The Role of ABC Efflux Transporter in Treatment of Pharmaco-Resistant Schizophrenia: A Review Article
Received: 23-Apr-2019 / Accepted Date: 24-Jun-2019 / Published Date: 01-Jul-2019 DOI: 10.4172/2167-065X.1000189
Abstract
A large percentage of schizophrenic patients respond poorly to antipsychotic treatment. This could be explained by inefficient drug transport across the blood-brain barrier due to ABC efflux transporter, in particular, P-glycoprotein, mediated efflux. P-glycoprotein (P-gp) activity and expression in the blood-brain barrier can be affected by genetics (polymorphism), inflammation and pharmacotherapy. The level of expression of P-gp at BBB is thought to be one of the factors contributing for pharmaco-resistant schizophrenia. Despite the differences in the experimental set-up that partly explain the controversies regarding the interaction between P-gp and antipsychotics, it is feasible to say that the majority of the antipsychotics have shown (mostly weak) affinity as a P-gp substrate and that most have a weak inhibitory effect on P-gp in vitro. The three major Single Nucleotide Polymorphisms (SNPs) in protein coding region at C3435T, G2677T, and C1236T of ABCB1 at BBB have been associated with efflux pump efficiency and with predicting changes in the function of P-gp that determines the inter-cerebral concentration and therapeutic response in human studies to anti-psychotics unlike serum concentration of these agents. The effects of ABCB1 polymorphisms and their significance remain unclear due to contradictory and inconsistent study results, and so far they have not been able to incontestably explain differences in the pharmacokinetics of substrate drugs. P-gp modulators/inhibitors/chemosensitizers are seemed to have low potency, weak effectiveness, and poor selectivity, and would have to be given chronically at high doses to block transporter function effectively in human which bears an increased risk of severe side effects. Owing to these complications, no transporter inhibitors are currently in clinical use to improve brain delivery of anti-psychotic for treatment-resistant schizophrenia. In general, the functional significance of P-gp efflux transporters as drug carriers are constantly increasing in current medical practice and as they represent a key factor in clinical outcome. However, direct evidence for a major role of P-gp in pharmacokinetics has been lacking, and thus requires further standardized research in future in particular in tackling pharmaco-resistant schizophrenia.
Keywords: ABC efflux transporter; Blood-brain barrier; Antipsychotic; Pharmaco-resistant
Introduction
Schizophrenia is a chronic and disabling brain disease [1] and serious mental disorder with an annual incidence of 0.23 per 1000 persons [2,3] and a prevalence rate over life of around 1% [4-9].
The effectiveness of drug treatments for Central Nervous System (CNS) disorders (epilepsy, depression, and schizophrenia) is limited by poor therapeutic outcomes or drug resistance might due insufficient drug enter brain [10]. This makes up to 12.9%-50% of schizophrenic patients are pharmaco-resistant schizophrenia [5,11,12]. That is why the issues of efficacy and safety of antipsychotic drugs are still far from having idealistic values [13].
Even though the causes of pharmaco-resistant schizophrenia are likely to be multifactorial, the ABC drug efflux transporter Pglycoprotein (P-gp) expressed at the blood-brain barrier, in particular, might play an important role [7,8,14].
The brain uptake of the majority of antipsychotics, as well as many other psychotropic drugs and endogenous compounds is hampered by the activity of P-glycoprotein [15]. Moreover, acting alone or in concert with drug metabolizing enzymes they can affect the pharmacokinetics and pharmacodynamics of antipsychotic drugs [16]. However, randomized controlled studies on these issues with psychotropic drugs, in particular antipsychotic in humans are still lacking.
To achieve efficacy, pharmaco-resistant schizophrenia frequently requires increased doses or an increase in the number of drugs administered which can often result in toxic drug-drug interactions and unwanted physiological side effect [17].
This current review focuses on the functional significance of membrane transporters as drug carriers: as their role is constantly increasing in current medical practice and as they represent a key factor in clinical outcome. It elaborately assesses the impact of efflux transporter, mainly P-gp, its polymorphism and its modulation on the drug therapy of schizophrenia, while the other ABC transporters were briefly reviewed. We used different searching terms like ABC transporters, efflux transporter, P-gp, MDR1 protein, ABCB1, Multi drug resistant associated proteins, MRP1, ABCC1, Breast cancer resistance proteins, BCRP, ABCG2, MRP2, ABCC2, Polymorphism of P-gp, P-gp substrate, anti-psychotic agents, pharmaco-resistant schizophrenia, P-gp modulator and P-gp inhibitor from searching engine like Medline, Pubmed and Google scholar.
ABC efflux transporter
The ABC (ATPbinding cassette) transporters constitute one of the largest super families of membrane proteins [18,19]. They are ubiquitous in all phyla [12] Drug transporters are those proteins that carry either endogenous compounds like electrolytes, nucleosides, amino acids, and glucose [14] or xenobiotics across the biological membranes. These play an important role in the uptake, bioavailability, efficacy, toxicity and clearance of drugs [15].
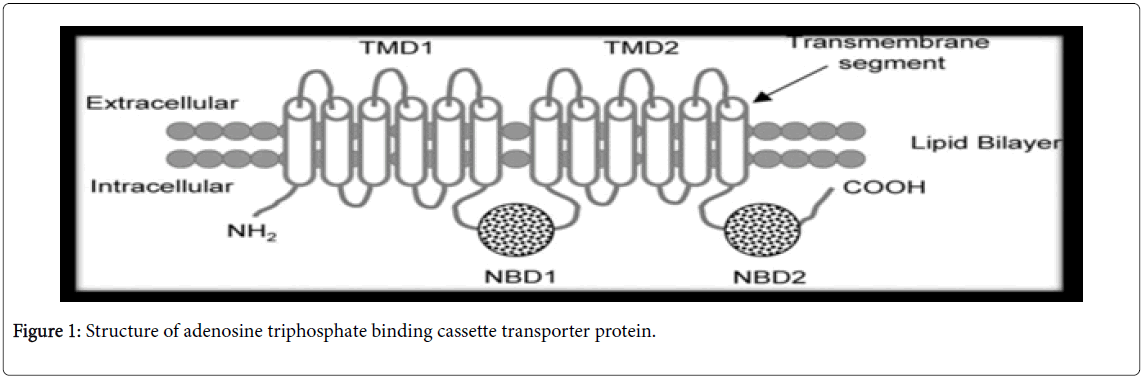
Drug transporters are membrane proteins present in various tissues such as the lymphocytes, intestine, liver, kidney, testis, placenta, and central nervous system [20,21]. They are classified into eight subfamilies (ABCA-ABCH). The multidrug resistance protein MDR1 (ABCB1), also called P-glycoprotein, the multidrug resistanceassociated proteins MRP1 (ABCC1) and MRP2 (ABCC2), and the breast cancer-resistance protein BCRP (ABCG2) are ATP-dependent efflux transporters [22] (Figure 1).
