Research Article Open Access
Degradation of the Azo Dye Reactive Red 195 by Paenibacillus spp. R2
| Mohd Zaini Nawahwi1*, Zaharah Ibrahim2 and Adibah Yahya2 | |
| 1Department of Biology, Faculty of Applied Science, University Teknologi MARA, 72000 Kuala Pilah, Negeri Sembilan Malaysia | |
| 2Department of Industrial Biotechnology, Faculty of Bioscience and Bioengineering, Universiti Teknologi Malaysia, 81310 Skudai, Johor, Malaysia | |
| Corresponding Author : | Mohd Zaini Nawahwi Department of Biology Universiti Teknologi MARA Kuala Pilah 72000 Negeri Sembilan, Malaysia Tel: 604832133 Fax: 604842449 E-mail: mohdzaini82@gmail.com |
| Received: August 06, 2012; Accepted: November 11, 2012; Published: November 11, 2012 | |
| Citation: Nawahwi MZ, Ibrahim Z, Yahya A (2013) Degradation of the Azo Dye Reactive Red 195 by Paenibacillus spp. R2. J Bioremed Biodeg 4:174. doi:10.4172/2155-6199.1000174 | |
| Copyright: © 2013 Nawahwi MZ, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
Azo dyes are a widespread class of poorly biodegradable industrial pollutants. In anaerobic environments, azo bonds are reductively cleaved yielding carcinogenic aromatic amines, many of which are assumed to resist further metabolism by anaerobes bacteria. The latter compounds generally require aerobic conditions for their degradation. A reactive group of azo dye called SFRed (C.I: Reactive Red 195) was found to be degraded using Gram negative, coccobacillus, facultative anaerobe bacteria, Paenibacillus sp. R2 to α-ketoglutaric acid with transient accumulation of 4-aminobenzenesulphonic acid (sulphanilic acid), 4-amino, 3-hydronapthalenesulphonic acid and 4-amino, 5-hydronapthalene 2,7 disulphonic acid as a degradation intermediate in anaerobic facultative batch culture. Colour and total organic carbon (TOC) was successfully removed more than 95% and up to 50% respectively. There is no significant correlation between pH and oxygen depletion since there is slightly change in pH was observed (pH from 7.21 to 7.25) though the anaerobiosis was found developed throughout the experiment (redox potential from 0.7 to 1.6 mV). The anaerobic metabolism of glucose as co-metabolite also shown to provide the electrons required for the
initial reductive cleavage of the azo group. This finding suggest that it is possible to mineralize the azo dye in the environment; thereby, avoiding accumulation of toxic intermediates in the water.
| Keywords |
| Paenibacillus sp; Azoreductase; Decolourization; Reactive red 195 |
| Introduction |
| Azo dyes, aromatic moieties linked together by azo (-N=N-) chromophores, represent the largest class of dyes used in textileprocessing and other industries such as cosmetic, food colorants, printing, and pharmaceutical industries [1]. As a characteristic of the textile-processing industry, a wide range of structurally diverse dyes is used within short periods in one and the same textile industry, and therefore, effluents from textile industry are extremely variable in composition [2]. SFRed (C.I: Reactive Red 195) is one of the reactive azo dyes that are commercially used for textile dyeing that contains reactive group which is often a heterocyclic aromatic ring substituted with chloride or fluoride, e.g. dichlorotriazine [3]. There are a few reports of aerobic decolorization of azo dyes as well as in contrast a wide range of organisms are able to reduce azo compounds under anaerobic conditions. This has been shown for purely anaerobic (e.g. Bacteroides spp., Eubacterium spp; Clostridium spp.), facultatively anaerobic (e.g. Proteus vulgaris, Streptococcus faecalis), and aerobic (e.g. Bacillus spp., Sphingomonas spp.) bacteria, yeasts, and even tissues from higher organisms [4-11]. Synthetic azo dye compounds are resistant to aerobic degradation by bacteria [12]. The strong electron-withdrawing nature of the azo group stabilizes these aromatic pollutants against conversions by oxygenases and obstructs the susceptibility of azo dye molecules to oxidative processes [13]. Only bacteria with specialised azo dye reducing enzymes were found to degrade azo dyes under fully aerobic conditions [14]. Due to their recalcitrance