Research Article Open Access
Ground Water Ingestion Dose Due to Intake of Radionuclide (Natural U and 226Ra) to Population Around Uranium Mining Complex at Jaduguda
N. K. Sethy1*, V. N. Jha1, S. K. Sahoo2, A. K. Shukla1, R. M. Tripathi1 and V. D. Puranik1
1Environmental Survey Laboratory, Health Physics Unit, Jaduguda Mines, Jharkhand-832102
2Environmental Assessment Division, Bhabha Atomic Research Centre, Mumbai-400085, India
- *Corresponding Author:
- Dr. N.K. Sethy
Environmental Survey Laboratory
Health Physics Unit
Jaduguda Mines
Jharkhand-832102
E-mail: sethybarc@rediffmail.com
Received Date: December 17, 2010; Accepted Date: February 22, 2011; Published Date: February 25, 2011
Citation: Sethy NK, Jha VN, Sahoo SK, Shukla AK, Tripathi RM, et al. (2011) Ground Water Ingestion Dose Due to Intake of Radionuclide (Natural u and 226ra) to Population Around Uranium Mining Complex at Jaduguda. J Ecosys Ecograph 1:104. doi:10.4172/2157-7625.1000104
Copyright: © 2011 Sethy NK, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
Monitoring of ground water for naturally occurring radionuclide around a uranium ore processing industry is an integral part of long term environmental surveillance program. Ground water around the uranium mining complex at various distances are collected and analyzed for natural occurring radionuclide. Concentration of uranium and 226Ra in ground water were found to be very low and present in background level. The subsequent annual ingestion dose to the members of public within 1.5km of the uranium industry is less than 1% of the stipulated dose limit by ICRP. Statistical analysis of uranium concentration during the ground water monitoring program from the period 2003- 2007 is found to be not following either normal or lognormal distribution. Concentration of 226Ra was log-normally distributed for the same period. Ground water ingestion dose due to intake of dissolved radionuclide (U and 226Ra) insignificant and less than 1% of the stipulated dose limit for members of public (1mSv).
Keywords
Ground Water; Uranium; 226Ra; Dose
Introduction
Ground water still remains a major source of drinking water in many parts of our country (India). Rapid industrialization and subsequent waste disposal has been a concern for ground water contamination. Unlike any other conventional industry uranium mining and ore processing has a novel way of disposing low level radioactive waste in a continuous surveillance engineered impoundment system popularly known as tailings pond (TP). Tailings in the form of slurry is the waste product of uranium industry containing radionuclide such as U(nat), 226Ra, 230Th, 210Po and other heavy metals and anions such as SO4 =, Cl-, hardness etc. is stored in the tailings pond for years. Seepage of contaminants from tailings pond to public domain through underground water table cannot be ruled out. Therefore, continuous monitoring of ground water around the tailings pond is an essential part of long term environmental monitoring program around a uranium mining and ore processing industry. Mining and processing of uranium in the east-Singbhum region of Jharkhand has been going on for last four decades. Uranium ore is mined from a cluster of underground and open cast mines spreading in the region and processed at centralized processing plant in Jaduguda using sulphuric acid based hydrometallurgy technique. Recovery of uranium from low grade ore leads to the generation of large quantity of waste i.e. tailings. The tailings slurry which is inherently acidic in nature is neutralized and classified into fine (65%) and coarse (35%) fraction. The course fraction is send back to mine for back filling where as in fine fraction the pH is raised to > 9.5 and finally discharged in to a engineered impoundment system called tailings pond. At the tailings pond the settling of solid takes place slowly and the supernatants liquid effluent is decanted out for further removal of dissolved toxins (226Ra, Mn and U). The activity of 210Po in the decanted effluent from tailings pond is appreciably low which attributed to the physicochemical characteristics of the environment and the related geochemistry [1]. Besides uranium ore processing the whispered rich mineralization of the Singbhum thrust belt (STB) region also host to a number of other mineral industry such as copper, and Iron [2].
