Research Article Open Access
Abuse of Pharmaceutical Drugs-antibiotics in Dairy Cattle in Kosovo and Detection of their Residues in Milk
Zana Sulejmani1*, Agim Shehi2, Zehra Hajrulai3 and Elida Mata21University of Prishtina, Faculty of Medicine, Department of Pharmacy - Kosovo
2University of Tirana, Faculty of Medicine, Institute of Public Health - Albania
3University of Skopje, Faculty of Veterinary Medicine, Veterinary Institute - Macedonia
- *Corresponding Author:
- Zana Sulejmani
University of Prishtina, Faculty of Medicine
Department of Pharmacy – Kosovo
E-mail: z_sulejmani@yahoo.com
Received date: March 08, 2012; Accepted date: July 12, 2012; Published date: July 15, 2012
Citation: Sulejmani Z, Shehi A, Hajrulai Z, Mata E (2012) Abuse of Pharmaceutical Drugs-antibiotics in Dairy Cattle in Kosovo and Detection of their Residues in Milk. J Ecosyst Ecogr 2:114. doi:10.4172/2157-7625.1000114
Copyright: © 2012 Sulejmani Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
Introduction: Pharmaceutical drugs-antibiotics are given to livestock by veterinarians for the prevention and treatment of disease, but also as a stimulant for animal growth and conversion. Regardless of the way the drugs are inserted into the body of the animal, there will always be residual risk in milk which exceed the maximum level allowed. Thus, it can lead to health disorders in humans and in some cases even death.
Aim of study: The main aim of this study is to discover the use of pharmaceutical drugs – antibiotics in dairy cows and to detect betalactam and sulfonamide residues in milk.
Methods and materials used: During the period January to August 2011, were collected 127 milk samples from cows treated with pharmaceutical drugs – antibiotics. Samples were analysed in the Veterinary Institute in Skopje using “Elissa” screening method and “HPLC” confirmatory method.
Results: The study shows that of 127 samples of milk analysed, over 70% of them (64 were contaminated with betalactam residues and 24 with sulphonamide residues) contains residues of drugs, while over 20% of them (15 being beta-lactams and 4 sulphonamides) have passed the maximum residue limit allowed.
Conclusion: 70% of the drugs given to dairy cattle in Kosovo are pharmaceutical – antibiotics and their residues are present in milk. We conclude that pharmaceutical drugs – antibiotics that are used in humans should not be used in animals too and their provision should be limited to a reasonable and lawful level.
Keywords
Pharmaceutical drugs; Antibiotics; Beta-lactams; Sulphonamides; Milk residues
Introduction
Milk is a food substance and nutritious liquid that is essential for normal growth and development of an organism.
Milk offered for sale is a specific food item that requires strict control and enforcement by all legal regulations and relevant state authorities, that must allow its marketing at home and abroad only through special and official permission [1].
Use of antibiotics in dairy farms, and the use of antimicrobial agents at sub-therapeutic levels in dairy animals have always been considered for application [2]. Even Kosovo, as a new and less developed state, is exposed to uncontrolled use of antibiotics in dairy cattle. So even in this country there is a need to monitor the presence of antibiotic residues in food products such as milk.
However, the visible absence of veterinary structures has caused the spread of uncontrolled use of medications to prevent and treat infections in dairy cattle, and, unfortunately, now its use has become so unprofessional that it interferes in the farmer’s way of giving antibiotics. This, not only, is damaging to the health and well-being of the animal but it affects greatly the health and wellbeing of human consumers too through consumption of animal products such as milk and meat, thus affecting general public health.
We’ve learned from many scientific studies that human diseases caused by food consumption can have serious negative implication on economies of countries leading to many other social consequences. Today, in our globalization age, geography is no longer a factor which can prevent serious disease spreading across borders. Liberalization of market, food systems and free movement of people and animals across borders have greatly reduced geographical barriers [3,4].
Antibiotics are vital drugs in the treatment of infectious diseases in humans and animals. They are used to treat and control diseases caused by bacteria, destroying them or stopping their reproduction, and thus overcoming the infection and allowing the infected body to heal. However, their efficiency can decrease from its broad, continuous, and inadequate use. This can happen not just in medicine but also in agriculture [5].
