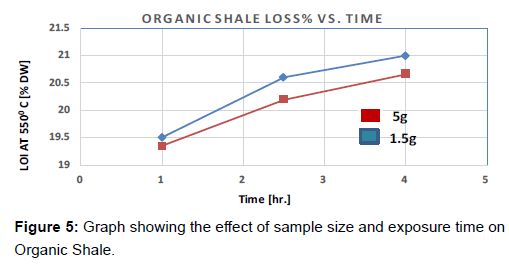
Figure 5 shows the 3 pairs of runs at 2 different sizes and at 3 exposure times. It can be seen that the higher the sample size, the lower is the weight percentage Loss-on-ignition. The two sample sizes had a close reading at 1 hour, with 5 g sample showing a lower reading (19.3%, compared to 19.5% for 1.5 g sample size). However, the rate of loss due to ignition started to increase. This steep increment continues until around 2.5 hours before it slows down. On comparing the two sizes, loss with respect to time is seen to be more in the smaller sample (1.5 g) than 5 g sample (Figure 5).
For the inorganic shale (Figure 6), the total weight loss for the organic matter at 550°C was very low compared to the organic shale (losses vary from 2.1% to around 3.6%). However, similar patterns were observed in weight loss with respect to sample size and exposure time. For both sizes, weight loss increased dramatically in the first two to three hours. The rate dropped in the following hours. Results start to converge after 4 hours. These observations may suggest that as the exposure time has an impact on weight loss only upto a certain time after which it ceases to effect the weight loss.
In addition, it was noticed that higher sizes always result in less sample weight losses. This phenomenon is expected because large sample sizes do not get combusted completely only the portions on the surface gets combusted, while the inner parts are left untreated. This also happens at the 1.5 g size, but in a less significant manner (Figure 5).
Comparison between the two methods
On comparing the Total Organic Carbon results of the Shimadzu TOC analyzer and the methods of Loss On Ignition (Table 2), it was noticed that there is a strong proximity between the two estimations for the “organic shale” sample (11.21% by the automated direct measurement, and 11.15% for Loss-on-ignition average for 5 g sample size, and 11.31% for Loss On Ignition for 1.5 g sample size). However, the values of Total Organic Carbon for the “Inorganic shale” were not of high precision. The results varied from 0.29% for the direct measurement to 1.44% (5 g size) and 1.74% (1.5 g size) for the Loss-onignition method. The inconsistency may be due to the high temperature effect on the sample content for the Loss on Ignition method. At high temperature the inorganic shales lose interstitial water due to collapse of crystal structure [2].
| Organic Shale | |||
|---|---|---|---|
| Parameter | Direct Method | LOI (5g) | LOI (1.5 g) |
| TC% | 11.23 | 12.28 | 12.44 |
| IC% | 0.02 | 1.13 | 1.13 |
| TOC% | 11.21 | 11.15 | 11.31 |
| Inorganic Shale | |||
| Parameter | Direct Method | LOI (5g) | LOI (1.5 g) |
| TC% | 0.88 | 1.73 | 2.03 |
| IC% | 0.022 | 0.29 | 0.29 |
| TOC% | 0.86 | 1.44 | 1.74 |
Table 2: Comparison between LOI and the direct method.
Based on the established source rocks quality values (Table 3), it can be noticed that if the TOC of source rock is greater than 1 percent, the rock can be a good source rock. If the rock contains greater than 4 percent Total Organic Carbon, then it has an excellent petroleum potential. Therefore, “organic shale” sample is an excellent source rock and its petroleum potential is very high, whereas “inorganic shale” sample does not have the potential to be a source rock. The values of TOC varied from less than 1 percent to just above 1 percent. By relying on the direct method for TOC estimation, which is 0.86 percent, it may be inferred that the inorganic shale rock is not productive as a source rock.
| Petroleum Potential (Quality) of an Immature Source Rock | ||||||||
|---|---|---|---|---|---|---|---|---|
| Petroleum Potential | Organic Matter | Bitumen | hydrocarbons | |||||
| TOC | Rock-Eval Pyrolysis | |||||||
| (wt.%) | S1 | S2 | (wt.%) | (ppm) | (ppm) | |||
| Poor | 0-0.5 | 0-0.5 | 0-2.5 | 0-0.5 | 0-500 | 0-300 | ||
| Fair | 0.5-1 | 0.5-1 | 2.5-5 | 0.05-0.10 | 500-1000 | 300-600 | ||
| Good | 1-4 | 1-2 | 5-10 | 0.10-0.20 | 1000-2000 | 600-1200 | ||
| Very Good | 2-4 | 2-4 | 10-20 | 0.20-0.40 | 2000-4000 | 1200-2400 | ||
| Excellent | >4 | >4 | >20 | >0.40 | >4000 | >2400 | ||
Table 3: Petroleum Potential (Quality) of immature source Rock [21].
Conclusion
This study which was conducted on shale source rocks using Total Organic Carbon TOC and the loss-on-ignition method (LOI), comes out with the following conclusions: The HTCO method that utilizes the automated measurement using Shimadzu Analyzer is a fast and interference-free method. Shale 1 designated as the organic shale was found to be rich in hydrocarbon content with a high potential to generate hydrocarbons. Shale 2 or inorganic shale would make for a poor source rock due to its low Total Organic Carbon.
