Review Article Open Access
The Role of Coffee in the Therapy of Parkinson's Disease
Andrew Tran1, Cecilia Yijun Zhang1 and Chuanhai Cao1-3*
1The USF-Health Byrd’s Alzheimer’s Center and Research Institute, Tampa, FL, USA
2The College of Pharmacy at University of South Florida, Tampa, FL, USA
3Department of Neurology at Morsani College of Medicine, Tampa, FL, USA
- Corresponding Author:
- Chuanhai Cao
Assistant Professor
College of Pharmacy University of South Florida
USF-Health Byrd Alzheimer’s Institut
,4001 E. Fletcher Ave. Tampa FL, USA 33613
Tel: +813-3960742
E-mail: ccao@health.usf.edu
Received date: October 14, 2015; Accepted date: December 29, 2015; Published date: December 30, 2015
Citation: Tran A, Zhang CY, Cao C (2015) The Role of Coffee in the Therapy of Parkinson’s Disease. J Alzheimers Dis Parkinsonism 5:203. doi: 10.4172/2161-0460.1000203
Copyright: © 2015 Tran A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Alzheimers Disease & Parkinsonism
Abstract
Active compounds in coffee have been reported to possess therapeutic effects in treating Parkinson’s disease (PD). PD is a neurological degenerative disorder affecting millions of people around the world and to this day, there is no known cure or effective method to prevent or delay the disease progress. The most effective drug being used to treat symptoms of PD is L-3,4-dihydroxyphenyalanine (L-DOPA). However, chronic use of L-DOPA diminishes its effectiveness as well as inducing dyskinesia. Current studies have shown that coffee consumption enhances the effects of L-DOPA and has a preventative role in both the onset and progression of PD. Coffee also has been reported to increase the plasma levels of granulocyte colony-stimulating factors (G-CSF), a protein that also possesses neuroprotective functions. G-CSF may be used in combination treatments with L-DOPA and coffee to synergize their function for treating PD. The underlying processes are not fully understood as to how coffee interacts with both L-DOPA and G-CSF in PD models. This review attempts to provide a possible mechanism for these interactions. Understanding the metabolic interactions between coffee, L-DOPA, and G-CSF may shed light on new therapeutic treatments to treat PD.
Keywords
Parkinson’s disease; Coffee; L-DOPA; G-CSF
Parkinson’s Disease
Cases of PD have been documented as far back as the seventeenth and eighteenth century. It was not until the early nineteenth century that the English doctor James Parkinson, whom the disease is named after, published the first, detailed essay on the degenerative disorder [1]. Those afflicted by PD exhibit motor symptoms that include tremors, shakes, and increasing difficulties in movement [2]. PD patients also experience degeneration of neuronal cells in the substantia nigra region of the brain. The degeneration results in the loss of dopaminergic terminals that extend into the striatum. The diminished concentration of dopamine (DA) is suggested to be the cause of the motor symptoms of PD [3].
There currently are no cures for PD. Studies have found a variety of drugs that are able to manage the symptoms of the disease. However, these drugs do not stop the progression of PD and have undesirable side effects [4,5]. The treatments are palliative and fail to address the underlying causes. Other studies have found variable successes through surgical means in partially reversing the effects of PD in animal models [6-8]. However, these procedures are invasive and an assessment of these procedures is needed to determine their safety in clinical trials and as a means of treating PD in humans.
Coffee and PD
Multiple studies on the effects of coffee in PD onset have found a preventative correlation, where coffee consumption delays and prevents the onset of PD [9]. The mechanism as to how coffee accomplishes this has yet to be fully understood. However, dopamine receptors and neurotropic factors like G-CSF may all be involved in the benefits. Combining coffee with other compounds like L-DOPA and G-CSF may provide more efficient means in treating and preventing PD. It is critical to understand the mechanisms to which coffee interacts with these compounds and in the human body to improve the efficacy of current PD treatments while also proposing new combination treatments. Therefore, we will summarize published information about all factors associated to PD, and propose a possible a treatment in this review.
