Case Report Open Access
Suspected Local Anesthetic Allergy of a Child: A Case Report
Matsumura T*, Miyamoto T and Fukayama HAnesthesiology and Clinical Physiology, Graduate School, Tokyo Medical and Dental University, Bunkyo, Tokyo, Japan
- *Corresponding Author:
- Matsumura T
Assistant Professor
Anesthesiology and Clinical Physiology
Graduate School, Tokyo Medical and Dental University
Bunkyo, Tokyo, Japan
Tel: 81358035549
Fax: 81358030206
E-mail: tomoanph@tmd.ac.jp
Received Date: August 02, 2017; Accepted Date: August 12, 2017; Published Date: August 18, 2017
Citation: Matsumura T, Miyamoto T, Fukayama H (2017) Suspected Local Anesthetic Allergy of a Child: A Case Report. Pediatr Dent Care 2: 139. doi: 10.4172/2573-444X.1000139
Copyright: © 2017 Matsumura T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Neonatal and Pediatric Medicine
Abstract
In dental practice, a certain number of patients complain of various symptoms after injection of local anesthetics. However, especially in children who cannot express their symptoms properly, a correct diagnosis of local anesthetic allergy is difficult. Here is a case of a boy with suspected local anesthetic allergy. We looked for an alternative local anesthetic on the basis of the medical history and the results of allergy tests
Keywords
Local anesthetics; Allergy; Allergy tests
Introduction
Local anesthetics (LAs) are essential in dental practice. Although hypersensitivity reactions are especially rare and account for less than 1% of all adverse reactions, exposure to allergens can cause a lifethreatening allergic reaction known as anaphylactic shock [1-4]. We sometimes encounter patients who believe that they have LA allergy on the basis of their experiences of developing various symptoms after injection of LAs. However, we often find it difficult to detect and to diagnose LA allergy partly owing to the low reliability of the allergy tests [5]. Here, we examined the use of LAs in a child with suspected LA allergy.
Case Report
An 8-year-old boy was planned for pulpectomy of the second deciduous molar in the lower jaw. He was referred to our hospital by his pediatric dentist because he was suspected of having LA allergy. He was medicated for asthma using inhaled steroids and was diagnosed as having allergies to mites, rice and ragweed. In his previous dental treatments, he did not show any allergic reactions to lidocaine. One year before the first visit to our department, his cavity was filled with temporary soft resin under local anesthesia induced using mepivacaine by his pediatric dentist. After 30 minutes of his dental treatment, when he left the clinic, rashes emerged around his neck and spread throughout his body in the next 6 hours. He was brought to a nearby emergency hospital, where he did not show other typical allergic symptoms such as choking, discomfort of gastrointestinal tracts and unconsciousness. Steroids were administered and his rashes disappeared. In the next morning, he developed sudden fever and he was diagnosed as having influenza.
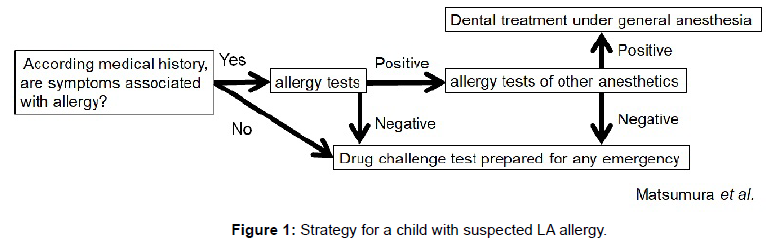
Prior to his next dental appointment, we planned his treatment on the basis of the literature [1-8] (Figure 1). In our searching for the cause of his skin rashes, we suspected him as having LA allergy not influenza, because he presented with typical symptoms of allergy such as rashes that are different from those in influenza. Moreover, type IV allergy, not type I allergy, was suspected owing to the time of onset. We performed drug lymphocyte stimulation tests (DLSTs) of five LAs; 2% Lidocaine with 1/80,000 Adrenaline, 3% Prilocaine-Felypressin (0.054 U), 3% Mepivacaine, 0.5% Ropivacaine and 1% Procaine. The results of his DLSTs in Table 1 show that he had positive reactions to Lidocaine and Mepivacaine, which suggest that he had type IV allergy to these LAs.
