Research Article Open Access
Concentrations of Trace Metals from Soils in Pretoria, South Africa
Olowoyo JO1*, van Heerden E1 and Fischer JL21Department of Biology, University of Limpopo, Medunsa Campus, P. O. Box 139 Medunsa, Pretoria, South Africa
- *Corresponding Author:
- Olowoyo JO
University of Limpopo, Department of Biology
Medunsa Campus. P. O. Box 139 Medunsa
Pretoria, South Africa
E-mail: woleolowoyo@yahoo.com
Received date: May 14, 2012; Accepted date: June 26, 2012; Published date: June 29, 2012
Citation: Olowoyo JO, van Heerden E, Fischer JL (2012) Concentrations of Trace Metals from Soils in Pretoria, South Africa. J Ecosyst Ecogr 2:113. doi:10.4172/2157-7625.1000113
Copyright: © 2012 Olowoyo JO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
Trace metals are common pollutants in urban industrial soils thus making the assessment of these pollutants important especially from developing countries. The present study investigated the level and source of trace metals from different sites in Pretoria in relation to vehicular emission. Soil samples were taken from ten different locations (0-5 cm and 5-15 cm) in the city and analyzed for trace metal content using Inductive Couple Plasma Mass Spectrophotometer (ICP-MS). The values of trace metals from the dust and top soil samples were in the range 12.8-145 μg/g for Pb, 33.9-140 μg/g for Cu, 43.6-101 μg/g for Zn, 0.24-3.49 μg/g for Sb, 39.3-97.6 mg/g for Fe and 0.09-0.98 μg/g for Cd. The result revealed a statistically different concentrations for trace metals in all the sites and layers (p<0.01). Increased concentrations of Pb, Cu, Zn and Sb were witnessed in areas associated high traffic areas. The pollution index (PI) revealed that some of the sites were enriched and polluted with metals associated with vehicular emissions. It was concluded from the study that vehicular emission played a significant role in releasing trace metals into the environment. This can pose a serious health risk to humans via inhalation of suspended dust, direct ingestion or through consumption of contaminated food stuff.
Keywords
Pollution; Trace metals; Vehicular emission; Urban
Introduction
Trace metals are released into the environment by natural processes and by anthropogenic activities [1]. Atmospheric pollution is one of the major sources of heavy metal availability in the environment. These sources include vehicular emissions, industrial discharges and other activities [2]. Emissions from road traffic have contributed greatly to the level of particulate matters both in developed and developing countries [3]. In urban city centers, it is not uncommon to see high number of industries and vehicles thus making urban centers a major harbor of resource consumption and chemical emissions which may lead to serious environmental problems if not controlled [4]. The release of pollutants such as trace metals in the urban centre is on the increase and may pose a serious threat to the quality of life in many of these urban centers [5].
Urban soils accumulate contaminants from diffuse or localized sources on continuous basis [6]. Heavy metals can accumulate in topsoil from atmospheric deposition by sedimentation, impaction and interception. The accumulation of these metals and the ability of trace metals to remain in urban soils for a prolonged period of time may pose serious environmental and health problems in the urban city centers [7].
Concerning the danger posed by trace metals, there could be a direct intake of heavy metals by people living in the urban city centers. Children and elderly people are more prone to the direct effect of heavy metals. Studies of metal concentrations from play–ground dusts ingested by children through the hand-mouth pathway have been demonstrated [2]. Metals can exert a range of toxic effects, including neurological, hepatic, renal and hematopoietic ones [8]. Furthermore, the non biodegradable nature and long biological half lives within the human body is of great concern.
Crop uptake is another route of human exposure to trace elements in most of the urban soils through food chain. Uptake of trace elements by plant varies and depends largely on several factors, such as soil pH and organic matter content [9]. Among crop types the leafy vegetables such as lettuce and cabbage have the greatest ability to take up trace elements from soil [10].