Beta-lactam antibiotics are most commonly used for the treatment of gram positive and gram negative bacterial infections. They have a broad spectrum of bactericidal action hampering the synthesis of bacterial cell-wall [6].
This group includes all antibiotics, which, in their core ring contain β-lactams, such as: penicillin, b) cephalosporin, c) carbapenems and d) monobactams [7].
Sulphonamides are among the oldest groups of antibiotic/ antimicrobial agents. They are widely used in the treatment of bacterial diseases of dairy cattle. They have a wide spectrum of bacteriostatic action, affecting gram positive and gram negative organisms [8,9].
Best-known sulphonamides are: sulfadiazine, sulfamethoxazole, sulphadimidine, sulphadoxine, sulfadimethoxine, sulfamerazine, sulphasalazine, silversulfadiazine and sulfisoxazole, which have the sulfonamide structure as their base [10,11].
Both these types of antibiotics dissolve quickly, easily distribute in all tissues and body fluids, by including the cerebrospinal fluid and fetal circulation. About 90% of these antibiotics bind to plasmatic proteins, and their maximum concentration seen after 3-6 hours. They metabolize in the liver and eliminate by glomerular filtration [12].
The use of antibiotics ignoring any criteria in all cases of disease is that from cold, viral infection, inflammation or wounding can have a wide negative effect with the drugs left behind in milk and edible tissues. These residues in milk and other food products of animal origin can cause many functional disorders in the human body, such as, for example: a) transfer of bacterial resistance to antibiotics in humans, b) effects of immunopathology, c) carcinogenic effects, d) permanent gene mutation, e) nephropathies or kidney disease, f) liver poisoning, g) reproductive disorders, h) bone marrow poisoning and i) allergic reactions [13].
The main purpose of this study is to discover the use of pharmaceutical drugs – antibiotics in dairy cattle. Other goal is to detect beta-lactam and sulphonamide residues in the milk manufactured in the Republic of Kosovo.
Materials and Methods
In order to carry out this research over 80% of dairy farms in the Republic of Kosovo were visited during period January to August of 2011.
In this time 127 samples of raw milk were collected, 39 of them during the winter period and 86 of them during the summer.
Samples were taken only from cows treated with pharmaceutical drugs – antibiotics that were last given to them no longer than 2 weeks before the sample collection.
Search and collection of milk samples was conducted in two seasons of the year: during the winter and the summer. This was done to identify the time period during which these drugs are mostly used.
It is observed that the greatest number of cows fell sick during the summer, apparently due to high temperatures that cause proliferation of bacteria and other parasites in water, food, soil and air, in turn, increasing the number of infections and various diseases in dairy cattle.
The assessment was made through a specific questionnaire, which identified problems in the treatment of cattle with drugs.
Analyses of the samples were done in the laboratories of the Veterinary Institute in Skopje using Elissa screening method and HPLC confirmatory method.
To examine the presence of beta-lactam and sulphonamide residues in the samples of raw milk the method used was ‘Elissa’ for beta-lactams manufactured by Randox-England and for sulphonamides produced by Europroxima in the Netherlands.
Elissa screening method is one of the fastest and most reliable analytical methods for detecting the presence of antibiotics. The testing is easy and shows high sensitivity and fast response.
For the preparation of raw milk samples and in order to detect possible beta-lactams these reagents are used: a) wash buffer (concentrated) of 1 × 32 ml, Randox-England, b) the standards of 6 species, Randox-England, c) conjugate (concentrated) of 3 vials, Randoks-England, d) one shot substrate of 1 × 15 ml, Randoks-England, e) a bottle of stop solution, Randoks-England and f) re-distilled water.
Whereas, in order to detect sulfonamides these reagents were used: a) ethylacetaat, Merck-Germany, b) iso-octane, Merck-Germany, c) chloroform, Merck-Germany, d) mixture of iso-octane and chloroform in a 2:3 ratio V/V, e) hydrochloric acid 0.1 mol/dm3, Merck-Germany, f) PBS has been prepared by digesting the mixture of 0.77 g Na2HPO4 + 1.88 g KH2PO4 + 8.94 g NaCl in one litre of distilled water and adjusted to pH of 7.4 ± 0.2 and g) sulfamerazine standard, Sigma-Aldrich.