Coffee
Coffee is one of the most consumed beverages around the world. There are over 1000 compounds found in coffee [10]. There is substantial evidence that compounds in coffee, in particular caffeine, play a preventative role in PD development. At the molecular level, caffeine has been reported to antagonize and block adenosine A2A receptors in the striatum. The blockade of receptors increases and improves motor functions in models of PD [11,12]. In rat and mice PD models, caffeine has shown to counteract the symptoms and amplify the effects of L-DOPA treatments [13,14]. These studies highlight the effectiveness of coffee as an A2A antagonist in potential symptomatic treatments for PD. Caffeine has also been reported to exhibit neuroprotective properties and prevent the deterioration of dopaminergic cells in animal PD models [13,15]. Clinical trials have also found an inverse relationship between coffee consumption and the onset of PD. Increasing consumption of coffee has been correlated with a lower risk of PD development [16-19]. Coffee consumption also had a positive correlation with an increase of G-CSF plasma levels. However, when only caffeine was tested, minimal changes in plasma G-CSF levels were observed [20]. The results suggest that a compound(s) in coffee other than caffeine is responsible for the significant changes in plasma G-CSF levels. Further research is necessary to identify these unknown compounds.
Coffee and PD treatment
Coffee consumption is correlated with the prevention and treatment of PD. By drinking the beverage daily, this method may provide a basis for treating PD. Studies show that increasing the amount of coffee from less than one cup to several cups a day significantly decreases the chances of developing PD [21-23]. A possible strategy is to drink at least a cup of coffee daily as a person ages to prevent the onset of PD. It is not recommended to drink a substantial amount of coffee, especially at a young age as this can lead to addictive behaviors. Consumption in moderation is key to balancing the benefits and side effects for any treatment.
PD and Dopamine Receptors
Dopamine receptors
DA is the primary neurotransmitter that binds to DA receptors found on neurons in the central nervous system. The receptors play a part in many neurological processes. There are five subtypes of DA receptors: D1, D2, D3, D4, and D5 [24]. Dysfunction and the loss of the D1 and D2 DA receptors are associated with the onset of Parkinson’s disease [25]. These two receptors have opposite roles, where D1 receptors activate adenylate cyclase whereas D2 receptors inhibit it [26]. In PD, these receptors deteriorate and or malfunction and DA production and binding is inhibited, producing the characteristic motor symptoms of PD. Treatments include a variety of drugs that depress PD symptoms and replenish decreased levels of DA as well as delaying the onset of side effects from other medications [27,28].
Dopamine agonist treatments
One method in treating the symptoms of PD uses DA agonists. These compounds work by entering the body and behaving similar to DA and activate signaling pathways. In a typical presynaptic neuron, DA release results in the signaling of the postsynaptic neurons via D1 and D2 DA receptors. In PD cases, this action is blocked or inhibited from denervation of DA generating cells [29]. When used solely in treating early PD, DA agonists reduce symptoms of the disease by stimulating these DA-deficient neurons. This method is not the most efficient, but does postpone the use of L-DOPA, a more effective form of treatment that has also been linked to causing motor fluctuations [30-32].
Coffee and Dopamine
L-DOPA treatments
L-DOPA was the first drug used in treating PD in 1967 [33,34]. Since, L-DOPA has remained the most effective drug for treating PD [28,35]. Unlike DA, L-DOPA is able to cross the blood brain barrier (BBB) and enter the central nervous system (CNS). After entering the CNS, L-DOPA is converted into DA via the enzyme aromatic L-amino acid decarboxylase [36]. Chronic use of L-DOPA results in decreased production of naturally occurring L-DOPA in the body and becomes less effective in treating PD symptoms [35,37]. Despite being the most effective method in treating PD, L-DOPA is complicated by inducing an involuntary muscle-movement disorder, dyskinesia, from chronic use due to the induction of neurotoxicity [38,39]. L-DOPA has been hypothesized to over stimulate D1 and D2 receptors, leading to a cascade of events that eventually manifests into dyskinesia. Over accumulation of DA converted from L-DOPA in the areas of the brain as well as outside of it also have been suggested to induce dyskinesia [28,40]. Aside from these long-term side effects, acute treatment of L-DOPA can also induce systemic side effects including vomiting, nausea, and confusion [41]. New treatments are therefore necessary in treating those afflicted by PD.
L-DOPA treatments with coffee
Combining the therapeutic effects of coffee with L-DOPA provides a method of treatment for PD. L-DOPA treatment in patients over time becomes less effective and induces numerous side effects, one being dyskinesia. Coffee has been shown to increase the effectiveness of L-DOPA and a combination treatment would require less L-DOPA administration for the same desired effect. This combination treatment provides the benefit of delaying or preventing the development of L-DOPA induced-dyskinesia. Further studies are necessary to determine the safe and optimal concentrations of L-DOPA and coffee and how often they should be consumed.