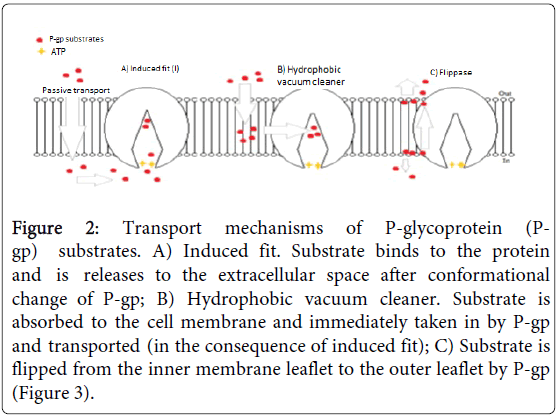
Among ABC membrane transporter, P-gp is the best studied and well characterized transport protein. Its most striking property is transport of a wide range of structurally different substrates [17]. The mechanism by which P-gp recognizes and transports its substrates has puzzled researchers and there are at least three different theories for the mechanism of function. These are “hydrophobic vacuum-cleaner” which transports the substrates immediately when they enter the membrane based on lipophilic nature of the substrate. The second one is an “induced fit” which is in line with the first one; however, the third theory is “flippase” which took greater recognition and acceptance [20] (Figure 2).
Figure 2: Transport mechanisms of P-glycoprotein (Pgp) substrates. A) Induced fit. Substrate binds to the protein and is releases to the extracellular space after conformational change of P-gp; B) Hydrophobic vacuum cleaner. Substrate is absorbed to the cell membrane and immediately taken in by P-gp and transported (in the consequence of induced fit); C) Substrate is flipped from the inner membrane leaflet to the outer leaflet by P-gp (Figure 3).
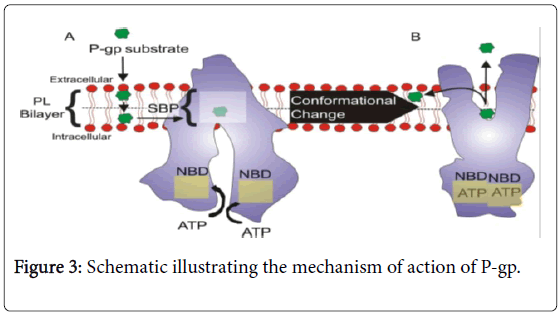
In its substrate-binding conformation, P-gp contains a large Substrate Binding Pocket (SBP), comprising mostly of hydrophobic and aromatic residues. Lipid soluble P-gp substrate molecules travel into the SBP from the inner leaflet of the phospholipid (PL) bilayer membrane. Substrate-P-gp interactions lead to the binding of two ATP molecules to the Nucleotide Binding Domain (NBD). ATP-binding causes dimerization of the NBDs, leading to a conformational change, resulting in an outward facing configuration. This outward facing arrangement facilitates the release of substrates into the extracellular environment or the outer leaflet of the PL bilayer, and sterically prevents the substrate from travelling into the intracellular space. Thus, P-gp acts as a unidirectional efflux pump (flippase) [23-25].
ABC transporters at the BBB
P-glycoprotein (P-gp), was discovered in 1976 [26]. In 1989, two independent research groups detected the ABC efflux transporter Pglycoprotein (ABCB1, formerly known as MDR1) at the human Blood Brain Brain (BBB). Since then, P-glycoprotein has been a main focus in the areas of BBB transporter and brain drug-delivery research [27].
ABCB1 or P-glycoprotein (P-gp) is the 170-kDa glycoprotein product of the MDR1 gene [28-31] that is highly expressed on the luminal membrane of the endothelial cells at the BBB. Therefore, P-gp is widely believed to be the most important ABC transporter in the CNS that plays a crucial role in both neuroprotection and pharmacoresistance [32].
The importance of P-glycoprotein for barrier function and brain protection is best highlighted by experiments using genetic knockout mice. In vivo dosing studies using P-glycoprotein knockout mice show 5-50 fold increased brain-to-plasma ratios of a large number of therapeutic drugs that are P-glycoprotein substrates and normally cannot cross the BBB to enter the brain [28]. Moreover, P-gp knockout animals display a disrupted BBB, and can be up to 100 folds more sensitive to many drugs, which often show neurotoxicity not seen in wild animals [32,33]. That is why P-glycoprotein is considered the most prominent element of selective, active barrier function that limits xenobiotics from entering the brain [22].
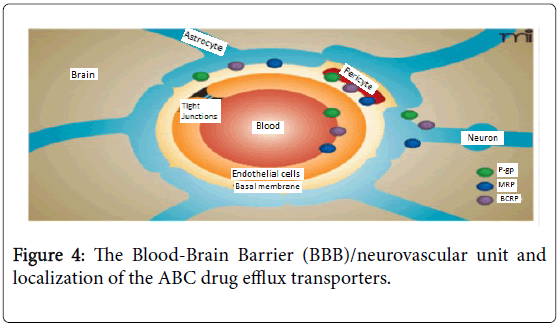
P-gp are the main ABC transporters expressed at the human BBB; they are both localized at the apical (or luminal) pole of the BBB where they transport drugs from the brain to the blood [34].
In general, three factors:-location, potency, and broad multispecificity: combine to make P-glycoprotein a key determinant of drug entry into the CNS [26]. 1) substantial protein expression; 2) localization in the luminal plasma membrane of brain capillary endothelial cells, at the interface between blood and CNS;3) highly effective and potent ATP-driven efflux transport against a concentration gradient; and 4) a remarkably broad substrate spectrum that covers a wide range of structurally diverse therapeutic drugs [32,33].