For the loss-on-ignition method, best results were obtained with small sample size. Larger sample sizes may result in incomplete combustion of the sample due to which the accurate values of the total organic carbon, TOC are not obtained. Samples should also not be exposed to very high temperatures as overheating may destroy the lattice structure of clay minerals resulting in higher weight loss percentages.
Sediment composition seems to be the main cause, which determines the value of correction factor between loss on-ignition and total organic carbon in case of the inorganic shale samples. A rise in clay content is closely followed by a rise in extra weight-loss [18]. Care must be taken in the interpretation of carbonate content via loss-on-ignition as carbonates are ignited at a temperature higher than 550ºC leading to increased carbon reading. In the HTCO method, inorganic carbonates are neutralized by HCl sparging at the start of the experiment therefore the results of HTCO method seem to be more reliable for inorganic carbon. In order to provide accurate organic and carbonate content determinations for a sediment, loss-on-ignition data must be corroborated by standard total carbon analysis.
References
- Schwarzkoff TA (1991) Source rock potential (TOC+ Hydrogen Index) evaluation by integrating well log and geochemical data. Organic Geochemistry 19: 545-555.
- Crain ER (2010) Crain’s Petrophysical Handbook. 3rd spectrum 2000 Mindware.
- Tissot BP, Welte DH (1984) Petroleum formation and occurrence: 2nd Edition. Springer-Verlag, 699.
- Espitalie′ J, Deroo G, Marquis F (1985) Rock- Eval Pyrolysis and its applications: Journal of the Institute of French Pe'trole 40: 563-579.
- Tissot BP (1984) Recent advances in petroleum geochemistry applied to hydrocarbon exploration. AAPG Bulletin 68: 546-563.
- Peters K (1986) Guidelines for evaluating petroleum source rock using programmed pyrolysis. AAPG Bulletin 70: 318-329.
- Kogel-Knabner I (1997) 13C and 15N NMR spectroscopy as a tool in soil organic matter studies. Geoderma 80: 243-270.
- Rumpel C, Janik LJ, Skjemstad JO, Kogel-Knabner I (2001) Quantification of carbon derived from lignite in soils using mid-infrared spectroscopy and partial least squares. Organic Geochemistry 32: 83l-839.
- Nguyen TT, Janik LJ, Raupach M (1991) Diffuse reflectance infrared fourier transform (DRIFT) spectroscopy in soil studies. Aust. J. Soil Res. 29: 49-67.
- Schumacher BA (2002) Methods for the determination of Total Organic Carbon (TOC) in soils and sediments. United States Environmental Protection Agency.
- Tiessen H, Moir JO (1993) Total and organic carbon. In: Soil Sampling and Methods of Analysis, M.E. Carter, Ed. Lewis Publishers, Ann Arbor, MI 187-211.
- Bremner JM (1949) Use of the Van Slyke-Neil manometric apparatus for the determination of organic and inorganic carbon in soil and of organic carbon in soil extracts. Analyst 74: 492-498.
- Allison LE (1965) Organic carbon. In: Methods of Soil Analysis: Part 2. In. Black CA et al., (edn.). Agronomy 9:1367-1378.
- Dean WE (1974) Determination of carbonate and organic matter in calcareous sediments and sedimentary rocks by loss on ignition: Comparison with other methods. JSed Petrol 44: 242-248.
- Bengtsson L, Enell M (1986) Chemical analysis. In Berglund BE (edn.), Handbook of Holocene Palaeoecology and Palaeohydrology, John Wiley & Sons Ldt., Chichester 423-451.
- Maher LJ (1998) Automating the dreary measurements for loss on ignition. INQUA Sub-Commission on Data-Handling Methods, Newsletter 18.
- Heiri O, Lotter AF, Lemcke G (2001) Loss-on-ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. Journal of Paleolimnology 25: 101-110.
- ASTM (2000) Standard test methods for moisture, ash, and organic matter of peat and other organic soils. American Society for Testing and Materials, West Conshohocken, US.
- Ming CA, Takako T (2003) High Sensitivity Total Organic Carbon Analysis. AD-0007-TOC. Shimadzu (Asia Pacific) Pt Ltd, Application Report.
- Veres DS (2002) A comparative Study between Loss on Ignition and Total Carbon Analysis on mineralogical sediments. StudiaUniversitatisBabeS-Bolyai, Geologia, XLVII 1: 171-182.
- Peters KE,Cassa MR (1994) Applied source rock geochemistry: In Magoon LB, Dow WG, (edn). The petroleum system-From source to trap: Tulsa, Okla, American Association of Petroleum Geologists Memoir 60: 93-117.
Relevant Topics
Recommended Journals
- Oil & Gas Research Journal
- Renewable Energy and Applications Journal
- Oceanography Journal
- Industrial Pollution Control Journal
- Coastal Zone Management Journal
- Climatology & Weather Forecasting Journal
- Geoinformatics & Geostatistics Journal
- Engineering and Technology Journal
- Petroleum & Environmental Biotechnology Journal
- Polymer Sciences Journal
Article Tools
Article Usage
- Total views: 19156
- [From(publication date):
December-2015 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 17596
- PDF downloads : 1560