Gold Laser Resection of the Concha Bullosa: Description of a New Technique
Received: 04-Mar-2012 / Accepted Date: 14-May-2012 / Published Date: 19-May-2012 DOI: 10.4172/2161-119X.1000114
251282Introduction
The concha bullosa, wherein the middle turbinate is pneumatized, represents the most common anatomic abnormality of the lateral nasal wall [1]. This congenital anomaly often results in an oversized middle turbinate which can predispose the patient to sinusitis. Subsequent results may include obstruction of normal sinus outflow tracts, polyposis within the turbinate, headaches from increased pressure within the pneumatized cavity of the turbinate, contact between mucosal surfaces, and pyocele of the middle turbinate secondary to retained secretions within the cavity [1-4].
Numerous methods have been described to surgically address concha bullosa, from crushing with endoscopic instruments, resection via marsupialization using combinations of blunt and sharp dissection or powered microdebriders, to laser treatment [5-7]. When considering Functional Endoscopic Sinus Surgery (FESS), the very small working space dictates that visibility and hemostasis are paramount, and this is also true of surgical management of concha bullosa. Other endoscopic methods may be effective in long-term disease control, but the ideal method will be both efficacious and facile for the surgeon, eliminating unnecessary intraoperative bleeding.
The use of lasers in endoscopic sinus surgery is not new; multiple lasers have been utilized with varying degrees of success. CO2, KTP- 532, argon laser, and pulsed diode laser have all been described in FESS for a variety of nasal complaints, each with its own limitations and successes [8-11]. The ideal laser for endoscopic sinus surgery is one that is able to ablate thin, vascular tissue of the nasal mucosa for hemostasis and polyp destruction, as well as relatively hypovascular bony tissue such as the concha bullosa or anterior ethmoid bulla. While a perfect laser for these applications remains elusive, the Lightforce Gold Laser (Medical Energy, Pensacola, Florida, USA) provides these features and, in our experience, is a facile tool for endoscopic sinus surgery and concha bullosa resection.
Methods
Retrospective chart review was undertaken of all patients having undergone Gold Laser resection of a concha bullosa by the senior author (N.K.W.) from 2007 – 2011. Indications for procedure, any concurrent surgical procedures, intraoperative and postoperative complications, and postoperative recurrent ipsilateral rhinosinusitis or headache symptoms were recorded.
The mean age of patients at the time of surgery was 52 years old (range: 15 to 77 years old) and mean follow-up time was 30 months (range: 1 to 51 months). Fourteen patients were female and 19 were male. Indications for concha bullosa resection, as well as any concurrent rhinologic pathologies, are given in Table 1. The most common indications were chronic ipsilateral maxillary sinusitis (31/33 patients) and nasal obstruction (29/33 patients), while other indications included recurrent ipsilateral acute sinusitis, headache, nasal polyposis, nasal mass, allergic fungal sinusitis and mucocele. Multiple indications were present in 22/33 patients. The most common concurrent non-surgical rhinologic diagnosis was allergic rhinitis (10/33 patients), with others including obstructive sleep apnea and rhinitis medicamentosa.
| Indication for resection | Patients No. (%) |
|---|---|
| Chronic ipsilateral maxillary sinusitis | 31 (93.9) |
| Ipsilateral nasal obstruction | 29 (87.9) |
| Ipsilateral nasal polyposis | 10 (30.3) |
| Recurrent ipsilateral acute sinusitis | 2 (0.06) |
| Allergic fungal sinusitis | 1 (0.03) |
| Mucocele | 1 (0.03) |
| Nasal mass | 1 (0.03) |
Table 1: Indications for concha bullosa resection. Percentages will not add up to 100 due to multiple patients presenting with multiple indications.