In this paper all standards and samples are applied microtitre plate replication. For detection of beta-lactams six standards applied on the microtitre plate have the following concentration: a) 0 ng/ml, b) 0.46 ng/ml, c) 1.0 ng/ml, d) 5.6 ng/ml, d) 10.1 ng/ml and f) 58.9 ng/ml. However, for detection of sulfonamides these standards have concentrations, such as: a) 0 ng/ml, b) 0.125 ng/ml, c) 0.5 ng/ml, d) 0.8 ng/ml, d) 2.5 ng/ml and f) 5.0 ng/ml.
To evaluate the screening method “ELISA” for the analysis of beta-lactams and sulphonamides in milk it is necessary to set some parameters on the basis of set criteria and conforming to the requirements and recommendations by the Rules of the Directive no. 96/23/EC [14]. Parameters to be determined are: a) selectivity, b) precision (accuracy), c) reproducibility, d) limit of detection and e) detection capability (CCβ) [15].
All these parameters are determined by various formulas and their final results are presented in Table 1.
| Parameters | Total of beta-lactams | Total of sulphonamides |
|---|---|---|
| Calibration curve | Y = -0125 + 0.7744 lnX | Y = -21.758 ln (x) + 40.88 |
| Regression coefficient | 0.879 | 0.9926 |
| Range of concentrations | 0.00-58.9 ng / ml | 0.125 to 5.0 ng / mL |
| IC50 (concentration at 50 % bounding) | 0.65 ng / ml | 0.75 ng / mL |
| Limit of detection (LOD) | 1.5 �?µg / ml | 13.3 �?µg / kg |
| Detection capability (CC�?�?) | 48.5 �?µg / kg | 54.8 �?µg / kg |
| Repeatability | 5.05% | 7.39% |
| Reproducibility (precision) | 6.28% | 8.82% |
| Recovery (accuracy) | 96.18% | 97.51% |
Table1: Validation of parameters of the screening method for the analysis of betalactams & sulphonamides.
Results were calculated by Excel program. The absorbance read becomes a percentage (%) of optical density relative to zero standards B0 and it is based on the calibration line assigned to each series of standard solutions and has the following formula:
y = a + b*lnX
Y-read signal expressed in% of optical density, X-concentration of the substance and a and b-coefficients.
In every batch of samples analyzed for values of R2 beta-lactams must be at least 0.8278, while the sulphonamides R2 ˃ 0.98.
Limit of detection is determined by the formula: LOD = Xaverage + 3SD.
The limit of detection (LOD) determined by ELISA method for beta-lactam residues in milk is 1.5 μg / ml, while for sulphonamide residues is 13.3 μg / kg.
Meanwhile, through the confirmatory analysis the presence of antibiotic residues continues to be noted in those samples in which the screening method has shown a detection limit higher than normal.
In our case we used the method of High-Performance Liquid Chromatography with Fluorescence Detection, HPLC/FD in order to detect sulphonamide residues only, since it has not been possible to specifically adjust the HPLC/FD device for beta-lactam detection.
In order to carry out the confirmatory method for sulphonamide residues these reagents were used: a) methanol, Sigma-USA, b) acetonitrile, Sigma-USA, c) water, Sigma-USA, d) acetic acid 98-100%, Merck-Germany, e) hydrochloric acid 36-37%, Merck-Germany, f) formic acid 90%, Merck-Germany, g) ammonia 25%, Merck- Germany, h) acetone, Merck-Germany, i) sodium acetate dissolved 0.01 M with pH = 3.5, j) fluorescamine for synthesis, Sigma-USA, k) sulfadimidine analytical standard, SDM 99.8%, Fluke-Switzerland, l) sulfadiazine analytical standard, 99.0% SDZ, Fluke-Switzerland, m) sulfamethoxazole analytical standard, SMX 99%, Sigma-USA and n) sulfisoxazole analytical standard, SXZ 99.95%, Farmabase-Italy.