In order to monitor the environment around the uranium facility, Environmental Survey Laboratory has been setup by Bhabha Atomic Research Centre, which routinely monitoring the environment for last four decades. Monitoring of ground water, surface water, effluents, soil, sediments, dietary items, vegetation etc. is a part of the integrated environmental surveillance program. Radionuclides present in drinking water are significant contributor of natural radiation exposure. The presence of radionuclide in ground water depends on several factors like rock and soil characteristics, biochemical interactions of the radionuclide with environmental components, aquifer properties, geographical conditions etc. Elevated levels can be expected in areas known for radioactive mineral deposits. Apart from this industrialization may also increase the levels particularly involving mining and processing of radioactive ore, fly ash generation, and phosphate fertilizers etc. Low concentration of this radionuclide is invariably present in most of the environmental media including drinking water. Elevated concentration may occur either naturally in uranium mineralized areas if solubility conditions are favorable or near the uranium ore processing sites if the discharge methodology is not adequate enough in restricting the radionuclide within a confined area. Monitoring of the adjoining ground water sources as well as sources far away from the uranium industry has been carried out on routine basis. The objective was to evaluate the public exposure due to intake of U(nat) and 226Ra through ground water from natural sources and the risk for population residing around uranium industry.
Materials and Methods
Ground water samples were collected from different drinking water sources around tailings ponds as well as away from tailings pond. Water samples are also collected from different bore wells situated in the vicinity of tailings pond. These bore wells are specially designed to trace the migration of contaminants from tailings pond to public domain and known as monitoring wells. Grab ground water samples (5lit) were collected from wells and tube wells situated at various distances in the public domain. Samples were brought to the laboratory, filtered immediately using Whatman-42/Millipore (0.25µm) filter paper and preserved in acidic medium. The details of sample collection and processing are given elsewhere [2-5]. Natural uranium was estimated by a combined method of solvent extraction followed by flurometry. About 100ml of water is evaporated to dryness and 20ml of 0.25N electronic grade pure H2SO4 is added and reflux for 30 minutes in a hot plate. It is then cooled and transferred to a separating funnel/tube. 20ml of alamine-benzene (2% alamine in 98% benzene) solution is added and the mixture and shaken for few minutes with occasionally opening the mouth of the separating tube to vent off the gases formed inside. The aqueous phase is discarded out and 0.1ml from organic phase is transferred to a platinum planchate. A small volume (0.1ml) of organic media containing the uranium is transferred to platinum disc, fused with 250 mg of NaF- Na2 CO3 (15:85) fusion mixture at 800°C for 3 minutes. Cooled and fluorescence intensity was measured in ECIL, make Fluorimeter [6]. Ultraviolet excitation wavelength of 3650A is irradiated on the platinum disc containing fused sample and emitted florescence of 5546A°wavelength is unique to uranium [7]. Intensity of fluorescence is proportional to the amount of uranium present in the sample. Standard (1µg/ml) and blank were processed simultaneously and uranium was estimated by using the formula

The uranium content of the original sample was obtained from the above equation by further applying the sampling parameters. The detection limit of this method is 0.1µg.
226Ra in water samples was estimated by emanometric method [8]. Due to very low level concentration of 226Ra observed in ground water, 2 liter of water sample was pre-concentrated to 100ml and loaded in a standard airtight bubbler for a known period of time. During the buildup period 226Ra decays and form 222Rn gas inside the bubbler which was transferred to a Lucas cell and alpha counted for estimation of 226Ra. 226Ra is estimated by allowing build up of its daughter 222Rn for a known period (nearly 4 weeks). The inbuilt radon was collected in a scintillation cell by vacuum transfer and counted after equilibrium (between radon & its progeny) is attended [8-11]. Leached sample aliquot (50ml) or concentrated water sample was loaded in radon bubbler. The radon already present in the solution was removed by using evacuation pump. After ensuring the radon free aliquot, the solution was allowed to retain for two weeks or more depending on the expected level of radium in the sample. During this period, 226Ra through alpha decay leads to the formation of its progeny 222Rn. The inbuilt radon was collected in a previously evacuated scintillation cell. The scintillation cell was left for minimum 200 minutes for 222Rn and its progeny equilibrium. The alpha counts can be measured at any time after that. The scintillation cell was coupled to photo multiplier tube. Alpha counts were recorded for a desired period in order to get counts above 95% confidence level. From the counts obtained, the radon activity transferred from the bubbler to the cell was calculated using the equation.