As a consequence of efflux transporters’ nature, in the case of CNS targeted drugs, brain permeability and transport activity needs to be determined early on in development, because it is a critical and often limiting factor in their success [35], study suggested that maximizing the CNS penetration of antipsychotics therapeutic agents may require at the least concurrent blockade of both P-gp and MRP [34] (Figure 4).
Impact of ABC efflux transporter on schizophrenia and its treatment
As it has been discussed so far, despite the huge impact that psychiatric illness has on society, treatment of the major psychiatric disorders remains very limited in its efficacy. Response rates of less than 50% are common for patients with schizophrenia to achieve adequate response to their antipsychotic drug treatment [35].
The lack of adequate response to antipsychotic drugs may be caused by inefficient drug transport across the Blood Brain Barrier (BBB) and consequently low availability of the drug in the brain [36]. Also antipsychotics were found to have affinity for P-gp at BBB, which can result in low availability of the drugs in the brain and consequently treatment resistance [37,38].
A role of BBB efflux transporters in drug resistance of mood disorders or schizophrenia is generally conceivable based on the fact that some antipsychotic drugs are substrates of P-gp. However, due to the lack of models for treatmentresistant psychiatric disorders, it is difficult to test the validity of the hypothesis, which therefore still remains rather speculative [36].
Pharmaco-resistant schizophrenia is a significant impediment to the successful management of the disease in which ABC efflux transporter mainly P-gp at BBB thought to be implicated in this phenomena [39].
Moreover, it was clear that ABC transporters were also affected by and contribute to CNS pathology [40]. Schizophrenia-related decrease in capillary and neuronal ABCB1 expression in the habenular complex might have consequences for brain concentrations of proinflammatory cytokines [41].
It has been shown that neuro-inflammation may also play an important role in schizophrenia [42-44]. Neuro-inflammation may increase P-gp activity or expression at the BBB in a subpopulation of schizophrenic patients, which could explain treatment resistance in schizophrenia [43] in particular during acute injuries. P-gp function has been reported to be reduced in the progression of neurodegenerative diseases [44] that might in line with the report chronic inflammation it leads to neuronal cell death. In addition, acute, sub-acute and chronic antipsychotic treatment could also induce treatment resistance by affecting P-gp activity. Thus this up regulation is considered as one factor contributing to MDR in the therapy of various CNS diseases [45].
Antipsychotic drugs have also been shown to stimulate the catalytic activity of P-gp providing additional evidence of the participation of Pgp mediated drug extrusion processes in restraining CNS penetration of these drugs [46].
On contrary to the above, P-glycoprotein transport activity is reduced after short exposure to TNF-α but increased with longer exposure times, indicating a complex, time-dependent regulatory mechanism. This makes generalizations difficult because patterns of responses can be dose, time, context, and model-dependent. Given that brain inflammation is a major component of most CNS disorders, future research will likely identify more pathways where proinflammatory mediators signal changes in BBB transporter expression and/or function [47].
Over 20% of patients with schizophrenia do not respond to treatment with antipsychotics [48]. In vitro studies have shown that the majority of clinically used antipsychotic drugs, such as amisulpride, aripiprazole, risperidone, quetiapine, clozapine and olanzapine, are transported substrates of P-gp at the BBB having a varying degree of affinity for the P-gp efflux transporter [49,50]. Results of in vivo study depicted that olanzapine brain concentration was 3-fold higher in the abcb1a knockout mice than as the comparative control mice [51] while risperidone was ten times [52,53].
The other studies demonstrated in their animal in vivo study, risperidone and 9-hydroxyrisperidone, and aripiprazole and, even more pronounced its active metabolite dehydroaripiprazole could be identified as substrates of P-gp at BBB [54,55].
In addition to its being substrate, risperidone was shown to inhibit transport of other compounds transported by P-gp. The finding that risperidone lowered P-gp activity suggests that long-term (subchronic) treatment with risperidone becomes more effective over time due to a self-induced increase in availability of the drug for the brain. This could also be attributed to its active metabolite 9- hydroxyrisperidone. This effect has, however, not been reported in clinical trials [56].
Interestingly, clozapine is among the most effective antipsychotic drugs and is generally used in treatment resistant schizophrenia, which may be due to its low affinity for P-gp, but it needs further research before drawing conclusions in this regard [57].
Olanzapine showed intermediate affinity and clozapine showed the least affinity of the drugs studied in vitro (i.e: quetiapine>risperidone>olanzapine>chlorpromzaine>haloperidol= clozapine) [58-60]. Furthermore, quetipine, risperidone, olanzapine, and clozapine stimulated the ATPase activity of P-gp in crude membrane preparations, which is an indication that the compounds interact with the substrate binding site(s) of P-gp [35] even though a study suggested that olanzapine and risperidone were the only agents that may inhibit P-gp activity in the BBB. Moreover, study suggested that loxapine is not a substrate for P-gp in vivo unlike moderate inhibitor in vitro [36].
To sum up, the translation of in vitro results to the human case could be complicated by differences in substrate specificity for P-gp across species. Despite the differences in the experimental set-up that partly explain the controversies regarding the interaction between P-gp and antipsychotics, it is feasible to say that the majority of the antipsychotics have shown (mostly weak) affinity as a P-gp substrate and that most have a weak inhibitory effect on P-gp in vitro [58].
Some data demonstrated that it can be more difficult to detect an interaction with P-gp, when drugs pass membranes efficaciously by rapid diffusion due to their lipophilic characteristics. In addition, the value of the studies using the knock-out mouse model is limited by the fact that a one dose model is mostly used, representing only the acute modulatory effect on P-gp. On contrary, such an in vivo model has a clear advantage, since the BBB is very complex making it difficult to study interactions between the BBB and drugs in vitro [59].
In spite of the findings of the preclinical work on the role of P-gp in the uptake of drugs used for the major psychiatric disorders, there are arguments pleading against a significant role for P-gp. For example, antipsychotic drugs like chlorpromazine, risperidone, olanzapine and haloperidol may significantly differ in their interaction with P-gp, but have a comparable clinical efficacy. Thus direct evidence for a major role of P-gp in pharmacokinetics has been lacking, [60].