Sulphonamide concentrations in the analyzed samples were calculated, through the calibration lines for specific sulphonamides, according to these two formula equations:
Y = AX + B
Y-signal read by the instrument with the appropriate concentration, X-concentration of the respective substance, A-avoiding the calibration curve and B – circuit that separates the (cuts off) curve from the axis, and:
w = {(ws ▪ V) / m} ▪ 1000
ws-the numerical value of the sulphonamide content in μg/ml in absorption test, V-the final volume, from which is transferred the test for analysis in ml and m-the numerical value of the mass in g.
Limits of detection and quantification can be determined through the ratio signal/noise, respectively, through these two equations:
LOD = 3 ▪ SD and LOQ = 10 ▪ SD
LOD-Limit of Detection, LOQ-Limit of Quantification and SDStandard Deviation.
The method precision for confirmatory analysis of sulphonamide residues is controlled by the values of repeatability and reproducibility of the method. They are calculated on the level of concentration of 100 μg/kg as is the value of the maximum residue limit (MRL) in conformity with the recommendations of the regulation no. 657/2002 [16].
Results
During this research study we have observed that for the treatment of sick cattle in Kosovo beta-lactam and sulphonamide antibiotics are used most commonly. However, not that often, other types of antibiotics are used too such as tetracyclines, aminoglycosides and macrolides.
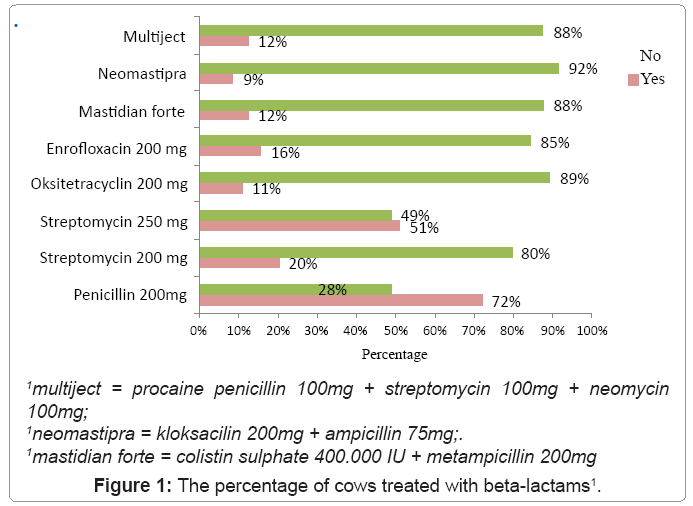
Figure 1 presents the percentage of cows treated with beta-lactams. It can be seen that penicillin 200mg is one of the beta-lactams mostly used. 93 cows were treated with pencillin being 72.1% of the total, a statistically significant difference from the number of cows not treated with this antibiotic (p<0.01).
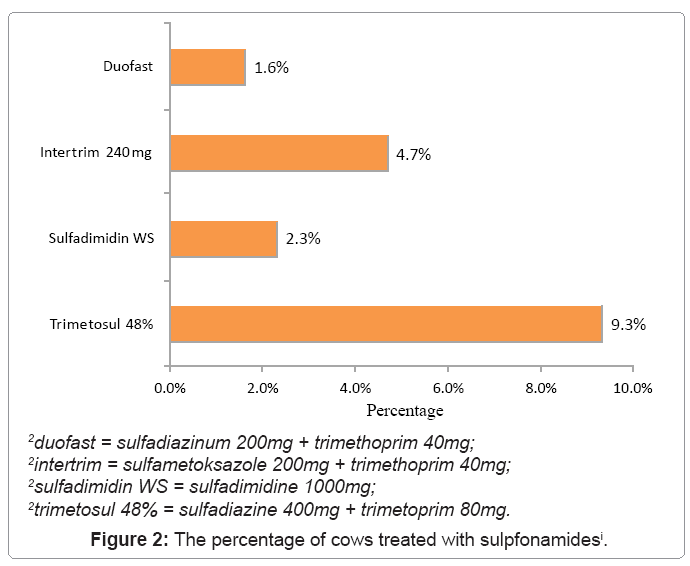
Figure 2 presents the number and percentage of cows treated with sulphonamide. It can be seen that “trimetosul 48%” is mostly used here. 12 cows or 9.3% of the total were treated with this drug, which shows a statistically significant difference when compared to other sulphonamides used (χ2 = 5.10; p = 0.01).
