Research Article Open Access
A Three Year Assessment of the Influence of Physical Habitat, Pyrethroids and Metals on Benthic Communities in Two Urban Calfornia Streams
Lenwood W. Hall1*, William D. Killen1, Ronald D. Anderson1 and Raymond W. Alden III21University of Maryland, Agricultural Experiment Station, Wye Research and Education Center, Queenstown, Maryland, USA
2Northern Illinois University, Dekalb, Illinois, USA
- *Corresponding Author:
- Lenwood W. Hall
University of Maryland, Agricultural Experiment Station
Wye Research and Education Center, P.O. Box 169
Queenstown, Maryland 21658, USA
Tel: 410-827-8056
Fax: 410-827-9039
E-mail: lwhall@umd.edu
Received date: July 01, 2013; Accepted date: August 15, 2013; Published date: August 20, 2013
Citation: Hall LW, Killen WD, Anderson RD, Alden III RW (2013) A Three Year Assessment of the Influence of Physical Habitat, Pyrethroids and Metals on Benthic Communities in Two Urban Calfornia Streams. J Ecosys Ecograph 3:133.doi:10.4172/2157-7625.1000133
Copyright: © 2013 Hall LW, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
This three year study was designed to characterize benthic communities (macroinvertebrates) and physical habitat annually in two urban stream areas (Arcade Creek and Salinas streams) in California. Concurrent water quality evaluations, physical sediment parameters, pyrethroids, and bulk metals [including simultaneously extracted metals (SEM) and acid volatile sulfides (AVS) ratios] were also measured in both study areas. The relationship of various benthic metrics to physical habitat metrics, pyrethroids, and metals was evaluated for the the three year data set (2009 -2011) using univariate analysis, stepwise multiple regression analysis and canonical correlation analysis. Habitat conditions were determined to be poor in Arcade Creek and extremely poor in the Salinas streams. Tolerant benthic taxa were dominant in both study areas but degraded communities more apparent in Salinas streams. Canonical correlation analysis of the three year data set for Arcade Creek indicated that, while benthic community diversity was inversely related to toxicants (metals and pyrethroids) in the sediments, community composition appeared to be more closely related to physical habitat quality. The statistical analyses conducted on the three year data set from Salinas streams detected fewer and weaker relationships between benthic metrics and environmental variables than did the analyses of data from Arcade Creek, likely due to the greater degree of overall environmental degradation of the Salinas streams. However, habitat (particularly sediment deposition) appeared to be the most significant stressor. In summary the tthree year data sets for both streams showed more significant relationships with benthic metrics and habitat metrics than with pyrethroid or metals.
Keywords
Bioassessments; Pyrethroids; Metals; Physical Habitat, Urban California Streams
Introduction
Various investigators have reported that urbanization resulted in a major negative impact on aquatic ecosystems [1]. Large areas of impervious surfaces and high levels of hydraulic connection of impervious surfaces to streams, through stormwater pipes or drains, are two characteristics of the urban environment that can lead to major negative impacts on urban/residential streams [2]. These two characteristics of urban streams cause decreased levels of evapotranspiration and infiltration and rapid delivery of water to these lotic water bodies. Human activities in the urban environment can degrade aquatic ecosystems by altering one or more of the following principal groups of attributes: water or sediment quality; habitat structure; flow regime; energy source (food); or biotic interactions [3]. Urbanization specifically leads to fundamental changes in the hydrologic, hydraulic, erosional, and depositional characteristics of fluvial systems causing increased channel instability [4]. Urbanization in the western United States was reported to produce lower Index of Biotic Integrity (IBI) scores than activities such as logging and larger cities were reported to have lower IBI scores than smaller cities [5-7]. States such as California that have experienced expanded population growth in many urban and residential areas provide an example of multiple stressors in aquatic environments that can contribute to degradation. Studies designed to evaluate the potential impact of multiple stressors in these aquatic environments are therefore needed.
Common approaches used to assess impairments in water bodies are chemical monitoring, toxicity testing and biological assessments (bioassessment). Bioassessment, formally defined as a quantitative survey of physical habitat and biological communities of a water body, is a well established approach for determining the ecological condition of stream and river systems [3,8-11]. Bioassessments are particularly useful and provide realism in providing an observed response to environmental stressors because the status of resident biological communities provides a realistic and integrated response to multiple stressors. Assessments of benthic invertebrate assemblages and physical habitat (bioassessments) have been conducted in wadeable streams in California’s Central Valley for a number of years [12-21]. To date, the State of California lacks numeric objectives for instream biota to adequately protect its resources. In 2010 California’s State Water Resources Control Board initiated a project to develop “Biological Objectives” for the entire state of California. Past and present bioassessment data from the State of California will therefore be critical for the success of the “Biological Objectives” project.
In recent years, pyrethroid insecticides - replacements for the organophosphates used for structural pest control, landscape maintenance and residential home and garden use - were reported at potentially toxic concentrations in Arcade Creek in Sacramento, California [22] and three small urban streams in Salinas, California (Gabilan Creek, Natividid Creek, and Alisal Creek - Salinas streams) [23]. The toxicity assessment of pyrethroids in these two stream areas was based on sediment toxicity test results with a single species, the amphipod Hyalella azteca, which is highly sensitive to pyrethroids in laboratory based clean water toxicity tests [24]. Uncertainty exists when using only one species - particularly a highly sensitive one - as a benthic barometer for suggesting impairment of general ecosystem health. By contrast, bioassessments that include assessing the status of the entire benthic assemblage in concert with physical habitat assessments, as described above, are a preferred approach for determining the ecological status of these streams. In addition, the assumption that pyrethroids are the only stressor in urban waterbodies is questionable as other investigators have reported that chemical stressors such as metals [25,26] and polycyclic aromatic hydrocarbons (PAHs) [27] may also be present at concentrations that are potentially toxic to aquatic life. Previous studies in both an urban and residential stream in California have demonstrated that physical habitat limitations, rather than chemical stressors such as pyrethroids and metals, were the primary stressors associated with resident benthic community impairment [20].
The goal of this three year study was to characterize benthic communities (bioassessments) and physical habitat in an urban stream in Sacramento, California (Arcade Creek) and an urban stream network in Salinas, California (Gabilan Creek, Natividid Creek, and Alisal Creek - Salinas streams). Basic water quality parameters, eight specific pyrethroids, Total Organic Carbon (TOC), grain size, and bulk metals [including simultaneously extracted metals (SEM) and acid volatile sulfides (AVS)] were evaluated in the sediment of each stream area in concert with the bioassessments. The relationship of 14 different benthic community metrics (i.e., taxa richness, abundance) to physical habitat metrics, pyrethroids, and metals was evaluated. Benthic community data were interpreted in the context of biological expectations for these urban streams.
Materials and Methods
Site selection
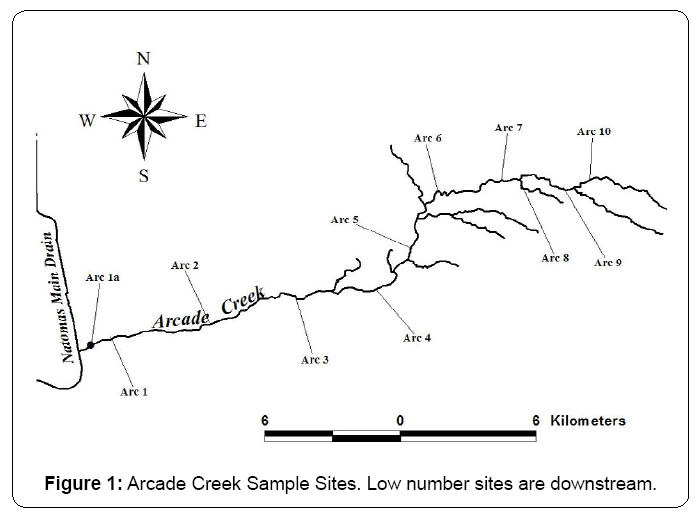
A total of 11 wadeable sites were sampled in Arcade Creek in late May to early June of 2009, 2010 and 2011 (Figure 1). All sites sampled except site ARC1a (the most downstream site) were previously sampled in 2000 as part of a bioassessment sampling program in urban areas [14]. Sites were selected to represent different types of habitat in this stream. Arcade Creek is located within the city of Sacramento in Sacramento County, California and is a tributary of the Sacramento River watershed. This creek is approximately 26 km long. Annual rainfall in the Arcade Creek area is approximately 55 cm a year. Arcade Creek flows from Orangevale, northeast of Greenback Lane and Kenneth Avenue to the American River via the Natomas East Main Drainage Canal. The watercourse is partially bordered with valley oaks, Fremont Cottonwood, small willows, and Oregon ash. Native vegetation persists in some natural and open areas providing habitat for wildlife. However, this habitat is vanishing due to bordering land development. Much of the area surrounding Arcade Creek is highly urbanized and consists of residential, commercial and light industry land use types with extensive impervious surface contributions from roads, parking lots and buildings.
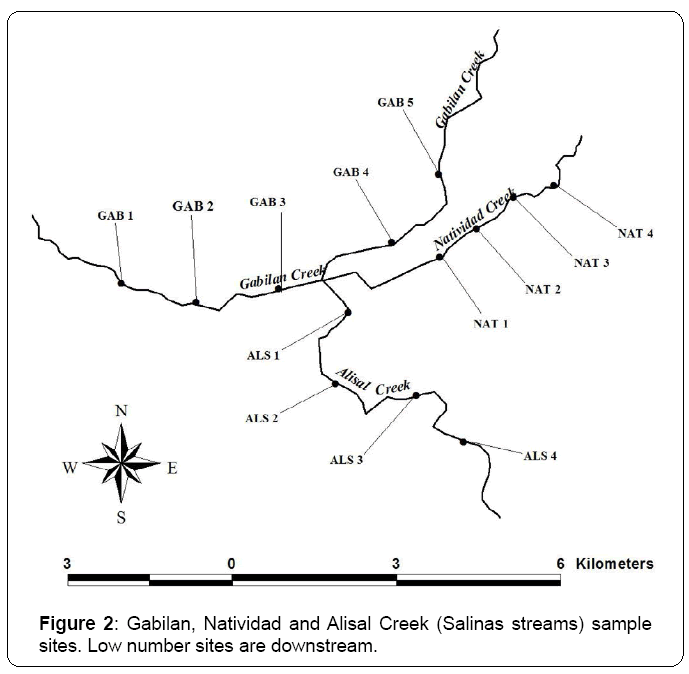
A total of 13 wadeable sites were sampled in Gabilan Creek, Natividad Creek, and Alisal Creek (Salinas streams) in May or early June of 2009, 2010 and 2011 (Figure 2). Salinas is the county seat of Monterey County and a major urban center with a population of approximately 150,000. Salinas is composed of residential, commercial and industrial areas. Salinas is also surrounded by agricultural lands. The farmland surrounding Salinas produces salad vegetables (e.g., lettuce and spinach) in addition to many other fruits and vegetables (e.g., strawberries, broccoli, cauliflower, celery, artichokes, and wine grapes). The length of each stream within the city limits of Salinas was as follows: 7.8 km for Gabilan Creek, 4.0 km for Natividad Creek and 5.5 km for Alisal Creek. Annual rainfall is approximately 33 cm and is largely limited to November through April. Gabilan Creek, Natividad Creek, and Alisal Creek are located within the city of Salinas. All three creeks originate in undeveloped land in hills northeast of the city, flow through agricultural lands, through the city and then back into agricultural lands. Water flow into the creeks varies significantly with season as large storm events in the winter produce the greatest amount of flow. Minimal flow is present during the summer.
Physical habitat assessments
Physical habitat was evaluated at each site concurrently with benthic collections, water quality evaluations, sediment parameters, pyrethroids, and metals. The physical habitat evaluation methods followed protocols described in Harrington and Born [28]. The physical habitat metrics used for this study were based on nationally standardized protocols [29]. A total of 10 continuous metrics scored on a 0-20 scale were evaluated. Other non-continuous metrics such as percent canopy, percent gradient, and substrate composition were also measured [28].
Benthic macroinvertebrate sampling
Benthic macroinvertebrates were collected in May through early June of 2009, 2010 and 2011 from three replicate samples at 11 sample sites in Arcade Creek and 13 sites in the Salinas streams as previously described (Figures 1 and 2). The sampling procedures were conducted in accordance with methods described in Harrington and Born [28]. Within each of these sample reaches, a riffle was located (if possible) for the collection of benthic macroinvertebrates. A tape measure was placed along the riffle and potential sampling transects were located at each meter interval of the tape. Using a random numbers table, three transects were randomly selected for sampling. Benthic samples were taken using a standard D-net with 0.5 mm mesh starting with the most downstream portion of the riffle. A 30.5 x 61 cm section of the riffle immediately upstream of the net was disturbed to a depth of 10 -15 cm to dislodge benthic macroinvertebrates for collection. Large rocks and woody debris were scrubbed and leaves were examined to dislodge organisms clinging to these substrates. Within each of the randomly chosen transects, three replicate samples were collected to reflect the structure and complexity of the habitat within the transect. If habitat complexity was lacking, samples were taken near the side margins and thalweg (deepest path) of the transect and the procedures described above were followed. All samples were preserved in 95% ethanol.
