Review Article Open Access
Drugs for Alzheimer's
Gonzalo Emiliano Aranda-Abreu1*, María Elena Hernández-Aguilar1, Marisol Herrera-Rivero2 and Luis Isauro García-Hernández11Centro de Investigaciones Cerebrales/Universidad Veracruzana, Cuerpo Académico de Neuroquímica, Xalapa,Veracruz, México
2Doctorado en Ciencias Biomédicas/Universidad Veracruzana, Centro de Investigaciones Biomédicas, Xalapa, Veracruz, México
- *Corresponding Author:
- Gonzalo Emiliano Aranda-Abreu
Centro de Investigaciones Cerebrales/Universidad Veracruzana
Cuerpo Académico de Neuroquímica
Av. Luis Castelazo Ayala
Km 3.5 Carretera Xalapa-Veracruz 91190; México
Tel: 52 228 8418900 Ext. 13616
Fax: 52 228 8418920
E-mail: garanda@uv.mx
Received November 14, 2011; Accepted January 09, 2012; Published January 08, 2012
Citation: Aranda-Abreu GE, Hernández-Aguilar ME, Herrera-Rivero M, García-Hernández LI (2012) Drugs for Alzheimer’s. J Addict Res Ther S5:003. doi:10.4172/2155-6105.S5-003
Copyright: © 2012 Aranda-Abreu GE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Addiction Research & Therapy
Abstract
Five drugs are currently approved for the treatment of Alzheimer’s disease; nevertheless, we have faced the challenge to respond to family members of these patients the reason why they observe a limited improvement from these treatments. Although both acetylcholinesterase inhibitors and memantine are correctly designed and they show an adequate performance, we believe the neuronal conditions resulting from the pathophysiology of the disease difficult their proper working in brains affected with Alzheimer’s. Patients should therefore undergo a neuronal rehabilitating therapy that enables these drugs for a more effective performance.
Introduction
Five drugs have been so far approved for the treatment of Alzheimer’s disease (AD) by the Food and Drug Administration (FDA), the most popular amongst them being the acetylcholinesterase inhibitors such as donepezil (aricept), galantamine (razadyne) and rivastigmine (exelon). Some of the more outstanding clinical characteristics of this drugs are that, for example, donepezil (aricept) is the most widespread used because it presents reduced side effects with a once a day dosage [1] and, importantly it can be used for treating severe AD cases [2]; galantamine (razadyne) with a twice a day dosage taken with meals, has proved to be involved in the inhibition of β-amyloid (Aβ) aggregation and citotoxicity [3], also showing a protective role against oxidative stress [4]; and rivastigmine (exelon) is only used at very low dosages because of the risk of severe gastric damage and hepatotoxicity associated with its chronic consumption [5]. We should also mention in this group tacrine which associated hepatotoxicity has left it out of the common clinical practice.
Aside acetylcholinesterase inhibitors we find memantine, a NMDA receptor antagonist [6] drug that is the only of its kind approved by the FDA for the treatment of moderate to severe AD [7]. Despite all the beneficial effects these drugs have shown on the cognitive and behavioral states of AD patients, it is noticeable that the treatment for AD does not come helpful to stop the cognitive decline as the neurodegeneration keeps progressing what gives any treatment a limited period of actual improvement of the symptomatology of this disease (Table 1).
Alzheimer´s Pathology
In 1979, it was raised the idea of manipulating the cholinergic system for the treatment of memory problems [8] and so the use of acetylcholinesterase inhibitors was introduced. It was thought these drugs could be helpful for treating Alzheimer’s symptoms, precisely memory loss. The first acetylcholinesterase inhibitor developed specifically for treating AD was tacrine [9] and latter galantamine arrived [10,11]. Nevertheless, neither of them showed the expected improvement on AD patients. After a while, scientists began to turn their attention to the effects dietary habits could exert on these patients, keeping in mind the role that lipids and other molecules play in processes such as myelogenesis and brain development [12].
Both the acetylcholinesterase inhibitors and memantine do function in a proper manner on the neuronal level under normal conditions, although this might change on the damaged and deteriorated AD neurons. To understand the basis of the AD treatment, we should remember the pathophysiology of the disease and its characteristic lesions: the neuritic plaques generated by Aβ aggregation [13] and the neurofibrillary tangles originated by an abnormal phosphorylation of tau [14].
Recent studies of postmortem brains from Alzheimer’s disease (AD) patients and transgenic mouse models of AD suggest that oxidative damage, induced by amyloid beta (Aβ), is associated with mitochondria early in AD progression. Furthermore, accumulation of Aβ at synaptic terminals might contribute to synaptic damage and cognitive decline in patients with AD [15].