In addition to the above concerns, changes in the ABC efflux transporters at the BBB in mental disorders are understudied with only few studies that suggested local changes of P-gp function in the brain of depressive or schizophrenic patients. Using positron emission tomography, a significantly lowered 11C-verapamil uptake (indicating increased P-gp activity) in prefrontal cortex and temporal lobes was found in patients with a schizophrenia and chronic use of antipsychotic.
This decrease of 11C-verapamil uptake in the brain of schizophrenic patients may be correlated with an increased activity of the P-gp pump. These results suggested a local increase in the activity and/or expression of P-gp at areas that are mostly affected by schizophrenia. Moreover, clinical findings of these studies speculated that pharmacoresistance schizophrenia may in fact be associated with increased P-gp function that may cause low uptake of antipsychotics. However, it is still unclear whether the observed increase in P-gp activity in schizophrenia is related to specific pathological alterations in these diseases or to the drugs used in the treatment of these diseases [61].
By-passing ABCB1 has recently been proposed as a therapeutic option in treatment resistant schizophrenia as it is regarded as a “major gatekeeper” of the brain. Therefore, the inter-individual variability of p-glycoprotein function at the BBB, in particular in schizophrenic patients, requires further research [62].
Targeting ABC efflux transporters to improve pharmacotherapy of schizophrenia
In addition to genotype, concomitantly administered drugs and disease state can affect the function of efflux transporter especially pglycoprotein. In response to the inefficiency in conventional delivery mechanisms, a great amount of research efforts has lately focused primarily on the development of new strategies to allow for a greater efficacy in the delivery of drug molecules to the CNS [63].
Increasing awareness of the impact of efflux transporters on success in the pharmacotherapy of CNS diseases promotes efforts to develop strategies to modulate transporter activity. P-gp inhibitors were explored for overcoming multidrug resistance and poor bioavailability problems of the therapeutic P-gp substrates [64].
P-glycoprotein inhibition significantly increased risperidoneinduced cataleptic effects, blockade of amphetamine induced locomotion, and effects on dopamine turnover as seen by increased striatal dopamine metabolite levels [65].
The first clinical case reports with co administration of verapamil to the individual antiepileptic drug regimen resulted in improved seizure control especially in patients with refractory status epilepticus. So it has a potential in increasing the efficacy of antipsychotics even though it yet remains the research gap to combat P-gp efflux at BBB. Moreover, recent advances in drug delivery technology like nano-carriers have provided promising solutions to overcome these challenges.
In general, modulation of P-gp in brain is possible by three possible ways: (i) direct inhibition by specific competitors, (ii) functional modulation, and (iii) transcriptional modulation [65]. Fortunately, unraveling intracellular signaling pathways and networks, and identifying molecular switches that regulate ABC transporters at the BBB, will provide new molecular targets for CNS therapy.
First generation inhibitors are pharmacological actives, which are in clinical use for other indications but have been shown to inhibit P-gp. Second and third generation inhibitors specifically modulate P-gp, with the second generation inhibitors lacking the pharmacological activity of its first generation counterparts and with a higher affinity for P-gp even though they show strong substrate for CYP 450 3A4.
Evolved third generation inhibitors were highly specific, lacked interactions with CYP450 3A4 system and required no alterations in the chemotherapy dose. Many natural product extracts are also upcoming as P-glycoprotein inhibitors. Fluoxetine is considered as fourth generation chemo sensitizer and exerts its ability to chemo sensitize of multi-pump type at low safe doses, well below its human safety range [65]. Moreover, itraconazole, flupentixol, paracetamol and haloperidol were reported in inhibition of P-gp but paracetamol and haloperidol are not actively transported as substrates.
However, devising strategies to deliver therapeutic drugs like antipsychotic into the CNS, while limiting entry of toxic chemicals and preserving an optimal extracellular environment by transiently modulating (i.e. decreasing activity) of P-gp at BBB, is a substantial challenge [66]. Little is known about the regulation of P-glycoprotein at the blood-brain barrier, and what were known are mechanisms that work over hours to days rather than minutes.
Owing to complications such a slow potency, weak effectiveness, poor selectivity and increased risk of severe side effects in particular at phase III clinical studies, no transporter inhibitors are currently in clinical use to improve brain delivery of CNS drugs [67].
ABC Efflux transporter polymorphism and its impact on drug therapy of schizophrenia
Evidence is now accumulating which strongly suggests that drug transporters are one of the determinants governing the pharmacokinetics profile of drugs in realizing personalized medicine [68].
Unlike the Collie breed of dogs or CF-1 mice, no inherent functional knockout of P-glycoprotein has been discovered in humans. Drug transporters modulate pharmacokinetics of drugs by controlling the influx and efflux of drugs in cells [69,70].
Although many factors, such as diet, race, environment, pharmacotherapy and disease state, may influence inter-individual variability in the pharmacokinetics of P-gp substrate drugs and therapeutic outcome of certain diseases treated with such drugs, the genetic polymorphisms in the MDR1 gene are one of the primary determinants [71].
SingleNucleotide Polymorphisms (SNPs) in efflux transporter genes have been associated with inter-individual differences in transport activity. Pharmacogenetics of P-gp has been suggested to critically influence the individual response to pharmacotherapy. More than 50 SNPs have been identified in the human MDR1 gene which has been linked to the therapeutic response in different CNS disorders including schizophrenia. Consequently, it may have to be taken into consideration when prescribing drugs with a low therapeutic index that are p-glycoprotein substrates.
ABCB1 gene is located in the human chromosome 7 band p21-21.1. Many Single-Nucleotide Polymorphisms (SNPs) have been identified in ABCB1, including several in the protein-coding region (rs1128503, 1236C>T; rs2032582, 2677G>T/A; rs1045642, 3435C>T) which are believed to affect the activity and/or expression level of the P-gp expression [72]. And Clinical efficiency of antipsychotic treatment has been correlated with ABCB1 genetic polymorphisms.
Frequency of the polymorphisms is significantly different across different ethnic group. The effects of these polymorphisms and their significance remain unclear due to contradictory and in consistent study results, and so far they have not been able to incontestably explain differences in pharmacokinetics of substrate drugs.
Unlike polymorphisms observed for some drug metabolizing enzymes and their effects on drug disposition, for which loss of function mutations or gene amplification manifests as distinct phenotypes in the population, the clinical relevance of transporter genetic variations are moderate or less well established [73].