Due to the physical nature of these urban streams, it was often difficult to locate a substantial number of riffles to sample. Therefore, the alternative sampling method for non-riffle areas was used as outlined in Harrington and Born [28]. The non-riffle method was used for all Salinas stream sites for all years. For the 11 Arcade Creek sites the non-riffle method was used for 7 to 10 sites per year. This involved sampling the best available 30.5 x 61 cm sections of habitat throughout the reach using the same procedures described above. Nine 30.5 x 61 cm sections were randomly selected for sampling (i.e, stratified random sampling). Groups of three 30.5 x 61 cm sections were composited for each replicate for a total of three replicates per site.
It should be noted that benthic communities were generally not collected from fine grain (depositional) areas where pyrethroids and metals were collected as described below because CDFG protocols require sampling in best available habitat while sediment collection protocols require targeting fine grain areas. However, both benthic taxa and chemical constituents were collected within the same 75m sample segment of the stream. Previous studies conducted by our laboratory in two different California urban streams have shown that benthic metrics from depositional areas and non-depositional areas were not significantly different [30,31]. Therefore, sampling of benthic communities in non-depositional areas is considered representative of depositional areas where chemistry measurements were conducted at the sample site.
Taxonomy of benthic macroinvertebrates
The goal of this study was to identify all benthic samples to the species level if possible. For taxa such as oligochaetes and chironomids, family and genus level, respectively, were often the lowest level of identification possible. Benthic macroinvertebrate subsampling (resulting in a maximum of 300 individuals) and identifications was conducted by California’s Department of Fish and Game (CDFG). The benthic macroinvertebrate samples were subsampled and sorted by personnel at the CDFG Laboratory located at Chico State University. Level 3 identifications (species level identifications) followed protocols outlined in Harrington and Born [28]. Slide preparations and mounting for taxa such as midges and oligochaetes followed protocols from the United States Geological Survey National Quality Control Laboratory [32].
Taxonomic information was used to develop benthic metrics. Benthic metrics for wadeable streams in California were developed by California Department of Fish and Game [28]. The process of metric selection is driven by the goal of representing different categories of ecological information (i.e., richness, composition, tolerances and trophic measures). The various metrics were selected to maximize the effectiveness of detecting degradation in concert with communicating meanful ecological information.
Water quality and sediment measurements
The following water quality parameters were measured at each stream site and year using procedures described in Kazyak [33]: temperature, pH, salinity, specific conductivity, dissolved oxygen, and turbidity [34].
Grain size [35] and TOC [36] were measured on sediment samples collected from each site and year. Depositional areas - fine grain areas most likely to contain hydrophobic pesticides such as pyrethroids - were specifically sampled at each site and three to five sediment samples from depositional areas were composited for the final sample. A stainless steel spoon (similar to a scoop) was used to collect the top 2-3 cm of sediment from each site. Approximately one liter of sediment was collected from each site for grain size and TOC determinations as well as pyrethroids and metals. All sampling equipment was cleaned between sites using nitric acid, ethanol and distilled water. Sediment samples were stored in a cooler on ice in the field and later transferred to a refrigerator before shipment to Alpha Analytical Laboratory in Mansfield, Massachusetts for grain size and TOC analysis.
Pyrethroid analysis
The pyrethroids bifenthrin, cypermethrin, cyfluthrin, deltamethrin, esfenvalerate, fenpropathrin, lambda-cyhalothrin and permethrin residues were extracted from sediment by shaking with methanol/ water mixture and hexane for one hour. The sample was centrifuged and an aliquot of the upper hexane layer evaporated to dryness and re-dissolved in a small volume of hexane. The hexane sample was then subjected to a silica solid phase extraction (SPE) procedure prior to residue determination by gas chromatography with mass selective detection using negative ion chemical ionisation (GC-MS/NICI). The limit of quantitation (LOQ) of the method was 0.12 - 0.32 ng/g dry weight for bifenthrin, cypermethrin, cyfluthrin, deltamethrin, esfenvalerate, fenpropathrin, lambda-cyhalothrin and 1.2 - 3.2 ng/g dry weight for permethrin [37].
Bulk metals and SEM/AVS analysis
The following bulk metals with existing Threshold Effects Levels (TELs), conservative protective benchmarks, as described by Buchman [38] were measured on composited sediment samples for each site using EPA method 6020m: arsenic (As), cadmium (Cd), chromium (Cr), copper (Cu), lead (Pb), nickel (Ni) and zinc (Zn). The method detection limit (MDL) for these seven metals ranged from 0.02 to 0.90 μg/g dry weight. Mercury (Hg) was also measured on all sediment samples using EPA method 245.7m. The MDL for mercury was 0.02 μg/g dry weight.
Simultaneously extracted metals (SEM) analysis was conducted for Ni, Cu, Zn, Cd, and Pb using EPA method 200.8m. The MDLs (μmol/ dry g) for these SEMs were as follows: Ni (0.02), Cu (0.009), Zn (0.02), Cd (0.0005), and Pb (0.006). Acid volatile sulfides (AVS) were evaluated on sediment samples from each site using procedures described by Plumb [39]. SEM/AVS ratios were then developed for each site to provide insight on the bioavailability of these metals in sediment. The principle of SEM/AVS is based on the observation that there are some components in sediment that bind certain metals such that they are no longer available and therefore not toxic to benthic organisms [40,41]. Sulfides in sediments have the ability to bind with divalent metals such as cadmium, copper, lead, mercury, nickel and zinc and may render these metals unavailable to the extent sulfides are available. Sediments from the study sites were therefore analyzed for the amount of AVS and for the amount of freely available divalent metals as simultaneously extractable metals (SEM). Assuming that sulfides would bind with metals on a 1:1 molar basis, dividing SEM by the amount of AVS would suggest that these metals are available when the ratio is greater than 1.
Statistical analysis
The statistical approach used was similar to that used for previous bioassessment/multiple stressor studies in California’s urban streams [20]. Data for the 14 key benthic metrics were averaged across the three transects sampled for each site in Arcade Creek and the three Salinas streams. These data were merged with data sets of habitat metrics, sediment concentrations of metals, concentrations of simultaneously extracted metals (SEM) to AVS ratios, and sediment pyrethoid concentrations for each site. The sediment concentration data for each pyrethroid were converted to toxicity units (TUs) by standardizing them to 1% TOC and dividing by the Hyalella LC50 values that were also standardized to 1% TOC [22]. Metals in sediment concentrations were also standardized to their relative toxicities by dividing the dry weight concentrations of each metal by their respective Threshold Effects Levels (TEL) values.
The potential associations between the benthic metrics and the toxicity units for pyrethroids and metals were explored by a series of regression techniques. Prior to this analysis, all data were unit deviate standardized to place all dependent and independent variables on the same relative scales, as well as to produce more normal distributions. Univariate general linear model regressions were conducted for each study area to determine whether there were indications of significant relationships (α=0.01) between benthic metrics and specific pyrethroids (expressed as TUs) and specific metals (both metal concentrations to TELs ratios and SEM to AVS ratios for each metal) [42]. All the detailed univariate analysis results for both streams for 2009, 2010 and 2011 are provided in detail in separate documents [34,42,43].
A series of stepwise multiple regressions were conducted to determine potential relationships between the benthic metrics and pyrethroids (in TUs), metals (metals to TEL ratios), and habitat metrics [44]. Stepwise regressions were conducted separately for each of these three groups of independent variables, as well as with all variables combined into the same model. A second series of stepwise regressions were conducted for the benthic metrics versus principal components of the environmental data (pyrethroids, metals, and habitat metrics) that were produced by a principal components analysis (PCA) with an orthogonal rotation (Proc Factor, principal components method with a “varimax” rotation) [44].
Additional analyses were used to confirm the results of the multiple regressions, due to concerns over multicollinearity between the independent variables, a common characteristic of environmental data sets. A pair of complementary multivariate models involving the principal components was also employed: Model 1 was designed to take the effects of the toxicants (pyrethroids TUs or metals to TELs) on the benthic metrics into account before the effects of the habitat metrics were assessed; and Model 2 was designed to take the effects of the habitat metrics on the benthic metrics into account before the effects of potential toxicants were assessed. In Model 1, the principal components from the PCA on environmental data that were most highly “loaded” by the toxicants (i.e. those PCs identified principally by salient factor loadings of pyrethroids and/or metals) were forced into multiple regression models [42] to remove their potential effects, and the residuals were re-analyzed by the two stepwise regression series: the benthic metrics versus the habitat metrics; and the benthic metrics versus the habitat Principal Components (i.e., PCs associated with the habitat metrics).
In the Model 2 analyses, the effects of the PCs that were most highly loaded by the habitat metrics were removed in a similar manner prior to re-assessing the effects of the toxicants by the two regression series: the benthic metrics versus the pyrethroids and metals; and the benthic metrics versus the PCs associated with pyrethroids and/or metals. In each case, if the significant relationships between the benthic and the environmental variables were observed to persist from the results of the original stepwise regression series to the results from Models 1 or 2, they were considered to be less confounded by the effects of other environmental variables and, therefore, more compelling. The final series of multivariate analyses involved canonical correlation analyses of the PCs of the benthic data versus the PCs of the environmental data for the three year data sets [42]. This 3 year data set for each stream addressed the concerns of limited statistical power associated with only a single year of data.
Results
Physical habitat
Based on 10 instream and riparian physical habitat metrics in (Table 1), total 3 year mean physical habitat scores in Arcade Creek ranged from 85 to 121 (maximum possible total score is 200). Various physical habitat metrics were highly variable among the 11 Arcade Creek sites. For example, epifaunal substrate ranged from 7 to 15, Embeddedness ranged from 0 to 13, velocity/depth/diversity ranged from 4 to 13 and Sediment deposition ranged from 0 to 13. In general, total physical habitat scores in this urban stream were considered to be poor.
| Site | Veloc | Chan | Freq | Left | Right | L Bank | R Bank | L Bank | R Bank | Total | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Epi | Depth | Sedim | Flow | Chan | Bends | Bank | Bank | Veget. | Veget. | Ripar | Ripar | |||
| Subs | Embed | Divers | Depos | Status | Alt | Riffles | Stab | Stab | Protect | Protect | Zone | Zone | ||
| ARC 1a | 11 | 0 | 4 | 0 | 15 | 12 | 0 | 9 | 9 | 7 | 7 | 5 | 5 | 86 |
| ARC 1 | 12 | 1 | 6 | 4 | 16 | 12 | 2 | 9 | 9 | 8 | 8 | 5 | 5 | 98 |
| ARC 2 | 15 | 11 | 8 | 11 | 12 | 8 | 11 | 9 | 9 | 7 | 8 | 2 | 2 | 113 |
| ARC 3 | 14 | 3 | 6 | 5 | 16 | 10 | 2 | 5 | 4 | 7 | 7 | 2 | 3 | 85 |
| ARC 4 | 7 | 13 | 13 | 10 | 14 | 16 | 9 | 5 | 6 | 9 | 7 | 7 | 5 | 121 |
| ARC 5 | 7 | 12 | 11 | 11 | 15 | 15 | 11 | 4 | 4 | 8 | 8 | 5 | 3 | 115 |
| ARC 6 | 11 | 8 | 11 | 9 | 16 | 14 | 9 | 5 | 5 | 8 | 8 | 5 | 5 | 114 |
| ARC 7 | 11 | 10 | 8 | 9 | 13 | 17 | 11 | 3 | 3 | 8 | 8 | 6 | 6 | 113 |
| ARC 8 | 10 | 9 | 11 | 13 | 15 | 13 | 9 | 5 | 4 | 7 | 8 | 4 | 4 | 112 |
| ARC 9 | 9 | 8 | 8 | 8 | 14 | 16 | 9 | 4 | 4 | 9 | 8 | 6 | 4 | 107 |
| ARC 10 | 8 | 8 | 7 | 8 | 10 | 17 | 6 | 4 | 4 | 9 | 9 | 5 | 5 | 99 |
Table 1: Scoring of 3 year mean individual physical habitat metrics (0-20 scale) and 3 year mean total habitat scores (maximum of 200) for sites in Arcade Creek (ARC) Sacramento, California sampled during spring 2009-2011.