Coffee and G-CSF in PD Treatment
G-CSF in current treatments
G-CSF, a neurotrophic factor, is a glycoprotein involved in stimulating bone marrow to release stem cells into the bloodstream [42]. G-CSF has been used to treat a variety of diseases including certain cancers [43,44] and neuronal structure defects [45]. G-CSF works through binding to its receptor and transposes its signals into the cell and activates signal pathways to neuron death. Receptors for G-CSF are expressed in the neurons of the brain and spinal cord. G-CSF has been found to promote the growth of nervous tissue, increase neuroplasticity, and prevent apoptosis [42,46]. Neurogenesis allows for new synapsis and neuron growth to combat the loss of nerve cells as a result of PD. Preventing apoptosis extends the longevity of cell life. Longer cell life, in particular immune and blood cells, provides for a stronger immune system and maintenance of resources that could also contribute in fighting and preventing the onset of PD.
G-CSF is normally produced in the human body. Chemical stimulants such as coffee or chemokines and sulfated polysaccharides have shown to increase the levels of the G-CSF in the blood [47-50]. Increasing the concentration of G-CSF may increase its effectiveness in treating PD. Animal models of Parkinson’s disease have shown that injections of recombinant human G-CSF stimulated the formation of new neuron synapses and even allowed mice injected with recombinant G-CSF to outperform those without injected G-CSF on cognitive tests [51,52]. Further research is necessary before clinical trials begin. However, these studies are promising and may eventually lead to clinical use of G-CSF in treating PD.
G-CSF treatments with coffee
Coffee has shown to not only enhance the effects of L-DOPA but also increase plasma G-CSF levels [20]. In this scheme, lower levels of L-DOPA can manage the symptoms of PD while G-CSF promotes the growth and development of nervous tissue that was loss from PD. Other compounds such as chemokines or injections of recombinant human G-CSF may be used to increase G-CSF levels [47-50]. The elevation in G-CSF can potentially halt and reverse the effects of PD and provide another possibility for treatment.
Treatment Mechanisms of Coffee in PD
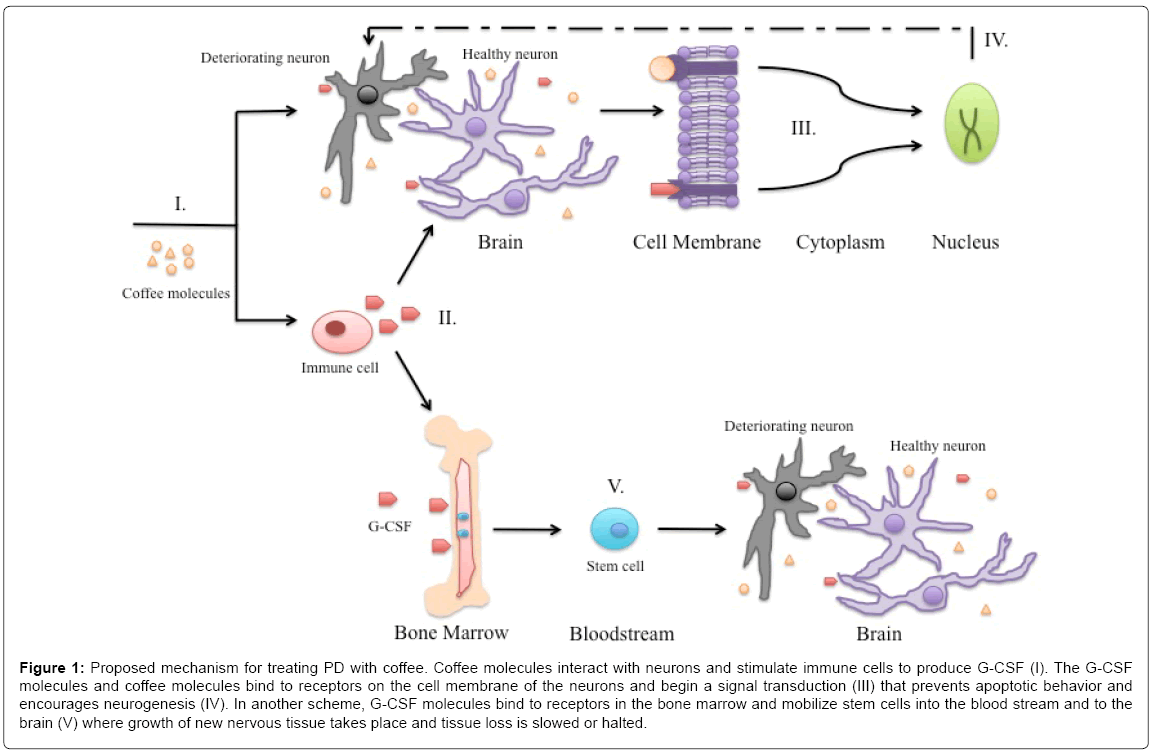
Based on many studies, coffee has shown to be a possible therapeutic agent in treating PD. A mechanism is proposed in the following diagram and we aim to discuss the effect coffee has on the neurons affected by PD.