Concurrent rhinologic surgeries of the study patients are given in Table 2. Concurrent rhinologic surgery was performed in 32 of 33 patients, with septoplasty (29/33 patients) and ipsilateral medial maxillary antrostomy (28/33 patients) being the most common procedures performed. Submucosal resection of the inferior turbinate, anterior ethmoidectomy, total ethmoidectomy, sphenoidotomy, and frontal recess exploration were also performed.
| Concurrent rhinologic surgery | Patients No. (%) |
|---|---|
| Septoplasty | 29 (87.9) |
| Medial maxillary antrostomy | 28 (84.8) |
| Anterior ethmoidectomy | 18 (54.5) |
| Submucosal resection of inferior turbinates | 14 (42.4) |
| Total ethmoidectomy | 5 (15.1) |
| Bilateral concha bullosa resection | 3 (0.09) |
| Frontal recess exploration | 1 (0.03) |
| Sphenoidotomy | 1 (0.03) |
Table 2: Concurrent rhinologic surgery. Percentages will not equal 100% due to patients undergoing multiple concurrent procedures.
Operative technique
After obtaining informed consent from the patient, general endotracheal anesthesia was induced, and the nose was prepped with betadine into a sterile field. A mixture of 1% lidocaine with 1:100,000 U epinephrine was injected into the nasal mucosa along the ipsilateral nasal septum and head of the inferior turbinate, as well as into the anterior face of the concha bullosa. Cotton balls soaked with 4% cocaine were then placed into the nostril, and at least 5 minutes was allowed to elapse for these agents to take effect before proceeding with surgery. The cotton balls were then removed, or pushed farther into the nasopharynx to minimize secretions or irrigation fluid entering the oropharynx. Utilizing a 0o rigid nasal endoscope, any blood and secretions were removed from the nostril with a Frazier-tipped suction.
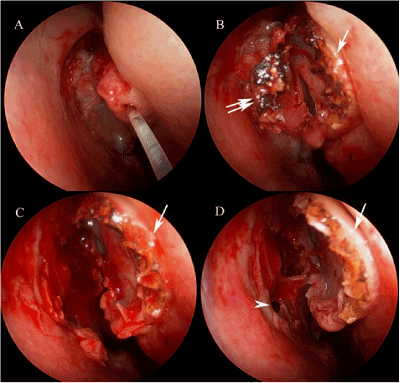
The Lightforce Gold Laser (Medical Energy, Inc., Pensacola, FL, USA) hand piece with chisel tipped fiber (Figure 1) was then inserted into the nostril under endoscopic visualization. The anterior mucosal surface of the concha bullosa was incised using the gold laser, at a setting of 6 W, from superior to inferior in a sagittal plane corresponding to the midpoint of the concha bullosa. The laser was then used to incise the underlying bone of the concha bullosa via the mucosal incision. Endoscopic scissors were then used to complete the superior and inferior bony cuts, with one scissor jaw outside the concha bullosa and the other in the cavity. This fully separates the lateral hemisphere of the concha bullosa from the medial hemisphere, which remains attached to the nasal wall via the root of the middle turbinate. This free-floating lateral hemisphere was then removed from the nose with Takahashi forceps. The gold laser was used to ensure hemostasis, though this was rarely needed. A small Merocel (Medtronic, Inc., Jacksonville, FL, USA) sponge was placed between the marsupialized concha bullosa and lateral nasal wall and ostiomeatal complex to prevent scarring or synechiae.
The patients were instructed to use nasal saline sprays four times daily beginning postoperative day two, and were placed on prophylactic antibiotics with anti-Staphylococcal activity to prevent toxic shock syndrome whilst the Merocel was in place. Any remaining Merocel was removed in the clinic on postoperative day five
Results
Thirty-two of 33 patients had an uneventful early postoperative course. One patient developed postoperative ipsilateral epistaxis, treated successfully with anterior nasal packing for 48 hours, after which symptoms resolved. No blood or blood product transfusion was required. This patient had no personal or family history of bleeding diatheses, yet was determined to have hemophilia B via hematologic workup subsequent to this postoperative bleed. Two additional patients developed recurrent ipsilateral maxillary rhinosinusitis in the early postoperative period (within the first 30 days postoperatively). Both reported suboptimal compliance with their nasal saline rinses and had significant residual Merocel in place at the postoperative visit that was unable to be completely removed in the clinic. Both patients were treated uneventfully with oral antibiotics and had good long-term outcomes.