The three major Single Nucleotide Polymorphisms mentioned, (SNPs) at C3435T, G2677T, and C1236T of ABCB1 have been associated with efflux pump efficiency that determines the intercerebral concentration and therapeutic response in human studies to psychotropic drugs [74].
The three most common SNPs in the protein coding region are rs1128503 (1236T>C, Gly412Gly), rs2032582 (2677T>G/A, Ser893Ala/ Thr), and rs1045642 (3435T>C, Ile1145Ile) are in high linkage disequilibrium. They were shown to associate with controversial results of antipsychotic efficacy [75].
Individuals carrying the favourable homozygous genotypes of rs1045642 and rs2032582 in one clinical study in South India displayed better response with increased dosage of antipsychotic while that carrying risk genotype manifested refractoriness on increased dosage [76].
It has been also suggested that ABCB1 1236TT, 2677T/T, 3435T/T genotypes, and 2677T/3435-T haplotype carriers have a better response to risperidone and its metabolite with lower frequency of extrapyramidal side effects in comparison with non-carriers of the genes in schizophrenic patients [76].
Carriers of ABCB1 1236T/2677T/3435T haplotype had higher serum and cerebrospinal fluid olanzapine concentrations than patients without this haplotype. It has been suggested that the P-gp C3435T genotype may also help to determine positive symptom reduction from olanzapine clinical trial. The T allele of 2677G/T/A polymorphism is related to better response to olanzapine treatment in women [77]. Additionally, suggested that ABCB1 G2677T and C3435T MDR1 genetic polymorphisms influence the development of metabolic abnormalities among female patients treated with olanzapine and risperidone.
Suzuki and his co-worker found that the T-allele of the ABCB1 C3435T genotype should be considered in future diagnostic development efforts for risperidone associated QT prolongation unlike C-allele of the ABCB1 C3435T and G2677T/A. Likewise, one study was also found an association between the C3435T polymorphism, with treatment response with the TT genotype predicting higher drug concentration in the brain. The G2677T/A variation were found to be less useful as a predictor of response to bromperidol [78].
Moreover, the 2677T and 3435T MDR1 genetic variants were significantly associated with the greater increase in fasting glucose level in blood when patients were using olanzapine. A study indicated that a possible influence of -759CT 5-HT2C and MDR1 G2677T and C3435T MDR1 genetic polymorphisms on the development of metabolic abnormalities among female patients treated with olanzapine/risperidone (i.e. lower P-gp function and higher SGA accessibility and its effect to the brain). Although exon 26 C3435T genotype also seems to influence MDR1 expression per se, it might be possible that observed associations are due to its LD with exon 21 G2677T or other mutations. Likewise, one study done in Chinese schizophrenic patient was came up with result that indicate those developing neutropenia and agranulocytosis after taking clozapine had high frequency of ABCB1 3435TT [79]. Side effects of drugs acting on the CNS may be caused by a weakened P-gp function. Thus, knowing the P-gp status could make it possible to predict central side effects of drugs both acting on the CNS and peripherally.
In summary, most studies on the genetic effects of the SNPs C3435T, G2677T/A and C1236T or ABCB1 haplotypes showed little or no effect on treatment response. A strong effect of a single transporter gene on a phenotypic response in complex disorders like schizophrenia is not amenable. A few reports on antipsychotic related side effects associated with polymorphisms have been published, and a few groups have communicated on the effect of ABCB1 polymorphisms and plasma concentrations of antipsychotics [80].
The contradictions in observations for the major polymorphisms of ABCB1 are partly accountable to differences in methodology and to ethnic differences between study groups. In most studies the sample size is too small to draw firm conclusions. Thus, none of the SNPs of ABCB1, nor any of the haplotypes, can definitely be connected to phenotypical variation, but instead some may serve as biological marker for pin pointing a disease [81].
In the near future it is to be expected that analysis of SNP patterns in large patient cohorts with identical phenotypic features and/or Meta analyses will identify SNP profiles that characterize susceptibility factors or the impact of the pharmacogenomics of ABC transporter genes [82]. Genome Wide Association Studies (GWAS) are the type of study designed to identify such a genetic variation possibly in all efflux transporter including P-gp because they all affects inter-cerebral drug concentration even though the other than P-gp others effects were not well studied. As a result, priority knowledge of ABCB1 genotypes can provide a significant input into evaluating the patient’s response to medication, and minimizing redundant dosing and refractoriness [83].
Conclusion
Concluding remarks and future directions
Among those patients taking antipsychotic, several patients suffered from pharmaco-resistant schizophrenia. Among many factors that contribute to pharmaco-resistant, active efflux of these agents by ABC efflux transporter, in particular by P-gp, from brain into periphery takes pivotal consideration recently. Consequently, the promising applicability to the future therapy of pharmaco-resistant schizophrenia due to efflux transporter took greater concern.
Majority of clinically used antipsychotic drugs such as amisulpride, aripiprazole, dehydroaripiprazole, risperidone, paliperidone, quetiapine, clozapine, olanzapine, chlorpromazine etc., are substrates of P-gp at the BBB having weak to moderate degree of affinity for the P-gp efflux transporter. Most of them are weak inhibitors of P-gp including thioxanthene derivative. Moreover the three major Single Nucleotide Polymorphisms (SNPs) of ABCB1 at C3435T, G2677T, and C1236T have been associated with efflux pump efficiency and with predicting tolerability of antipsychotic drugs in addition to the disease and drugs itself. So that concomitant administration of agents, that modulate ABC efflux transporter at BBB, with antipsychotic will be one possible way to overcome the pharmaco-resistant schizophrenia.
However, taking all findings into consideration published observations, even when made with the same probe drug and in the same racial group, is controversial. There are multiple possible explanations for these discordant results include differential experimental conditions, such as probe drug used, applied dose, steady-state versus single dose pharmacokinetics, small sample sizes, sample selection, or genetically heterogeneity due to ethnical diverse populations.
In order to determine conclusive result and the actual impact of all ABC efflux transporter on the schizophrenia and its treatment; multicenter (i.e., multiethnic), large sample size, multi-dose, long term cohort and well standardized study should be conducted to optimize its usefulness in individualized pharmacotherapy of drug resistant schizophrenia. And also identification of genetic variants and the complex regulatory pathways involved in P-gp modulation should be well elucidated in future. P-gp transport screening and its polymorphism has to be incorporated into the drug discovery process as recently recommended by the FDA.