Active compounds in coffee are able to penetrate the BBB and reach deteriorating neurons in the brain (Figure 1I). The coffee compounds, in particular caffeine, have neuroprotective qualities that stimulate the growth of the dopaminergic neurons. Caffeine may behave as an allosteric or competitive inhibitor on an enzyme that participates in converting L-DOPA to DA or in the following downstream signaling cascade when paired with L-DOPA. Further studies are needed to confirm and understand the molecular docking of these molecules and the enzymes responsible in this metabolic pathway. A possible candidate may be in the class of cytochrome P450 (P450, CYP) enzymes. Prior studies have confirmed that caffeine is metabolized by isoforms of CYP [53,54]. L-DOPA is converted to DA via a different enzyme, however, it has been reported that P450 mediates the formation from other precursors [55]. Caffeine acting as an inhibitor provides a possible explanation as to how it enhances the effects of L-DOPA in treating PD. In L-DOPA treatments, the drug’s effectiveness decreases from negative feedback. Too much of the drug attenuates the remaining neurons’ responses to DA in PD patients. The addition of coffee slows down the amount of L-DOPA into DA that could lead to overstimulation of dopaminergic receptors and prevent the attenuation from occurring. This process suggests an extended half-life of L-DOPA. L-DOPA has a half-life of 60–90 minutes [56]. Through inhibition, caffeine extends the time L-DOPA is present in the body before it is converted to DA. This too prevents impairment of normal neural responses form an accumulation of DA converted from L-DOPA.
Figure 1: Proposed mechanism for treating PD with coffee. Coffee molecules interact with neurons and stimulate immune cells to produce G-CSF (I). The G-CSF molecules and coffee molecules bind to receptors on the cell membrane of the neurons and begin a signal transduction (III) that prevents apoptotic behavior and encourages neurogenesis (IV). In another scheme, G-CSF molecules bind to receptors in the bone marrow and mobilize stem cells into the blood stream and to the brain (V) where growth of new nervous tissue takes place and tissue loss is slowed or halted.
The coffee compounds also have been shown to stimulate the production of G-CSF via immune cells (Figure 1II). G-CSF binds to receptors on the cell membrane of the neurons and a transmission of molecular signals from the neuron’s exterior is relayed to the nucleus (Figure 1III). Coffee molecules also may follow such a pathway. Signal transduction from either or both of the molecules target the nucleus and relay chemical signals that prevent apoptosis and extend the cells longevity (Figure 1IV). The mechanism may also apply to cells other than neurons, in particular those of the immune system. A recent study reported that the nervous system and immune system are connected with newly found lymphatic vessels in the brain [57]. Altered immunity has been suggested as a significant catalyst in a variety of neurological disorders like PD. Counteracting apoptosis extends the cell life of the immune system and provides a stronger defense towards PD.
G-CSF proteins can also bind to precursor cells in the bone marrow (Figure 1II). The interaction promotes the mobilization of stem cells in the bone marrow into the bloodstream (Figure 1V). The stem cells travel to the CNS and reach affected neurons in PD patients. In the brain, the stem cells can differentiate into neurons and replace those that are lost from PD.
Conclusion
So for coffee drinkers, there is a benefit to consuming the beverage. When used with L-DOPA, coffee has the potential to amplify the drug’s ability in treating the symptoms of PD. When paired with an increase in G-CSF levels, the combination treatment lay the groundwork for the possibility of not only relieving symptoms of PD, but as well as reversing the effects of the neurological disorder. It is important to note that further studies are needed to determine long-term effects of the combination treatment and what concentrations of coffee are optimal in treating PD. The history of mankind has used coffee for the several hundred years. Many of the compounds of coffee have been isolated and studied since. However, it is still necessary to continue research on coffee to find more effective ways to use it.