Total 3 year mean physical habitat scores in (Table 2) based on instream and riparian metrics in the Salinas streams ranged from 29 to 81. Most of the individual habitat metric scores from the 13 sites were extremely low particularly for Embeddedness, Sediment deposition, frequency of bends/ riffles, and all the riparian habitat metrics. Total habitat scores for the Salinas streams were very poor. These extremely poor total habitat scores were some of the lowest we have reported in California streams [20,45].
| Site | Veloc | Chan | Freq | Left | Right | L Bank | R Bank | L Bank | R Bank | Total | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Epi | Depth | Sedim | Flow | Chan | Bends | Bank | Bank | Veget. | Veget. | Ripar | Ripar | |||
| Subs | Embedd | Divers | Depos | Status | Alt | Riffles | Stab | Stab | Protect | Protect | Zone | Zone | ||
| GAB 1 | 3 | 0 | 5 | 1 | 16 | 2 | 1 | 2 | 1 | 2 | 1 | 1 | 1 | 37 |
| GAB 2 | 1 | 0 | 3 | 0 | 16 | 0 | 0 | 2 | 1 | 2 | 2 | 0 | 1 | 29 |
| GAB 3 | 7 | 0 | 4 | 2 | 15 | 2 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 34 |
| GAB 4 | 3 | 1 | 5 | 3 | 9 | 9 | 6 | 4 | 2 | 8 | 2 | 6 | 1 | 58 |
| GAB 5 | 7 | 6 | 2 | 6 | 10 | 8 | 5 | 6 | 5 | 6 | 8 | 4 | 5 | 79 |
| NAT 1 | 4 | 0 | 3 | 0 | 10 | 12 | 0 | 5 | 1 | 8 | 0 | 5 | 1 | 48 |
| NAT 2 | 11 | 4 | 7 | 7 | 14 | 11 | 3 | 3 | 6 | 5 | 6 | 1 | 3 | 81 |
| NAT 3 | 8 | 3 | 5 | 4 | 8 | 16 | 3 | 5 | 5 | 5 | 6 | 5 | 5 | 79 |
| NAT 4 | 4 | 0 | 4 | 0 | 10 | 17 | 1 | 4 | 4 | 9 | 9 | 7 | 6 | 73 |
| ALS 1 | 2 | 0 | 4 | 7 | 15 | 2 | 1 | 4 | 1 | 5 | 0 | 2 | 0 | 44 |
| ALS 2 | 1 | 0 | 2 | 2 | 14 | 4 | 0 | 4 | 3 | 5 | 2 | 4 | 3 | 45 |
| ALS 3 | 0 | 0 | 3 | 1 | 10 | 6 | 2 | 3 | 2 | 3 | 1 | 1 | 1 | 32 |
| ALS 4 | 1 | 0 | 2 | 0 | 12 | 4 | 0 | 1 | 4 | 1 | 6 | 0 | 5 | 36 |
Table 2: Scoring of 3 year mean individual physical habitat metrics (0-20 scale) and 3 year mean total habitat scores (maximum of 200) for sites sampled during spring 2009- 2011 in Salinas streams, Salinas, California.
Total organic carbon and grain size
The mean % TOC concentrations from the 3 year Arcade Creek data set ranged from 0.05 to 3.49 % with a 3 year mean of 0.74% (Table 3). TOC concentrations were higher at downstream sites ARC 1a and ARC 1 (3.49% and 2.7%) when compared with the upstream sites.
| Site ID | % TOC | % Sand | % Gravel | % Silt | % Clay |
|---|---|---|---|---|---|
| ARC 1a | 3.49 | 4.37 | 0.10 | 62.4 | 33.1 |
| ARC 1 | 2.70 | 29.4 | 0.03 | 52.2 | 18.4 |
| ARC 2 | 0.05 | 17.4 | 0.57 | 68.1 | 13.9 |
| ARC 3 | 0.59 | 63.7 | 0.30 | 29.3 | 6.73 |
| ARC 4 | 0.09 | 30.6 | 0.50 | 56.8 | 12.1 |
| ARC 5 | 0.10 | 50.0 | 0.03 | 39.4 | 10.6 |
| ARC 6 | 0.42 | 54.0 | 0.00 | 36.9 | 9.09 |
| ARC 7 | 0.26 | 43.2 | 0.03 | 47.1 | 9.67 |
| ARC 8 | 0.12 | 21.4 | 0.10 | 56.5 | 22.0 |
| ARC 9 | 0.16 | 27.2 | 0.67 | 44.4 | 27.7 |
| ARC 10 | 0.18 | 44.7 | 0.00 | 38.4 | 16.2 |
| GAB 1 | 2.01 | 13.5 | 0.00 | 59.4 | 27.1 |
| GAB 2 | 1.80 | 16.0 | 0.00 | 60.7 | 23.1 |
| GAB 3 | 2.11 | 12.1 | 0.00 | 58.2 | 29.6 |
| GAB 4 | 2.26 | 32.5 | 0.00 | 50.6 | 16.9 |
| GAB 5 | 2.36 | 34.2 | 0.00 | 45.5 | 20.1 |
| NAT 1 | 2.14 | 18.1 | 0.00 | 55.5 | 26.4 |
| NAT 2 | 1.78 | 20.0 | 0.00 | 57.2 | 22.7 |
| NAT 3 | 1.76 | 25.1 | 0.00 | 54.9 | 19.9 |
| NAT 4 | 1.21 | 6.11 | 0.00 | 68.4 | 25.5 |
| ALS 1 | 1.60 | 11.9 | 0.00 | 53.7 | 34.4 |
| ALS 2 | 1.71 | 13.1 | 0.00 | 62.8 | 24.1 |
| ALS 3 | 1.40 | 29.9 | 0.00 | 50.4 | 19.7 |
| ALS 4 | 1.15 | 19.5 | 0.00 | 56.5 | 24.0 |
Table 3: Three year mean TOC and grain size values for Arcade Creek in Sacramento, California and Salinas streams, Salinas, California during spring 2009-2011 sample events.
Three year mean % TOC values from Salinas stream sites ranged from 1.15 to 2.36% with a mean value of 1.79% (Table 3). There was no apparent spatial trend in TOC concentrations in the Salinas streams as the highest TOC value (2.36% at GAB 5) and the lowest value (1.15% at ALS 4) were both reported at upstream sites.
Three year mean % sand in Arcade Creek ranged from 4.4 to 54 % with a mean value of 35% (Table 3). The 3 year mean % gravel, % silt and % clay values for Arcade Creek were 0.21%, 48.3% and 16.3%, respectively.
For the Salinas streams, the 3 year mean percent sand ranged from 6.1 to 34.2% with a mean value of 19.4% (Table 3). The Salinas streams mean 3 year values were 0%, 56.4% and 24.11% for % gravel, % silt and % clay, respectively.
Bulk metals and SEM/AVS
The 3 year mean Arcade Creek bulk metal concentrations exceeded the Threshold Effect Levels (TELs) for at least one metal for all sites except ARC 3 and ARC 6 (Table 4). The downstream Arcade Creek sites ARC 1a and ARC 1 had the highest number of metals TEL exceedances (6 to 7 exceedances). The metals with the highest number of TEL exceedances in Arcade Creek were nickel (9 exceedances) and chromium (5 exceedances).
| Site | As | Cd | Cr | Cu | Pb | Hg | Ni | Zn |
|---|---|---|---|---|---|---|---|---|
| ARC 1a | 6.48 | 0.835 | 58.7 | 71.5 | 64.2 | 0.139 | 42.1 | 341 |
| ARC 1 | 4.99 | 1.023 | 52.8 | 60.5 | 55.4 | 0.107 | 37.2 | 288 |
| ARC 2 | 3.31 | 0.236 | 22.4 | 36.7 | 10.8 | 0.010 | 31.2 | 73.5 |
| ARC 3 | 2.39 | 0.134 | 24.6 | 16.9 | 16.3 | 0.024 | 15.1 | 59.1 |
| ARC 4 | 2.67 | 0.113 | 35.4 | 32.0 | 6.81 | 0.010 | 35.5 | 85.7 |
| ARC 5 | 3.04 | 0.060 | 33.5 | 17.3 | 5.58 | 0.010 | 23.2 | 45.0 |
| ARC 6 | 2.20 | 0.062 | 25.2 | 14.3 | 6.98 | 0.016 | 14.7 | 42.7 |
| ARC 7 | 9.87 | 0.110 | 49.5 | 33.4 | 8.98 | 0.017 | 49.1 | 67.6 |
| ARC 8 | 4.92 | 0.083 | 49.1 | 29.5 | 10.1 | 0.016 | 43.6 | 68.4 |
| ARC 9 | 3.72 | 0.113 | 44.3 | 28.5 | 8.59 | 0.020 | 38.2 | 65.2 |
| ARC 10 | 2.70 | 0.052 | 37.2 | 18.7 | 5.89 | 0.015 | 29.6 | 43.8 |
| GAB 1 | 7.57 | 1.231 | 72.0 | 49.3 | 28.5 | 0.089 | 81.3 | 308 |
| GAB 2 | 6.42 | 1.071 | 54.9 | 39.5 | 30.0 | 0.133 | 55.8 | 232 |
| GAB 3 | 8.71 | 0.939 | 55.9 | 39.0 | 20.8 | 0.054 | 42.9 | 225 |
| GAB 4 | 4.79 | 0.361 | 24.8 | 19.2 | 7.56 | 0.055 | 15.6 | 102 |
| GAB 5 | 4.43 | 0.338 | 24.8 | 17.9 | 8.34 | 0.044 | 14.1 | 109 |
| NAT 1 | 6.55 | 0.443 | 29.9 | 24.3 | 11.2 | 0.045 | 21.1 | 121 |
| NAT 2 | 5.21 | 0.344 | 27.2 | 18.5 | 8.18 | 0.030 | 19.0 | 85.4 |
| NAT 3 | 5.97 | 0.331 | 27.3 | 23.0 | 8.42 | 0.030 | 18.8 | 93.6 |
| NAT 4 | 7.30 | 0.398 | 41.3 | 18.9 | 11.1 | 0.032 | 26.5 | 90.7 |
| ALS 1 | 8.39 | 1.130 | 68.5 | 45.8 | 46.1 | 0.073 | 55.7 | 229 |
| ALS 2 | 8.64 | 0.823 | 45.0 | 36.8 | 16.6 | 0.032 | 27.1 | 250 |
| ALS 3 | 7.82 | 0.640 | 37.1 | 29.3 | 13.3 | 0.035 | 27.1 | 144 |
| ALS 4 | 7.31 | 0.951 | 44.0 | 25.5 | 12.8 | 0.040 | 42.1 | 132 |
| TEL | 5.9 | 0.596 | 37.3 | 35.7 | 35 | 0.174 | 18 | 123.1 |
Table 4: Three year mean bulk metal concentrations in sediment (μg/g dw) for sites sampled in Arcade Creek in Sacramento, California and in Salinas streams, Salinas, California in spring 2009-2011. Metals concentrations exceeding Threshold Effects Levels (TELs) are in bold.
Three year mean Salinas stream bulk metal concentrations exceeded the TEL for at least one metal for all sites except GAB 4 and GAB 5 (Table 4). The Salinas stream sites with the highest number of metals TEL exceedances were ALS 1 (7 excedances) and GAB 1, GAB 2, GAB 3, and ALS 2 (6 exceedances). Metals with the highest number of TEL exceedances were nickel (11 exceedances) and arsenic (10 exceedances).