in aerobic environments, the azo dyes eventually end up in anaerobic treatment process which is much less specific [15]. This anaerobic reduction implies decolourisation as the azo dyes are converted to usually colourless but potentially harmful, mutagens and carcinogens aromatic amines which cannot be regarded as environmentally safe end products [16]. Aromatic amines are generally not further degraded under anaerobic conditions [17]. The aromatic amines are considered to be stable biotransformation products of azo dye metabolism by anaerobes [17]. Anaerobic treatment must therefore be considered merely as the first stage of the complete degradation of azo dyes. The second stage involves conversion of the produced aromatic amine biodegradation under aerobic conditions.This observation coupled with the fact that many aromatic amines are completely degraded under aerobic conditions has led to the proposal that anaerobic-aerobic systems might be effective in achieving the complete biodegradation of azo dyes [3]. Results presented in this study suggesting that the biodegradation of Reactive Red 195 under facultative anaerobic conditions was degraded into aromatic amines which can be further degraded into safe end byproducts. |
| Materials and Methods |
| Isolation of dye-degrading bacteria |
| Four bacterial strains (Rl, R2, Rc, Rd) isolated in this study were previously isolated from textile wastewaters. Filter Sterilized Textile wastewater (FSTW) was prepared by diluting the filter sterilized raw textile wastewater collected from a textile industry in Johor in distilled water at 1:1 ratio and supplemented with glycerol (1 g/L). Cultures were incubated overnight at 37°C prior to isolation of pure strains by single colony isolation onto the same agar medium. Direct isolation was conducted via serial dilution (100-10-6) followed by spread plate onto Filter Sterilized Textile Wastewater (FSTW) or Autoclaved Textile Wastewater (AWW) agar medium. Potential bacteria that can grow and produce clear zone on the agar medium was selected for further screening. |
| Screening of bacteria for color removal |
| Pure bacteria were inoculated (10% v/v) into Chemically Synthetic Medium (CDM) broth containing (g/L): K2HPO4 (7), KH2PO4 (2), MgSO4.7H2O (0.1), CaCl2 (0.02), (NH4)2SO4, (1), Glycerol (1), and Reactive Red 195 dye (0.1), adjusted to pH 7 prior to sterilization. Bacteria were grown under facultative anaerobic conditions (1:1 ratio of medium to the total volume of container) for 24 h at 37°C, prior to monitor the decolorization activity. Other parameters were also screened for their removal such as sulfate, phosphate, nitrate and COD. The culture (10mL) was taken out gradually every 24 hours and HACH reagent was mix and read by using HACH Spectrophotometer. The decrease of these parameters can increase the quality of the wastewater treated. |
| Identification of bacteria using 16S rRN |
| The total genomic DNA of R2 was isolated using Promega WIZARD® Genomic DNA Purification kit. The forward primers, FD1 (5’- AGAGTT TGATCC TGGCTCAG -3’) and the reverse primer, RD1 (5’- AAGGAGGTGATCCAGCC -3’) were used to amplify the 16S rRNA sequence of R2. PCR conditions included an initial denaturation for 5 min at 95°C followed by 30 cycles of 1 min at 95°C, 1min at 50°C and 2 min at 72°C. The amplicons were purified and sequenced. The 16S rRNA sequence of R2 was analyzed using Basic Local Alignment Search Tool (www.ncbi.nlm.nih.gov/BLAST). |
| Phylogenetic analyses of R2 16S rRNA gene sequences |
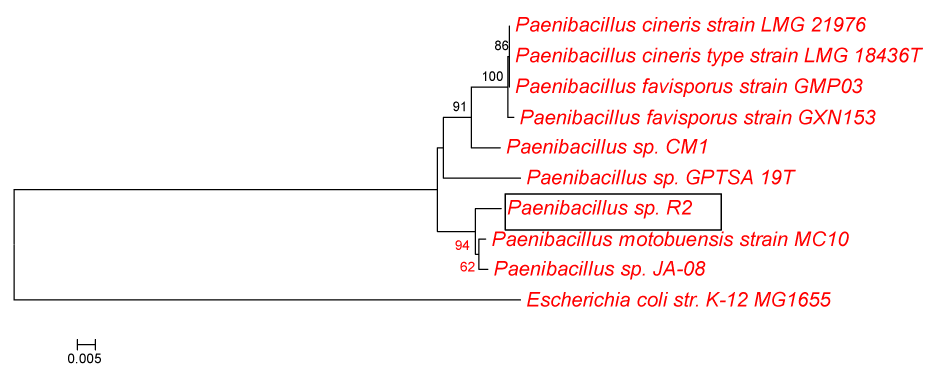
| Phylogenetic and molecular evolutionary analyses were conducted using MEGA version 4 [18]. The evolutionary history was inferred using the Neighbor-Joining method [19]. The bootstrap consensus tree inferred from 500 replicates is taken to represent the evolutionary history of the taxonomy analyzed [20]. Branches corresponding to partitions reproduced in less than 50% bootstrap replicates are collapsed. The percentage of replicate trees in which the associated taxonomy clustered together in the bootstrap test (500 replicates) is shown next to the branches [20]. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Maximum Composite Likelihood method [21] and are in the units of the number of base substitutions per site. All positions containing gaps and missing data were eliminated from the dataset (Complete deletion option). There were a total of 1420 positions in the final dataset. This tree was rooted with gram negative bacteria Eschericia coli strain K12 MG1655. |