References
- Lees AJ (2007) Unresolved issues relating to the shaking palsy on the celebration of James Parkinson's 250th birthday. MovDisord 22 Suppl 17: S327-334.
- Calne DB (1993) Treatment of Parkinson's disease. N Engl J Med 329: 1021-1027.
- Calabresi P, Picconi B, Tozzi A, Di Filippo M (2007) Dopamine-mediated regulation of corticostriatal synaptic plasticity. Trends Neurosci 30: 211-219.
- Iravani MM, McCreary AC, Jenner P (2012) Striatal plasticity in Parkinson's disease and L-dopa induced dyskinesia. Parkinsonism RelatDisord 18 Suppl 1: S123-125.
- Zhang C, Steiner JP, Hamilton GS, Hicks TP, Poulter MO (2001) Regeneration of dopaminergic function in 6-hydroxydopamine-lesioned rats by neuroimmunophilin ligand treatment. J Neurosci 21: RC156.
- Bjorklund A (1992) Dopaminergic transplants in experimental parkinsonism: cellular mechanisms of graft-induced functional recovery. CurrOpinNeurobiol2:683-689.
- Nikkhah G, Cunningham MG, Jödicke A, Knappe U, Björklund A (1994) Improved graft survival and striatal reinnervation by microtransplantation of fetalnigral cell suspensions in the rat Parkinson model. Brain Res 633: 133-143.
- Rylander D, Bagetta V, Pendolino V, Zianni E, Grealish S, et al. (2013) Region-specific restoration of striatal synaptic plasticity by dopamine grafts in experimental parkinsonism. ProcNatlAcadSci U S A 110: E4375-4384.
- Costa J, Lunet N, Santos C, Santos J, Vaz-Carneiro A (2010) Caffeine exposure and the risk of Parkinson's disease: a systematic review and meta-analysis of observational studies. J Alzheimers Dis 20 Suppl 1: S221-238.
- Moon JK, Shibamoto T (2009) Role of roasting conditions in the profile of volatile flavor chemicals formed from coffee beans. J Agric Food Chem 57: 5823-5831.
- Fenu S, Morelli M (1998) Motor stimulant effects of caffeine in 6-hydroxydopamine-lesioned rats are dependent on previous stimulation of dopamine receptors: a different role of D1 and D2 receptors. Eur J Neurosci 10: 1878-1884.
- Kuwana Y, Shiozaki S, Kanda T, Kurokawa M, Koga K, et al. (1999) Antiparkinsonian activity of adenosine A2A antagonists in experimental models. AdvNeurol 80: 121-123.
- Chen JF, Xu K, Petzer JP, Staal R, Xu YH, et al. (2001) Neuroprotection by caffeine and A(2A) adenosine receptor inactivation in a model of Parkinson's disease. J Neurosci 21: RC143.
- Morelli M, Carta AR, Jenner P (2009) Adenosine A2A receptors and Parkinson's disease. HandbExpPharmacol: 589-615.
- Iida M, Miyazaki I, Tanaka K, Kabuto H, Iwata-Ichikawa E, et al. (1999) Dopamine D2 receptor-mediated antioxidant and neuroprotective effects of ropinirole, a dopamine agonist. Brain Res 838: 51-59.
- Fall PA, Fredrikson M, Axelson O, Granérus AK (1999) Nutritional and occupational factors influencing the risk of Parkinson's disease: a case-control study in southeastern Sweden. MovDisord 14: 28-37.
- Ross GW, Abbott RD, Petrovitch H, Morens DM, Grandinetti A, et al. (2000) Association of coffee and caffeine intake with the risk of Parkinson disease. JAMA 283: 2674-2679.
- Tan EK, Tan C, Fook-Chong SM, Lum SY, Chai A, et al. (2003) Dose-dependent protective effect of coffee, tea, and smoking in Parkinson's disease: a study in ethnic Chinese. J NeurolSci 216: 163-167.
- Sääksjärvi K, Knekt P, Rissanen H, Laaksonen MA, Reunanen A, et al. (2008) Prospective study of coffee consumption and risk of Parkinson's disease. Eur J ClinNutr 62: 908-915.
- Cao C, Wang L, Lin X, Mamcarz M, Zhang C, et al. (2011) Caffeine synergizes with another coffee component to increase plasma GCSF: linkage to cognitive benefits in Alzheimer's mice. J Alzheimers Dis 25: 323-335.