| Local anesthetics | Value | S.I (%) | Result | |||
|---|---|---|---|---|---|---|
| Aminoalkalymides | Test prior to first treatment | Lidocaine product (2% lidocaine added 1/80000 adrenaline) |
899 | 204 | Positive | |
| Mepivacaine | 914 | 208 | Negative | |||
| Prilocaine-felypressin product | 1622 | 114 | Negative | |||
| Repivacaine | 1778 | 126 | Negative | |||
| Test after treatment | Prilocaine-felypressin | 291 | 87 | Negative | ||
| Amino alkalymides | Prior to first treatment | Procaine | 1934 | 137 | Negative | |
Table 1: Results of DLST. An S.I. of more than 180 indicates a positive result.
After the DLSTs, a challenge test was undertaken. For a drug challenge test, we chose Prilocaine-felypressin among the dental anesthetics to which the patient showed negative reactions in the DLSTs. During and after injecting 0.9 mL of prilocaine-felypressin, his vital signs were monitored and rescue medication was prepared in case of severe hypersensitive reactions. After 15 minutes of close observation without immediate responses, pulpectomy was performed, which required about an hour. We continued monitoring for further one hour after the dental treatment and no symptoms of hypersensitivity reactions were observed. We asked his mother to check his condition carefully at home, but no abnormalities were reported 1 month later.
One month after the previous treatment, we planned the next dental treatment. Considering his possible sensitization to prilocainefelypressin in the previous treatment, we performed DLST again. With negative results in this test, we decided to use prilocaine-felypressin in the following treatments. In our hospital, he underwent dental treatment three times in total. Each time, we monitored his vital signs and checked his general condition, and no adverse reactions were observed. Finally, he was sent back to his primary care dentist and there has been no report of adverse events from the dentist so far.
Discussion
For our patient, we planned the treatment with priority in finding alternative LAs. Under general anesthesia, dental treatments can be performed without LAs; however, the drugs used in general anesthesia may also induce adverse reactions. Moreover, since dental treatments will continue throughout the lifetime of the patients, multiple treatments under general anesthesia would be a big burden for them both physically and mentally. Therefore, searching for safe alternative LAs are considered to be a beneficial in when planning dental surgeries for children with suspected LA allergy.
In clinical practice, two types of allergic reaction to LAs are recognized: IgE-mediated type I (immediate-type) allergy and T-cell mediated type IV (delayed-type) allergy [9]. In type I allergy, a rapid increase in vascular permeability as well as contraction of smooth muscles leads to symptoms such as urticaria, bronchospasm and, in severe cases, shock. Type IV allergy is most often recognized as a major cause of contact dermatitis. The underlying pathomechanism of LA allergy most often involves type IV allergy but type I allergy may also be involved [6,10].
The reported incidence of allergic reactions to LAs is low, which accounted for less than 1% of all adverse reactions associated with LAs [11]. In patients who were suspected of having LA allergy, some of them thought that all the symptoms after injection of LAs were caused by allergy. According to previous reports, only 15-37% of adult patients had past histories indicative of true events associated with LAs [1]. Therefore, a detailed analysis of medical history may prevent unnecessary tests, and a thorough investigation of the cause of allergy is strongly recommended to support or rule out the diagnosis of LA allergy.
Prior to an in vivo test, we performed an in vitro test i.e., DLST, which is a test for the diagnosis of type IV allergy. In DLST, we calculate the [H3]-thymidine uptake of lymphocytes obtained from the patient’s peripheral blood, which were stimulated by culture with a specific drug or vehicle. Stimulation index (SI) is defined as the count per minute (cpm) of the stimulated lymphocytes/cpm of the negative control.
As the in vitro tests for type I allergy, the leucocyte histamine release test and radioallergosorbent test are known [12]. However, we did not perform these tests because they have not been well established yet. Although in vitro tests are safer than in vivo tests, their validities are often questioned. Therefore, combining in vitro and in vivo tests is recommended for evaluation of drug allergy [13].