Mitochondrial dysfunction is observed in Alzheimer’s disease (AD) brain, and the amyloid-beta (Aβ) peptide is known to induce mitochondrial dysfunction. The relative degree of mitochondrial dysfunction in different regions of the brain in AD is not completely understood. Moreover, the relationship between levels of synaptic mitochondrial Aβ and mitochondrial dysfunction has not been clearly established. It has been showed that hippocampal and cortical mitochondria showed the highest levels of mitochondrial dysfunction, while striatal mitochondria were moderately affected, and amygdalar mitochondria were minimally affected. Synaptic mitochondria were more impaired than non-synaptic mitochondria in the AD mouse models. The AβPP/PS1 mice showed more impairment in the cognitive interference task of working memory than the AβPP mice. The association between mitochondrial Aβ levels and mitochondrial dysfunction in mouse models of AD supports a primary role for mitochondrial Aβ in AD pathology. Moreover, the degree of cognitive impairment in AD transgenic mice can be linked to the extent of synaptic mitochondrial dysfunction and mitochondrial Aβ levels, suggesting that a mitochondrial Aβ-induced signaling cascade may contribute to cognitive impairment. Therapeutics that target this cascade could be beneficial in the treatment of AD [16,17].
| Group | Drug | AD stage | Beneficial Effect | Risk | Dosage |
|---|---|---|---|---|---|
| Acetilcholinesterase inhibitor |
Tacrine | Mild-to-moderate | Improves cognition and behavior | Hepatotoxicity | Initial:10 mg/6h Maintenance: 30 mg/6h |
| Galantamine | Mild-to-moderate | Improves learning and memory, cortical protection, inhibition of Aß aggregation | Nausea, vomiting, weight loss | Solution-Initial: 4mg/12h Maintenance: 8mg/12h Capsules-Initial: 8mg/day Maintenance: 16mg/day Maximum:24mg/day |
|
| Donepezil | Moderate-to-severe | Improves cognition. Good tolerante. |
Muscle weakness |
Initial: 5mg/day Maintenance:10 mg/day |
|
| Rivastigmine | Mild-to-moderate | Improves cognition. Good tolerance |
Gastric damage Hepatotoxicity |
Oral-Initial: 1,5 mg/12h Maintenance:3-6mg/12h Transdermic-Initial: 4,6 mg/day Maintenance:9,5 mg/day |
|
| NMDA receptor antagonist |
Memantine | Moderate-to-severe | Decreases Aß aggregation, prevents synaptic dysfunction, inhibits tauhyperphosphorylation | Hallucinations Confusion Debilitation |
Initial: 5mg/day Maintenance: 20 mg/day |
Table 1: Drugs approved for the AD treatment.
Loss of synapses and synaptic damage are the best correlates of cognitive decline identified in patients with Alzheimer’s disease (AD), and mitochondrial oxidative damage and synaptic pathology have been identified as early events in the progression of AD. The progressive accumulation of amyloid β (Aβ) in synapses and synaptic mitochondria are hypothesized to cause synaptic degeneration and cognitive decline in patients with AD [18].
Microtubule Associated Protein Tau (MAPT)
This protein owes its name to its function as it associates to microtubules to provide stability [19,20], giving neurons their characteristic morphology. Thus, when tau expression is inhibited the neuronal morphology dramatically changes [21-24].
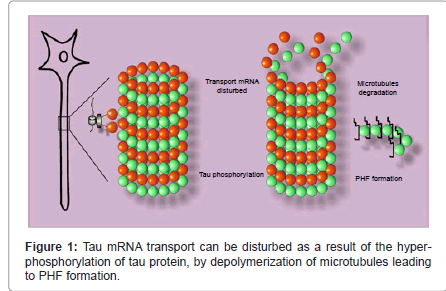
Tau’s mRNA transports anterogradely directed by the presence of a uracil rich 3'-UTR (Un-Translated Region) [25]. A protein HuD binds to this region to stabilize the RNA molecule and then attaches to a kinesin which will transport the formed RNA-protein complex along the axon [26,27] (Figure 1).
Tau’s translation occurs in several sites along the neuronal axon; this implies a movement of the RNA-protein complex together with those proteins involved in the translation process [28]. The hyperphosphorylation and the subsequent detachment of tau from microtubules disturb the antero- and retrograde transports in growing axons because of a displacement of the transporter protein [29]. These hyperphosphorylated, microtubule dissociated tau proteins then form paired helical filaments (PHFs) which will constitute neurofibrillary tangles inside neurons with a destabilized membrane [30,31] and lost polarity [32]. It is thought that a disregulation of tau transport might be the cause for PHF generation [33].
Amyloid cascade
The amyloid precursor protein (APP) is a type I membrane of small family with a large extracellular domain and a short cytoplasmic one, APP presents three main isoforms (695, 571 and 770 residues) and is the only protein containing the Aβ sequence. The 695 residues is the most abundant isoform in neurons, but other brain cells also express variable amounts of APP and non-neuronal cells express mainly the 751 and 770 residues APP isoforms. The APP gene is located in chromosome 21 and over 25 mutations to this gene have been describes as responsible for familial forms of AD [34,35].