| Batch culture experiments |
| Culture bacteria of Paenibacillus spp. R2 was incubated at 37°C under facultative anaerobes condition (Schott Bottle filled up with CDM and incubation without shaking). After an optimization processes, the CDM broth have been modified which contains the following chemicals (g/L): K2HPO4 (7), KH2PO4 (2), MgSO4.7H2O (0.1), CaCl2 (0.02), NH4Cl2 (0.5), Glucose (1), and Reactive Red 195 dye (0.1), and was adjusted to pH 7 prior to sterilization. The 1 L Schott bottle bacterial culture was incubated for a total 350 hours period in incubator without any agitation. Samples were withdrawn using syringe without any introduction of air to maintained facultative anaerobic condition. Initially at primary phase, which redox potential tend to decrease from 2.5 to 0.5 mV; samples were taken at 4 hours interval from 0 to 96 h. Meanwhile, after redox potential tend to become steady state at/or less than 0.5 mV (secondary phase), samples were taken daily from 96 h to 350 h. |
| Localization and detection of azoreductase activity |
| Fresh overnight culture of Paenibacillus spp. R2 was harvested after growth for 24 hours in CDM broth without shaking at 37°C. At this time, the cell culture had reached its late exponential phase. The cells were harvested by centrifugation, and washed 2-3 times in cold potassium phosphate buffer (50 mM, pH 7.5). Supernatant (S) was separated from the cell pellets in 1mL eppendorf tube. Meanwhile, the cells were then re-suspended in the same potassium phosphate buffer, and pellet maintained as resting cells (Rc). The remaining cell suspension was sonicated for a total of 2 minutes, with 2 minutes intervals after each 15 seconds of treatment. The cells were incubated on ice during sonication using an ultrasonic processor (Vibra CelFM, Sanies&Materials Inc.) of 600 W at 0% amplitude. The homogenate (H) was centrifuged at 14000 rpm at 4°C for 30 minutes. The supernatant labeled as crude Cell Free Extract (CFE) while pellet contained as Cell Debris (CD). All bacterial fractions were test for their azo reductase activity using following assay condition. |
| Assay for azoreductase activity |
| The azoreductase activity was detected and measured using a modified procedure by [22]. The homogenate or cell extract (150 μL) was added to a sparged anaerobic solution containing Tris-HCI buffer (50 mM, pH 7.5), RR195 (0.03 mM) and FAD (0.05 mM). The reaction mix was flushed with nitrogen gas to create an anaerobic condition. For aerobic assay, sparging with nitrogen gas to the reaction mix was not required. The reaction (1 mL) was initiated by the addition of NADH (0.8 mM), followed by 10 minutes incubation. The reaction was followed using UV-Visible spectrophotometer (Shimadzu UV 2450) at the RR195 dye maximum wavelength absorbance of 517 nm. The azoreductase activity was calculated from the decrease in absorption at 517 nm. One unit (U) of the enzyme activity is defined as the amount of enzyme catalyzing the reduction of 1 ppm of dye per minute. |
| pH and temperature optimum and stability of the azoreductase |
| For determination of the pH dependence of azoreductase, the buffer used for the assay was set to pH values of 5.5 to 8.5. NADH solution and dyestuff solution were prepared with the corresponding buffers. For the determination of the temperature optimum, all solutions were mixed after brought to the corresponding temperature and the spectrophotometer (Shimadzu UV 2450) was temperature controlled during measurement. Enzyme stability was tested at five different pH values and five different temperatures. For this test, 150 μL of enzyme solution was mixed with 850 μL of buffer and incubated in a thermocontrolled water bath. Samples were taken after 10 minutes incubation, immediately frozen at -20°C, and subsequently analyzed. Buffers used for incubation were as follows: For pH values of 5.5, a 50 mM sodium acetate buffer system, pH of 6.5-7.5, phosphate buffer system and for pH 8.5, Tris-HCL buffer system were used respectively. |