Seven patients developed recurrent ipsilateral maxillary rhinosinusitis greater than one month postoperatively, with mean time to onset 14.3 months (range: 4 – 48 months). All were treated uneventfully with oral antibiotics, and had only one recurrent episode of sinusitis in the study period.
Four patients developed recurrent polyposis greater than one month postoperatively, with mean time to onset 30 months (range: 18 – 48 months). All patients with recurrence reported non-compliance with their prescribed topical nasal steroid sprays and oral antihistamine regimens for at least six months prior to recurrence. Three of four patients were treated successfully with one-week oral prednisone taper and re-institution of topical nasal steroids and oral antihistamines. One patient eventually required repeat FESS for recurrent nasal polyps on the contralateral side from the concha bullosa resection. This patient had undergone prior FESS three times for allergic fungal sinusitis and nasal polyposis, and had an endoscopic frontal sinus mucocele resection at the time of concha bullosa treatment. With reinstatement of topical nasal steroids and oral antihistamines, the patient is doing well one year after his revision FESS for contralateral disease.
In all patients there were no instances of cerebrospinal fluid leakage, ocular or orbital injury, anosmia, synechiae, exposed bone intranasally, lateralization of the middle turbinate, persistent ipsilateral headaches or nasal obstruction postoperatively.
Discussion
There have been many technological advances in recent years in the field of FESS. Among these are the development of smaller, lighter endoscopes and cameras, the advent of high-definition cameras and monitors, and a remarkable variety of endoscopic surgical instruments. Concha bullosa resection, which is frequently performed in conjunction with other endoscopic surgical procedures, has undoubtedly benefited from some of these advances as well. Paramount to successful and safe endoscopic sinus surgery is visibility, which is achieved partly through the use of cameras and monitors capable of delivering high-quality images, but also through surgical technique and surgical instruments capable of maintaining hemostasis.
Preoperative and intraoperative use of topical and injected vasoconstrictive agents has long been the backbone of hemostatic maintenance in FESS, and remains an integral tool. In addition, minimizing trauma, both intentional surgical trauma as well as inadvertent trauma incurred via repetitive withdrawal and reinsertion of multiple surgical instruments, is necessary to achieve this aim. While some authors have described novel methods of handling multiple instruments simultaneously within the nose [12], another technique used to reduce such inadvertent mucosal trauma is to utilize a single instrument capable of performing multiple tasks.
The Lightforce Gold Laser hand piece incorporates a suction tip, as well as the flexible quartz laser fiber, thus allowing this single instrument to cut soft tissue and thin bone, coagulate bleeding vessels, and evacuate smoke, blood, secretions, or irrigation fluid from the operative field without changing instruments. The 980 nm wavelength of the Indium-Gallium-Arsenide Phosphate III laser contained within the Lightforce Gold unit has a reported burn depth of 0.3 mm [13], and can be used in two ways. Placing the tip of the laser fiber in contact with tissue allows precise cutting of vascular nasal mucosal surfaces whilst coagulating the small vessels within the tissue as it cuts, avoiding bleeding. The chisel shape of the fiber tip allows the surgeon, by gently increasing applied pressure with the instrument, to puncture thin bony lamina, such as concha bullosa or ethmoidal cells, while maintaining the same hemostatic ability. The requirement of a subtle, but conscious, increase in force by the operating surgeon makes it very difficult to unintentionally puncture even thin bone. Withdrawal of the tip of the laser fiber 1-2 mm away from the tissue surface defocuses the beam to an area of approximately 2 mm, producing a superficial coagulating effect to control any unexpected bleeding. The shallow depth of burn injury of the Lightforce Gold Laser and precision handling by the operative surgeon both obviate the thick, fibrinous exudate formation in the postoperative healing period reported by Kennedy, and other authors [14,15].
The ability to maintain a virtually bloodless field not only increases the safety and decreases the technical difficulty of the procedure, but increases patient comfort postoperatively as well. Only a small amount of absorbable packing material is left in the nasal cavity, and postoperative nausea is less likely as there is almost no blood to be inadvertently swallowed intraoperatively by the patient.