The 3 year mean SEM/AVS ratios for Arcade Creek showed that for at least 9 of the 11 sites ratios were greater than 1 but for 2 of these sites (ARC 3 and ARC 6) no metals exceeded a TEL (Table 5). Therefore, these data suggest that metals are bioavailable and potentially toxic at 7 sites where SEM/AVS ratios exceed 1 and at least 1 metal exceeds a TEL.
| (concentrations in µmole/g dry weight) | Total | SEM/ | ||||||
|---|---|---|---|---|---|---|---|---|
| Station | AVS | Ni | Cu | Zn | Cd | Pb | SEM | AVSa |
| ARC 1a | 3.916 | 0.078 | 0.122 | 1.690 | 0.004 | 0.096 | 1.989 | 3.06 |
| ARC 1 | 3.311 | 0.062 | 0.174 | 1.475 | 0.004 | 0.079 | 1.734 | 0.74 |
| ARC 2 | 0.016 | 0.039 | 0.082 | 0.126 | 0.002 | 0.013 | 0.260 | 7.74 |
| ARC 3 | 0.014 | 0.026 | 0.070 | 0.261 | 0.002 | 0.024 | 0.382 | 7.16 |
| ARC 4 | 0.014 | 0.024 | 0.063 | 0.180 | 0.001 | 0.008 | 0.251 | 3.61 |
| ARC 5 | 0.027 | 0.025 | 0.075 | 0.080 | ND | 0.005 | 0.125 | 1.49 |
| ARC 6 | 0.848 | 0.020 | 0.120 | 0.184 | 0.001 | 0.007 | 0.250 | 1.33 |
| ARC 7 | 0.012 | 0.024 | 0.038 | 0.138 | 0.001 | 0.008 | 0.209 | 4.66 |
| ARC 8 | 0.017 | 0.008 | 0.015 | 0.060 | ND | 0.001 | 0.073 | 2.16 |
| ARC 9 | 0.021 | 0.020 | 0.027 | 0.043 | ND | 0.003 | 0.092 | 4.01 |
| ARC 10 | 0.061 | 0.037 | 0.029 | 0.037 | ND | 0.004 | 0.098 | 0.88 |
| GAB 1 | 2.101 | 0.214 | 0.325 | 2.404 | 0.007 | 0.045 | 2.884 | 4.55 |
| GAB 2 | 1.268 | 0.100 | 0.155 | 1.178 | 0.006 | 0.046 | 1.430 | 2.59 |
| GAB 3 | 1.028 | 0.086 | 0.130 | 0.901 | 0.004 | 0.026 | 1.103 | 2.71 |
| GAB 4 | 1.133 | 0.060 | 0.103 | 0.427 | 0.002 | 0.106 | 0.663 | 0.90 |
| GAB 5 | 0.477 | 0.057 | 0.162 | 0.513 | 0.003 | 0.021 | 0.700 | 1.01 |
| NAT 1 | 0.986 | 0.078 | 0.092 | 0.723 | 0.002 | 0.017 | 0.880 | 0.99 |
| NAT 2 | 0.561 | 0.064 | 0.060 | 0.507 | 0.002 | 0.015 | 0.647 | 1.83 |
| NAT 3 | 0.260 | 0.213 | 0.057 | 0.532 | 0.002 | 0.014 | 0.815 | 6.34 |
| NAT 4 | 0.019 | 0.049 | 0.042 | 0.184 | 0.002 | 0.009 | 0.285 | 36.1 |
| ALS 1 | 0.622 | 0.109 | 0.175 | 1.280 | 0.005 | 0.063 | 1.573 | 12.3 |
| ALS 2 | 0.398 | 0.075 | 0.137 | 1.688 | 0.003 | 0.022 | 1.880 | 10.8 |
| ALS 3 | 0.560 | 0.071 | 0.112 | 0.683 | 0.003 | 0.020 | 0.889 | 16.0 |
| ALS 4 | 0.740 | 0.085 | 0.092 | 0.549 | 0.004 | 0.011 | 0.709 | 19.6 |
aBold SEM/AVS ratios >1 suggest metals are bioavailable and may be toxic. The mean of the three SEM/AVS values for each year was used to calculate this value.
Table 5: Three year mean concentrations of acid volatile sulfides (AVS), simultaneously extracted metals (SEM), and the mean SEM/AVS ratio in sediment from the spring 2009-2011 Arcade Creek in Sacramento, California and Salinas Streams, Salinas, California sample sites.
For Salinas streams, the 3 year mean SEM/AVS ratios exceeded 1 at 11 of the 13 sites. However, for one of these sites (GAB 5) there were no exceedances of a metal TEL. Therefore, these results suggest that metals are bioavailable and potentially toxic at 10 sites where SEM/AVS ratios exceed 1 and at least 1 metal exceeds a TEL (Table 5).
Pyrethroids
The three year mean pyrethroid concentrations in Arcade Creek normalized to 1% TOC in (Table 6) showed that the highest concentrations of all pyrethroids were found at downstream sites [ARC 1a and ARC 1, see (Figure 1)]. These downstream sites are located in areas that are primarily composed of fine grained material (depositional areas) in sediment and would be expected to contain pyrethroids if they were present in the stream [46]. The range of concentrations by pyrethroid normalized to 1% TOC were as follows: bifenthrin (0.43 to 25.8 ng/g); fenpropathrin (ND to 0.05 ng/g); lambda-cyhalothrin (0.05 to 1.19 ng/g); permethrin (ND to 8.66 ng/g); cyfluthrin (0.03 to 4.97 ng/g); cypermethrin (0.18 to 3.48 ng/g); esfenvalerate (0.01 to 0.70) and deltamethrin (ND to 1.53 ng/g).
| Sample | % | Bifenthrin | Fenpropathrin | Lambda-cyhalothrin | Permethrin | Cyfluthrin | Cypermethrin | Esfenvalerate | Deltamethrin |
|---|---|---|---|---|---|---|---|---|---|
| ID | TOC | @ 1% TOC | @ 1% TOC | @ 1% TOC | @ 1% TOC | @ 1% TOC | @ 1% TOC | @ 1% TOC | @ 1% TOC |
| ARC 1a | 3.49 | 25.8 | 0.03 | 1.13 | 8.66 | 4.97 | 3.37 | 0.70 | 1.53 |
| ARC 1 | 2.70 | 21.5 | 0.05 | 1.19 | 8.38 | 4.93 | 3.48 | 0.64 | 1.26 |
| ARC 2 | 0.05 | 1.71 | ND | 0.07 | ND | 0.41 | 2.85 | 0.05 | ND |
| ARC 3 | 0.59 | 7.67 | 0.01 | 0.49 | 1.76 | 1.43 | 1.08 | 0.17 | 0.87 |
| ARC 4 | 0.09 | 1.68 | 0.02 | 0.07 | 1.21 | 0.70 | 0.41 | 0.04 | ND |
| ARC 5 | 0.10 | 2.73 | 0.03 | 0.10 | 0.83 | 0.77 | 1.88 | 0.05 | ND |
| ARC 6 | 0.42 | 5.70 | 0.02 | 0.47 | 2.06 | 1.62 | 1.73 | 0.14 | 0.70 |
| ARC 7 | 0.26 | 6.52 | 0.02 | 0.47 | 5.24 | 1.30 | 1.63 | 0.16 | 0.85 |
| ARC 8 | 3.12 | 0.43 | ND | 0.05 | ND | 0.03 | 0.19 | 0.01 | ND |
| ARC 9 | 0.16 | 1.10 | ND | 0.07 | 0.37 | 0.47 | 0.18 | 0.02 | ND |
| ARC 10 | 0.18 | 1.23 | ND | 0.06 | 0.65 | 1.06 | 0.18 | 0.03 | ND |
| GAB 1 | 2.01 | 3.91 | 1.25 | 1.09 | 13.4 | 1.95 | 1.59 | 0.71 | 0.76 |
| GAB 2 | 1.80 | 5.32 | 3.11 | 1.53 | 15.2 | 3.49 | 3.20 | 0.89 | 0.77 |
| GAB 3 | 2.11 | 5.70 | 3.92 | 4.34 | 40.6 | 1.46 | 4.21 | 3.20 | 0.33 |
| GAB 4 | 2.26 | 3.22 | 1.75 | 2.03 | 10.3 | 0.47 | 0.72 | 0.14 | 0.10 |
| GAB 5 | 2.36 | 3.35 | 1.89 | 1.68 | 9.45 | 1.16 | 1.46 | 0.20 | 0.38 |
| NAT 1 | 2.14 | 8.93 | 12.1 | 2.49 | 9.26 | 2.96 | 3.45 | 0.34 | 0.39 |
| NAT 2 | 1.78 | 8.06 | 7.72 | 1.96 | 8.97 | 1.34 | 1.57 | 0.22 | 0.26 |
| NAT 3 | 1.76 | 9.39 | 8.76 | 2.29 | 13.9 | 1.42 | 2.63 | 0.37 | 0.28 |
| NAT 4 | 1.21 | 10.4 | 16.4 | 12.1 | 656 | 0.28 | 77.0 | 0.07 | 0.33 |
| ALS 1 | 1.60 | 3.73 | 2.73 | 2.88 | 30.7 | 1.38 | 3.73 | 2.19 | 1.03 |
| ALS 2 | 1.71 | 8.04 | 8.06 | 8.19 | 60.8 | 2.27 | 11.3 | 5.25 | 0.53 |
| ALS 3 | 1.40 | 8.39 | 9.57 | 2.19 | 8.21 | 1.02 | 2.88 | 2.16 | 1.10 |
| ALS 4 | 1.15 | 4.88 | 3.15 | 5.72 | 28.2 | 1.43 | 7.16 | 1.56 | 0.22 |
Table 6: Three year mean pyrethroid concentrations (ng/g dry weight) normalized to 1% total organic carbon (TOC) for Arcade Creek in Sacramento, California and Salinas streams, Salinas, California sample sites during spring 2009-2011.
The three year mean pyrethroid concentrations in Salinas streams normalized to 1% TOC in (Table 6) showed no consistent spatial trend among the three streams from upstream to downstream; however, site NAT 4 (Figure 2) had the highest concentrations for 5 of the 8 pyrethroids. The range of concentrations by pyrethroid were as follows: bifenthrin (3.22 to 10.4 ng/g); fenpropathrin (1.25 to 16.4 ng/g); lambda cyhalothrin (1.09 to 12.1 ng/g); permethrin (8.21 to 656 ng/g); cyfluthrin (0.28 to 3.49 ng/g); cypermethrin (0.72 to 77 ng/g); esfenvalerate (0.07 to 5.25 ng/g); and deltamethrin (0.10 to 1.10 ng/g).
Three year mean TUs based on Hyalella azteca for Arcade Creek in (Table 7) showed that bifenthrin was the only individual pyrethroid with a TU greater than 1 at any site. Bifenthrin TUs were greater than 1 at 5 sites and the highest TUs were reported at the 2 most downstream sites. The sum of pyrethroid TUs exceeded a value of 1 at 6 of the 11 Arcade Creek sites.
| Sample ID | % TOC | Bifen TU | Fen TU | Lam-cy TU | Perm TU | Cyflu TU | Cyper TU | Esfen TU | Delt TU | Sum TU | Stream Rank |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ARC 1a | 3.49 | 4.96 | NA | 0.25 | 0.08 | 0.46 | 0.89 | 0.05 | 0.19 | 8.03 | 3 |
| ARC 1 | 2.70 | 4.13 | NA | 0.26 | 0.08 | 0.46 | 0.92 | 0.04 | 0.16 | 6.78 | 4 |
| ARC 2 | 0.05 | 0.33 | NA | 0.01 | ND | 0.04 | 0.75 | 0.00 | ND | 0.53 | 20 |
| ARC 3 | 0.59 | 1.47 | NA | 0.11 | 0.02 | 0.13 | 0.28 | 0.01 | 0.11 | 2.46 | 15 |
| ARC 4 | 0.09 | 0.32 | NA | 0.02 | 0.01 | 0.06 | 0.11 | 0.00 | ND | 0.49 | 21 |
| ARC 5 | 0.10 | 0.52 | NA | 0.02 | 0.01 | 0.07 | 0.49 | 0.00 | ND | 1.16 | 19 |
| ARC 6 | 0.42 | 1.10 | NA | 0.10 | 0.02 | 0.15 | 0.45 | 0.01 | 0.09 | 2.14 | 18 |
| ARC 7 | 0.26 | 1.25 | NA | 0.10 | 0.05 | 0.12 | 0.43 | 0.01 | 0.11 | 2.16 | 17 |
| ARC 8 | 0.12 | 0.08 | NA | 0.01 | ND | 0.01 | 0.05 | 0.00 | ND | 0.18 | 24 |
| ARC 9 | 0.16 | 0.21 | NA | 0.02 | 0.00 | 0.04 | 0.05 | 0.00 | ND | 0.38 | 23 |
| ARC 10 | 0.18 | 0.24 | NA | 0.01 | 0.01 | 0.10 | 0.05 | 0.00 | ND | 0.40 | 22 |
| GAB 1 | 2.01 | 0.75 | NA | 0.24 | 0.12 | 0.18 | 0.42 | 0.05 | 0.10 | 2.69 | 14 |
| GAB 2 | 1.80 | 1.02 | NA | 0.34 | 0.14 | 0.32 | 0.84 | 0.06 | 0.10 | 3.31 | 11 |
| GAB 3 | 2.11 | 1.10 | NA | 0.96 | 0.38 | 0.14 | 1.11 | 0.21 | 0.04 | 4.71 | 6 |
| GAB 4 | 2.26 | 0.62 | NA | 0.45 | 0.10 | 0.04 | 0.19 | 0.01 | 0.01 | 2.38 | 16 |
| GAB 5 | 2.36 | 0.64 | NA | 0.37 | 0.09 | 0.11 | 0.38 | 0.01 | 0.05 | 2.71 | 13 |
| NAT 1 | 2.14 | 1.72 | NA | 0.55 | 0.09 | 0.27 | 0.91 | 0.02 | 0.05 | 4.23 | 7 |
| NAT 2 | 1.78 | 1.55 | NA | 0.44 | 0.08 | 0.12 | 0.41 | 0.01 | 0.03 | 3.21 | 12 |
| NAT 3 | 1.76 | 1.81 | NA | 0.51 | 0.13 | 0.13 | 0.69 | 0.02 | 0.04 | 3.74 | 9 |
| NAT 4 | 1.21 | 2.00 | NA | 2.70 | 6.08 | 0.03 | 20.25 | 0.00 | 0.04 | 31.6 | 1 |
| ALS 1 | 1.60 | 0.72 | NA | 0.64 | 0.28 | 0.13 | 0.98 | 0.14 | 0.13 | 3.55 | 10 |
| ALS 2 | 1.71 | 1.55 | NA | 1.82 | 0.56 | 0.21 | 2.98 | 0.34 | 0.07 | 8.14 | 2 |
| ALS 3 | 1.40 | 1.61 | NA | 0.49 | 0.08 | 0.10 | 0.76 | 0.14 | 0.14 | 3.76 | 8 |
| ALS 4 | 1.15 | 0.94 | NA | 1.27 | 0.26 | 0.13 | 1.88 | 0.10 | 0.03 | 4.94 | 5 |
Table 7: Three year mean Toxic unit (TU) calculations for pyrethroids (1% TOC normalized and based on Hyalella) by site for Arcade Creek and Salinas Streams sites in spring 2009-2011. The sum of TUs by site and ranking by stream and all sites is also included. Toxic units > 1.0 are in bold type.