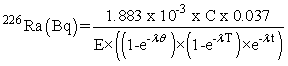
C = is the net counts obtained after subtraction of the background
E = Efficiency of the cell (85%)
t = counting delay in minutes
T = counting duration in minutes
θ = Build up period in minutes
λ = Decay constant of 222 Rn (1.258 x 10-4 min -1)
The minimum detectable 226Ra activity in the sample solution loaded in radon bubbler is affected by many factors like buildup period, efficiency and background count rate of the Lucas cell, counting duration etc. Considering the background of the Lucas cell as 0.5cpm and efficiency 85% the minimum detectable activity estimated to be 6.8mBq when maximum buildup period is allowed [9].
Result and Discussions
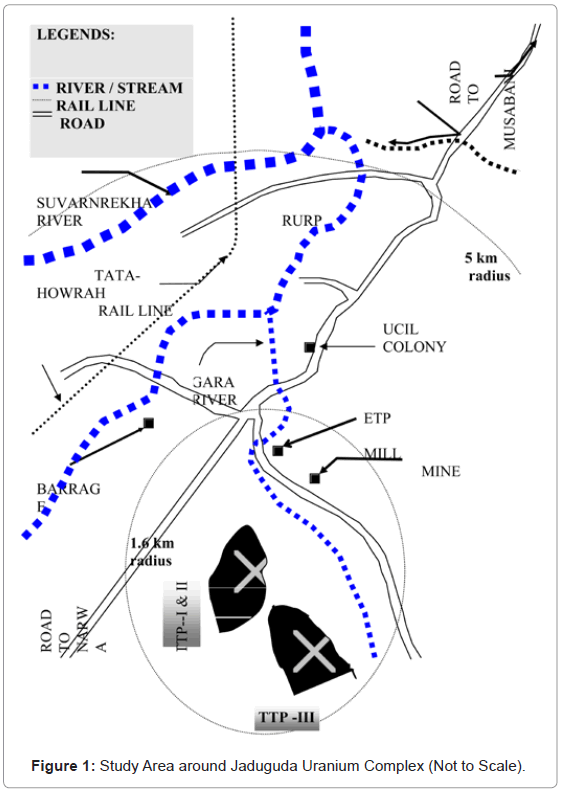
There are three tailings ponds at Jaduguda out of which two are saturated and abandoned where as one is in operation. Ground water samples from different distances from tailings pond around the uranium mining complex were collected. The sampling area was mainly divided in to three distance zones i.e. less than 1.6 km, 1.6 to 5km and > 5km form the uranium facility. Most of the sampling locations were selected in the downstream direction of ground water flow and with respect to tailings discharge. The study area is presented in Figure 1. Last five year (2003-2007) analytical result of ground water for U (nat) and 226Ra are presented below in Table 1 and 2.
| Distances from T.P (km) | Year | U(nat) (mg.m-3) | |
|---|---|---|---|
| 2003 | Range | Mean | |
| < 1.6 | 0.5-11(8) | 2.8 | |
| 1.6 - 5.0 | 0.5-20(20) | 2.9 | |
| > 5.0 | 0.5-4.7(20) | 1.6 | |
| 2004 | |||
| < 1.6 | 0.5-7.6 (15) | 2.4 | |
| 1.6 - 5.0 | 0.5-19 (15) | 4.8 | |
| > 5.0 | 0.5-4.3 (10) | 1.7 | |
| 2005 | |||
| < 1.6 | 0.5-7.6(16) | 2.3 | |
| 1.6 - 5.0 | 0.5-19 (19) | 4.6 | |
| > 5.0 | 0.5-4.3 (11) | 1.9 | |
| 2006 | |||
| < 1.6 | 0.5-2.3 (5) | 1.1 | |
| 1.6 - 5.0 | 0.5-1.2 (4) | 0.80 | |
| > 5.0 | 0.5-33.3 (17) | 4.2 | |
| 2007 | |||
| < 1.6 | 0.5-1.5 (15) | 0.8 | |
| 1.6 - 5.0 | 0.5-3.1 (9) | 1.3 | |
| > 5.0 | 0.5-3.5 (10) | 1.0 | |
*Figures in parentheses are the numbers of samples
Table 1: Concentration of U (nat) in ground water samples in various distance zones.