β-amyloid peptides generate from the amyloid precursor protein (APP), after it has been cleaved by the sequential actions of two membrane enzymes: the β- and the γ-secretases. γ -secretase is a tetrameric complex which cleaves APP within its transmembrane domain, releasing an intact 39 to 43 residues long β -amyloid peptide [36]. Most β -amyloid peptides are 40 aminoacids long (Aβ40), while a minor proportion (~10%) are 42 (Aβ42). Compared to Aβ40, Aβ42 is a little more hydrophobic, toxic and prone to aggregation; the latter variant is more widely found as a constituent of amyloid plaques in the AD brain [37]. According to the amyloid hypothesis, the excessive and chronic aggregation of Aβ, particularly Aβ42, triggers a pathogenic cascade leading to the pathophysiology of AD [38]; however, the evidence shows that amyloid plaques are also associated with mild neuronal alterations in normal aging and no necessarily correlate with the level of cognitive decline in an AD mouse model. This suggests an independence of Aβ toxicity from its accumulation in the brain [39-41] (Figure 2).
Drugs for AD Work at the Synapse Level
There is no cure for AD, however drug treatments are available to help with the symptomatology in several aspects of the disease.
The drugs approved by the Food and Drugs Administration (FDA) for the treatment of AD, these are mainly divided into two groups: the acetycholinesterase inhibitors and the NMDA receptor antagonists (this last represented by memantine). We should considerer here that these drugs are designed to diminish the symptoms originated by neurodegeneration but that neither of them targets the plaques and/ or tangles to destroy them or to stop the processes responsible for their formation and progress; they provide cognitive improvement by differents means. It is also convenient here to say that these are not the only drugs that have shown beneficial effects on AD patiens, nevertheless no other drug has been approved for AD treatment so far [42].
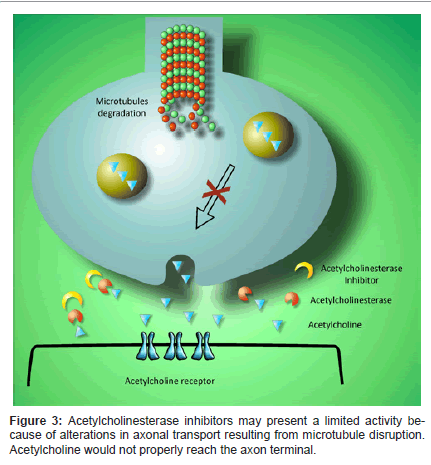
Acetylcholinesterase inhibitors allow a prolonged maintenance of acetylcholine in the synaptic cleft, which enables the neurotransmitter for a proper postsynaptic binding (Figure 3). When acetylcholinesterase is inhibited, cholinergic function rises by an increase of neurotransmitter concentration at the synapse. Acetylcholinesterase is the main degrading enzyme of acetylcholine in the brain; in AD, it has proved an increased activity in certain brain regions [43]. It has also been determined an association of acetylcholinesterase with amyloid plaques that may have an effect on AD’s pathology [44].
Donepezil and galantamine exert a rapid inhibiting action on acetylcholinesterase, while rivastigmine works more slowly. AD patients present an increase in acetylcholinesterase levels during the first months of treatment [44].
Although acetylcholinesterase inhibitors potentiate the brain on normal conditions [45], in the deteriorated AD brain their function could get a lot more difficult due to a disturbance in protein transport along the neuronal axon [46,47]. The alterations on axonal transport that lead to the loss of synapses related with cognitive decline might be due to an axonal misdistribution of mitochondria resulting from Aβ aggregation [18,48]. It has been hypothesized that amyloid peptides could be responsible for the disruption of the cytoskeleton which would contribute to a malfunctioning of axonal transport and subsequent alterations in the processing of membrane proteins such as APP [49].
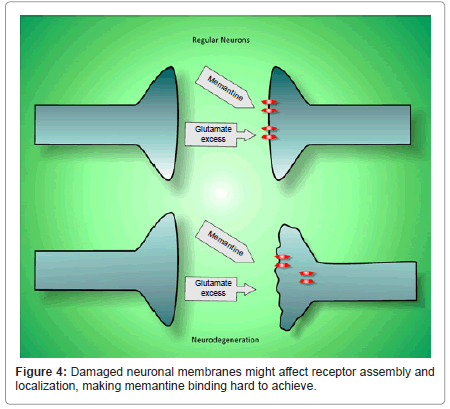
Glutamate activates several types of metabotropic and ionotropic receptors (AMPA, kainate and NMDA). This neurotransmitter is involved in Alzheimer’s neurotoxicity because it has been estimated that an increased activity of NMDA receptors in AD leads to neuronal loss [50] which might be exacerbated by the excitotoxicity of amyloid plaques. The progressive neuronal loss then increases cognitive impairment [51]. Memantine is a non-competitive, voltagedependent N-methyl-D-aspartate (NMDA) receptor antagonist with moderate affinity. In contrast to other competitive antagonists, memantine is well tolerated by patients [52]. A lot of research has been done to test the effects of memantine in animal models and clinical trials, data suggesting a neuroprotective effect in vascular dementia and its helpful use in the treatment of conditions such as CNS trauma and amyotrophic lateral sclerosis (ALS) [53]. Although memantine has been widely used for treating the symptoms of moderate to severe AD [54], we believe in the deteriorated AD brain it would exert a very limited improvement (Figure 4).