For the Salinas streams, the 3 year mean TU values exceeded 1 for 4 of the 8 pyrethroids as presented in (Table 7).
The number of sites with TUs greater than 1 by pyrethroid were as follows: 8 sites for bifenthrin; 3 sites for lambda-cyhalothrin; 1 site for permethrin; and 4 sites for cypermethrin. The sum of pyrethroid TUs exceeded a value of 1 for all 13 sites and the sum of pyrethroid TUs ranged from 2.38 to 31.6. It should be noted that all of the TU calculations were based on using Hyalella which is a highly sensitive species to pyrethroids [24].
Benthic communities
The five most dominant taxa in Arcade Creek in Table 8 showed that these dominant taxa comprised 53%, 61% and 46% of the taxa collected in 2009, 2010 and 2011, respectively. These dominant taxa were mainly represented by chironomidae and tubificinae. All of these dominant taxa were generally considered to be tolerant to environmental degradation [28,47]. The total number of taxa collected by year in Arcade Creek were as follows: 101 taxa in 2009; 92 taxa in 2010 and 110 taxa in 2011.
| Site ID | Year | Taxon | Higher Taxa | Total | Total % | Cumulative % |
|---|---|---|---|---|---|---|
| ARC | 2009 | Rheocricotopus sp. | Chironomidae | 1408 | 14.7 | 14.7 |
| Eukiefferiella sp. | Chironomidae | 1178 | 12.3 | 26.9 | ||
| Nais communis/ variabilis | Naidinae | 1133 | 11.8 | 38.7 | ||
| Unid Imm Tubificid | Tubificinae | 850 | 8.85 | 47.6 | ||
| Cricotopus sp. | Chironomidae | 536 | 5.58 | 53.1 | ||
| ARC | 2010 | Tubificinae | Tubificinae | 1496 | 17.0 | 17.0 |
| Nais communis/ variabilis | Naidinae | 1373 | 15.6 | 32.6 | ||
| Eukiefferiella sp. | Chironomidae | 1171 | 13.3 | 45.9 | ||
| Cricotopus sp. | Chironomidae | 897 | 10.2 | 56.1 | ||
| Dero digitata | Naidinae | 413 | 4.69 | 60.8 | ||
| ARC | 2011 | Tubificinae unid.imm. | Tubificinae | 1438 | 15.7 | 15.7 |
| Nais communis/ variabilis | Naidinae | 826 | 9.00 | 24.7 | ||
| Rheotanytarsus | Chironomidae | 686 | 7.47 | 32.1 | ||
| Simulium | Simuliidae | 685 | 7.46 | 39.6 | ||
| Physa | Physidae | 602 | 6.56 | 46.2 | ||
| SS | 2009 | Tubificid unid. Imm. | Tubificinae | 2346 | 21.1 | 21.1 |
| Physa sp. | Physidae | 1758 | 15.8 | 36.9 | ||
| Nais communis/ variabilis | Naidinae | 991 | 8.90 | 45.7 | ||
| Cricotopus sp. | Chironomidae | 949 | 8.52 | 54.3 | ||
| Chironomus sp. | Chironomidae | 646 | 5.80 | 60.1 | ||
| SS | 2010 | Tubificinae | Tubificinae | 2610 | 29.9 | 29.9 |
| Pisidium sp. | Sphaeriidae | 1983 | 22.7 | 52.6 | ||
| Dugesia tigrina | Planariidae | 1268 | 14.5 | 67.1 | ||
| Nais communis/ variabilis | Naidinae | 331 | 3.79 | 70.9 | ||
| Enchytraeidae | Enchytraeidae | 278 | 3.18 | 74.1 | ||
| SS | 2011 | Nais communis/ variabilis | Naidinae | 1645 | 15.5 | 15.5 |
| Tubificinae unid.imm. | Tubificinae | 1444 | 13.6 | 29.2 | ||
| Dugesia tigrina | Tubificinae | 1245 | 11.8 | 40.9 | ||
| Cricotopus | Chironomidae | 739 | 6.98 | 47.9 | ||
| Chironomus | Chironomidae | 708 | 6.69 | 54.6 |
Table 8: Total and taxon abundance by year for benthic macroinvertebrates in Arcade Creek (ARC) and the Salinas Streams (SS) sites. Only the top five dominant taxa are listed for each year and stream. A detailed presentation of all benthic taxa data are available [34,42,48].
The five most dominant taxa in Salinas streams comprised 60%, 74% and 55% of the taxa collected in 2009, 2010 and 2011, respectively (Table 8). As reported above for Arcade Creek, these dominat taxa were generally considered to be tolerant of environmental degradation [28,47]. The number of taxa collected by year in the Salinas streams were 76 taxa in 2009, 68 taxa in 2010 and 71 taxa in 2011. It is noteworthy that the number of different taxa collected in Salinas streams was lower than in Arcade Creek.
Among the 14 benthic metrics in (Table 9) for Arcade Creek, taxonomic richness ranged from 23 to 40; % Dominant taxa ranged from 18 to 37; Shannon diversity ranged from 1.9 to 2.9; % Tolerant taxa ranged from 38 to 58; and abundance ranged from 1,273 to 6,965. Lower taxonomic richness and Shannon diversity scores and lack of EPT taxa at the most downstream site (ARC 1a (Figure 1)) would suggest that benthic communities at this site is more impaired than the other sites.
| Site | ARC1a | ARC1 | ARC2 | ARC3 | ARC4 | ARC5 | ARC6 | ARC7 | ARC8 | ARC9 | ARC10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxonomic Richness | 23.3 | 35.0 | 38.0 | 40.0 | 33.0 | 28.7 | 33.3 | 37.0 | 33.3 | 32.3 | 31.0 |
| % Dominant Taxon | 36.8 | 30.2 | 20.9 | 18.0 | 25.3 | 29.3 | 22.4 | 14.5 | 19.7 | 30.2 | 37.1 |
| # Ephemeroptera Taxa | 0.0 | 0.0 | 2.3 | 0.7 | 2.0 | 2.3 | 1.0 | 1.0 | 0.7 | 2.0 | 0.3 |
| # EPT Taxa | 0.0 | 0.3 | 2.3 | 1.0 | 3.0 | 2.3 | 1.0 | 1.7 | 0.7 | 2.3 | 0.3 |
| EPT Index (%) | 0.0 | 0.0 | 2.0 | 0.7 | 6.7 | 13.0 | 3.0 | 1.0 | 0.3 | 1.3 | 0.0 |
| Shannon Diversity | 1.9 | 2.3 | 2.5 | 2.7 | 2.3 | 2.1 | 2.7 | 2.9 | 2.7 | 2.3 | 2.1 |
| Tolerance Value | 8.7 | 8.7 | 7.4 | 7.6 | 7.6 | 7.5 | 7.4 | 7.2 | 7.6 | 7.4 | 8.0 |
| % Tolerant Taxa (8-10) | 57.7 | 58.0 | 37.7 | 50.7 | 40.0 | 42.3 | 47.7 | 41.3 | 47.7 | 43.3 | 47.0 |
| % Collector-Filterers | 1.0 | 4.3 | 18.7 | 11.0 | 12.3 | 4.0 | 5.3 | 15.7 | 13.0 | 14.3 | 3.3 |
| % Collectors Gatherers | 79.0 | 71.7 | 53.7 | 74.7 | 52.3 | 60.7 | 59.7 | 61.3 | 55.7 | 53.3 | 75.0 |
| % Grazers | 9.3 | 7.3 | 2.0 | 8.3 | 1.0 | 1.0 | 1.3 | 1.7 | 0.3 | 0.3 | 0.7 |
| % Predators | 6.0 | 10.3 | 2.3 | 1.3 | 0.7 | 0.3 | 0.0 | 1.0 | 7.7 | 0.7 | 0.7 |
| % Shredders | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | 0.3 | 0.0 |
| Abundance (#/sample) | 4222 | 5006 | 3696 | 1273 | 5220 | 6965 | 5021 | 1931 | 3044 | 4244 | 6780 |
Table 9: Three year mean benthic metrics by site for the 11 Arcade Creek sites sampled in spring 2009-2011.
The three year mean values for 14 benthic metrics in Salinas streams by site presented in (Table 10) showed the following ranges for key metrics: taxonomic richness ranged from 16 to 29; % Dominant taxa ranged from 27.7 to 61.2; Shannon diversity ranged from 1.2 to 2.3; % Tolerant taxa ranged from 45 to 67; and abundance ranged from 1,154 to 7,662. There were no obvious spatial trends in the metrics. However, the overall general condition of the benthic communities in the Salinas streams appeared to be lower than Arcade Creek based on lower values for taxonomic richness and Shannon Diversity, higher % Dominant taxa scores and lack of EPT taxa. This result is not surprising based on the very poor physical habitat conditions in this stream as previously discussed.
| Site | GAB1 | GAB2 | GAB3 | GAB4 | GAB5 | NAT1 | NAT2 | NAT3 | NAT4 | ALS1 | ALS2 | ALS3 | ALS4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxonomic Richness | 25.3 | 20.7 | 21.7 | 29.3 | 29.0 | 25.3 | 26.0 | 19.3 | 24.3 | 21.7 | 16.0 | 17.3 | 19.0 |
| % Dominant Taxon | 27.7 | 35.8 | 30.8 | 32.1 | 38.6 | 38.6 | 30.3 | 61.2 | 29.7 | 43.1 | 53.8 | 45.1 | 56.6 |
| # Ephemeroptera Taxa | 0.0 | 0.0 | 0.0 | 0.0 | 0.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| # EPT Taxa | 0.0 | 0.0 | 0.0 | 0.0 | 1.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| EPT Index (%) | 0.0 | 0.0 | 0.0 | 0.0 | 1.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Shannon Diversity | 2.3 | 1.9 | 1.8 | 2.2 | 2.1 | 2.0 | 2.1 | 1.3 | 2.3 | 1.6 | 1.2 | 1.8 | 1.4 |
| Tolerance Value | 7.6 | 6.9 | 8.5 | 8.1 | 8.2 | 8.1 | 7.8 | 6.2 | 7.9 | 8.2 | 8.8 | 8.5 | 9.1 |
| % Tolerant Taxa (8-10) | 45.3 | 54.7 | 52.3 | 45.0 | 51.3 | 54.0 | 54.7 | 65.0 | 53.0 | 55.7 | 66.7 | 52.0 | 58.7 |
| % Collector-Filterers | 9.7 | 4.0 | 20.3 | 7.0 | 1.3 | 24.3 | 29.3 | 9.3 | 2.0 | 28.7 | 41.3 | 28.3 | 20.0 |
| % Collectors Gatherers | 70.0 | 58.0 | 69.0 | 81.3 | 89.0 | 70.7 | 48.7 | 34.7 | 90.0 | 50.7 | 53.3 | 64.3 | 74.7 |
| % Grazers | 0.3 | 0.3 | 0.0 | 0.0 | 0.0 | 0.3 | 0.7 | 0.0 | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 |
| % Predators | 15.3 | 35.7 | 9.3 | 9.0 | 2.0 | 2.7 | 15.0 | 51.7 | 3.7 | 9.7 | 1.3 | 2.7 | 1.3 |
| % Shredders | 0.0 | 0.0 | 0.0 | 0.0 | 1.3 | 0.0 | 0.3 | 0.0 | 0.7 | 0.0 | 0.0 | 0.0 | 0.0 |
| Abundance (#/sample) | 1517 | 2640 | 6576 | 6418 | 7662 | 3927 | 6063 | 4979 | 2329 | 4203 | 7103 | 1154 | 3098 |
Table 10: Three year mean benthic metrics by site for the 13 Salinas Streams sites sampled in spring 2009-2011.