| Distances from T.P (km) | Year | 226Ra (Bq.m-3) | |
|---|---|---|---|
| 2003 | Range | Mean | |
| < 1.6 | 3.5-20 (18) | 7.5 | |
| 1.6 - 5.0 | 3.5-72.0(20) | 15.6 | |
| > 5.0 | 3.5-65.3(20) | 12.1 | |
| < 1.6 | 2004 | 3.5-41.4(15) | 11.0 |
| 1.6 - 5.0 | 3.5-29 (17) | 7.5 | |
| > 5.0 | 3.5-25 (11) | 11.5 | |
| < 1.6 | 2005 | 3.5-41.4 (16) | 12.6 |
| 1.6 - 5.0 | 3.5-29 (19) | 7.9 | |
| > 5.0 | 3.5-41.4(10) | 16.3 | |
| < 1.6 | 2006 | 4.0-16.0 (6) | 6.5 |
| 1.6 - 5.0 | 3.5-53 (13) | 9.2 | |
| > 5.0 | 3.5-95 (10) | 22.5 | |
| < 1.6 | 2007 | 3.5-26 (10) | 7.4 |
| 1.6 - 5.0 | 3.5-6.0 (14) | 4.0 | |
| > 5.0 | 3.5- 19.3 (10) | 6.6 | |
*Figures in parentheses are the numbers of samples
Table 2: Concentration of 226Ra in ground water samples in various distance zones.
Natural uranium in ground water
Toxicity of uranium has been established by animal studies and human data from uranium miners and workers with accidental exposures indicate that uranium affects the proximal tubules of the kidney; at very high acute doses, tubular degeneration and necrosis (that is, death of tissue) may occur a few days after the intake of uranium [12]. Kidney is critical organ for uranium toxicity [13,14]. The uranyl ion forms bicarbonate, citrate and UO3 (CO2)3 complexes in blood plasma [15]. The UO2 ++ ion binds with the red blood cells. During the purification of blood in kidney, it is filtered from the blood and then recombines with the cell surface ligands. Studies on uranium toxicity studies in human have been described elsewhere [16,17]. Though, intake of uranium by members of the public can occur through various routes. However the principal route of ingestion of uranium is through drinking water [18] and to a lesser extent through the foodstuff. Intake of uranium through drinking water by population residing around the uranium mining area has been considered in the present study. United States Environmental Protection Agency [19] has classified uranium as a group- A human carcinogen. It has prescribed maximum contaminant level goal (MCLG) for uranium as 0 (zero) in 1991 (zero tolerance). In drinking water, EPA suggests maximum contaminant level as (MCL) of 30µg.l-1 [21]. In Canada, the proposed interim maximum acceptable level is (IMAC) of 20µg.l-1, whereas World Health Organization [21] strictly recommended a reference level as 2µg.l-1 [22].
Concentration of uranium natural found in the ground water around Jaduguda is summarized in Table 1. The < 1.6km distance zone is very close to the tailings pond. Concentration of natural uranium in ground water samples was varied from 0.5 to 11mg.m-3 with a mean concentration of 2.8mg.m-3 in the year 2003. The mean concentrations of uranium in the following years were 2.4, 2.3, 1.1 and 0.8mg.m-3 in 2004, 2005, 2006 and 2007 respectively in the same distance zones. The second sector which starts at 1.5km away from tailings Pond and stretches up to 5km. Concentration of natural uranium in ground water samples in this zone was varied from 0.5 to 20mg.m-3 with a mean concentration of 2.9mg.m-3 in the year 2003. The mean concentrations of uranium in 1.5-5km distance zone in the following years were 4.8, 4.6, 0.80 and 1.3 mg.m-3 in the year 2004, 2005, 2006 and 2007 respectively. The last distance zone is > 5.0 km and situated far away from tailings pond. Concentration of natural uranium in ground water samples was varied from 0.5 to 4.7mg.m-3 with a mean concentration of 1.6mg.m-3 in the year 2003. The maximum mean concentrations of uranium in same distance zone was 4.2 1.0mg.m-3 in the year 2006.