In general, the role of P-gp in pharmaco-resistant schizophrenia was under studied even though majority of antipsychotic drugs are substrate for this efflux transporter. As a result, great focus should be given to P-gp efflux transporter at BBB in future if it is necessary to tackle drug resistant schizophrenia.
Authors’ Contributions
DA critically reviewed the manuscript; DA, GM, KT and ET determined the searching terms, searching engine and compile the necessary literatures. All authors reviewed all necessary literatures.
References
- Doorduin J, de Vries EF, Dierckx RA, Klein HC (2008) PET imaging of the peripheral benzodiazepine receptor: Monitoring disease progress and therapy response in neurodegenerative disorders. Curr Pharm Des 14: 3297-3315.
- Gesteira A, Barros F, MartÃn A, Pérez V, Cortés A, et al. (2010) Pharmacogenetic studies on the antipsychotic treatment-current status and perspectives. Actas Esp Psiquiatr 38: 301-316.
- Naumovska Z, Nestorovska AK, Filipce A, Sterjev Z, Brezovska K, et al. (2015) Pharmacogenetics and antipsychotic treatment response. Pril 36: 53-67.
- Reynolds GP (2007) The impact of pharmacogenetics on the development and use of antipsychotic drugs. Drug Discov Today 12: 953-959.
- McIlwain ME, Harrison J, Wheeler AJ, Russell BR (2011) Pharmacotherapy for treatment-resistant schizophrenia. Neuropsychiatr Dis Treat 7: 135-149.
- Haslemo T (2012) Pharmacokinetic variability of olanzapin-A study based on therapeutic drug monitoring data. Oslo 4-36.
- Huo R, Tang K, Wei Z, Shen L, Xiong Y, et al. (2012) Genetic polymorphisms in CYP2E1: Association with schizophrenia susceptibility and risperidone response in the chinese han population. PLoS One 7: e34809.
- Hoosain FG, Choonara YE, Tomar LK, Kumar P, Tyagi C, et al. (2015) Bypassing P-glycoprotein drug efflux mechanisms: Possible applications in pharmaco-resistant schizophrenia therapy. Biomed Res Int 2015: 484963.
- Dima L (2009) Pharmacokinetic interactions of new antipsychotics with other psychotropic drugs. Bulletin Transilvania Univ Brasov 2: 31-38.
- Loscher W (2015) Transport dementia: ABC transporters in brain diseases. General information program, Oslo.
- Loscher W, Potschka H (2005) Drug resistance in brain diseases and the role of drug efflux transporters. Nat Rev Neurosci 6: 591-602.
- de Klerk OL (2011) The guarded brain: The role of P-glycoprotein at the blood-brain barrier in major psychiatric disorders and antidepressant treatment. Cent Nerv 11: 197-209.
- de Klerk OL, Bosker FJ, Nolte IM, Den JA, Dierckx R, et al. (2011) The role of P-glycoprotein in psychiatric disorders: A trustful guard of the brain? Cent Nerv Syst Agents Med Chem 11:197-209.
- de Klerk OL, Willemsen AT, Bosker FJ, Bartels AL, Hendrikse NH, et al. (2010) Regional increase in P-glycoprotein function in the blood-brain barrier of patients with chronic schizophrenia: A PET study with [(11)C] verapamil as a probe for P-glycoprotein function. Psychiatry Res 183:151-156.
- de Klerk OL, Willemsen AT, Roosink M, Bartels AL, Hendrikse NH, et al. (2009) Locally increased P-glycoprotein function in major depression: A PET study with [11C] verapamil as a probe for P-glycoprotein function in the blood-brain barrier. Int J Neuropsychopharmacol 12: 895-904.
- Girardin F (2006) Membrane transporter proteins: A challenge for CNS drug development. Dialogues Clin Neurosci 8: 311-321.
- Emmert D, Campos CR, Ward D, Lu P, Namanja HA, et al. (2014) Reversible dimers of the atypical antipsychotic quetiapine inhibit P‑glycoprotein-mediated efflux in vitro with increased binding affinity and in situ at the blood-brain barrier. ACS Chem Neurosci 5: 305-317.
- Al‑Shawi MK (2011) Catalytic and transport cycles of ABC exporters. Essays Biochem 50: 63-83.
- Adkins CE, Barbe KR, Lockman PR (2013) Crossing the barrier: The role of the blood-brain barrier in treating mental illness. Mol Psychatr 4: 46-49.
- Suvanto S (2014) P-Glycoprotein characteristics and investigation of P-glycoprotein mediated drug-drug interactions with in vitro methods. Am J Trop Med Hyg 87: 64-69.
- Choudhuri S, Klaassen CD (2006)Â Structure, function, expression, genomic organization, and single nucleotide polymorphisms of human ABCB1 (MDR1), ABCC (MRP), and ABCG2 (BCRP) efflux transporters. Int J Toxicol 25: 231-259.
- Veda P, Apilak W, Watshara S, Virapong P, Chanin N (2015) Classification of P-glycoprotein-interacting compounds using machine learning methods. Excli J 14: 958-970.
- Sharoom FJ (2008) ABC multi drug transporter: Structure, function and role in chemoresistance. Pharmacogenom 9: 105-127.
- O’Brien FE, Gerard CG, Dinan TG, Cryan JF, Griffin BT (2012) Human P-glycoprotein differentially affects antidepressant drug transport: Relevance to blood-brain barrier permeability. Int J Neuropsychopharmacol 16: 2259-2272.
- O'Brien FE (2013) P-glycoprotein inhibition as a strategy to increase drug delivery across the blood-brain barrier: focus on antidepressants. Int J Neuropsychopharmacol 1: 1-10.
- Aller SG, Yu J, Ward A, Weng Y, Chittaboina S, et al. (2009) Structure of P-glycoprotein reveals a molecular basis for poly-specific drug binding. Science 323: 1718-1722.
- Lakshmi PK, Srivalli KMR (2012) Overview of P-glycoprotein inhibitors: A rational outlook. Braz J Pharm Sci 48: 353-367.
- Hartz AM, Bauer B (2010) Regulation of ABC transporters at the blood-brain barrier: New targets for CNS therapy. Mol Interv 10: 293-304.
- Li YH, Wang WH, Li Y, Yang L (2006) MDR1 gene polymorphisms and clinical relevance. Yi Chuan Xue Bao 33: 93-104.