Neuroprotection and Potential New Drugs
There is currently no cure for AD and no treatment capable of eradicate its symptomatology, therefore our best weapons against the disease might be neuroprotection and the maintenance of a healthy neuronal state. As an old Hindu proverb says: “the fish dies by its mouth”, dietary habits importantly participate in several pathologies and AD is not the exception; thus, a healthy diet could be helpful to prevent AD as much as other diseases. AD presents a higher prevalence in industrialized countries, which might be the result of several environmental, dietary and social factors [55].
In order to keep neurons in a healthy state it is important to maintain the integrity of the plasma membrane and to avoid oxidative stress [56]. On this matter, attention has been turned to omega-3 acids as they are essential for neurogenesis [57] and the maintenance of cell membranes [58,59], they also reduce the oxidative stress-induced damage and take BDNF back to its normal levels [60].
Other commonly used drugs for the treatment of several symptoms of AD have shown some extra advantages and other drugs are currently being developed for this purpose. Examples are the antidepressants of the selective serotonin reuptake inhibitors (SSRIs) group such as escitalopram, used to treat depression in AD patients, which could also help to create neuronal connections in the AD brain [61,56]. Antipsychotic agents are useful to treat the associated psychotic symptoms of AD. Recently, agonists of the metabotropic glutamate receptors (mGlu) have been developed as novel antipsychotic agents lacking of the adverse effects of conventional antipsychotics. Selective positive allosteric modulators (PAMs) of mGlu2 receptors mimic the antipsychotic activity of mGlu2/3 receptor antagonists in animals. Experiments performed in mixed and pure neuronal cultures exposed to synthetic Aβ to investigate the distinct influence of mGlu2 and mGlu3 receptors indicate that a selective potentiation of mGlu2 receptors enhances neuronal vulnerability to Aβ toxicity while the dual activation of mGlu2 and mGlu3 receptors is protective against it [62].
Nowadays, several research groups focus their efforts on the development of new multitargeted drugs against AD [62-65] such as memoquin, an acetylcholinesterase inhibitor, free-radical scavenger and inhibitor of Aβ aggregation [66], and other quinone derivatives that are being widely used to develop new drugs which are effective simultaneously towards BACE1 activity, inhibition of Aβ aggregation and disaggregation of preformed Aβ fibrils [67].
Commentary
In our experience with AD patients, family members have repeatedly manifested their worry about treatments appearing to be ineffective despite their accuracy following physician’s instructions. It is known that AD drugs have a limited period of efficacy in these patients and that they do not stop the neurodegeneration process, reason why the cognitive decline keeps progressing over time. We believe a neuronal rehabilitation would enable these drugs to function in a better manner and thus become more effective to treat the disease, prolonging their positive effects over cognitive decline on AD patients.
We previously proposed a neuronal rehabilitation therapy which considers several aspects involved in the pathophysiology of AD [56]. This therapy first considers the repair of plasma membranes, which are very important because they are responsible for maintaining the cellular contents organized. Although there is no drug serving this purpose, fatty acids such as the omega-3 found in fish are capable of incorporating into membranes, being highly important for the cerebral cortex development [68].Unsaturated fatty acids like decosahexaenoic acid (DHA) are a great example of the relevance fatty acids have on health. DHA is particularly abundant in brain and retinal tissues; it corresponds to nearly 50% of all membrane acylated chains, thus a reduction in DHA affects membrane physical properties such as the domains involved in signaling processes, called rafts [69]. These rafts or cholesterol and sphingolipid rich microdomains are the major regulators of membrane organization. Because lipid rafts can move laterally and cluster into larger patches, they have been proposed to play a role in the redistribution of specific molecules to specialized cellular structures. Rafts have shown to favor formation and maintenance of synaptic receptor clusters in neurons of the CNS [70] and they are altered in AD patients [71].
Another important aspect to consider is oxidation. Resveratrol is a polyphenol with a great antioxidant activity and it has also demonstrated an ability to reduce amyloid plaques [72,73]; therefore, it is believed resveratrol could be beneficial for AD patients [74,75]. To restore memory processes, ginkgo biloba could be useful and provide some improvement for the AD patient’s quality of life [76,77] despite controversial results obtained from different studies. Neuronal rehabilitation also involves the reestablishment of neuronal connections and for this purpose we consider fluoxetine or escitalopram could be useful as they have demonstrated a role in the formation of new synapses and neuronal reconnection [78,79].