In the past few years, the study of urban soil has emerged an important area in environmental research due to the level of pollution. Studies on metal flows in the anthroposhere have indicted the traffic sector as the major contributor of diffuse metal emission from either rural or urban centers [11]. Hjortenkrans et al. [12] reported on the presence of trace metals in urban soils and related the source of Zn, Cd, Pb and Cu to traffic density on the roads. In a separate study, Dolan et al. [13] traced the source of these metals in urban soils to burning of fossil fuel in internal combustion engines, wear and tear of tires, leakage of oil from vehicles and corrosion of batteries and metallic parts. Therefore, monitoring of the metals contents from dust samples maybe an efficient way of obtaining information on the current environmental state of large areas.
Pretoria is the administrative capital city of South Africa, with a population of about 2.4 million people. It enjoys a dry, sunny climate, with the exception of occasional late afternoon downpours in the summer months of October to April. Temperatures are usually fairly mild due to the city’s high altitude (1271 m) with the average maximum daytime temperature in January of 21.5°C, dropping to an average maximum of around 11°C in July. Snow is rare. Mean annual precipitation is 784 mm. Industries, vehicles, veld fires, and the burning of coal by households without electricity causes air pollution, which occasionally rises during the winter. With the present population figure, economic activities in the city and the hosting of the world cup in 2010, the road network in Pretoria metropolis has increased accommodating the number of automobile transportation and creating a lot of ancillary roadside work. It is not uncommon to see hawkers selling their goods next to roadsides and standing next to traffic lights. Improved road accessibility creates a variety of ancillary employment which range from vehicle repairs and welders to auto-electricians, battery chargers and dealers in other facilitators of motor transportation. These activities send trace metals into the air and the metals subsequently are deposited into nearby soils, which are absorbed by plants on such soils. The South African government in 2005 introduced the sale of unleaded gasoline with a view to reducing the release of metals associated with vehicular emission in the environment with particular reference to lead. The present work investigated the concentrations of trace metals from soil with a view to determining the effect of vehicular emission on the level of trace metals in the city.
Methodology
Study sites
University campus: Located within the University of Limpopo, Medunsa Campus. Generally, the passage of vehicles (mainly private vehicles) through this area is very low.
Residential area: Situated towards the south-west of the city centre. There are some human activities around this area that may serve as sources of pollution. These include the movement of cars (mainly private vehicles) in and out of the area and burning of coal by households.
Mining area: Around a mining industry. There is a continuous gas flaring both day and night in this area with a long stack emitting gasses and particulate matter into the environment.
Industrial area: It houses a number of companies which includes automotive industries like BMW, Nissan and Toyota production plants and the brewing industry, South African Breweries (SAB) as well as several other smaller industries. As a result of the clustering of industries in this area, the traffic in the area comprises of heavy trucks, private and commercial vehicles on a daily basis and thus could be classified as a high traffic area. A cloudy atmosphere is a common sight in this particular area.
Cement industry: The factory has a high chimney for fume emissions. It is a common phenomenon to see delivery trucks departing from the factory. The source of emission from this area might not only come from the cement company itself, but may be influenced by truck emissions and the general traffic congestion on the opposite busy road.
Commercial and high traffic center: It is a high traffic area, noted for the presence of heavy trucks, buses and small cars. This is the commercial hub of the city which influences the economy of the city. This consequently leads to the influx of people with a high number of automobiles in this area.
Metal smelting area: Several metal smelting companies are located in this area with the presence of some other smaller industries. There is also a thermoelectric power station in this area. The atmosphere in this area always looks cloudy.
Union buildings: This is generally a restricted area as it is situated very close to the seat of SA Government known as Union Buildings. The choice of this was with a view of assessing the effect of high altitude in the dispersion of trace elements as pollutants in the city.
Local airport: The purpose for the selection of this site was to assess the influence of the aviation industry on the release of trace elements in the local vicinity.
Taxi rank: This site is located in the commercial nerve centre of the city of Tshwane. The area is a high traffic centre with hundreds of trucks, buses and cars passing through it on hourly basis.
Soil sample collection
Samples of soil were taken from of 0-5 cm (dust) and 5-15 cm (top soil) with the aid of a soil auger of 7.0 cm in diameter. In each of the sites, 5 samples were taken and the mean result presented as a composite soil sample. From each sites 5 composite samples were analyzed and the mean result presented. The soil samples were allowed to dry by spreading it on laboratory table and grounded in a closed environment. After grinding, the soil samples collected were sieved using a 2 mm mesh.