- Ascherio A, Zhang SM, Hernán MA, Kawachi I, Colditz GA, et al. (2001) Prospective study of caffeine consumption and risk of Parkinson's disease in men and women. Ann Neurol 50: 56-63.
- Benedetti MD, Bower JH, Maraganore DM, McDonnell SK, Peterson BJ, et al. (2000) Smoking, alcohol, and coffee consumption preceding Parkinson's disease: a case-control study. Neurology 55: 1350-1358.
- Hernán MA, Takkouche B, Caamaño-Isorna F, Gestal-Otero JJ (2002) A meta-analysis of coffee drinking, cigarette smoking, and the risk of Parkinson's disease. Ann Neurol 52: 276-284.
- Cools AR, Van Rossum JM (1976) Excitation-mediating and inhibition-mediating dopamine-receptors: a new concept towards a better understanding of electrophysiological, biochemical, pharmacological, functional and clinical data. Psychopharmacologia 45:243-254.
- Fuxe K, Manger P, Genedani S, Agnati L (2006) Thenigrostriatal DA pathway and Parkinson's disease. J Neural TransmSuppl: 71-83.
- Kebabian JW, Calne DB (1979) Multiple receptors for dopamine. Nature 277: 93-96.
- Bracco F, Battaglia A, Chouza C, Dupont E, Gershanik O, et al. (2004) The long-acting dopamine receptor agonist cabergoline in early Parkinson's disease: final results of a 5-year, double-blind, levodopa-controlled study. CNS Drugs 18: 733-746.
- Jenner P (2008) Molecular mechanisms of L-DOPA-induced dyskinesia. Nat Rev Neurosci 9: 665-677.
- Girault JA, Greengard P (2004) The neurobiology of dopamine signaling. Arch Neurol 61: 641-644.
- Mones RJ, Elizan TS, Siegel GJ (1971) Analysis of L-dopa induced dyskinesias in 51 patients with Parkinsonism. J NeurolNeurosurg Psychiatry 34: 668-673.
- Kanda T, Jackson MJ, Smith LA, Pearce RK, Nakamura J, et al. (1998) Adenosine A2A antagonist: a novel antiparkinsonian agent that does not provoke dyskinesia in parkinsonian monkeys. Ann Neurol 43: 507-513.
- Westin JE, Vercammen L, Strome EM, Konradi C, Cenci MA (2007) Spatiotemporal pattern of striatal ERK1/2 phosphorylation in a rat model of L-DOPA-induced dyskinesia and the role of dopamine D1 receptors. Biol Psychiatry 62: 800-810.
- Fahn S (2008) The history of dopamine and levodopa in the treatment of Parkinson's disease. MovDisord 23 Suppl 3: S497-508.
- Hornykiewicz O (2002) L-DOPA: from a biologically inactive amino acid to a successful therapeutic agent. Amino Acids 23: 65-70.
- Walkinshaw G, Waters CM (1995) Induction of apoptosis in catecholaminergic PC12 cells by L-DOPA. Implications for the treatment of Parkinson's disease. J Clin Invest 95: 2458-2464.
- Schwarz EJ, Alexander GM, Prockop DJ, Azizi SA (1999) Multipotential marrow stromal cells transduced to produce L-DOPA: engraftment in a rat model of Parkinson disease. Hum Gene Ther 10: 2539-2549.
- Curtis L, Lees AJ, Stern GM, Marmot MG (1984) Effect of L-dopa on course of Parkinson's disease. Lancet 2: 211-212.
- Cenci MA (2014) Presynaptic Mechanisms of l-DOPA-Induced Dyskinesia: The Findings, the Debate, and the Therapeutic Implications. Front Neurol 5: 242.
- Asanuma M, Miyazaki I, Ogawa N (2003) Dopamine- or L-DOPA-induced neurotoxicity: the role of dopamine quinone formation and tyrosinase in a model of Parkinson's disease. Neurotox Res 5: 165-176.
- Obeso JA, Rodríguez-Oroz MC, Rodríguez M, Lanciego JL, Artieda J, et al. (2000) Pathophysiology of the basal ganglia in Parkinson's disease. Trends Neurosci 23: S8-19.
- Godwin-Austen RB, Tomlinson EB, Frears CC, Kok HW (1969) Effects of L-dopa in Parkinson's disease. Lancet 2: 165-168.