Benthic metrics vs. pyrethroids, metals and habitat metrics for both streams
Interpretation of the relationships of benthic metrics to environmental stressors requires an understanding of how different benthic metrics are expected to respond to impairment. In the statistical analyses (Table 11), the expected response of the various benthic metrics to impairment is as follows according to Harrington and Born [28].
| Benthic Metric | Response to Impairment |
|---|---|
| Taxonomic Richness | Decrease |
| % Dominant Taxon | Increase |
| Emphemeroptera Taxa | Decrease |
| EPT Taxa | Decrease |
| EPT Index (%) | Decrease |
| Shannon Diversity | Decrease |
| Tolerance Value | Increase |
| % Tolerant Taxa | Increase |
| % Collectors/Filterers | Increase |
| % Collectors/Gatherers | Increase |
| % Grazers | Variable |
| % Predators | Variable |
| % Shredders | Decrease |
| Abundance | Decrease |
Table 11: Expected response of the various benthic metrics.
Arcade creek
The results of the stepwise multiple regression analyses of the 3 year data sets from Arcade Creek are shown in (Table 12). The benthic metric Taxonomic richness displayed an inverse relationship to total pyrethroid TUs, whereas % Dominant taxon was directly related to it (Table 12a, column a.1). Both of these relationships were confirmed by the Model 2 analyses. It is not surprising that the metric Taxonomic richness, which tends to decrease with environmental impairment, also displayed an inverse relationship with PC1, the principal component that was heavily loaded by all pyrethroids, mercury, and zinc (Table 12a, column a.2.), while the % Dominant taxon, a metric which tends to increase with stress, was directly related to PC1. Both of these relationships were confirmed by the Model 2 analyses. The % Dominant taxon metric was also directly related to deltamethrin and Vegetative protection and inversely related to lamda-cyhalothrin, but these relationships were not confirmed by Model 1 and 2 analyses, respectively.
| Benthic Metrics | a.1. Significant Environmental Variables (R2) | a.2. Significant Environmental Principal Components* |
|---|---|---|
| Taxonomic Richness | -Total TUs*** (0.25) | -PC1***(0.29) |
| % Dominant Taxon | +Total TUs*** (0.31), +Vegetative protection (0.11), -Lambda-cyhal. (0.11), +Deltamethrin (0.08) | +PC1***(0.28) |
| Ephemeroptera Taxa | +Frequency of riffles/bends ** (0.32) | +PC2** (0.16) |
| EPT Taxa | +Embeddedness ** (0.30) | +PC2** (0.18) |
| EPT Index (%) | -Permethrin (0.20), +Cypermethrin (0.18), +Embeddedness ** (0.16) | NS |
| Shannon Diversity | -Zn *** (0.21), +%Fines (0.11) | -PC1*** (0.18) |
* The environmental variables with the largest loadings for the environmental principal components (proportion of variance explained by PCs are in parentheses) are: PC1
(0.49) = +TUs for all pyrethroids, +Mercury to TEL and +Zinc to TEL; PC2 (0.12) = +Total Score, +Embeddedness, +Frequency of riffles/bends, +Sediment deposition,
+Velocity depth regimes, and -%Fines; PC3 (0.10) = -%Canopy Cover, +Bank Stability, +Cadmium to TEL, +Lead to TEL, +Zinc to TEL, and +Copper to TEL; PC4 (0.08) =
+Nickel to TEL, +Arsenic to TEL, +Chromium to TEL, and +Total metals to TEL; PC5 (0.05) = -Riparian vegetative zone, +Epifaunal substrate/available cover, +%Boulder,
-Channel alteration, and +%Cobble; PC6 (0.04) = +Channel Flow.
** Variables that remained significant after the toxicant-loaded principal components (PC1, PC3 and PC4) were forced into the stepwise regressions prior to the testing of
the habitat variables (Model 1 regression confirmation; see text for details).
*** Variables that remained significant after the habitat principal components (PC2, PC5, and PC6) were forced into the stepwise regressions prior to the testing of the toxic
variables (Model 2 regression confirmation; see text for details).
Table 12: Results of stepwise multiple linear regression models for the three year data sets: a.1. benthic metrics versus toxicity units for pyrethroids, habitat metrics, and metals to TEL ratios for Arcade Creek; and a.2. benthic metrics versus principal components of the environmental data for Arcade Creek; b.1. benthic metrics versus toxicity units for pyrethroids, habitat metrics, and metals to TEL ratios for the three Salinas streams; and b.2. benthic metrics versus principal components of the environmental data for the three Salinas streams. Only variables that were significant at α=0.01 were included in the models (NS= not significant; NA=not analyzed because few, if any, of the samples had the benthic metric present). The direction of the relationship for each significant variable is indicated (+ = direct; - = inverse), as is the contributed R2 value.
| Benthic metrics | a.1. Significant Variables (R2) | a.2. Principal Components |
|---|---|---|
| Tolerance Value | +Mercury *** (0.39) | -PC2**(0.20) |
| % Tolerant Taxa (8-10) | +Mercury*** (0.53) | -PC2**(0.24), + PC6**(0.14) +PC1*** (0.11), |
| % Collectors/Filterers | +Embeddedness (0.17), -Velocity depth regimes (0.11) | NS |
| % Collectors/Gatherers | -Embeddedness** (0.30) | -PC2** (0.21) |
| % Grazers | +Total TUs*** (0.68), -Riparian vegetative zone** (0.11) | +PC1*** (0.57), +PC5** (0.12) |
| % Predators | +Lead *** (0.29) | NS |
| % Shredders | NA | NA |
| Abundance (#/sample) | NS | NS |
Regression Models for Arcade Creek.
** Variables that remained significant after the toxicant-loaded principal components (PC1, PC3 and PC4) were forced into the stepwise regressions prior to the testing of
the habitat variables (Model 1 regression confirmation; see text for details).
*** Variables that remained significant after the habitat principal components (PC2, PC5, and PC6) were forced into the stepwise regressions prior to the testing of the toxic
variables (Model 2 regression confirmation; see text for details).
Table 12a: Regression models for Arcade Creek.
Shannon Diversity, another diversity-related benthic metric, displayed an inverse relationship with zinc to TEL and PC1, the principal component that was positively loaded by toxicants, including pyrethroids, mercury and zinc. The Model 2 analyses confirmed both of these relationships. However, causality cannot be inferred by the inverse relationship with zinc, since this metal is very highly correlated (p≤0.0001) to all of the pyrethroids and all of the metals except arsenic and nickel. Shannon Diversity was also shown to be directly related to % Fines, but this relationship was not confirmed by the Model 1 analysis.
The Ephemeroptera taxa metric was directly related to Frequency of riffles/bends, and EPT taxa and EPT index were both directly related to the habitat metric Embeddedness. All of these relationships were confirmed by the Model 1 analyses. These benthic metrics, which tend to decrease with environmental impairment, also displayed direct relationships with PC2, the principal component that was positively associated with habitat metrics such as Total score, Embeddedness, Frequency of riffles/bends, Sediment deposition, Velocity depth regimes and coarser sediments (Table 12a, column a.2.). These relationships were confirmed by the Model 1 analyses on the principal components. The EPT index metric was also inversely related to permethrin and directly related to cypermethrin, but neither of these relationships was confirmed by the Model 2 analyses.
The benthic metrics Tolerance value and % Tolerant taxa were both apparently directly related to mercury to TEL, and the relationships were confirmed by the Model 2 analysis. As was the case of the apparent inverse relationship between Shannon Diversity and zinc, causality cannot be inferred by these apparent direct relationships, since mercury is very highly correlated (p<0.0001) to all of the pyrethroids and all of the metals except arsenic and nickel. Both the Tolerance value and the % Tolerant taxa metrics were inversely related to PC2, while the latter metric was also directly related to PC1 and PC6. These relationships were confirmed by the Model 1 and 2 analyses. Thus, these benthic metrics, which tend to increase with environmental impairment, appeared to have an inverse relationship with the principal component for habitat metrics indicative of environmental quality (PC2) and, at least with % Tolerant taxa, a direct relationship with the principal component that was positively loaded by toxicants (PC1).
The benthic metric % Collectors/Filterers was directly related to Embeddedness and inversely related to Velocity depth regimes. Neither of these relationships was confirmed by the Model 1 analyses. There were no significant relationships detected in the analyses of principal components.
The metric % Collectors/gatherers was inversely related to Embeddedness and to PC2, the principal component that was positively loaded by Total score, Embeddedness, Sediment deposition, Frequency of riffles/bends and Velocity depth regimes and negatively loaded by % Fines. Both of these relationships were confirmed by Model 1 analyses. Thus, this benthic metric that tends to increase with impairment appears to be inversely related to habitat metrics that reflect relative habitat quality.
The metric % Grazers was directly related to total pyrethroids and inversely related to Riparian vegetative zone. The direct relationship with total pyrethroids was confirmed by the Model 2 analysis, while the relationship with Riparian vegetative zone was confirmed by the Model 1 analysis. This benthic metric was shown to be directly related to PC1, the principal component that was positively loaded by toxicants (all pyrethroids and certain metals) and to PC5, the principal component that was negatively loaded by Riparian vegetative zone and Channel alteration, and positively loaded by Epifaunal substrate/available cover, % Boulder and % Cobble. These relationships were confirmed by the Model 1 and 2 analyses, respectively. Thus, this benthic metric appears to have a direct relationship to habitats associated with toxicants and to have an inverse/negative relationship to vegetative buffer zones.
The benthic metric % Predators was directly related to lead to TEL. This relationship was confirmed by the Model 2 analysis. No significant relationships were observed between this benthic metric (which has a variable response to impairment) and the environmental principal components. The stepwise regressions did not indicate any significant relationships for the benthic metric Abundance.
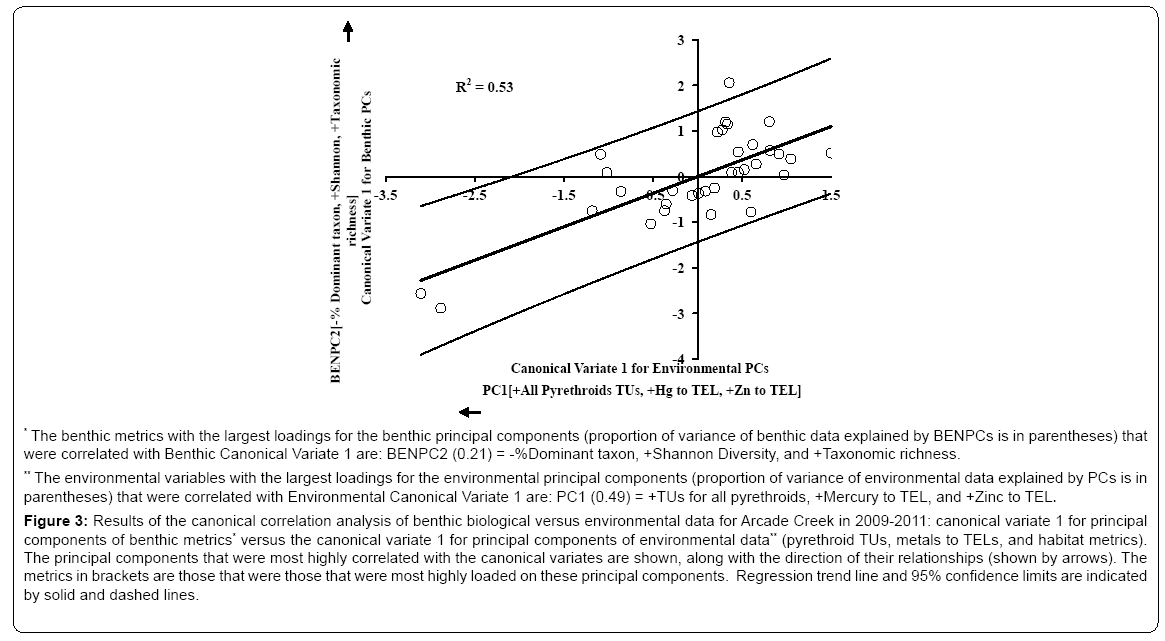
For the Arcade Creek 2009-2011 data, the canonical correlation analysis of principal components based on benthic metrics versus the principal components based on environmental variables (pyrethroid TUs, metals to TELs and habitat metrics) produced two highly significant pairs of canonical variates. The results are presented in (Figures 3 and 4). The environmental principal component PC1 (representing all pyrethroids and the metals mercury and zinc) was inversely correlated with the independent variable Canonical Variate 1 for Environmental PCs (Figure 3). The benthic principal component BENPC2 (representing a positive gradient of Shannon Diversity and Taxonomic richness and a negative gradient of % Dominant taxa) was positively (directly) correlated with the dependent variable Canonical Variate 1 for Benthic PCs. Thus, samples displaying greater diversity were associated with lower levels of toxicants and vice versa. The squared canonical correlation value for this relationship is 0.53, suggesting that more than half of the spatiotemporal variation in benthic metrics indicative of diversity may be related to the toxicants loaded on PC1.