226Ra in ground water
Radium is a naturally-occurring radioactive metal. Its most common isotopes are 226Ra, 224Ra, and 228Ra. It occurs at low levels in virtually all rock, soil, water, plants, and animals [23]. Inhaled or ingested radium increases the risk of developing such diseases as lymphoma, bone cancer, and diseases that affect the formation of blood, such as leukemia and aplastic anemia [23]. Absorbed radium behaves similarly to calcium and is deposited in the tissues of the body, especially bone [24]. In ground water it can arise from natural sources, resulting from interaction of ground water with radium bearing materials like rocks, soil, ore bodies etc. It can also arise indirectly from man’s exploration of radioactive minerals particularly of uranium. In the case of natural unmined ore deposits radium can enter the ground water by way of leaching of the radium bearing rock strata by the ground water aquifers. Elevated concentration may occur either naturally in uranium mineralized areas, if solubility conditions are favorable or near the uranium ore processing /waste disposal sites if the discharge methodology is not adequate enough in restricting the radionuclide within a confined area. Radium can enter ground water by direct seepage of the effluent from the waste depository. Earlier studies indicate there are several spring wells in the valley of uranium mineralized hills of this region with reportedly higher concentration of 226Ra [25,26].
Distribution of Radium in ground water is presented in the Table 2. Within < 1.6 km, concentration of 226Ra in ground water samples was varied from 3.5 to 20Bq.m-3 with a mean concentration of 7.5Bq.m-3 in the year 2003. The mean concentrations of 226Ra in the same distance zone in the following years were 11.0, 12.6, 6.5 and 7.4Bq.m-3 in 2004, 2005, 2006 and 2007 respectively. The second sector which starts at 1.5 km away from tailings Pond and stretches up to 5km. Concentration of 226Ra in ground water samples was varied from 3.5 to 72Bq.m-3 with a mean concentration 15.6Bq.m-3 in the year 2003. The mean concentrations of radium in the following years in the same distance area were 7.6, 7.9, 9.2 and 4.0Bq.m-3 in 2004, 2005 2006 and 2007 respectively. The last distance zone is > 5.0 km and situated far away from tailings pond and concentration of 226Ra in ground water samples was varied from 3.5 to 65.3Bq.m-3 with a mean concentration of 12.0Bq.m-3 in the year 2003. The maximum (22.5Bq.m-3 ) mean concentrations of uranium was observed in the year 2006.
Intake and dose assessment
Data generated over the years for natural uranium in ground water at various distance zones are summarized in Table 1. Statistical analysis of raw data is presented in Table 3. It is evident from Table 2 that, natural uranium is not following either normal or lognormal distribution. Natural uranium in ground water was observed in low concentration range. Though mass concentration of natural uranium was measured in ground water samples it can be expressed in activity concentration using conversion factor of 25Bq.m-1. Uranium level (Bq.m-3) = Measured mass concentration (mg.m-3) x Conversion factor (25Bq.m-1). Considering the daily intake of water for Indian reference man 4.05l.d-1 [27,28], dose conversion factor of 0.045µSv.Bq-1 (ICRP 68) the annual ingestion dose to the adult individual residing around the tailings pond due to natural uranium was estimated. Since the data distribution in the three zones can be approximated by log normal the intake and ingestion dose should be based on geometric mean. Accordingly the annual intake from drinking water in the three zones discussed can be estimated to be 41.8, 44.4 and 41.8Bq.Y-1 with an ingestion dose of 1.88, 2.0 and 1.88µSv.Y-1 respectively. This is far lower than the WHO (2003) recommended guideline of 100µSv.Y-1 for ingestion from the intake of a single radionuclide.
| Distance from TP | N | 226Ra (Bq.m-3) | |
|---|---|---|---|
| GM | GSD | ||
| < 1.5km | 58 | 5.7 | 2.0 |
| 1.5-5km | 67 | 6.3 | 2.3 |
| > 5km | 67 | 7.9 | 2.3 |
| Distance from TP | N | U (mg.m-3) | |
| GM | GSD | ||
| < 1.5km | 58 | 1.1 | 2.6 |
| 1.5-5km | 59 | 1.6 | 3.1 |
| > 5km | 68 | 1.1 | 2.6 |
Table 3: Overall Summary of Natural Uranium and 226Ra concentration in Ground water (2003-2007).