In AD, neuro-inflammatory processes are of great importance. Non-steroidal anti-inflammatory drugs (NSAIDs) have a protective effect on these patients, as shown by studies performed on rats where quisqualic acid was injected into the right nucleus basalis, excitotoxin induced cholinergic degeneration, an intense glial reaction and production of inflammatory mediators, but seven days of treatment with nimesulide (10 mg/kg/day, i.m.) strongly attenuated the microglial reaction, reduced the number of inducible nitric oxide synthase positive cells and completely abolished increase in prostaglandin-E2 formation [80]. Cyclooxygenase-2 (COX-2) is involved in the inflammatory component of ischemic cascade, playing an important role in the delayed progression of brain damage. In another study, the neuroprotective effect of nimesulide was still evident 30 days after an ischemic episode, providing evidence that COX-2 inhibitors confer a long-lasting neuroprotection. Oral administration of nimesulide was also able to significantly reduce brain damage, suggesting its protective effects are independent of the route of administration [81].
Induction of the endogenous neurogenesis is very important to help any neurological patient to improve therapeutic results, thus we believe a neuronal rehabilitation therapy for AD must also include activities pursuing this objective such as physical exercise and therapy, and mental games to exercise the brain while we rehabilitate neurons and their connections.
Conclusion
Memantine, tacrine, donepezil, rivastigmine and galantamine are the FDA approved drugs for the treatment of AD; nevertheless, the purpose for which they were designed is hard to achieve in the context of an ongoing neurodegenerative process, making it very difficult for current drug treatments to maintain a prolonged beneficial effect on AD patients.
We believe one has to be in the understanding that the only way these drugs might show better treatment outcomes is to look for a neuronal rehabilitation in AD patients [56] while new and better drugs able to target β-secretase activity, dissolution of amyloid plaques, and the restoration of axonal transport and neuronal polarity, are being developed. To cure AD with a single drug might be a hard task to accomplish; therefore, the treatment should be multidisciplinary and targeted to the care of these patients from the cellular level to the physical and psychosocial aspects, making use not only of drug treatments but also of neurogenesis induction, neuronal membrane rehabilitation and neuroprotection all of which could be importantly influenced by diet, sleep, and physical and mental exercise.
References
- Tsuno N (2009) Donepezil in the treatment of patients with Alzheimer's disease. Expert Rev Neurother 9: 591-598.
- Winblad B (2009) Donepezil in severe Alzheimer's disease. Am J Alzheimers Dis Other Demen 24: 185-192.
- Matharu B, Gibson G, Parsons R, Huckerby TN, Moore SA, et al. (2009) Galantamine inhibits beta-amyloid aggregation and cytotoxicity. J Neurol Sci 280: 49-58.
- Melo JB, Sousa C, Garção P, Oliveira CR, Agostinho P (2009) Galantamine protects against oxidative stress induced by amyloid-beta peptide in cortical neurons. Eur J Neurosci 29: 455-464.
- Birks J, Grimley Evans J, Iakovidou V, Tsolaki M (2009) Rivastigmine for Alzheimer's disease. Cochrane Database Syst Rev CD001191.
- Martinez-Coria H, Green KN, Billings LM, Kitazawa M, Albrecht M, et al. (2010) Memantine improves cognition and reduces Alzheimer's-like neuropathology in transgenic mice. Am J Pathol 176: 870-880.
- Voisin T, Vellas B (2009) Diagnosis and treatment of patients with severe Alzheimer's disease. Drugs Aging 26: 135-144.
- Kent S (1979) Can drugs improve memory? Geriatrics 34: 77, 80, 83 passim.
- Crismon ML (1994) Tacrine: first drug approved for Alzheimer's disease. Ann Pharmacother 28: 744-751.
- Scott LJ, Goa KL (2000) Galantamine: a review of its use in Alzheimer's disease. Drugs 60: 1095-1122.
- Sramek JJ, Frackiewicz EJ, Cutler NR (2000) Review of the acetylcholinesterase inhibitor galanthamine. Expert Opin Investig Drugs 9: 2393-2402.
- Salvati S, Attorri L, Avellino C, Di Biase A, Sanchez M (2000) Diet, lipids and brain development. Dev Neurosci 22: 481-487.
- Bertram L, Tanzi RE (2008) Thirty years of Alzheimer's disease genetics: the implications of systematic meta-analyses. Nat Rev Neurosci 9: 768-778.
- Iqbal K, Liu F, Gong CX, Alonso Adel C, Grundke-Iqbal I (2009) Mechanisms of tau-induced neurodegeneration. Acta Neuropathol 118: 53-69.
- Reddy PH, Beal MF (2008) Amyloid beta, mitochondrial dysfunction and synaptic damage: implications for cognitive decline in aging and Alzheimer's disease. Trends Mol Med 14: 45-53.