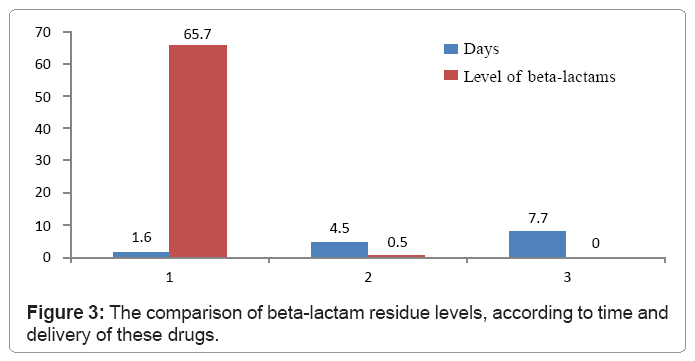
Figure 3 present the comparison of beta-lactams residue levels, according to time and delivery of these drugs. It can be seen that in the first days of their delivery beta-lactam residue level is high and it continues to be so over the following days. By mid-time of their receipt their level significantly lowers, while in last days since their first use the residue level becomes incalculable.
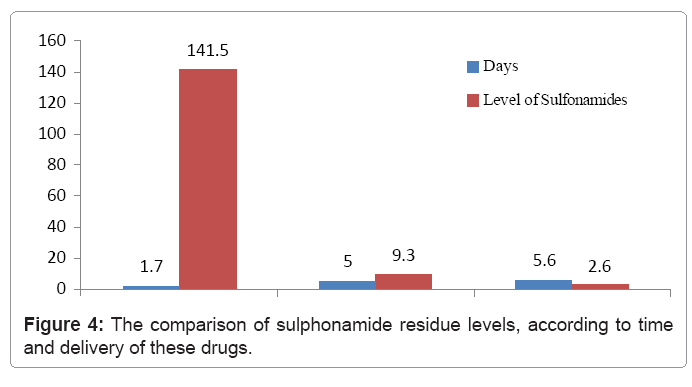
Figure 4 present the comparison of sulphonamide residue level according to time and delivery of these drugs. It can be clearly seen that sulphonamide residue level remains high in the first days of their delivery and this high level continues during the following days. By mid-time the drug’s residue level significantly lowers, while in last days since their first use their residue level is incalculable.
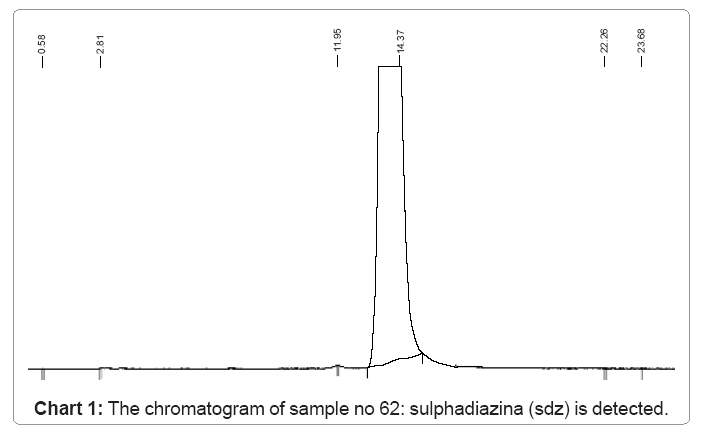
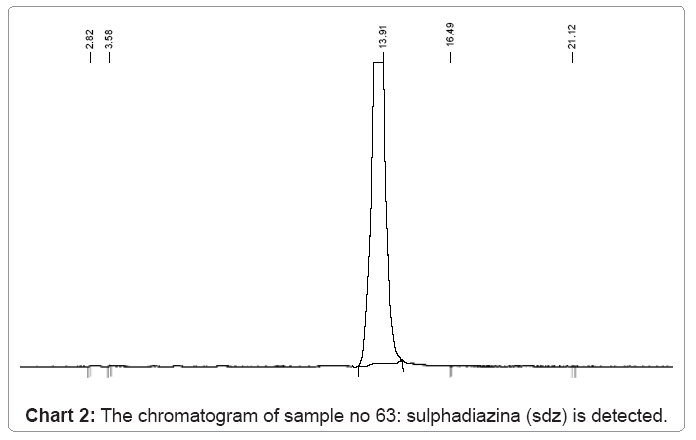
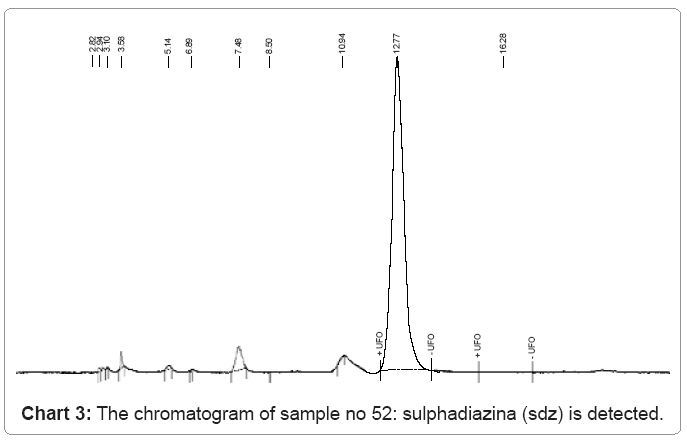
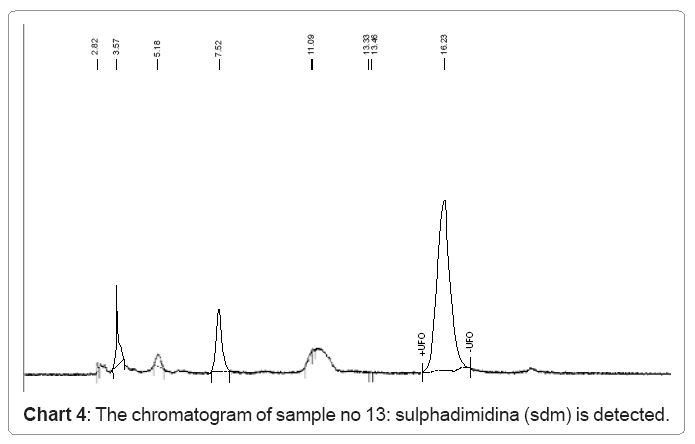
The following charts 1, 2, 3 and 4 show chromatograms of sulphonamide residues in 4 samples of raw milk being above the limit of detection (LOD). This limit for the method HPLC/FD is 100 μg/kg.
During result calculation using the above-mentioned formulas we note that:
a) In the sample no.62 sulfadiazine SDZ is detected having a value of 1114.5 μg/kg, which is passes the limit,