| Analytical Methods |
| Decolorization activity was expressed as the percentage of color removed by the bacteria using a modified method described by Yatome et al. [23]. Azo dye of Reactive Red reduction was monitored at an absorbance maximum (λmax) of 517 nm, using UV-Visible spectrophotometer (Shimadzu UV 2450) in 1-cm quartz cuvette. The pH and ORP value was determined immediately after sampling to avoid any change due to the CO2 production, using a pH meter and ORP meter (Hanna Instrument). All the other analytical determinations such as sulfate, phosphate, nitrate and Chemically Oxygen Demand (COD) were performed as described in Standard Methods for Examination of Water and Wastewater (APHA, 1985). Total Organic Carbon (TOC) was measured using TOC analyzer Multi N/C 2100 provided by Analytic Jena. |
| Meanwhile, samples for High Performance Liquid Chromatography (HPLC) were concentrated using equal volume of ethyl acetate of solvent extraction method [2]. A High Performance Liquid Chromatography (HPLC) Agilent 1100 Series model, equipped with an auto sampler and Water Hypersil C18, 5 μm (4.6 m×250 mm) reverse phase column was used to separate individual compounds of intermediates that were detected using UV-Vis Detector. The mobile phase (HPLC grade) consisted of methanol (50%) and nano pure water (50%) in ratio of 1:1. The column was run at flow rate 0.5 mL/min, without controlling the temperature and the eluant was monitored at wavelength 254 nm using isocratic elution. |
| Chemicals |
| Most chemical were analytical and HPLC grade, obtained from various companies such as Amresco, Fluka, Merck, Oxoid, Promega, Riedel-daHaen and Sigma Aldrich. All Kits for molecular purposes were obtained from Promega, Fermentas, and Vivantis. Both primers were synthesized and 16SrRNA sequencing was performed by First Base (Malaysia) Sdn. Bhd. |
| Nucleotide sequence accession number |
| The 16S rRNA gene sequence of strain R2 has been deposited in GenBank nucleotide sequence databases under accession no: GQ244430 |
| Results |
| Isolation and screening study |
| Four isolates designated R1, R2, Rc and Rd, showed their ability to remove colour. All of these isolates were further grown on Filter Sterilized Textile Wastewater agar (FSTW agar), without the addition of any carbon and nitrogen sources. They showed the ability to grow on the FSTW agar after 48 h of incubation at 37°C. Screening results for colour removal was done under two different conditions, facultative anaerobic and aerobic condition. Bacteria R2 give the highest colour removal under facultative anaerobic condition, which is more than 63%. Meanwhile, Rd showed the reverse trends that is, colour removal occurred under aerobic condition, but only 28.5%. Subsequently, the forthcoming decolourisation studies were carried out under facultativeanaerobic condition using pure culture of R2. |
| Sequence analyses of gene encoding for the 16S rRNA from bacterium R2 |
| The phylogenetic tree (Figure 1) constructed by the MEGA4 [18] displays R2’s position in relation to other members of the Paenibacillus spp., and 98% most closely related to Paenibacillus spp., JA-08 and Paenibacillus motobuensis strain MC10. |
| Degradation of RR195 by Paenibacillus spp.R2 |
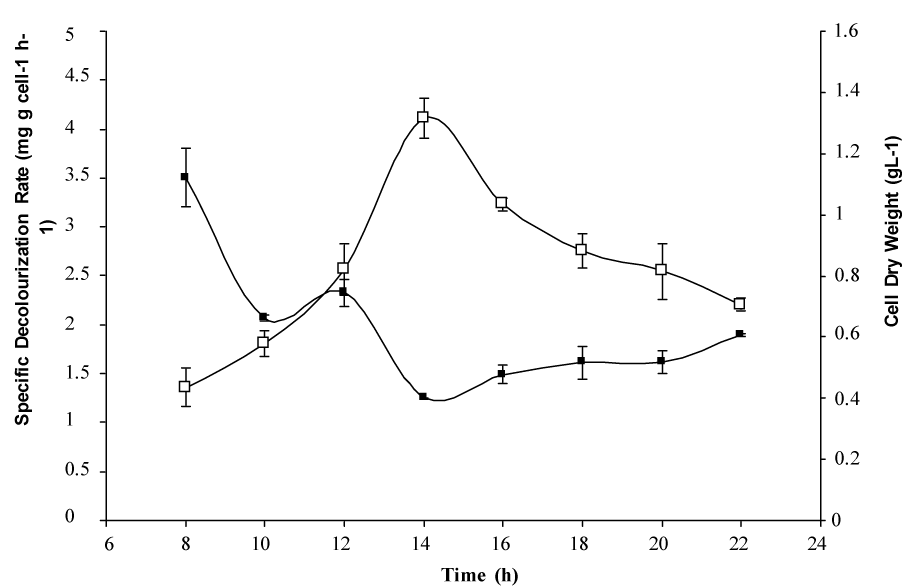
| Further decolourization studies under optimized conditions in chemically defined medium supplemented with (gL-1) of glucose (1), NH4Cl (0.5), Reactive red 195 (0.1), adjusted to pH 7 with inoculum (10% v/v) and grown under partial anaerobic condition at 37°C, showed that bacterium R2 successfully removed more than 95% colour and up to 50% of Total Organic Carbon (TOC). While the colour was removed more than 70%, the ORP values were decreased from 2.5 mV to 0.47 mV and the pH maintained from 7.21-7.25 in the phosphate buffered system (Table 1). In addition, data in figure 2 show that the decolorization activity of Paenibacillus spp. R2 decreased slightly from early-log phase (3.29 mg g cell-1 h-1) to early-stationary phase (1.24 mg g cell-1 h-1). Meanwhile, biomass concentration or cell dry weight of the bacteria increased from 0.48 to 1.36 g L-1. First-order rate constants (k), resulting from fitting equation [1.0] [24] to the whole curve (monoazo dyes) is, k=4 0.0091 h-1. Based on the k-values, RR195 which contains triazine as a reactive group had a slow rate of decolourisation as compared to other reactive dye [24]. |