Statistical summary of concentration data of radium (2003-2007) is presented in Table 3. The radium found to be log normally distributed in all distance zones. Geometric mean values are taken for calculation of ingestion dose due to 226Ra (Table 5). The USEPA [19] have given a Maximum Contamination Level (MCL) of 185mBq.l-1 (226Ra and 228Ra combined) based on the updated dosimetry and risk considerations [29,30]. The 226Ra concentration found to be much lower than MCL. Samples from ground water sources in the vicinity of tailings pond were found to have comparatively lower concentration of radionuclide as compared to the areas away from tailings pond.
Conclusions
Concentration of U and 226Ra in ground water around the mining and ore processing industry is found to be very low. The low concentration of radionuclide in ground water represents the natural background of the area. Highest ground water ingestion dose (Table 4) due to intake of dissolved U is 27.2µSv.y-1 (95th percentile) in the 1.5-5km distance from the uranium industry. Similarly the highest ingestion dose due to 226Ra (Table 5) is 3.3µSv.y-1 is beyond 5km from uranium complex. However, within 1.5km of the uranium mining complex the ingestion dose due to both U and 226Ra is less than 1% of the limit for the members of public (1000µSv).
| Percentile | U(mg.m-3) < 1.5km | Dose mSv/y | U(mg.m-3) 1.5 - 5km | Dose mSv/y | U(mg.m-3) >5km | Dose mSv/y |
|---|---|---|---|---|---|---|
| 50th | 0.5 | 0.83 | 0.5 | 0.83 | 0.5 | 0.83 |
| 95 th | 6.34 | 10.5 | 16.3 | 27.2 | 4.6 | 7.7 |
Table 4: Annual Ground water Ingestion Dose (µSv) due to Natural Uranium (2003-07).
| Distance from TP | GM conc. Of 226Ra (Bq.m-3) | Annual Dose mSv/y |
|---|---|---|
| < 1.5km | 5.7 | 2.3 |
| 1.5-5km | 6.3 | 2.6 |
| > 5km | 7.9 | 3.3 |
Table 5: Annual Ground water Ingestion Dose (μSv) due to 226Ra (2003-07).
Acknowledgements
Thanks are due to Shri H.S. Kushwaha, Ex Director, Health, Safety and Environment group, BARC, for his support and encouragement during this study. Suggestions received from colleagues of Health Physics Unit, Jaduguda, are highly acknowledged. Thanks are due to the Uranium Corporation of India Limited, for facilities and assistance provided during this investigation.
References
- The Radiochemistry of Polonium (1961), National Academy of Sciences, National Research Council, Nuclear Science Series, USA.
- Sahoo SK, Mohapatra S, Sethy NK, Patra AC, Shukla AK et al. (2010) Natural Radioactivity in Roadside Soil along Jamshedpur-Musabani Road: A Mineralised and Mining Region, Jharkhand and Associated Risk. Radiat Prot Dosimetry 140: 281-286.
- Tripathi RM, Radha R, Sastry VN, Krisnamurty TM (1997) Dietary intake of managanese b the adult population of Mumbai. Sci. Total Environ 208: 149- 159.
- Tripathi RM, Mishra S, Radha R, Sastry VN, Krisnamurty TM (2000) Daily intake of Managanese by the adult population of Mumbai. Sci. Total Environ 250: 43-50.
- Popek EM (2003) Sampling and Analysis of Environmental Chemical Pollutants. Academic Press, Elsevier.
- Eappan KP, Markose PM (1986) Amine extraction for uranium estimation. Bulletin of Radiation Protection 9: 83-86.
- Kolthoff IM, Philip, Eiving J (1962) Treatise on Analytical chemistry. Part II 9: 102-111.
- Raghavayya M (1990) Estimation of 226Ra by Emanometry, Bulletin of Radiation Protection, Indian Association of Radiological Protection 3: 11-17.
- Jha VN, Tripathi RM, Sethy NK, Sahoo SK, Shukla AK et al. (2010) Bioaccumulation of 226Ra by plants growing in fresh water ecosystem around the uranium industry at Jaduguda, India. J Environ Radioact 101: 717-722.
- Markose PM (1990) Studies on the Environmental behavior of Radium from Uranium mill tailings. Ph.D. Thesis, University of Mumbai, PP.62.
- Markose PM, Eappan KP, Venkataraman S, kamat PR (1987) Distribution of radium and chemical toxxins in the environment of a uranium complex. In the Nature Radiation Environment III, Rice University, Houstion, USA.