Soil sample analysis
The soil pH was determined in 0.01M CaCl2 (1:2 soil-solution ratio) and in distilled water using a pH meter fitted with glass electrode (Jenwal Model 3015 digital). For the determination of trace metals content in soil, the grounded soil samples were further sieved to pass through a mesh <60 μm. The total metal content of the soil was determined by digesting the samples with a mixture of 10 ml HNO3 and 5 ml HCl and the resulting solution was analysed for trace element content with the aid of ICP-MS.
Quality assurance
For the purpose of quality assurance, the analysis soil samples were carried out in triplicate and also with the analysis of NCS DC 73309 (GSD-11 soil) that contains metals. The result and the recovery rate are presented in Table 1 below:
| Elements | Certified Value | Measured | % Recovery |
|---|---|---|---|
| Cd | 2.30 | 2.23 | 97 |
| Cu | 78.6 | 80.1 | 102 |
| Zn | 373.00 | 367.01 | 98 |
| Cr | 40.00 | 34.06 | 85 |
| Pb | 63.6 | 62.2 | 98 |
Measured data in micrograms per gram
Table 1: Comparing the values of lichens in standard values with measured data in μg/g.
Statistical analysis
All statistical analyses were carried out mainly on the log transformed data using SPSS 13.0 for windows. Differences in the concentration of the various trace metals between sites were determined using either the student t-test (for dichotomous variables) or the analysis of variance (ANOVA). Duncan’s Multiple Range Test was used to separate the significant means between the sites. In that case, it was possible to determine statistical differences between the study sites. A Pearson correlation test was also performed in order to assess the strength of metal relationships.
Results and Discussion
The pH of the soils from the study sites varied from 4.92 ± 0.12 to 5.12 ± 0.23 (CaCl2) and 4.99 ± 0.01 to 5.23 ± 0.02 (H2O) for dust samples, 5.62 ± 0.02 to 6.15 ± 0.03 (CaCl2) and 5.98 ± 0.03 to 6.32 ± 0.05 (H2O) for top soil. There were significant differences in the value determined for the pH from the sites (p<0.05). The observed pH level recorded form the study revealed that some of the trace metals might become easily accessible and mix with dust with respect to the pH level from some of the sites. At a relatively low pH, the mobility of trace metals is substantially enhanced due to the acidic nature of the soil and the displacement of exchangeable caution from the exchange site. These metals will not be adsorbed or occluded by the carbonates, organic matters or secondary minerals and as such may miss easily with soil dust [14].
The highest concentration for lead was recorded from the dust (Site 6) with a mean value of 145 ± 0.12 μg/g with the lowest concentration occurring in site 1 with a mean value of 16.2 ± 0.01 μg/g. It was gathered from the result that areas associated with high traffic density ( Sites 5, 6, 9 and 10) recorded higher concentration for this metal (Figure 1) and the differences recorded form all the sites were significant (p<0.05). Khan et al. [15] reported higher concentration (176.05 ± 17 μg/g) for lead in the city of Hyderabad in Pakistan. The increased concentration of lead from all the areas associated with high traffic density suggested an anthropogenic source for this trace metal. Lead pollution in the environment including soil and dust comes from combustion of gasoline that contains tetraethyl lead as anti – knock agent [16]. Also, the high levels of lead from some sites in this study can be attributed to the burning of fossil fuels and traffic [17]. Report from other researchers suggested a long history of metal contamination in urban environment due to re –suspension which may in turn account for the presence of lead in this present study [2].