| At = A0 . e− kt [1.0] |
| ln(At) = ln(A0)-kt |
| with: At=light absorbance at λmax at a given time (t) |
| A0=light absorbance at λmax at time 0 |
| k=first order rate constant (h-1) |
| t=time |
| Localization and detection of enzymatic activities |
| The enzyme activity of whole cells/resting cells (0.006 U/mL) and supernatant (0.003 U/mL) were significantly lower than the enzyme activity of the same dyes determined with other cellular component (0.020-0.030 U/mL) under anaerobic assay condition (Table 2). But, the highest enzyme activity was found in cell free extract which is 0.033 U/mL as well as homogenate (0.030 U/mL). Otherwise, under aerobic assayed condition (Table 2), the azo reductase activities were significantly high to whole cell supernatant and resting cells of Paenibacillus spp. R2, which are 0.026 U/mL and 0.015 U/mL, respectively. While the azo reductase activities present in membrane fraction/cell debris and homogenate were slightly low (0.001-0.002 U/mL). No enzyme activity was detected (<0.001 U/mL) in the cytoplasmic fraction/cell free extract of strain R2. This azoreductase enzyme was found to be stable at pH 7 and temperature of 37°C. The enzyme activity tend to decrease more than 20% when the pH was varies from pH slightly acidic and alkaline. While, same trends was observed when it was exposed at lower and higher temperature. |
| Detection and analysis of HPLC products |
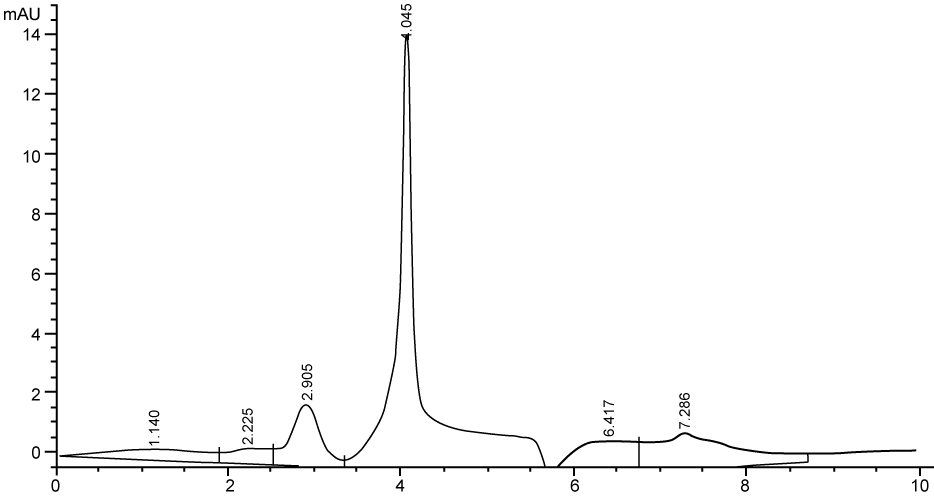
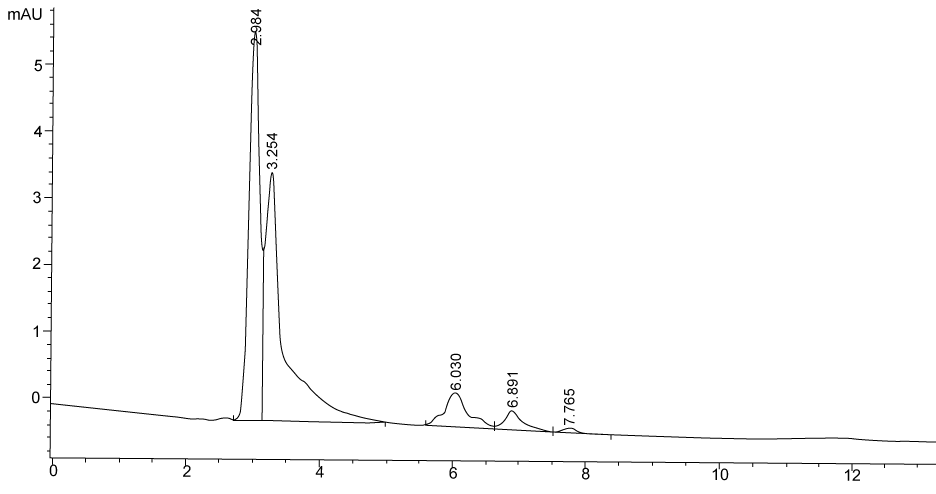
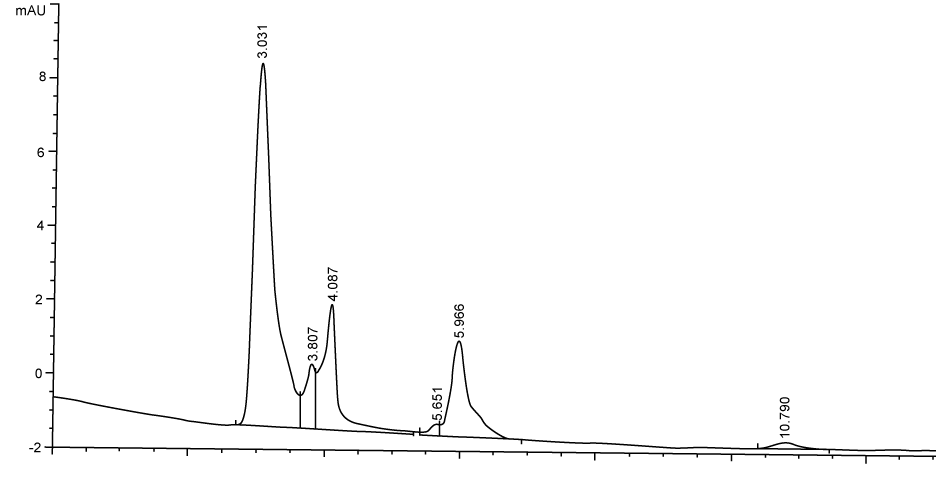
| Figure 3 shows the HPLC chromatograms of RR195 decolourization before the treatment processes. It was shown that the compound which is unknown metabolite had retention time, Rt=4.045. At the beginning of the anaerobic incubation, the parent compound, Reactive Red 195 (RR195) was not detected under these chromatographic conditions. While (Figure 4) shows that the retention time of the parent compound was disappeared (Rt=4.045) and produced lower retention time of 2.984 (A) and 3.254 (B) suggested were 4-amino, 3-hydronapthalenesulphonic acid (± 0.44%) and 4-aminobenzenesulphonate (sulpanilic acid) (± 0.6%) respectively. The sulphanilic acid was suggested can further be degraded by ring opening mechanism of aromatic ring into α-ketoglutaric acid, C with retention time of 3.031(± 0.36%) as shown in Figure 5. The α-ketoglutaric acid is one of TCA cycles intermediates which is a common metabolic in organism [25]. Furthermore, the fact that the UV-detectable area in Figure 4 shifted towards lower retention times (Rt=2.984 and 3.254) which means that these by-products of RR195 decolourization were more polar than the parent compounds (Rt=4.045) shown in Figure 3. While (Figure 4) also showed that when such by-products were degraded aerobically they formed less aromatic, more-polar compounds, since the UV-detectable area decreased in Figure 4 and was shifted towards more lower retention times (Rt=3.031) than in Figure 4(Rt=3.254). |
| Discussion |
| Factors affecting colour removal |
| Previously, it was observed that physiological factors like pH, oxygen level and temperature had significant effects on the rate of decolourization. This finding on colour removal seems to indicate that glucose is probably an essential co metabolites for cell growth, since the was a decrease in specific reduction activity from early log phase to early stationary phase, during which cell growth is the dominant physiological activity [3]. The growth promotes certain metabolite pathways or accumulation of certain coenzyme or reducing equivalents (eg. NADH and/or flavin) which lead to the decolourization by the reduction of azo bond(s) [26]. |