- Schneider A, Krüger C, Steigleder T, Weber D, Pitzer C, et al. (2005) The hematopoietic factor G-CSF is a neuronal ligand that counteracts programmed cell death and drives neurogenesis. J Clin Invest 115: 2083-2098.
- Sheridan WP, Begley CG, Juttner CA, Szer J, To LB, et al. (1992) Effect of peripheral-blood progenitor cells mobilised by filgrastim (G-CSF) on platelet recovery after high-dose chemotherapy. Lancet 339: 640-644.
- Crawford J, Ozer H, Stoller R, Johnson D, Lyman G, et al. (1991) Reduction by granulocyte colony-stimulating factor of fever and neutropenia induced by chemotherapy in patients with small-cell lung cancer. N Engl J Med 325: 164-170.
- Copp AJ, Stanier P, Greene ND (2013) Neural tube defects: recent advances, unsolved questions, and controversies. Lancet Neurol 12: 799-810.
- Souza LM, Boone TC, Gabrilove J, Lai PH, Zsebo KM, et al. (1986) Recombinant human granulocyte colony-stimulating factor: effects on normal and leukemic myeloid cells. Science 232: 61-65.
- Laterveer L, Lindley IJ, Hamilton MS, Willemze R, Fibbe WE, et al. (1995) Interleukin-8 induces rapid mobilization of hematopoietic stem cells with radioprotective capacity and long-term myelolymphoid repopulating ability. Blood 85:2269-2275.
- Moore MA, Hattori K, Heissig B, Shieh JH, Dias S, et al. (2001) Mobilization of endothelial and hematopoietic stem and progenitor cells by adenovector-mediated elevation of serum levels of SDF-1, VEGF, and angiopoietin-1. Ann N Y AcadSci 938: 36-45.
- Sweeney EA, Priestley GV, Nakamoto B, Collins RG, Beaudet AL, et al. (2000) Mobilization of stem/progenitor cells by sulfated polysaccharides does not require selectin presence. ProcNatlAcadSci U S A 97: 6544-6549.
- Frenette PS, Weiss L (2000)Sulfatedglycans induce rapid hematopoietic progenitor cell mobilization: evidence for selectin-dependent and independent mechanisms. Blood 96:2460-2468.
- McCollum M, Ma Z, Cohen E, Leon R, Tao R, et al. (2010) Post-MPTP treatment with granulocyte colony-stimulating factor improves nigrostriatal function in the mouse model of Parkinson's disease. MolNeurobiol 41: 410-419.
- Sanchez-Ramos J1, Song S, Sava V, Catlow B, Lin X, et al. (2009) Granulocyte colony stimulating factor decreases brain amyloid burden and reverses cognitive impairment in Alzheimer's mice. Neuroscience 163: 55-72.
- Kot M, Daniel WA (2008) Caffeine as a marker substrate for testing cytochrome P450 activity in human and rat. Pharmacol Rep 60: 789-797.
- Tassaneeyakul W, Birkett DJ, McManus ME, Tassaneeyakul W, Veronese ME, et al. (1994) Caffeine metabolism by human hepatic cytochromes P450: contributions of 1A2, 2E1 and 3A isoforms. BiochemPharmacol 47: 1767-1776.
- Bromek E, Haduch A, Gołembiowska K, Daniel WA (2011) Cytochrome P450 mediates dopamine formation in the brain in vivo.J Neurochem 118: 806-815.
- Brooks DJ (2008) Optimizing levodopa therapy for Parkinson's disease with levodopa/carbidopa/entacapone: implications from a clinical and patient perspective. Neuropsychiatr Dis Treat 4:39-47.
- Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, et al. (2015) Structural and functional features of central nervous system lymphatic vessels. Nature 523: 337-341.
Relevant Topics
- Advanced Parkinson Treatment
- Advances in Alzheimers Therapy
- Alzheimers Medicine
- Alzheimers Products & Market Analysis
- Alzheimers Symptoms
- Degenerative Disorders
- Diagnostic Alzheimer
- Parkinson
- Parkinsonism Diagnosis
- Parkinsonism Gene Therapy
- Parkinsonism Stages and Treatment
- Stem cell Treatment Parkinson
Recommended Journals
Article Tools
Article Usage
- Total views: 24962
- [From(publication date):
December-2015 - Sep 01, 2025] - Breakdown by view type
- HTML page views : 23741
- PDF downloads : 1221