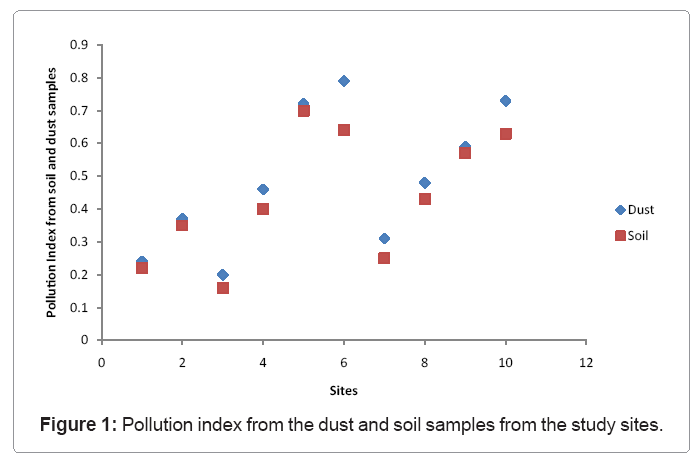
Pollution index from the dust and soil samples from the study sites.Concentrations of Cu exhibited a similar pattern with those noted for lead from all the sites. An increased concentration for Cu was witnessed in all the areas associated with high traffic density and to some extent in the industrial areas. The lowest concentration for Cu was recorded from site 3 from the top soil with a value of 33.9 ± 0.14 μg/g while the highest concentration was recorded from the soil dust from site 10 with a value of 121 ± 0.20 μg/g. The differences in the concentrations of this element in all the sites were significant statistically (p< 0.05). A strong positive correlation was noted for this element and other elements such as Pb, Zn and Sb (Table 3). The source of Cu from the dust and soil samples may be due to corrosion of metallic parts of cars derived from engine wear, thrust burning, brushing and bearing metals.
The average concentration of Zn from the dust was in the range 51.7 ± 0.07 μg/g-101 ± 0.43μg/g. From the soil sample the average concentration of Zn was in the range 43.6 ± 0.56 μg/g–95.3 ± 0.98 μg/g. The highest concentration of Zn was also noticed from areas associated with traffic (Sites 4, 5, 6, 9 and 10). Elik [18] noted that higher concentrations of zinc in heavy traffic zones may indicate the fragmentation of car tyres. Zinc is mainly derived from tyre dust, because it is used as filler during tyre manufacturing and hence released onto the soil during the wear and tear of tyres [15]. The average concentration of Zn both from the soil dust and soil samples from this study are still very low when compared to its acceptable value of 300 mg/kg [19]. The highest mean concentration of 410μg/g and 715 μg/g reported for Zn from studies conducted in Jordan and China respectively was clearly greater than Zn concentrations obtain from this study [17,20]. However, similar trends in the deposition of Zn from high traffic centers were also noted.
The concentration of Sb increased seven times fold in areas associated with traffic densities (Sites 5, 6, 9 and 10) when compared to low traffic areas (Site 1 and 2) (Tables 2 and 3). The lowest concentration for Sb was reported from the soil sample from site 3 with a mean value of 0.24 ± 0.03 μg/g while the highest concentration was from soil dust from site 6 with a mean value of 3.49 ± 0.11μg/g. Despite Sb short history as traffic-emitted metals, the concentration increased more from roadside soil dust when compared to soil samples from all the sites. The major source of road traffic related Sb is brake linings. Though, according to Hjortenkrans et al. [17] Sb was introduced to gasoline fuel in order to provide strength and stability for lead. The significant increase of Sb in roadside soil dust stresses the need for metal transport studies as well as effect of this metal in the environment.