Among LAs with negative results in DLST, we decided to use prilocaine-felypressin for the drug challenge test because it is available in general dental clinics. Prilocaine-felypressin also belongs to the same group of amide-type anesthetics inducing mepivacaine and lidocaine, which means that cross sensitivity is possible. Although the underlying pathomechanisms of type I and type IV allergy are independent, a few cases were difficult to differentiate clinically [11]. Moreover, allergic reactions may vary from mild contact dermatitis to anaphylactic shock [9]. According to the medical history and the results of DLST of our patient, we suspected that he had type IV allergy, but the results did not confirm that he did not have type I allergy. Therefore, we performed the challenge test prepared for any emergency and continued monitoring after treatment.
If memory T cells have been sensitized at the first use of an alternative LA, hypersensitivity would be possible upon re-exposure. Performing DLST after the first use of an alternative LA is important for detecting sensitization. Moreover, careful observation of the patient’s condition is necessary even after subsequent dental treatments.
Conclusion
For children suspected of having LA allergy, it is worth trying to find alternative LAs. However, it is difficult to find the safest LAs, because of the risk of sensitization to alternative LAs and the low reliability of the allergy tests. Paying attention to the patient’s general condition is required during dental treatment using the alternative LAs.
References
- De Shazo RD, Nelson HS (1979) An approach to the patient with a history of local anesthetic hypersensitivity: experience with 90 patients. J Allergy Clin Immunol 63: 387-394.
- Bhole MV, Manson AL, Seneviratne SL, Misbah SA (2012) IgE-mediated allergy to local anaesthetics: separating fact from perception: a UK perspective. Br J Anaesth 108: 903-911.
- Fisher MM, Bowey CJ (1997) Alleged allergy to local anaesthetics. Anaesth Intensive Care 25: 611-614.
- Gall H, Kaufmann R, Kalveram CM (1996) Adverse reactions to local anesthetics: analysis of 197 cases. J Allergy Clin Immunol 97: 933-937.
- Saito M1, Yagi M, Uno K, Takanaka K (2008) Comparative Study of the Usefulness of the Drug-Induced Lymphocyte Stimulation Test and the Leukocyte Migration Test in Drug Allergies. Biol Pharm Bull 31: 299-304.
- Troise C, Voltolini S, Minale P, Modena P, Negrini AC (1998) Management of patients at risk for adverse reactions to local anesthetics: analysis of 386 cases. J Investig Allergol Clin Immunol 8: 172-175.
- Grzanka A, Wasilewska I, Sliwczynska M, Misiołek H (2016) Hypersensitivity lo local anesthetics. Anaesthesiol Intensive Ther 48: 128-134.
- Tomoyasu Y, Mukae K, Suda M, Hayashi T, Ishii M, et al. (2011) Allergic reactions to local anesthetics in dental patients: analysis of intracutaneous and challenge tests. Open Dent J 5: 146-149.
- Finucane BT (2003) Allergies to local anesthetics - the real truth. Can J Anaesth 50: 869-874.
- Adriani J (1972) Etiology and management of adverse reactions to local anesthetics. Int Anesthesiol Clin 10: 127-151.
- Thyssen JP, Menne T, Elberling J, Plaschke P, Johansen JD (2008) Hypersensitivity to local anaesthetics–update and proposal of evaluation algorithm. Contact Dermatitis 59: 69-78.
- Malinovsky JM, Chiriac AM, Tacquard C, Mertes PM, Demoly P (2016) Allergy to local anesthetics: Reality or myth? Presse Med 45: 753-757.
- Mayorga C, Doña I, Perez-Inestrosa E, Fernández TD, Torres MJ (2017) The Value of In Vitro Tests to Diminish Drug Challenges. Int J Mol Sci 18: 1222.
Relevant Topics
- About the Journal
- Birth Complications
- Breastfeeding
- Bronchopulmonary Dysplasia
- Feeding Disorders
- Gestational diabetes
- Neonatal Anemia
- Neonatal Breastfeeding
- Neonatal Care
- Neonatal Disease
- Neonatal Drugs
- Neonatal Health
- Neonatal Infections
- Neonatal Intensive Care
- Neonatal Seizure
- Neonatal Sepsis
- Neonatal Stroke
- Newborn Jaundice
- Newborns Screening
- Premature Infants
- Sepsis in Neonatal
- Vaccines and Immunity for Newborns
Recommended Journals
Article Tools
Article Usage
- Total views: 6818
- [From(publication date):
specialissue-2017 - Aug 23, 2025] - Breakdown by view type
- HTML page views : 5689
- PDF downloads : 1129