- Komoto C, Nakamura T, Yamamori M, Ohmoto N, Kobayashi H, et al. (2007) Reversal effects of Ca2+ antagonists on multidrug resistance via down-regulation of MDR1 mRNA. Kobe J Med Sci 53: 355-363.
- Jagodinsky JC, Akgun U (2015) Characterizing the binding interactions between P-glycoprotein and eight known cardiovascular transport Substrates. Pharma Res Perspect 3: e00114.
- Qosa H, Miller DS, Pasinelli P, Trotti D (2015) Regulation of ABC efflux transporters at blood-brain barrier in health and neurological disorders. Brain Res 1628: 298-316.
- Bernacki J, Dobrowolska A, Nierwinska K, Malecki A (2008) Physiology and pharmacological role of the blood-brain barrier. Pharmacol Rep 60: 600-622.
- Marquez B, Van Bambeke F (2011) ABC multidrug transporters: Target for modulation of drug pharmacokinetics and drug-drug interactions. Curr Drug Targets 12: 600-620.
- Bauer B, Hartz AM, Fricker G, Miller DS (2005) Modulation of P-glycoprotein transport function at the blood-brain barrier. Exp Biol Med 230: 118–127.
- Sjostedt N (2011) Investigating drug transport at the blood-brain barrier and the effects of p-glycoprotein on the brain distribution of drugs. Biol Med Model 6: 2.
- Vallabhaneni V, Julie LD, Mark EB, Abraham ZS, Rick AF, et al. (1999) Choroid plexus epithelial expression of MDR1 P glycoprotein and multidrug resistance-associated protein contribute to the blood-cerebrospinal-fluid drug-permeability barrier. Proc Natl Acad Sci 96: 3900-3905.
- Banks WA (1999) Physiology and pathology of the blood-brain barrier: Implications for microbial pathogenesis, drug delivery and neurodegenerative disorders. J Neurovirol 5: 538-555.
- Bebay W, Chetty M (2008) Differential pharmacological regulation of drug efflux and pharmaco-resistant schizophrenia. BioEssays 30: 183-188.
- Bernstein HG, Hildebrandt J, Dobrowolny H, Steiner J, Bogerts B, et al. (2015) Morphometric analysis of the cerebral expression of ATP-binding cassette transporter protein ABCB1 in chronic schizophrenia: Circumscribed deficits in the habenula. Schizophr Res 177: 52-58.
- Boulton DW, DeVane CL, Liston HL, Markowitz JS (2002) In vitro P-glycoprotein affinity for atypical and conventional antipsychotics. Life Sci 71: 163-169.
- Bozina N, Kuzman MR, Medved V, Jovanovic N, Sertic J, et al. (2008) Associations between MDR1 gene polymorphisms and schizophrenia and therapeutic response to olanzapine in female schizophrenic patients. J Psychiatr Res 42: 89-97.
- Brzozowska N, Li KM, Wang XS, Booth J, Stuart J, et al. (2016) ABC transporters P-gp and Bcrp do not limit the brain uptake of the novel antipsychotic and anticonvulsant drug cannabidiol in mice. Peer J 4: e2081.
- Chang HH, Chou CH, Yang YK, Lee IH, Chen PS (2015) Association between ABCB1 polymorphisms and antidepressant treatment response in taiwanese major depressive patients. Clinical Psychopharmacol Neurosci 13: 250-255.
- Cox AG (2010) Pharmacogenomics and drug transport/efflux. Miura M, Satoh S (eds.) Concepts in Pharmacogenomics pp:1-24.
- Doorduin J, Klein HC (2009) P-glycoprotein activity in the rat brain is affected by HSV-1 induced neuroinflammation and antipsychotic treatment: Neuropharmacol 85: 548-553.
- Doorduin J, deVries EF, Willemsen AT, deGroot JC, Dierckx RA, et al. (2009) Neuroinflammation in schizophrenia related psychosis: A positron emission tomography study. J Nucl Med 50: 1801-1807.
- Doorduin J, deVries EF, Willemsen AT, deGroot JC, Dierckx RA, et al (2010) Evaluation of [11C]-DAA1106 for imaging and quantification of neuroinflammation in a rat model of herpes encephalitis. Nucl Med Biol 37: 9-15.
- Fortuna A, Alves G, Falcao A (2011) In vitro and in vivo relevance of the P-glycoprotein probe substrates in drug discovery and development: Focus on rhodamine 123, digoxin and talinolol. J Bioequiv Availab S2: 1-24.
- Franke RM, Gardner ER, Sparreboom A (2010) Pharmacogenetics of drug transporters. Curr Pharm Des 16: 220-230.
- Hodges LM, Markova SM, Chinn LW, Gow JM, Kroetz DL, et al. (2011) Very important pharmacogene summary: ABCB1 (MDR1, Pglycoprotein). Pharmacogenet Genomics 21: 152-161.
- Holthoewer D, Hiemke C, Schmitt U (2010) Induction of drug transporters alters disposition of risperidone-A study in mice. Pharmaceutics 2: 258-274.
- Ieiri I (2012) Functional significance of genetic polymorphisms in P-glycoprotein (MDR1, ABCB1) and breast cancer resistance protein (BCRP, ABCG2). Drug Metab Pharmacokinet 27: 85-105.
- Jelen AM, Salagacka A, Zebrowska MK, Mirowski M, Talarowska M, et al. (2015) The influence of C3435T polymorphism of the ABCB1 gene on genetic susceptibility to depression and treatment response in polish population-preliminary report. Int J Med Sci 20: 974-979.
- Karlsson L, Green H, Zackrisson AL, Bengtsson F, Jakobsen Falk, et al. (2013) ABCB1 gene polymorphisms are associated with fatal intoxications involving venlafaxine but not citalopram. Int J Legal Med 127: 579-586.
- Karlsson L, Schmitt U, Josefsson M, Carlsson B, Ahlner J, et al. (2010) Blood-brain barrier penetration of the enantiomers of venlafaxine and its metabolites in mice lacking P-glycoprotein. Eur Neuropsychopharmacol 20: 632-640.
- Kirschbaum KM, Henken S, Hiemke C, Schmitt U (2008) Pharmacodynamic consequences of P-glycoprotein dependent pharmacokinetics of risperidone and haloperidol in mice. Behav Brain Res 188: 298-303.