| Elements | Sites | F – Test | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Pb | 17.8±0.05 | 41.2±0.56 | 13.45±0.12 | 43.6±0.11 | 102±0.11 | 145±0.12 | 31.1±0.11 | 62.3±0.01 | 98.1±0.07 | 101±0.02 | ** |
| Cu | 53.3±0.04 | 71.4±0.67 | 46.32±0.12 | 83.2±0.12 | 140±0.13 | 108±0.98 | 51.1±0.23 | 82.3±0.05 | 98.0±0.04 | 121±0.20 | * |
| Zn | 54.3±0.04 | 56.3±0.23 | 51.71±0.07 | 95.7±0.23 | 101±0.43 | 95.5±0.64 | 81.7±0.27 | 81.4±0.06 | 88.9±0.04 | 86.3±0.23 | ** |
| Mnα | 1.05±0.78 | 1.06±1.12 | 1.10±0.45 | 1.88±0.67 | 1.56±0.45 | 1.76±0.54 | 8.96±0.31 | 1.01±0.01 | 1.32±0.24 | 1.34±0.34 | * |
| Sb | 0.87±0.01 | 0.41±0.01 | 0.34±0.11 | 0.76±0.12 | 3.23±0.65 | 3.49±0.11 | 0.89±0.01 | 0.69±0.02 | 2.12±0.12 | 3.31±0.08 | * |
| Feα | 56.7±0.65 | 72.1±0.02 | 39.9±0.12 | 76.2±0.11 | 91.3±0.23 | 98.2±0.34 | 49.2±0.12 | 73.6±0.12 | 91.3±0.42 | 97.6±0.05 | * |
| Caα | 1.75±1.12 | 4.86±0.78 | 5.62±1.12 | 2.23±0.14 | 5.72±0.13 | 9.12±0.81 | 1.23±0.05 | 5.43±0.34 | 2.33±0.11 | 2.67±0.89 | ** |
| Crα | 0.43±0.02 | 0.19±0.65 | 0.34±010 | 0.45±0.06 | 1.21±0.63 | 1.12±0.11 | 1.91±0.13 | 0.97±0.12 | 0.82±0.01 | 1.12±0.24 | ** |
| Cd | 0.22±0.12 | 0.45±0.02 | 0.11±0.01 | 0.76±0.03 | 0.37±0.17 | 0.98±0.01 | 0.45±0.07 | 0.65±0.04 | 0.43±0.11 | 1.12±0.01 | * |
α: values presented in mg/g.
Table 2: Mean concentration of elements from the soils of the study area at depth 5–15 cm in μg/g. Significance of comparison means by ANOVA (F-test) are indicated (*p<0.05 and **p<0.01)”.
| Elements | Sites | F -test | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Pb | 16.2±0.01 | 39.9±0.08 | 12.8±0.03 | 34.7±0.12 | 98.8±0.22 | 108±0.11 | 25.1±0.11 | 59.5±0.23 | 102.7±0.06 | 99.3±0.36 | * |
| Cu | 49.3±0.25 | 68.2±0.15 | 33.9±0.14 | 79.4±0.18 | 138±0.45 | 98.8±0.69 | 44.4±0.52 | 79.5±0.35 | 88.9±0.58 | 109±2.36 | * |
| Zn | 48.2±1.12 | 57.2±0.63 | 43.6±0.56 | 88.9±1.85 | 95.3±0.98 | 87.0±0.54 | 72.9±0.52 | 79.2±0.75 | 86.9±0.58 | 84.2±0.36 | * |
| Mnα | 1.12±0.89 | 1.16±0.32 | 1.17±0.21 | 1.75±0.03 | 1.34±0.02 | 1.70±0.01 | 1.72±0.03 | 0.99±0.02 | 1.25±0.01 | 1.29±0.03 | ** |
| Sb | 0.78±0.01 | 0.32±0.00 | 0.24±0.03 | 0.66±0.02 | 2.14±0.03 | 2.42±0.00 | 0.53±0.02 | 1.34±0.01 | 1.99±0.01 | 2.83±0.06 | * |
| Feα | 38.5±0.42 | 69.8±0..38 | 39.3±0.11 | 63.0±0.50 | 88.6±0.36 | 84.1±0.35 | 44.8±0.92 | 65.9±0.33 | 88.4±0.44 | 89.6±0.22 | * |
| Caα | 1.83±0..6 | 5.23±0.01 | 7.46±0.04 | 3.22±0.12 | 6.76±0.09 | 10.2±0.65 | 3.26±0.24 | 5.23±0.08 | 3.44±0.23 | 4.87±0.05 | ** |
| Crα | 0.32±0.99 | 0.17±0.08 | 0.28±0.31 | 0.37±0.28 | 1.10±0.26 | 0.98±0.22 | 1.89±0.33 | 0.79±0.45 | 0.73±0.22 | 0.98±0.31 | ** |
| Cd | 0.18±0.01 | 0.32±0.02 | 0.09±0.01 | 0.42±0.03 | 0.28±0.02 | 0.56±0.04 | 0.23±0.02 | 0.22±0.01 | 0.25±0.02 | 0.48±0.01 | * |
α: values presented in mg/g.