| Identification of bacteria using 16S rRNA |
| Since the BLASTn result revealed that the sequence from isolated R2 has 98% identity with Paenibacillus sp., it can possibly suggested that bacteria R2 belongs to the genus Paenibacillus. In addition Paenibacillus azoreducens CM1 [27] is a known dye degrading bacteria isolated from industrial wastewater, having 95% sequence homology with bacterium R2. Thus, it can be suggested that Paenibacillus sp. R2 has the ablity to decolourize textile wastewater by reductive cleavage of azo linkages in the azo dyes compounds. |
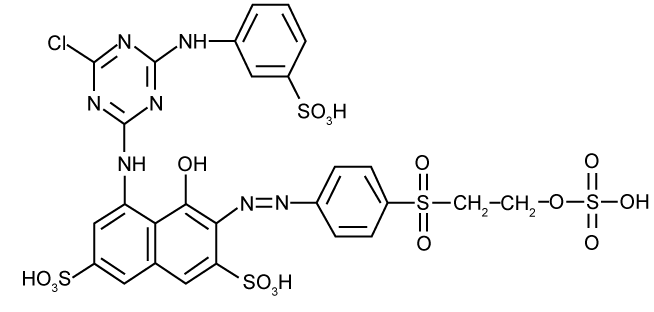
| There is more than 95% decrease in absorbance (λmax=517 nm) of azo dyes decolorization, RR195 by Paenibacillus sp. R2 caused the reaction products were usually colourless. But, in this experiment, a new absorption peak was found at 336 nm, close to λmax, in which a shift from red to yellow was observed. In the cases of monoazo dyes, the reaction followed first-order kinetics as shown for the example of RR195. In contrast, dyes with more than one azo linkage displayed multiphase kinetics [3]. Based on the k-values, RR195 which contains triazine as a reactive group (Figure 6) had a slow rate of decolourisation as compared to other reactive dye [24]. |
| Localization of azoreductase enzyme |
| Location of the enzyme system(s) responsible for the reduction of sulfonated azo dyes by Paenibacillus sp. strain R2 was determined. Majority of the azoreductase enzyme produced by Paenibacillus sp. R2 was probably located inside the cell (intracellular enzyme) which is oxygen sensitive enzyme. Recently, the most generally accepted hypothesis for bacterial reduction of azo dyes is that many bacterial cells possess a rather unspecific cytoplasmic azo reductases which transfers electrons via soluble or bound-flavins to the azo dyes. |
| The oxygen inhibition on enzymatic reduction of azo dyes for bacterial species containing azoreductase system has also been observed [28], and may be attributed to a predominant competition for NADH utilization by aerobic respiration, which triggers an electron transfer from NADH to oxygen to form ATP [29]. Since NADH acts as the electron donor for the reduction of azo bonds that leads to bacterial decolorization of azo dyes, the consumption of NADH by oxidative phosphorylation would become a negative effect on the azoreductasedriven decolorization. |
| Azoreductase was found to be stable at pH 7. This is due to azoreductase configuration and its active site was preserved and stable at pH 7 which promoted optimum decolourization processes. The decolourization was optimum when pH of the system is between pH 6-8. The optimum pH for colour removal is often at a neutral pH value or a slightly alkaline pH value and the rate of colour removal tends to decrease rapidly at strongly acid or strongly alkaline pH values [30]. Meanwhile, the enzyme shows high activity within the range of 30-40°C. The assay was carried out under anaerobic condition at pH 7. The temperature required to produce the maximum rate of colour removal tends to correspond with the optimum cell culture growth temperature of 35-45°C. The decline in colour removal activity at higher temperatures can be attributed to the loss of cell viability or to the denaturation of the azo reductase enzyme [31]. |
| Analysis of reactive red 195 biodegradation |
| The HPLC analysis performed to supernatants taken during the different duration of the anaerobic phases showed the correspondence of the evolution of decolorization metabolite to the color removal. The chromatographic peak areas corresponding to the dye-degradation metabolite increased as the incubation time of anaerobic phase increased. The UV-absorbing area of the metabolite formed in anaerobic phase significantly decreased in the subsequent phase. It should be noted that due to the unavailability of authentic standards, the chromatographic peaks appearing in this sample taken during the anaerobic phase could not be identified or quantified [2]. Another factor is precision which means that all measurements of an analyte should be very close together with the standard. All quantitative results should be of high precision and there should be no more than a ± 2% variation in the assay system [32]. |