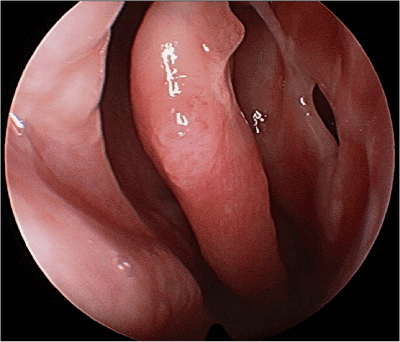
The use of the Lightforce Gold Laser in concha bullosa resection provides a simple, facile method to achieve long-term successful results (Figure 2), and is a further addition to the rhinologic surgeon's armamentarium.
References
- Peric A, Baletic N, Sotirovic J (2010) A case of an uncommon anatomic variation of the middle turbinate associated with headache. Acta Otorhinolaryngol Ital 30: 156-159.
- Erkan AN, Canbolat T, Ozer C, Yilmaz I, Ozluoglu LN (2006) Polyp in concha bullosa: a case report and review of the literature. Head Face Med 2: 11.
- Okuyucu S, Akoglu E, Dagli AS (2008) Concha bullosa pyocele. Eur Arch Otorhinolaryngol 265: 373-375.
- Smith KD, Edwards PC, Saini TS, Norton NS (2010) The prevalence of concha bullosa and nasal septal deviation and their relationship to maxillary sinusitis by volumetric tomography. Int J Dent.
- Peynegre R, Bossard B, Koskas G, Borsik M, Bouaziz A, et al. (1989) [Endoscopic surgery of the turbinates: Preliminary results]. Ann Otolaryngol Chir Cervicofac 106: 537-540.
- Kieff DA, Busaba NY (2009) Reformation of concha bullosa following treatment by crushing surgical technique: Implication for balloon sinuplasty. Laryngoscope 119: 2454-2456.
- Christmas DA Jr, Mirante JP, Yanagisawa E (2007) Endoscopic view of a powered partial resection of a concha bullosa. Ear Nose Throat J 86: 132-133.
- Gerlinger I, Lujber L, Jarai T, Pytel J (2003) KTP-532 laser-assisted endoscopic nasal sinus surgery. Clin Otolaryngol Allied Sci 28: 67-71.
- Johnson LP (1990) Nasal and paranasal application of lasers. In: Davis, R.K., ed. Lasers in Otolaryngology Head & Neck Surgery. Philadelphia, PA: Saunders: 145-155.
- Parkin JL, Dixon JA (1985) Argon laser treatment of head and neck vascular lesions. Otolaryngol Head Neck Surg 93: 211-216.
- Ohyama M (1988) Flexible fiberoptic applications of the Nd:YAG laser: Applications in chronic sinusitis. In: Joffe, S.N., Oguro, Y., eds. Advances in Nd:YAG Laser Surgery. New York, NY: Springer-Verlag 166-169.
- Manickavasagam J, Segaram S, Harkness P (2010) Functional endoscopic sinus surgery chopstick technique. Laryngoscope 120: 975-977.
- Ida JB, Worley NK, Amedee RA (2009) Gold laser adenoidectomy: long-term safety and efficacy results. Int J Pediatr Otorhinolaryngol 73: 829-831.
- Kennedy DW (2006) Technical innovations and the evolution of endoscopic sinus surgery. Ann Otol Rhinol Laryngol Suppl 196: 3-12.
- Knappe V, Frank F, Rohde E (2004) Principles of lasers and biophotonic effects. Photomed Laser Surg 22: 411-417.
Citation: Winters R, Worley NK (2012) Gold Laser Resection of the Concha Bullosa: Description of a New Technique. Otolaryngology 2:114. DOI: 10.4172/2161-119X.1000114
Copyright: © 2012 Winters R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 18150
- [From(publication date): 6-2012 - Oct 10, 2025]
- Breakdown by view type
- HTML page views: 13462
- PDF downloads: 4688