- Kirschbaum KM, Uhr M, Holthoewer D, Namendorf C, Pietrzik C (2010) Pharmacokinetics of acute and sub-chronic aripiprazole in P-glycoprotein deficient mice. Neuropharmacol 59: 474-479.
- Kuzman MR, Medved V, Bozina N, Grubisin J, Jovanovic N (2010) Association study of MDR1 and 5-HT2C genetic polymorphisms and antipsychotic-induced metabolic disturbances in female patients with Schizophrenia. Pharmacogenomics J 11: 35-44.
- Linnet K, Ejsing TB (2008) A review on the impact of P-glycoprotein on the penetration of drugs into the brain. Focus on psychotropic drugs. Eur Neuropsychopharmacol 18: 157-169.
- Ma Q, Lu AY (2011) Pharmacogenetics, pharmacogenomics, and individualized medicine. Pharmacol Rev63: 437-459.
- Mackenzie B, Souza RP, Likhodi O, Tiwari AK, Zai CC, et al. (2010) Pharmacogenetics of antipsychotic treatment response and side effects. Therapy 7: 191-198.
- Miller DS, Bauer B, Hartz AM (2008) Modulation of P-glycoprotein at the blood-brain barrier: Opportunities to improve central nervous system pharmacotherapy. Pharmacol Rev 60:196-209.
- Najjar S, Pearlman DM, Alper K, Najjar A, Devinsky O (2013) Neuroinflammation and psychiatric illness. J Neuroinflammation 10: 43.
- Nnadi CU, Malhotra AK (2008) Individualizing antipsychotic drug therapy in schizophrenia: The promise of pharmacogenetics. Curr Psychiatry Rep 9: 313-318.
- Pacchioni AM, Gabriele A, Donovan JL, DeVane CL, See RE (2010) P-glycoprotein inhibition potentiates the behavioural and neurochemical actions of risperidone in rats. Int J Neuropsychopharmacol 3: 1067-1077.
- Peer D, Margalit R (2006) Fluoxetine and reversal of multidrug resistance. Cancer Lett 237: 180-187.
- Peng R, Zhang H, Zhang Y, Wei DY (2015) Effects of the ABCB1 (1199G>A) Polymorphism on steroid sex hormone-induced P-Glycoprotein expression, ATPase activity, and hormone efflux. Med Sci 3: 124-137.
- Potschka H, Loscher W (2007) Efflux transporters in the brain. Springer-Verlag Berlin Heidelberg; pp: 462-483.
- Pouget JG, Tahireh A (2014) Pharmacogenetics and outcome with antipsychotic drugs. Dialogues Clin Neurosci 16: 555-566.
- Reed A, Huie K, Perloff ES, Cassella JV, Takahashi LH (2012) Loxapine P-glycoprotein Interactions in vitro. Drug Metab Lett 6: 26-32.
- Schinkel AH, Wagenaar E, Mol CA (1996) P-glycoprotein in the blood-brain barrier of mice influences the brain penetration and pharmacological activity of many drugs. J Clin Invest 97: 2517-2524.
- Suzuki Y, Tsuneyama N, Fukui N, Sugai T, Watanabe J, et al. (2014) Effect of risperidone metabolism and P-glycoprotein gene polymorphism on QT interval in patients with schizophrenia. Pharmacogenomics J 14: 452-456.
- Tandon B, Vishal R, Bano G, Gupta S, Gilliani Z, et al. (2006) P-glycoprotein: Pharmacological relevance. Indian J Pharmacol 38: 13-24.
- Uhr M, Holsboer F, Müller MB (2002) Penetration of endogenous steroid hormones corticosterone, cortisol, aldosterone and progesterone into the brain is enhanced in mice deficient for both MDR1A and MDR1B P-glycoproteins. J Neuroendocrinol 14: 753-759.
- Uhr M, Tontsch A, Namendorf C, Ripke S, Lucae S, et al. (2008) Polymorphisms in the drug transporter gene ABCB1 predict antidepressant treatment response in depression. Neuron 57: 203-209.
- Umamaheswaran G, Kumar DK, Adithan C (2014) Distribution of genetic polymorphisms of genes encoding drug metabolizing enzymes & drug transporters-A review with Indian perspective. Indian J Med Res 139: 27-65.
- Vijayan NN, Mathew A, Balan S, Natarajan C, Nair CM, et al. (2012) Antipsychotic drug dosage and therapeutic response in schizophrenia is influenced by ABCB1 genotypes: A study from a South Indian perspective. Pharmacogenomics 13: 1119-1127.
- Wang JS, Zhu HJ, Markowitz JS, Donovan JL, deVane CL (2006) Evaluation of antipsychotic drugs as inhibitors of multidrug resistance transporter P-glycoprotein. Psychopharmacol 187: 415-423.
- Wang JS, Zhu HJ, Donovan JL, Yuan HJ, Markowitz JS, et al. (2009) Aripiprazole brain concentration is altered in P-glycoprotein deficient mice. Schizophr Res 110: 90-94.
- Weide K, Loovers H, Pondman K, Bogers J, Straaten V, et al. (2016) Genetic risk factors for clozapine-induced neutropenia and agranulocytosis in a Dutch psychiatric population. Pharmacogenomics J 17: 471-478.
- Wen PC, Verhalen B, Wilkens S, Mchaourab HS, Tajkhorshid E (2013) On the origin of large flexibility of P-glycoprotein in the inward-facing state. J Biol Chem 288: 19211-19220.
- Zhao R, Raub TJ, Sawada GA, Kasper SC, Bacon JA, et al. (2009) Breast cancer resistance protein interacts with various compounds in vitro, but plays a minor role in substrate efflux at the blood-brain barrier. Drug Metab Dispos 37: 1251-1258.
Citation: Alemayehu D, Melisie G, Taye K, Tadesse E (2019) The Role of ABC Efflux Transporter in Treatment of Pharmaco-Resistant Schizophrenia: A Review Article. Clin Pharmacol Biopharm 8: 189. DOI: 10.4172/2167-065X.1000189
Copyright: © 2019 Alemayehu D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 4041
- [From(publication date): 0-2019 - Oct 23, 2025]
- Breakdown by view type
- HTML page views: 3168
- PDF downloads: 873