| The chromatograms demonstrate that anaerobic treatment generated UV-absorbing compounds which were more polar than the parent compounds. The anaerobic degradation of reactive azo dye has been shown to produce several aromatic and ionic by-products [33]. In addition, the chromatograms also demonstrate that aerobic treatment reduced the levels of these UV-absorbing compounds and increased their polarity. Degradation in the aerobic stage may result in the formation of oxidised and very polar derivatives (e.g., aldehydes, carboxylic acids) having a lower aromaticity, as suggested by Noertemann et al. [34] in a study of 6-aminonaphthalene-2-sulphonic acid degradation. The fate of aromatic amines in the aerobic stage cannot be conclusively determined. Partial or complete removal of many aromatic amines can be suspected from the decrease or disappearance of the sometimes unidentified- peaks in HPLC-chromatograms [3]. |
| Mechanism of colour removal |
| Decolourization usually occurs favorably under facultative anaerobic condition indicated by lower redox potential of the system. This phenomenon may be due to either the electron-withdrawing nature of the azo bond and their resistance to oxygenases attack, or because oxygen is a more effective electron acceptor, therefore having more preference for reducing equivalents than the azo dye [16,35]. However, certain aerobic bacteria are able to reduce azo compounds and produce aromatic amines in the presence of specific oxygencatalysed enzymes called azo reductases [36]. In the early stage of cell growth, the specific decolourization rate was relatively high due to high organics or inorganics compounds that may be involved in biodegradation mechanisms by acting as electron shuttles between the reducting equivalents and the azo dyes. The reduction equivalents that are formed during anaerobic oxidation of these carbon sources are finally used for the reduction of the azo bond [36]. In contrast, towards the ends of log phase, the specific decolourization rate was lower due to the limitation of carbon source as co metabolites and the accumulation of intermediates such as aromatic amines which may cause inhibition to the azoreductase enzyme [36] and (or) normally attributed to the loss of cell viability [31]. |
| Several different mechanisms have been proposed for reduction or degradation of azo dyes and similar compounds [37]. A description of a nonspecific azo reductase system involved in azo dye reduction has been provided for selected bacterial species, and it has been shown that the relevant gene is relatively conserved in various anaerobic and facultative bacteria. In this research it was also hypothesized that coenzyme reducing equivalents (e.g., NADH) involved in normal electron transport through oxidation of organic substrates (glucose) may act as electron donors for the reduction of azo dyes. This would likely explain the observation that azo dye reduction occurs more readily as a co-metabolic event when additional readily degradable substrates (glucose) are provided. There were at least two possible ways for glucose to enhance reduction of sulfonated azo dyes. It could act as a donor of reducing equivalents (e.g., via NADH or FADH2), or its addition could result in more actively respiring cells, thus rapidly removing the oxygen in culture medium and enabling corresponding enzymes to transfer reducing equivalent to azo dyes [15]. |