Table 3: Mean concentration of elements from the soils of the study area at depth 5–15 cm in μg/g. Significance of comparison means by ANOVA (F-test) are indicated (*p<0.05 and **p<0.01)”.
As with other trace metals, the highest concentration of Cd were recorded from areas associated with high traffic density (Sites 4, 6 and 10). From the dust sample, the concentration of Cd ranged from 0.11 ± 0.01μg/g-1.12 ± 0.01μg/g. The concentration of Cd ranged from 0.09 ± 0.01 μg/g-0.56 ± 0.04 μg/g in the soil samples. Differences in the concentration of cadmium from all the sites were significant statistically (p< 0.05). The concentration of Cd from this study is comparable to studies conducted by Baptista et al. [21] in Luanda where the mean value recorded for Cd from the street dust was 1.15 μg/g. Varrica et al. [22] also reported a mean value of 1.1 μg/g in road side dust from the metropolitan area of Palermo city in Italy. One of the sources of Cd occurs through its release as a combustion product in the accumulators of motor vehicles or in carburetors [23]. This may have accounted for an increase in the level of this trace element in all areas associated with traffic density. Though, metal production and smelting are also known to be a source of atmospheric cadmium emission and the production of batteries [24]. The metal concentration of the urban soil could represent the long term contamination from various sources in the city environment but with particular reference to vehicular emission.
The highest concentrations for all the elements were recorded for Fe, Ca, Mg and Cr (Table 1). The trend in the concentration were in the order Fe>Ca>Mg>Cr from all the sites. Apart from Cr, the remaining trace elements (Fe, Ca and Mg) are more associated to crustal elements, though the effect of anthropogenic sources could not be excluded totally. This was evident in the elemental concentration for most of these metals in areas associated with vehicular emission. Iron was the most abundant metal content from this study. The increased concentration of iron form all the sites associated with vehicular emission may have suggested that traffic density played a significant role in releasing this element as pollutants in the environment. The possible source of iron particles could be brake lining material in form of break dust [25]. The concentration of iron form this study exceeded the baseline range of 50 μg/g-500 μg/g (Allen 1989).
Pearson correlation coefficient was used to establish the interrelationship between trace metals level from soil dust and soil samples (Table 4). It was obvious from the matrix that there was a positive and strong correlation among elements that are associated with vehicular emission. The maximum correlation value of 0.94 was noted between Pb and Sb. Similarly high correlation matrix of 0.71, 0.78, 0.83, 0.84 and 0.87 was observed between Zn and Sb, Pb and Zn, Cu and Sb, Pb and Fe, Pb and Cu respectively (Table 4). The strong correlation coefficient from these metals may imply that these elements originated from the same source which in this case could be linked to vehicular emission. The values obtained for correlations coefficients for these metals is in agreement with the results from Cal-Prieto et al. [26] of a positive correlation for all the metals listed above. The poor correlation values for some of the other metals may indicate an independent or multiple sources (Table 4).
| Metals | Pb | Cu | Zn | Mn | Sb | Fe | Ca | Cr | Cd |
|---|---|---|---|---|---|---|---|---|---|
| Pb | 1 | 0.87 | 0.78 | 0.10 | 0.94 | 0.84 | -0.10 | -0.34 | 0.61 |
| Cu | 0.87 | 1 | 0.82 | 0.10 | 0.83 | 0.73 | 0.08 | -0.56 | 0.58 |
| Zn | 0.78 | 0.82 | 1 | 0.46 | 0.71 | 0.67 | -0.16 | -0.35 | 0.62 |
| Mn | 0.10 | 0.10 | 0.46 | 1 | 0.07 | 0.08 | -0.48 | 0.15 | 0.55 |
| Sb | 0.94 | 0.83 | 0.71 | 0.07 | 1 | 0.82 | -0.16 | -0.23 | 0.61 |
| Fe | 0.84 | 0.73 | 0.67 | 0.08 | 0.82 | 1 | 0.19 | 0.02 | 0.39 |
| Ca | -0.10 | 0.08 | -0.16 | -0.48 | -0.16 | 0.19 | 1 | 0.19 | -0.46 |
| Cr | -0.31 | -0.56 | -0.35 | 0.14 | -0.23 | 0.02 | 0.19 | 1 | -0.32 |
| Cd | 0.61 | 0.58 | 0.62 | 0.55 | 0.61 | 0.39 | -0.46 | -0.32 | 1 |
Table 4: Showing the correlation coefficient in all the metals.