- Dragicevic N, Mamcarz M, Zhu Y, Buzzeo R, Tan J, et al. (2010) Mitochondrial amyloid-beta levels are associated with the extent of mitochondrial dysfunction in different brain regions and the degree of cognitive impairment in Alzheimer's transgenic mice. J Alzheimers Dis 2: S535-550.
- Muller WE, Eckert A, Kurz C, Eckert GP, Leuner K (2010) Mitochondrial dysfunction: common final pathway in brain aging and Alzheimer's disease--therapeutic aspects. Mol Neurobiol 41: 159-171.
- Calkins MJ, Reddy PH 2011. Amyloid beta impairs mitochondrial anterograde transport and degenerates synapses in Alzheimer's disease neurons. Biochim Biophys Acta 1812: 507-513.
- Tucker RP (1990) The roles of microtubule-associated proteins in brain morphogenesis: a review. Brain Res Brain Res Rev 15: 101-120.
- Matus A (1990) Microtubule-associated proteins and the determination of neuronal form. J Physiol (Paris) 84: 134-137.
- Caceres A, Kosik KS (1990) Inhibition of neurite polarity by tau antisense oligonucleotides in primary cerebellar neurons. Nature 343: 461-463.
- Caceres A, Potrebic S, Kosik KS (1991) The effect of tau antisense oligonucleotides on neurite formation of cultured cerebellar macroneurons. J Neurosci 11: 1515-1523.
- Hanemaaijer R, Ginzburg I (1991) Involvement of mature tau isoforms in the stabilization of neurites in PC12 cells. J Neurosci Res 30: 163-171.
- Shea TB, Beermann ML, Nixon RA, Fischer I (1992) Microtubule-associated protein tau is required for axonal neurite elaboration by neuroblastoma cells. J Neurosci Res 32: 363-374.
- Aronov S, Marx R, Ginzburg I (1999) Identification of 3'UTR region implicated in tau mRNA stabilization in neuronal cells. J Mol Neurosci 12: 131-145.
- Aranda-Abreu GE, Behar L, Chung S, Furneaux H, Ginzburg I (1999) Embryonic lethal abnormal vision-like RNA-binding proteins regulate neurite outgrowth and tau expression in PC12 cells. J Neurosci 19: 6907-6917.
- Aronov S, Aranda G, Behar L, Ginzburg I (2001) Axonal tau mRNA localization coincides with tau protein in living neuronal cells and depends on axonal targeting signal. J Neurosci 21: 6577-6587.
- Aronov S, Aranda G, Behar L, Ginzburg I (2002) Visualization of translated tau protein in the axons of neuronal P19 cells and characterization of tau RNP granules. J Cell Sci 115: 3817-3827.
- Dubey M, Chaudhury P, Kabiru H, Shea TB (2008) Tau inhibits anterograde axonal transport and perturbs stability in growing axonal neurites in part by displacing kinesin cargo: neurofilaments attenuate tau-mediated neurite instability. Cell Motil Cytoskeleton 65: 89-99.
- Cowan CM, Bossing T, Page A, Shepherd D, Mudher A (2010) Soluble hyper-phosphorylated tau causes microtubule breakdown and functionally compromises normal tau in vivo. Acta Neuropathol 120: 593-604.
- Uchida T, Ishiguro K (1990) [Phosphorylation of tau protein]. Nippon Ronen Igakkai Zasshi 27: 280-286.
- Ginzburg I (1991) Neuronal polarity: targeting of microtubule components into axons and dendrites. Trends Biochem Sci 16: 257-261.
- Lippens G, Sillen A, Landrieu I, Amniai L, Sibille N, et al. (2007) Tau aggregation in Alzheimer's disease: what role for phosphorylation? Prion 1: 21-25.
- Thinakaran G, Koo EH (2008) Amyloid precursor protein trafficking, processing, and function. J Biol Chem 283: 29615-29619.
- Hung AY, Selkoe DJ (1994) Selective ectodomain phosphorylation and regulated cleavage of beta-amyloid precursor protein. EMBO J 13: 534-542.
- De Strooper B (2003) Aph-1, Pen-2, and Nicastrin with Presenilin generate an active gamma-Secretase complex. Neuron 38: 9-12.
- Selkoe DJ (2001) Alzheimer's disease: genes, proteins, and therapy. Physiol Rev 81: 741-766.
- Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 297: 353-356.
- Aizenstein HJ, Nebes RD, Saxton JA, Price JC, Mathis CA, et al. (2008) Frequent amyloid deposition without significant cognitive impairment among the elderly. Arch Neurol 65: 1509-1517.
- Fagan AM, Mintun MA, Mach RH, Lee SY, Dence CS, et al. (2006) Inverse relation between in vivo amyloid imaging load and cerebrospinal fluid Abeta42 in humans. Ann Neurol 59: 512-519.
- Pike KE, Savage G, Villemagne VL, Ng S, Moss SA, et al. (2007) Beta-amyloid imaging and memory in non-demented individuals: evidence for preclinical Alzheimer's disease. Brain 130: 2837-2844.