| In many intestinal bacterial isolates, a flavin compound (riboflavin, flavin adenine dinucleotide, of flavin mononucleotide (FMN)) is required for azoreductases activity [15]. The most generally accepted hypothesis for this phenomenon is that many bacterial cells possess a rather unspecific cytoplasmic favin-dependent reducatses (flavin reductases) which transfers electron under anaerobic conditions via (soluble) flavins to the azo dyes [15]. In the present study under anaerobic condition, a rather high decolorization of Reactive Red 195 azo dyes was observed when incubated them with Cytoplasmic Fraction Extract (CFE) of Paenibacillus sp. strain R2 in anaerobic buffer with NADH as a source of reduction equivalents (Table 2) with the presence of Flavin Adenine Dinucleotide (FAD). A possible explanation for this phenomenon is that FAD is reduced enzymatically to FADH2 by NADH and then spontaneously reduces the RR195 sulfonated azo dyes to the corresponding amines [15]. In contrast, it was shown that the addition of NADH and FAD did not lead to enhancement of the reduction rates of sulfonated azo dyes by whole cells, resting cells (Rc) and supernatant (S) of Paenibacillus sp. strain R2 (Table 2). Thus, this has generally been explained by the low permeability of the cell membranes for the highly polar sulfonated azo compounds [11]. |
| Moreover, the bacterial membrane is hardly permeable for flavin containing cofactors and restricts the transfer of reducing equivalents by flavins from cytoplasm of intact cells to extracellular sulfonated azo dyes [11]. It was clearly demonstrated in this study that the total activity of flavin reductase, which hypothesized to function under adequate conditions as flavin-dependent azo reductase, was present in the cytoplasmic fraction. Therefore, it appears reasonable that, with intact cells (Rc), intracellular enzymes like flavin reductases are of little importance for reduction of extracellular sulfonated azo compounds by strain R2. These results supported the hypothesis of Russ and coworkers that the reduction of sulfonated azo dyes by reduced flavins formed by cytosolic flavin-dependent azo reductases is mainly observed in vitro and in vivo is of insignificant importance [15]. Thus, in the intact cells, other enzyme systems, which does not require transport of the azo dyes though the cell membrane, are presumably responsible for the unspecific reduction of various sulfonated azo dyes by Paenibacillus sp. strain R2. In addition, it could be demonstrated in the cell-fractioning experiments that the azoreductase activity was almost restrictively present in the membrane fraction, cell debris (CD), (Table 2) These results suggest the existence of a non-specific azoreductase in Paenibacillus sp. strain R2 membranes that catalyze the reduction of endogenous reducing equivalent, maybe responsible for the reduction of exogenous reducing equivalents which then transfer reduction equivalents to sulfonated azo dyes outside the cells. |
| Meanwhile, the rather high reduction rate of RR195-sulfonated azo dyes in the present of oxygen (aerobic condition) found in whole cells (Rc) and supernatant (S) experiment (Table 2) Thus, we cannot eliminate the possibility that some unknown enzymatic activities are involved in the aerobic reduction of sulfonated azo dyes by whole cells of Paenibacillus sp. strain R2. We are currently attempting to explore the other extracellular electron transferring mechanisms of strain R2 which are involved for the reduction of the azo dye to clarify this mechanism. |
| Conclusion |
| In summary, we successfully isolated a coccobacillus, gram negative and facultative anaerobic decolourizing bacterium; Paenibacillus spp. strain R2 that can degrade azo dyes to yellowish coloured intermediates of aromatic amines and/or even to partially mineralize them into simpler compound that is a common metabolic in organism, which are safe and less toxic to the environment. We also have shown that an azoreductase is responsible for azo dye reduction by Paenibacillus spp. strain R2. Future investigations should focus on the application of this highly potential azo dye-degrading bacterium for the treatment of textile dyeing effluents. |
| Acknowledgments |
| The author is thankful to the Universiti Teknologi Malaysia for the UTM-PTP scholarship awarded and Ministry of Science and Technology (MOSTI) of Malaysia for the funding throughout this research. |
References
|
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
| Figure 4 | Figure 5 | Figure 6 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 20022
- [From(publication date):
January-2013 - Nov 13, 2025] - Breakdown by view type
- HTML page views : 15011
- PDF downloads : 5011