Pollution Assessment
The integrated pollution index (IPI) was calculated for all the sites under study using the method proposed by [27]. The pollution index was then calculated using the formula; IPI=(Cd/3+Cu/100+P b/100+Zn/300/4). The scatter diagram for the PI from the dust and soil samples is presented in figure 1. The IPI from all the study sites was <1, however, sites that were associated with high traffic presented higher values for IPI, suggesting the impact of vehicular emission on the release of these trace metals as pollutants in the study area. Generally, higher concentrations were recorded for metals that are associated with vehicular emission in street dust, therefore suggesting an anthropogenic source for all these metals. The pollution index PI was also calculated using the formula: Pi=Ci/Si, where Ci represents the concentration of heavy metal i in soil while Si indicates the relevant standard value for this metal. Based on the Pi the soil pollution levels were classified as follows: Pi<1 unpolluted, Pi>2 medium pollution and Pi>3 heavy pollution (Table 5) [28].
| Elements | Sites | F- Test | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Pb | 2.70 | 6.24 | 2.04 | 6.61 | 15.45 | 21.97 | 4.71 | 9.44 | 14.86 | 15.30 | ** |
| Cu | 8.08 | 10.82 | 7.02 | 12.61 | 21.21 | 16.36 | 7.74 | 12.47 | 14.85 | 18.33 | * |
| Zn | 1.17 | 1.21 | 1.11 | 2.06 | 2.17 | 2.05 | 1.76 | 1.75 | 1.91 | 1.86 | * |
| Cr | 5.38 | 2.36 | 4.25 | 5.63 | 15.13 | 14.00 | 23.88 | 12.13 | 10.25 | 14 | * |
| Cd | 0.11 | 0.23 | 0.06 | 0.38 | 0.19 | 0.49 | 0.23 | 0.34 | 0.22 | 0.56 | * |
Table 5: Pollution Index calculated for some of the elements within the study sites.
Conclusion
Dust is known as one of the most pervasive and important factor affecting human health in the urban environment. From the urban soils studied, trace metals concentration were largely associated with vehicular emission and might have contributed to the level of these metals from the urban soil dust. The positive correlation noted for Sb and all other trace metals associated with vehicular emission may call for further research to investigate the impact of this metal on the environment. Despite the introduction of unleaded fuel in the city since 2005, the levels of trace elements associated with vehicular emission are still very high in the city.
Since metals have been found as a pollutant in the study, future research should focus on making comprehensive assessments of soil quality in other big cities in the country, as this may allow for better city planning and sustainable management of urban resources. This is very important because the elevated levels of these metals may pose a serious threat to those that are selling goods around the traffic lights and for the group of people that rely on urban farming for survival.
References
- Komarnick GJ (2005) Lead and cadmium in indoor air and the urban environment. Environ Pollut 136: 47-61.
- Li X, Poon CS, Liu PS (2001) Heavy metal contamination of urban soils and street dusts in Hong Kong. Applied Geochemistry 16: 1361-1368.
- Charron A, Harrison RM, Quincey P (2007) What are the sources and conditions responsible for exceedences of the 24 h PM10 Limit value (50 µg/m3) at a heavily trafficked London site? Atmospheric Environment 41: 1960-1975.
- Davydova S (2005) Heavy metals as toxicants in big cities. Microchem J 79: 133-136.
- González RC, González-Chávez MC (2006) Metal accumulation in wild plants surrounding mining wastes. Environ Pollut 144: 84-92.
- Fabietti G, Biasioli M, Barberis R, Ajmone-Marsan F (2010) Soil contamination by organic acid and inorganic pollutants at the regional scale: the case of Piedmont, Italy. Journal of Soil Sediments 10: 290-300.
- Wong CS, Li X, Thornton I (2006) Urban environmental geochemistry of trace metals. Environ Pollut 142: 1-16.