- Alpha Emitter Uranium, Health physics Society, 2002.
- Diamond GL, Marrow PE, Panner BJ, Gelein RM, Baggs RB (1989) Reversible uranyl fluoride nephrotoxicity in the Long-Evans rat. Fundam. Appl. Toxiccol.13: 65-78.
- Kocher DC (1989) Relationship between kidney burden and radiation dose from chronic ingestion of uranium: implications for radiation standards for public. Health Physics 57: 9-15.
- Durbin PW (1984) Metabolic model for uranium. In Biokinetics and Analysis in Man 1984. Edited by R. H. Moore. United States Uranium Registry; USUR-05, HEHF-47, F1-F65. Avalaible from the National Technical Information Service, 5285 Port Royal Road, Springfield, VA 22151, USA.
- Hursh JB, Spoor NL (1973) Data on Man. In: Uranium, Plutonium, Transplutonium Elements, H.C. Hodge, J.N. Stannard and J.B. Hursh, Ed. Springer-Verlag, Berlin: 197-239.
- Lussenhop AJ, Gallimore JC, Sweet WH, Struxness EG, Robinson J (1958) The toxicity in man of hexavalent uranium following intravenous administration. Am. J. Roent 79: 83-100.
- Morris SC, Meinhold AF (1995) Probabilistic risk assessment of nephrotoxic effect of uranium in drinking water. Health Phys 69: 897-908.
- USEPA (1991) National Primary and Secondary Drinking Water Regulations, Final Rule, Selenium, January. U.S. EPA, Rules and Regulations,
- USEPA (1996) Proposed Guidelines for Carcinogen Risk Assessment. U.S. Environmental Protection Agency, Federal Register 61:17960 -18011.
- WHO, 2003. Guidelines for Drinking Water Quality. WHO, Geneva.
- Shin DC, Kim YS, Moon JY, Park HS, Kim JY et al. (2002) International trends in risk management of groundwater radionuclides. J Environ Toxicol 17: 273–284.
- USEPA reference information on radium, (October, 2010).
- Illinois Department of Public Health (2008) Division of Environmental Health, Radium in water, Environmental health fact sheet.
- Basu SK, Jha VN, Markose PM, Khan AH (1999) An assessment of ground water quality around Jaduguda after thirty years of UCIL operations Proceedings of 24th Annual Conference of IARP (Radiation Protection and Environment Journal) Kakrapar, Surat.
- Jha VN, Jha, Giridhar (2001) Evaluation of effluent management practices and its environmental impact around Jaduguda after thirty year of UCIL operations Proc. of International conf. on Radiation Protection Measurement and Dosimetry: Current Practices and Future trend 24: 481-483.
- Dang HS, Jaiswal DD, Parameswaran M, Krishnamony S (1994) Physical, Anatomical, Physiological and Metabolic Data for Reference Asian Man-A Proposal. BARC Report No. BARC/1994/FE-043.
- Dang HS, Jaiswal DD, Nair S (2001) Daily Dietary Intake of trace Elements of Radiological and Nutritional importance by the Adult Indian Population. Journal of Radioanlytical and Nuclear Chemistry 249: 1 95-101.
- US EPA ( 2000) National primary drinking water regulation radionuclide, Final Rule.
- US EPA (2000) Preliminary health risk reduction and cost analysis; revised national primary drinking water standards for radionuclide; review draft.
- International Commission on Radiological Protection Report (1994) Dose Coefficients for Intakes of Radionuclides by Workers. ICRP Publication 68, Annals of ICRP, Pergamon Press.
- Khan AH, Jha VN, Jha S, Kumar R (2000): Management of Radioactivitywaste and Environmental Impact of Mining and Processing of Uranium ore. International symposium on Geo- environment reclamation, Nagpur.PP.20-22.
- Sethy NK, Jha VN, Shukla AK, Khan AH (2004) Chronic exposure from natural uranium through drinking water to kidney. Proc. of 13th National Symposium on Environment, NEHU, Shillong, India
- ICRP (1994) Dose Coefficients for Intakes of Radionuclides by Workers. ICRP Publication 68. Ann. ICRP 24 (4).
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 15240
- [From(publication date):
March-2011 - Dec 08, 2025] - Breakdown by view type
- HTML page views : 10521
- PDF downloads : 4719