b) In sample no.63 sulfadiazina SDZ is also detected with a value of 627 μg/kg,
c) In sample no.52 sulfadiazina SDZ has a value of 227.6 μg/kg and
d) In sample no.13 sulfadimidina SDM is detected with a value of 109.9 μg/kg.
Discussion
Of the whole drugs given to dairy cattle in Kosovo by veterinarians and farmers 70% were pharmaceutical-antibiotics. Their residues were present in milk at high levels, enough to cause severe damage to the entire public health.
Out of 127 samples of milk, 64 were contaminated with beta-lactam residues, and 24 with sulphonamide residues. Where as 19 samples already passed the limit of detection, the majority 15, being beta-lactam residues, the rest, 4, sulphonamide residues.
In other words, the study confirms that over 70% of the total of milk samples analysed contains drug residues, with 20% of them already passing the maximum residue limit allowed.
According to a survey conducted in Macedonia in 2000 by Velev 79% of drugs - antibiotics used in veterinary medicine are pharmaceutical drugs [17].
Khaskheli et al. argue that in Pakistan, during 2006, 36.5% of the total of their raw milk samples were contaminated with beta-lactam residues [18].
Adesiyun and several co-authors have studied the spread of antibiotic residues in raw milk of cattle in Trinidad, in 1997, indicating that 10.8% of all their analyzed samples were contaminated [19].