- Rimmer DL, Vizard CG, Pless-Mulloli T, Singleton I, Air VS, et al. (2006) Metal contamination of urban soils in the vicinity of a municipal waste incinerator: One source among many. Sci Total Environ 356: 207-216.
- Logan TJ, Goins LE, Lindsay BJ (1997a) Field assessment of trace element uptake by six vegetables from N-Viro soil. Water and Environmental Research 69: 28-33.
- Chaney RL, Malik M, Li YM, Brown SL, Brewer EP, et al. (1997) Phytoremediation of soil metals. Current Opinions in Biotechnology 8: 279-284.
- Sorme L, Bergback B, Lohm U (2001) Century perspective of heavy metal use in urban areas. Water, Air and soil Pollution 1: 197-211.
- Hjortenkrans D, Bergbark B, Haggerud A (2006) New metal emission patterns in road traffic environments. Environ Monit Assess 117: 85-98.
- Dolan LMJ, Bohemen H, Whelan P, Akbar KF, O’Malley V, et al. (2006) Towards the Sustainable Development of Modern Road Ecosystem. In: The Ecology of Transportation Managing Mobility for the Environment, Davenport J and Davenport JL (Eds.). Springer Netherlands, UK 275-331.
- Olivia SR, Espinosa AJF (2007) Monitoring of heavy metals in top soils, atmospheric particles and plant leaves to identify possible contamination sources. Microchem J 86: 131-139.
- Khan MN, Wasim AA, Sarwar A, Rasheed MF (2011) Assessment of heavy metal toxicants in the roadside soil along the N-5, National Highway, Pakistan. Environ Monit Assess 182: 587-595.
- Tuzen M (2003) Determination of heavy metal in soil, mushroom and plant samples by atomic absorption spectrometry. Microchem J 74: 289-297.
- Al-Khashman OA (2007) The investigation of metal concentrations in street dust samples in Aqaba city, Jordan. Environ Geochem Health 29: 197-207.
- Elik A (2003) Heavy metal accumulation in street dust samples in Sivas. Communication in Soil Science and Plant Analysis 34: 145-156.
- Faiz Y, Tufail M, Javed MT, Chaudhry MM, Siddique N (2009) Road dust pollution of Cd, Cu, Ni, Pb and Zn along Islamabad Expressway, Pakistan. Microchem J 92: 186-192.
- Lu X, Wang L, Lei K, Huang J, Zhai Y (2009) Contamination assessment of copper, lead,zinc, manganese and nickel in street of Baoji, NM China. J Hazard Mater 161: 1058-1062.
- Baptista LF, DeMiguel E (2005) Geochemistry and risk assessment of street dust in Luanda, Angola: a tropical urban environment. Atmospheric Environment 39: 4501-4512.
- Varrica D, Dongarra G, Sabatino G, Monna F (2003) Inorganic geochemistry of roadway dust from the metropolitan area of Palermon, Italy. Environmental Geology 44: 222-230.
- Divrikli U, Horzum N, Soylak M, Elci L (2006) Trace heavy metal contents of some spices and herbal plants from western Anatolia, Turkey. International Journal of Food Science and Technology 41: 712-716.
- Al-Khashman OA, Shawabkeh RA (2009) Metal distribution in urban soil around steel industry beside Queen Alia Airport, Jordan. Environ Geochem Health 31: 717-729.
- Adachi K, Tainosho Y (2004) Characterization of heavy metal particles embedded in tire dust. Environ Int 30: 1009-1017.
- Cal-Prieto MJ, Carlosena A, Andrade JM, Martinez ML, Muniategui S, et al. (2001) Antimony as a tracer of the anthropogenic influence on soils and eustarine sediments. Water, Air, Soil Pollution 129: 333-348.
- Kloke A (1979) Content of arsenic, cadmium, chromium, fluorite, lead, mercury and nickel in plants grown on contaminated soil. Paper presented at United Nations ECE symposium.
- Muller G (1969) Index of geoaccumulation in sediments of the Rhine River, Geographical Journal 2: 109-118.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 16309
- [From(publication date):
June-2012 - Dec 22, 2025] - Breakdown by view type
- HTML page views : 11415
- PDF downloads : 4894