Figure 3: Results of the canonical correlation analysis of benthic biological versus environmental data for Arcade Creek in 2009-2011: canonical variate 1 for principal components of benthic metrics* versus the canonical variate 1 for principal components of environmental data** (pyrethroid TUs, metals to TELs, and habitat metrics). The principal components that were most highly correlated with the canonical variates are shown, along with the direction of their relationships (shown by arrows). The metrics in brackets are those that were those that were most highly loaded on these principal components. Regression trend line and 95% confidence limits are indicated by solid and dashed lines.
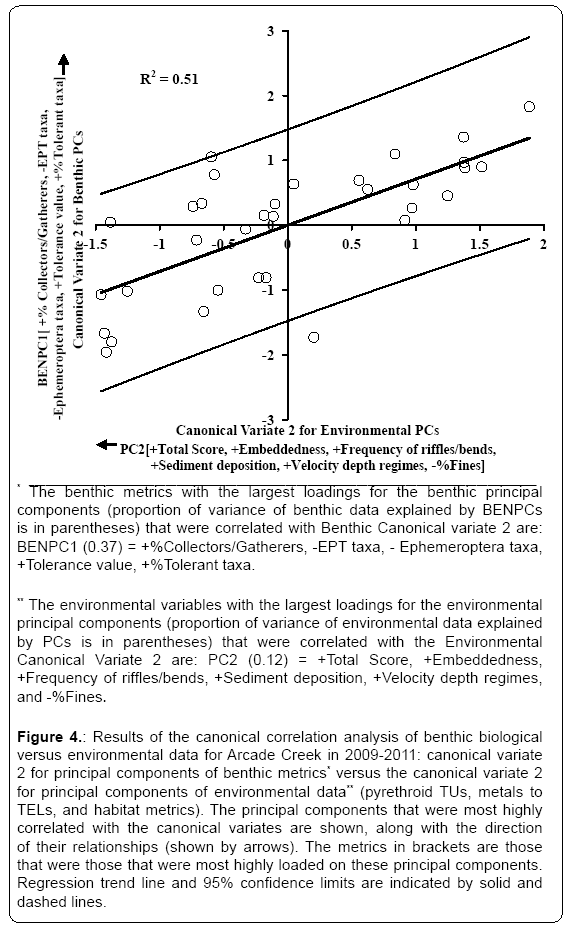
Figure 4: Results of the canonical correlation analysis of benthic biological versus environmental data for Arcade Creek in 2009-2011: canonical variate 2 for principal components of benthic metrics* versus the canonical variate 2 for principal components of environmental data** (pyrethroid TUs, metals to TELs, and habitat metrics). The principal components that were most highly correlated with the canonical variates are shown, along with the direction of their relationships (shown by arrows). The metrics in brackets are those that were those that were most highly loaded on these principal components. Regression trend line and 95% confidence limits are indicated by solid and dashed lines.
The second pair of significant canonical variates are shown in (Figure 4). The environmental principal component PC2 (habitat metrics indicative of environmental quality e.g., Total Score, Embeddedness, Frequency of riffles/bends, Sediment deposition, Velocity depth regimes, and coarser sediment) was negatively correlated with Canonical Variate 2 for Environmental PCs (Figure 4). The benthic principal component BENPC1 benthic metrics that are indicative of impaired environments (e.g. % Collectors/gatherers, Tolerance value, and % Tolerant taxa) was positively correlated with Canonical Variate 2 for Benthic PCs, whereas the benthic metrics that are indicative of less stressed conditions (e.g., EPT taxa and Ephemeroptera taxa that were negatively loaded on BENPC1) were negatively correlated with Canonical Variate 2 for Benthic PCs. Thus, the samples showing negative scores on both canonical variate 2 for Benthic PCs and Environmental PCs tended to display metric values that were indicative of healthier benthic communities and greater habitat quality; whereas samples showing positive scores on both axis had metrics that were indicative of more stressed benthic communities and poorer habitat quality. The squared canonical correlation value for the relationship between the canonical variates was 0.51, indicating that more than half of the spatiotemporal variation in the benthic metrics loaded on BENPC1 may be related to the habitat metrics loaded on PC2.
Salinas streams
The results of the stepwise multiple regression analyses of the three year Salinas streams data are shown in Table 12b. The benthic metric Taxonomic richness was inversely related to bifenthrin and directly related to % Canopy cover. This benthic metric was inversely related to the PC6, the principal component that was positively loaded by deltamethrin and bifenthrin. None of these relationships were confirmed by the Model 1 or 2 analyses.
| Benthic Metrics | b.1 Significant Variables (R2) | b.2 Principal Components |
|---|---|---|
| Taxonomic Richness | -Bifenthrin (0.15), +% Canopy cover (0.11) | -PC6 (0.19) |
| % Dominant Taxon | NS | NS |
| Ephemeroptera Taxa | NA | NA |
| EPT Taxa | NA | NA |
| EPT Index (%) | NA | NA |
| Shannon Diversity | -Esfenvalerate (0.14) | -PC5 (0.16) |
| Tolerance Value | -Canopy (0.11) | NS |
| % Tolerant Taxa (8-10) | +Esfenvalerate (0.12) | NS |
| % Collectors/Filterers | +Esfenvalerate (0.22) | +PC5 (0.22) |
| % Collectors/Gatherers | -Sediment deposition** (0.15) | NS |
| % Grazers | +Sediment deposition** (0.27), -Zinc (0.21), +Lead (0.15) | NS |
| % Predators | +Velocity depth regimes (0.14), +Cypemethrin (0.12), -Arsenic (0.13) | NS |
| % Shredders | NA | NA |
| Abundance (#/sample) | NS | NS |
* The environmental variables with the largest loadings for the environmental principal components (proportion of variance explained by PCs are in parentheses) are: PCI
(0.41) = +All of the metals to TELs, and to a lesser extent, +Channel flow, -Channel alteration, and -% Canopy cover; PC2(0.13) = -%Fines, +Embeddedness, +Sediment
deposition, +Cobble, +Total Score, +Epifaunal substrate, +Boulder, and +Frequency of riffles/bends; PC3(0.08) = +Cypermethrin TU; PC4(0.06) = +Lambda-cyhal. TU and
+Permethrin TU; PC5 (0.05) = +Esfenvalerate TU; PC6 (0.04) = +Deltamethrin TU and +Bifenthrin TU.
** Variables that remained significant after the toxicant-loaded principal components (PC1, PC3, PC4, PC5, and PC6) were forced into the stepwise regressions prior to the
testing of the habitat variables (see text for details).
***Variables that remained significant after the habitat principal component (PC2) was forced into the stepwise regressions prior to the testing of the toxic variables (see text
for details).
Table 12b: Regression models for Salinas streams
Shannon Diversity was inversely related to esfenvalerate, as it was to PC5, the principal component that was positively loaded by this pyrethroid. Neither of these relationships were confirmed by the Model 2 analyses. On the other hand, % Tolerant taxa and % Collectors/filterers, two benthic metrics expected to increase with impairment, were shown to be directly related to esfenvalerate. The latter benthic metric was also shown to be directly related to PC5, the principal component that was positively loaded by esfenvalerate. However, none of these relationships were confirmed by the Model 2 analyses.
Tolerance value, another benthic metric that would be expected to increase with environmental impairment, was inversely related to % Canopy cover. This relationship was not confirmed by the Model 1 analysis.
The metric % Collectors/gatherers was inversely related to Sediment deposition, while the metric % Grazers was directly related to this habitat metric. Both of these relationships were confirmed by the Model 1 analyses. The % Grazers metric was also shown to be inversely related to zinc to TEL and directly related to lead to TEL. However, neither of these relationships was confirmed in the Model 2 analyses.
Finally, the benthic metric % Predators was directly related to Velocity depth regime and cypermethrin and inversely related to arsenic to TEL. However, these relationships were not confirmed by the Model 1 and Model 2 analyses. None of the other analyses between benthic metrics and principal components for environmental variables were significant.
There were no significant multivariate relationships indicated by the canonical correlation analysis of the principal components based on the benthic metrics versus the principal components for the environmental variables for the Salinas streams data sets.
Discussion
Arcade creek
Analyses of the 3 year Arcade Creek data set indicated a number of relationships between benthic metrics and environmental variables. The stepwise regressions indicated that the benthic metrics indicative of benthic community diversity such as Taxonomic richness and Shannon Diversity were inversely related to toxicants represented on PC1 (pyrethroids, mercury and zinc). Conversely, % Dominant taxon, displaying an inverse relationship with community diversity, showed postive relationships with these same environmental variables. Ephemeroptera taxa, EPT taxa, and EPT index, benthic metrics that tend to increase in less stressed environments, were directly related to habitat metrics (PC2) and especially Embeddedness, Frequency of riffles/bends, as highlighted by the multiple regression performed on separate variables. Conversely, % Collectors/gatherers, a metric that tends to increase with stress, was inversely related to metrics related to habitat quality. Tolerance value and % Tolerant taxa, associated with stressed environments, appeared to be directly related to certain toxicants, such as mercury, but also inversely related to the principal component associated with habitat metrics indicative of environmental quality. The % Grazers metric also displayed a direct relationship with toxicants such as the total pyrethroid TUs and the principal component associated with the pyrethroids, mercury and zinc.
The multivariate canonical correlation analysis of the Arcade Creek data highlighted two major types of significant relationships between the environmental data and the benthic metrics. The overall relationship identified by the first set of canonical variates indicated that diversity of benthic communities (higher Shannon Diversity and Taxonomic richness; and lower % Dominant taxon) tended to be inversely related to the overall degree of contamination by toxicants (pyrethroids and certain metals). The samples collected from sites with lower concentrations of toxicants tended to display greater levels of benthic community richness and diversity and vice versa. However, no single toxicant could be statistically identified as being primarily responsible for this spatiotemporal pattern, since most of the toxicants were shown to be highly correlated with each other and many of the samples were observed to have toxicant concentrations that could be significant from a toxicological perspective.
For example, bifenthrin was the pyrethroid that most frequently exceeded a toxicity unit of 1, and the total sum of TUs for all pyrethroids averaged more than a value of “1” for 6 of the 11 sites. Likewise, nickel and chromium were the metals that displayed concentrations that most frequently exceeded their respective TELs, particularly at downstream sites ARC 1a and ARC 1. Moreover, it should be emphasized that none of the univariate or multivariate statistical analyses indicated than the toxicants found in the highest concentrations relative to their potential toxicities (e.g. bifenthrin, nickel or chromium) were uniquely related to benthic community diversity or associated in any specific manner that could suggest causality in Arcade Creek.
One area of uncertainty that should be recognized with this study is that there were likely other chemicals present in Arcade Creek that were not measured. For example, other investigators have reported polycyclic aromatic hydrocarbons (PAHs) at potentially toxic concentrations in urban watersheds in Australia [27]. Potentailly toxic concentrations of PAHs exceeding Threshold Effect Levels have also been reported in sediment for two urban California streams [48].
The second major type of relationship indicated by the canonical correlation analysis of the 2009-2011 Arcade Creek data was that benthic community composition was related to metrics indicative of relative habitat quality. Certain benthic community components sensitive to environmental impairment (e.g., Ephemeroptera taxa and EPT taxa, primarily mayfly larvae) were found to be directly related to conditions indicative of relatively greater habitat quality (e.g., higher values of the habitat metrics Total score, Embeddedness, Frequency of riffles/ bends, Sediment deposition, Velocity depth regimes; and lower levels of % Fines). In contrast, benthic metrics associated with communities that were more tolerant of environmental impairment (e.g., Tolerance value, % Tolerant taxa, % Collectors/filterers) tended to increase as these indicators of habitat quality declined. Thus, while overall diversity of the benthic communities were apparently related to concentrations of toxicants, the specific composition of the communities appeared to be more closely related to physical habitat quality. Of course, both of these types of stressors are often highly correlated with each other, since both are related to anthropogenic activities.