Other study carried out in Turkey in 2010 by Kaya and Filazi, confirms the presence of beta-lactam residues in 44% of samples among 200 of them analyzed [20].
In their work in Iran in 2010 Mohammad Hossein Movasag and several co-authors have shown beta-lactam residues in raw milk to be 5% [21].
Moreover, a study conducted by Shitandi in 2001 in Kenya, confirms that out of the total of 1109 raw milk samples analyzed in Kenya, 21% showed contamination with antibiotic residues [22].
In 1991 in Germany, Sanders and several co-authors analyzed around 3000 samples of milk and thereby proved the presence of sulphonamide in 1.1% of samples [23].
Also, in Germany in 2007, on another survey conducted by Kress and several co-authors during a program for identification of positive samples of milk, 1.6% of their cases showed a presence of sulphonamide residues [24].
One other survey made in the Netherlands, in 2000, by Abjean applying the examination method in 1100 milk samples of cattle, proves that 9 of the samples or 0.81% of the total were contaminated with sulphonamide residues. However, the survey, failed to show any presence of antibiotic residues when it came to applying the confirmatory HPLC method [25].
A study conducted by Tolentino and several co-authors in 2005 in Mexico using the screening method shows the number of samples detected with sulphonamide residues amounted to 51.3% of the total of analysed samples [26].
Smedley in 1994 when analysing samples using confirmatory method showed that 8 out of the total number were contaminated with sulphonamide residues [27].
Whereas, a study conducted in Turkey in 2007 by Alkan analysed 46 samples using HPLC confirmatory method, and confirmed the presence of sulphonamide residues only in a single sample being above the maximum limit [28].
In a survey by Hyun-Hee Chung in the Republic of Korea in 2008, out of 269 analysed samples using HPLC confirmatory method the presence of sulphonamides was verified only in 4 [10].
Conclusion
70% of the drugs given to dairy cattle in Kosovo are pharmaceuticalantibiotics. Their residues are present in milk at levels high enough to cause severe damage to the entire public health.
From the above article we can conclude that pharmaceutical drugsantibiotics manufactured for human use should not be used in animals too, and the provision of antibiotics should be limited to a reasonable and lawful way
Veterinarians who treat animals in farms are obliged to follow the conditions and regulations for keeping and raising animals, and they should use antibiotics as well as other drugs which are approved for animal use only
Measures must be taken to monitor the presence of drug residues in live animals and animal products, which have pharmacological effects and are harmful to human health.
Veterinarian during his treatment of farm animals, as an evidence of a safe conduct must mark the date, time and mode of application of drugs, the matriculation of the treated animal and the drug warranty. The owner or supervisor of the treated animal must administer veterinary medicine only under veterinary control always following the instructions for use given by the manufacturers and checking the expiry date. In this case the owner of the animal is obliged to keep records of medical prescriptions too.
Acknowledgement
We would like to express our thankfulness to all of those farmers, veterinarians, owners and their families that helped us without laziness in collecting milk samples.
References
- Donna LS (1998) Pasteurize Why? The dangers Of Consuming Raw Milk. Cornell University, Dairy Science Facts. NY, USA.
- Angulo FJ, Griffin PM (2000) Changes in antimicrobial Resistance in Salmonella enterica serovar typhimurium. Emerg Infect Dis 6: 436.
- Boor KJ (2001) Fluid Dairy product quality and safety: looking to the future. J Dairy Sci 84: 1-11.
- Brown P, Will RG, Bradley R, Asher DM, Detwiler L (2001) Bovine spongiform encephalopathy and variant Creutzfeldt-Jakob disease: background, evolution, and current concerns. Emerg Infect Dis 7: 6-14.
- Prescott LM, Harley JP, Klein DA (1993) Microbiology. Brown Publishers, 2nd Ed.
- Droumev DR (1983) Review of antimicrobial agents promoting available. Vet Growth. ResRes Commum 7: 85-99.
- Ghidini SM, Zanardi E, Varisco G, Chizzolini R (2002) Prevalence of Molecules of ß-lactam Antibiotics in Bovine Milk, in Emily Romagna and Lombardy. ANN Fac Medi Vet di Parma, Italy 22: 245-252.