- Herrera-Rivero M, Aranda-Abreu GE (2011) Therapeutics of Alzheimer’s Disease. In Advanced Understanding Neurodegenerative Diseases, ed. RC-C Chang, 193-212, Rijeka, Croatia: Intech.
- Ballard CG (2002) Advances in the treatment of Alzheimer's disease: benefits of dual cholinesterase inhibition. Eur Neurol 47: 64-70.
- Poirier J (2002) Evidence that the clinical effects of cholinesterase inhibitors are related to potency and targeting of action. Int J Clin Pract Suppl 6-19.
- Stix G (2009) Turbocharging the brain. Sci Am 301: 46-55.
- Terwel D, Dewachter I, Van Leuven F (2002) Axonal transport, tau protein, and neurodegeneration in Alzheimer's disease. Neuromolecular Med 2: 151-165.
- Marx A, Nugoor C, Panneerselvam S, Mandelkow E (2010) Structure and function of polarity-inducing kinase family MARK/Par-1 within the branch of AMPK/Snf1-related kinases. FASEB J 24: 1637-1648.
- Wicklund L, Leao RN, Stromberg AM, Mousavi M, Hovatta O, et al. (2010) Beta-amyloid 1-42 oligomers impair function of human embryonic stem cell-derived forebrain cholinergic neurons. PLoS One 5: e15600.
- Henriques AG, Vieira SI, da Cruz E Silva EF, da Cruz E Silva OA (2010) Abeta promotes Alzheimer's disease-like cytoskeleton abnormalities with consequences to APP processing in neurons. J Neurochem 113: 761-771.
- Parsons CG, Stoffler A, Danysz W (2007) Memantine: a NMDA receptor antagonist that improves memory by restoration of homeostasis in the glutamatergic system--too little activation is bad, too much is even worse. Neuropharmacology 53: 699-723.
- Miguel-Hidalgo JJ, Alvarez XA, Cacabelos R, Quack G (2002) Neuroprotection by memantine against neurodegeneration induced by beta-amyloid(1-40). Brain Res 958: 210-221.
- Mobius HJ (1999) Pharmacologic rationale for memantine in chronic cerebral hypoperfusion, especially vascular dementia. Alzheimer Dis Assoc Disord 13 Suppl 3: S172-178.
- Jain KK (2000) Evaluation of memantine for neuroprotection in dementia. Expert Opin Investig Drugs 9: 1397-1406.
- Reisberg B, Doody R, Stoffler A, Schmitt F, Ferris S, et al. (2003) Memantine in moderate-to-severe Alzheimer's disease. N Engl J Med 348: 1333-1341.
- Bilbul M, Schipper HM (2011) Risk profiles of Alzheimer disease. Can J Neurol Sci 38: 580-592.
- Aranda-Abreu GE, Hernandez-Aguilar ME, Denes JM, Hernandez LI, Rivero MH (2011) Rehabilitating a brain with Alzheimer's: a proposal. Clin Interv Aging 6: 53-59.
- Beltz BS, Tlusty MF, Benton JL, Sandeman DC (2007) Omega-3 fatty acids upregulate adult neurogenesis. Neurosci Lett 415: 154-158.
- Bourre JM (2004) Roles of unsaturated fatty acids (especially omega-3 fatty acids) in the brain at various ages and during ageing. J Nutr Health Aging 8: 163-174.
- Valentine RC, Valentine DL (2004) Omega-3 fatty acids in cellular membranes: a unified concept. Prog Lipid Res 43: 383-402.
- Wu A, Ying Z, Gomez-Pinilla F (2004) Dietary omega-3 fatty acids normalize BDNF levels, reduce oxidative damage, and counteract learning disability after traumatic brain injury in rats. J Neurotrauma 21: 1457-1467.
- Daubert EA, Condron BG (2010) Serotonin: a regulator of neuronal morphology and circuitry. Trends Neurosci 33: 424-434.
- Caraci F, Molinaro G, Battaglia G, Giuffrida ML, Riozzi B, et al. (2011) Targeting group II metabotropic glutamate (mGlu) receptors for the treatment of psychosis associated with Alzheimer's disease: selective activation of mGlu2 receptors amplifies beta-amyloid toxicity in cultured neurons, whereas dual activation of mGlu2 and mGlu3 receptors is neuroprotective. Mol Pharmacol 79: 618-626.
- Mandrekar-Colucci S, Landreth GE (2011) Nuclear receptors as therapeutic targets for Alzheimer's disease. Expert Opin Ther Targets 15: 1085-1097.
- Hoozemans JJ, Veerhuis R, Rozemuller JM, Eikelenboom P (2011) Soothing the inflamed brain: effect of non-steroidal anti-inflammatory drugs on Alzheimer's disease pathology. CNS Neurol Disord Drug Targets 10: 57-67.