The importance of physical habitat for aquatic biota such as benthic macroinvertebrates has been documented by other investigators [49]. Altered physical habitat structure is considered one of the major stressors of aquatic systems throughout the United States resulting in extinctions, local expiration and population reductions of aquatic fauna [49,50]. Other studies in California water bodies also reported that physical habitat is a major stressor to aquatic life [51].
Salinas streams
The stepwise regressions for the 3 year data sets from the Salinas streams indicated various significant but relatively weak relationships (i.e., displaying relatively low R2 values) between benthic metrics and toxicants (pyrethroids and/or metals), but none were confirmed by the Model 2 analyses. The benthic metric % Grazers was directly related to the habitat metric Sediment deposition, while the metric % Collector/ gatherers was inversely related to it. These relationships with physical habitat were confirmed by the Model 1 analyses and represent the most significant results from analysis of the Salinas stream data.
In general, the canonical correlation analysis of the 3 year data set for the Salinas streams did not produce significant results. It is plausible that the overall degradation of the habitats in these streams did not produce sufficiently distinct major patterns in the biological communities that could be detected by this multivariate technique. As discussed above for the Arcade Creek data, adequatic physical habitat is a critical element for robust benthic communities [49]. Therefore, it is not surprising that the highly degraded habitat for the Salinas streams was a highly influential factor stressing the benthic communities.
Acknowledgements
We thank the Pyrethroid Working Group for supporting this study. California Department of Fish and Game are acknowleged for identification of benthic species and development of metrics. Morse Laboratories is acknowledged for pyrethroid analysis. Alpha Analytical Laboratory is acknowledged for TOC, grain size and metals analysis.
References
- Grimm NB, Grove JM, Pickett STA, Charles LR (2000) Integrated approaches to long-term studies of urban ecological systems. BioSci 50: 571-584.
- Walsh CJ, Roy AH, Feminella JW Cottingham PD, Groffman PM, et al., (2005) The urban stream syndrome; current knowledge and the search for a cure. J N Am Benthol Soc 24: 706-723.
- An KG, Choi SS (2003) An assessment of aquatic ecosystem health in a temperate watershed using the index of biological integrity. J Environ Sci Health A Tox Hazard Subst Environ Eng 38: 1115-1130.
- Rhoads BL (1995) Stream power: a unifying theme for urban geomorphology. In: Herricks EE (ed), Stormwater Runoff and Receiving Systems, Impact Monitoring and Assessment, 65-76. Lewis Publishers, Baco Raton, FL, USA.
- Kleindl WJ (1995) A benthic index of biotic integrity for Puget Sound lowland streams, Washington USA. MS Thesis, University of Washington, Seattle, WA, USA.
- Fore LS, Karr JR, Wisseman RW (1996) Assessing invertebrate responses to human activities: Evaluating alternative approaches. J N Am Benthol. Soc 15: 221-231.
- Karr Jr (1998) Rivers as sentinels: Using the biology of rivers to guide landscape management. In Naiman RJ and Bilby RE (eds.). River Ecology and Management: Lessons from the Pacific Coastal Ecoregion, 502-28. Springer, New York, NY, USA.
- Yoder CO, Rankin ET (1995) Biological criteria program development and implementation in Ohio. In: Davis, WS and Simon TP (eds.), Biological Assessment and Criteria: Tools of Water Resource Planning and Decision Making, pp 109-44. Lewis Publishers, Boca Raton, FL, USA.
- Barbour MT, Gerritsen J, Griffith GE, Frydenberg R, Mccarron E, et al., (1996) A framework for biological criteria for Florida streams using benthic macroinvertebrates. J North American Benthological Society 15: 185-211.
- Wright JF, Sutcliffe DW, Furse MT (2000) Assessing the biological quality of freshwater: RIVPACS and other techniques. Freshwater Biological Association, Ableside, Cumbria, UK.
- Baily RC, Norris RH, Reynoldson TD (2004) Bioassessment of freshwater ecosystems: using the reference condition approach. Kluwer Academic Publishers, Boston, MA. USA.
- Bacey J (2007) Biological assessment of urban and agricultural streams in the California Central Valley (Fall 2002 through Spring 2004). California Environmental Protection Agency, California Department of Pesticide Regulation, Sacramento, CA, USA.
- Brown L, May J (2004) Periphyton and macroinvertebrate communities at five sites in the San Joaquin river basin, California during June and September 2001. U. S. Geological Survey Report No. 2004-5098, Sacramento, CA, USA.
- Hall LW Jr, Killen WD (2001) Characterization of benthic communities and physical habitat in an agricultural and urban stream in California’s Central Valley. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- Hall LW Jr, Killen WD (2002) Characterization of benthic communities and physical habitat in agricultural streams in California’s San Joaquin Valley. Final report prepared by University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- Hall LW Jr, Killen WD, Anderson RD (2003) Characterization of benthic communities and physical habitat in agricultural streams in California’s San Joaquin Valley in 2002. Final Report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- Hall LW Jr, Killen WD (2004) Characterization of benthic communities and physical habitat in agricultural streams in California’s San Joaquin Valley in 2003. Final Report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- Hall LW Jr, Killen WD (2005) Temporal and spatial assessment of water quality, physical habitat, and benthic communities in an impaired agricultural stream in California's San Joaquin Valley. J Environ Sci Health A Tox Hazard Subst Environ Eng 40: 959-989.
- Hall LW Jr, Killen WD (2005) Characterization of benthic communities and physical habitat in agricultural streams in California’s San Joaquin Valley in 2004. Final Report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- Hall LW Jr, Killen WD, Anderson RD, Alden WR III (2009) The influence of physical habitat , pyrethroids, and metals on benthic community condition in an urban and residential stream in California. Human and Ecological Risk Assessment 15: 526-553.
- Tetra Tech Inc (2003) The status and future of biological assessment for California Streams. Prepared for the California State Water Resources Control Board Division of Water Quality, Sacramento, CA, USA.
- Amweg EL, Weston DP, You J, Lydy MJ (2006) Pyrethroid insecticides and sediment toxicity in urban creeks from California and Tennessee. Environ Sci Technol 40: 1700-1706.
- Ng CM, Weston DP, You J, Lydy MJ (2008) Patterns of pyrethroid contaminantion and toxicity in agricultural and urban stream segments. In:.Gan J, Spurlock F, Hendley P, et al (eds), Synthetic Pyrethroids: Occurrence and Behavior in Aquatic Environments. pp 355-369. American Chemical Society, Washington, DC, USA
- Giddings JM (2006) Compilation and evaluation of aquatic toxicity data for synthetic pyrethroids. Report Compliance Services International Study, Rochester, USA.
- Crunkilton R., Kleist J, Ramcheck J, DeVita W, Villeneueve D (1997) Assessment of the response of aquatic organisms to long-term in-situ exposures to urban runoff. In: Roesner LS (ed). Effects of Watershed Development and Mangement on Aquatic Ecosystems 18-33 ASCE, USA.
- Pettigrove V, Hoffman A (2003) Major sources of heavy metal pollution during base flows from sewered urban catchments in the city of Melbourne. Proceedings, Third South Pacific Conference on Stormwater and Aquatic Resource Protection combined with the 10th Annual Conference of the Australian Chapter of the International Erosion Control Association, Auckland, New Zealand.
- Pettigrove V, Hoffman A (2003) Toxicity in Melbourne’s streams and wetlands: An emerging threat to healthy aquatic ecosystems. Proceedings Australian water Association Convention and Exhibition, Paper #214. Perth, WA, Australia, April 6-10.
- Harrington J, Born M (2000) Measuring the health of California streams and rivers. – A methods manual for water resource professionals, citizen monitors and natural resource students. Report. Sustainable Land Stewardship International Institute, Sacramento, CA, USA.
- Kim JY, Lee BT, Shin KH, Lee KY, Kim KW, et al. (2007) Ecological health assessment and remediation of the stream impacted by acid mine drainage of the Gwangyang mine area. Environ Monit Assess 129: 79-85.
- Hall LW Jr, Anderson RD, Killen WD (2013) Mapping of depositional and non-depositional areas in Salinas, California streams with concurrent pyrethroid and benthic macroinvertebrate assessments. J Environ Sci Health A Tox Hazard Subst Environ Eng 48: 1-13.
- Hall LW Jr, Killen WD, Anderson RW (2012) Mapping of depositional and non-depositional areas in Pleasant Grove Creek with concurrent pyrethroid and benthic macroinvertebrate assessments in 2012. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- Moulton SR II, Carter JL, Grotheer SA, Cuffney TF, Short TM (2000) Methods of analysis by the U. S. Geological Survey National Water Quality Laboratory – Processing, taxonomy and quality control of benthic macroinvertebrate samples. Report 00-212, U. S. Geological Survey, Sacramento, CA, USA.
- Kazyak PF (1997) Maryland Biological Stream Survey Sampling Manual. Maryland Department of Natural Resources, Chesapeake Bay Research and Monitoring Division, Annapolis, MD, USA.
- Hall LW Jr, Killen WD, Anderson RD (2012) An assessment of the potential influence of physical habitat, pyrethroids, and metals on benthic communities in urban California streams in 2011 and 2009-2011. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- American Society for Testing and Materials (ASTM). (1998) Standard test method for particle size analysis of soils. Method D422-63, Philadelphia, PA, USA
- USEPA (United States Environmental Protection Agency) (2004) SW-846, Method 9060A: Total Organic Carbon. Report. U. S. EPA Office of Water, Washington, DC, USA
- Robinson NJ (2005) Draft Method: Residue analytical method for the determination of residues of bifenthrin, cypermethrin, cyfluthrin, deltamethrin, esfenvalerate, fenpropathrin, lambda-cyhalothrin and permethrin in sediment. Report for the Pyrethroid Working Group, Syngenta Crop Protection, Greensboro, NC, USA
- Buchman MF (1999). NOAA Screening Reference Tables. NOAA HAZMAT Report 99-1. Coastal Protection and Restoration Division, National Oceanic and Atmospheric Administration, Seattle, WA. USA
- Plumb RH (1981) Procedures of Handling and Chemical Analysis of Sediments and Water Samples. US Army Corps of Engineers, US Army Engineer Waterways Experimental Station, Vicksburg, MS, USA
- Ditoro DM, Mahony JD, Hansen DJ, Scott KJ, Hicks MB, et al. (1990) Toxicity of cadmium in sediment: The role of acid volatile sulfide. Environ Toxicol Chem 9: 1488-1502.
- Ditoro DM, Mahony JD, Hansen DJ, Scott KJ, Carlson AR, et al. (1992) Acid volatile sulfide predicts the acute toxicity of cadmium and nickel in sediments. Environ. Sci. Tech. 26: 96-101.
- Hall LW Jr, Killen WD, Anderson RD (2010) An assessment of the potential influence of physical habitat, pyrethroids, and metals on benthic communities in urban California streams in 2009. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA
- Hall LW Jr, Killen WD, Anderson RD et al. (2010) An assessment of the potential influence of physical habitat, pyrethroids, and metals on benthic communities in urban California streams in 2010. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA.
- SAS Institute Inc. (2008) Statistical Analysis Program Version 9.2, Cary, NC, USA.
- Hall LW Jr, Killen WD, Anderson RD (2009). Characterization of benthic communities and physical habitat in agricultural streams in California’s San Joaquin Valley in 2009. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD, USA
- Hall LW Jr, Anderson RD, Killen WD (2012). Mapping the spatial extent of depositional areas in agricultural, urban and residential California streams: Implications for pyrethroid toxicity. Human and Ecological Risk Assessment 18: 368-392.
- Stribling JB, White JS, Jessup BJ, et al. (1998) Development of a benthic index of biotic integrity for Maryland streams. Report. Tetra Tech Incorporated, Owings, MD, USA.
- Hall, LW Jr., Killen WD, Anderson RD, Alden RW (2008) An assessment of benthic communities with concurrent physical habitat, pyrethroids, and metals analysis in a urban and residential stream in California in 2006 and 2007. Final report prepared by the University of Maryland, Wye Research and Education Center, Queenstown, MD.
- Rankin, ET (1995) Habitat indices in water resource quality assessments. In WS Davis and TP Simon editors. Biological Assessment and Criteria: Tools for Water Resource Planning and Decision Making. Lewis Publishers, Boca Raton, FL, USA.
- Karr JR, Fausch KD, Angermeier PL, Yant PR, Schlosser IJ (1986) Assessing biological integrity in running waters: A method and its rationale. Illinois Natural History Survey Special Publication 5, Champaign, IL.
- Hall, LW Jr, Killen WD, Alden RW (2007) Relationship of farm level pesticide use and physical habitat on benthic community status in a California agricultural stream. Human and Ecological Risk Assessment 13: 843-869.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 21347
- [From(publication date):
December-2013 - Nov 09, 2025] - Breakdown by view type
- HTML page views : 16553
- PDF downloads : 4794