- Carol A, Gonzalez M, Usher KM (2009) Determination of sulfonamides in Milk Using Solid-Phase Extraction and Liquid Chromatography-Tandem Mass Spectrometry. Department of Chemistry West Chester University, USA.
- Martindale (2008) The Extra Pharmacopoeia, 30th ed.
- Chung HH, Lee JB, Chung YH, Lee KG (2009) Analysis of sulfonamide and quinolone antibiotic residues in Korean milk using microbial assays and high performance liquid chromatography. Food Chemistry 113: 297-301.
- There Drug Bull (1995) Co-trimoxazole use restricted. 33: 92-93.
- Richelle RG (2007) Investigation of Safe-Level Testing for Beta-lactam, Sulfonamide, and Tetracycline Residues in Commingled Bovine Milk. Doctoral thesis, Salve Regina University, USA.
- Nisha AR Antibiotic residues - A Global Health Hazard. Department of pharmacology and Toxicology College of Veterinary and Animal Sciences. Veterinary World 1: 375-377.
- (1996) Council Directive 96/23/EC of 29 April 1996 on Certain Measures to monitor substances and residues thereof in live animals and animal Products Directive 85/358/EEC and repealing Decisions 89/187/EEC and 86/469/EEC and and 91 / 664/EEC. Off J Eur Comm 10-32.
- Reybroeck W (2010) Screening for residues of antibiotics and chemotherapeutics in milk and honey. Doctoral thesis. Department of Veterinary Public Health and Food Safety Laboratory of Chemical Analysis, Ghent University.
- Commission Decision 2002/657/EC of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical Methods and the interpretation of results OJ L 221.
- Velev R (2000) Palette of registrated drugs and Supporting Medical Supplies for the use in the veterinary medicine in the Republic of Macedonia. Macedonian Veterinary Review 29: 55-64.
- Khaskheli M, Malik RS, Arain MA, Soomro AH, Arain HH (2008) Detection of beta-lactam antibiotic residues in market milk. J Nutrition 7: 682-685.
- Adesiyun AA, Webb LA (1997) Prevalence of antimicrobial residues in preprocessed and processed Cow’s milk in Trinidad. J Food Safety 16: 301-310.
- Kaya ES, Filazi A (2010) Determination of antibiotics residues in milk samples. Kafkas Univ Vet Factors Derg 16:31-35.
- Movassagh HM, Karami AR (2010) Determination of Antibiotic residues in Bovine Milk in Tabriz, Iran. Global Veterinary Studies 5: 195-197.
- Shitandi A, Sternesjo A (2001) Detection of antimicrobial drug residues in Kenyan milk. J Food Safety 21: 205-214.
- Sanders P, Guillot P, Dagorn M, Delmas JM (1991) Liquid chromatographic Calf determination of chloramphenicol in tissues: Studies of Stability in muscle. Kidney and Liver. J Assoc off Anal Chem 74: 483-486.
- Kress C, Seidler C, Kerp B, Schneider E, Usleber E (2007) Experience with an identification and quantification program for inhibitor-positive milk sample. Anal Chim Acta 586: 275-279.
- Abjean JP, Delepine B, Hurtaud-Pessel D (2000) Qualitative or quantitative Methods for residue analysis? A Strategy for drug residue monitoring. Proceedings of the Conference EuroResidue IV. Veldhoven, The Netherlands 8-10 May.
- Tolentino RG, Perez MA, Gonzales GD, León SV, López MG, et al. (2005) Determination of the presence of 10 antimicrobial residues in mexican pasteurized milk. Intercsiencia 30.
- Smedley MD (1994) Liquid chromatographic determination of multiple sulfonamide residues in Bovine Milk: Collaborative Study. J AOAC Int 77: 1112-1122.
- Alkan A (2007) The confirmation of used commercial kits in the detection of antibiotics in milk with HPLC (High Pressure Liquid Chromatography). Master thesis. Graduate School of Engineering and Sciences of Izmir Institute of Technology, Izmir, Turkey
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 19061
- [From(publication date):
September-2012 - Dec 07, 2025] - Breakdown by view type
- HTML page views : 14275
- PDF downloads : 4786