- Piau A, Nourhashemi F, Hein C, Caillaud C, Vellas B (2011) Progress in the development of new drugs in Alzheimer's disease. J Nutr Health Aging 15: 45-57.
- Bolognesi ML, Bartolini M, Tarozzi A, Morroni F, Lizzi F, et al. (2011) Multitargeted drugs discovery: balancing anti-amyloid and anticholinesterase capacity in a single chemical entity. Bioorg Med Chem Lett 21: 2655-2658.
- Ortega A, Rincon A, Jimenez-Aliaga KL, Bermejo-Bescos P, Martin-Aragon S, et al. (2011) Synthesis and evaluation of arylquinones as BACE1 inhibitors, beta-amyloid peptide aggregation inhibitors, and destabilizers of preformed beta-amyloid fibrils. Bioorg Med Chem Lett 21: 2183-2187.
- Connor WE, Neuringer M (1988) The effects of n-3 fatty acid deficiency and repletion upon the fatty acid composition and function of the brain and retina. Prog Clin Biol Res 282: 275-294.
- Stillwell W, Shaikh SR, Zerouga M, Siddiqui R, Wassall SR (2005) Docosahexaenoic acid affects cell signaling by altering lipid rafts. Reprod Nutr Dev 45: 559-579.
- Stetzkowski-Marden F, Recouvreur M, Camus G, Cartaud A, Marchand S, et al. (2006) Rafts are required for acetylcholine receptor clustering. J Mol Neurosci 30: 37-38.
- Martin V, Fabelo N, Santpere G, Puig B, Marin R, et al. (2010) Lipid alterations in lipid rafts from Alzheimer's disease human brain cortex. J Alzheimers Dis 19: 489-502.
- Karuppagounder SS, Pinto JT, Xu H, Chen HL, Beal MF, et al. (2009) Dietary supplementation with resveratrol reduces plaque pathology in a transgenic model of Alzheimer's disease. Neurochem Int 54: 111-118.
- Marambaud P, Zhao H, Davies P (2005) Resveratrol promotes clearance of Alzheimer's disease amyloid-beta peptides. J Biol Chem 280: 37377-37382.
- Vingtdeux V, Dreses-Werringloer U, Zhao H, Davies P, Marambaud P (2008) Therapeutic potential of resveratrol in Alzheimer's disease. BMC Neurosci 2: S6.
- Rossi L, Mazzitelli S, Arciello M, Capo CR, Rotilio G (2008) Benefits from dietary polyphenols for brain aging and Alzheimer's disease. Neurochem Res 33: 2390-2400.
- Janssen IM, Sturtz S, Skipka G, Zentner A, Velasco Garrido M, et al. (2010) Ginkgo biloba in Alzheimer's disease: a systematic review. Wien Med Wochenschr 160: 539-546.
- Weinmann S, Roll S, Schwarzbach C, Vauth C, Willich SN (2010) Effects of Ginkgo biloba in dementia: systematic review and meta-analysis. BMC Geriatr 10: 14.
- Wang JW, David DJ, Monckton JE, Battaglia F, Hen R (2008) Chronic fluoxetine stimulates maturation and synaptic plasticity of adult-born hippocampal granule cells. J Neurosci 28: 1374-1384.
- Lucas G, Du J, Romeas T, Mnie-Filali O, Haddjeri N, et al. (2010) Selective serotonin reuptake inhibitors potentiate the rapid antidepressant-like effects of serotonin4 receptor agonists in the rat. PLoS One 5: e9253.
- Scali C, Prosperi C, Vannucchi MG, Pepeu G, Casamenti F (2000) Brain inflammatory reaction in an animal model of neuronal degeneration and its modulation by an anti-inflammatory drug: implication in Alzheimer's disease. Eur J Neurosci 12: 1900-1912.
- Candelario-Jalil E, Alvarez D, Merino N, Leon OS (2003) Delayed treatment with nimesulide reduces measures of oxidative stress following global ischemic brain injury in gerbils. Neurosci Res 47: 245-253.
Relevant Topics
- Addiction Recovery
- Alcohol Addiction Treatment
- Alcohol Rehabilitation
- Amphetamine Addiction
- Amphetamine-Related Disorders
- Cocaine Addiction
- Cocaine-Related Disorders
- Computer Addiction Research
- Drug Addiction Treatment
- Drug Rehabilitation
- Facts About Alcoholism
- Food Addiction Research
- Heroin Addiction Treatment
- Holistic Addiction Treatment
- Hospital-Addiction Syndrome
- Morphine Addiction
- Munchausen Syndrome
- Neonatal Abstinence Syndrome
- Nutritional Suitability
- Opioid-Related Disorders
- Relapse prevention
- Substance-Related Disorders
Recommended Journals
Article Tools
Article Usage
- Total views: 15318
- [From(publication date):
specialissue-2013 - Nov 22, 2025] - Breakdown by view type
- HTML page views : 10516
- PDF downloads : 4802