Research Article Open Access
Impact of Water Quality on Species Composition and Seasonal Fluctuation of Planktons of Batticaloa lagoon, Sri Lanka
Harris J.M* and Vinobaba PDepartment of Zoology, Faculty of Science, Eastern University, Sri Lanka
- *Corresponding Author:
- Harris J.M
Department of Zoology, Faculty of Science
Eastern University, Sri Lanka
E-mail: harriseusl@gmail.com
Received date: August 05, 2012; Accepted date: September 17, 2012; Published date: September 19, 2012
Citation: Harris JM, Vinobaba P (2012) Impact of Water Quality on Species Composition and Seasonal Fluctuation of Planktons of Batticaloa lagoon, Sri Lanka. J Ecosyst Ecogr 2:117. doi:10.4172/2157-7625.1000117
Copyright: © 2012 Harris JM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Abstract
This study examines whether planktons of the Batticaloa lagoon could be considered as bio-indicator of areas subjected to various anthropogenic and geomorphologic influences. However the physico-chemical parameters of the lagoon plays vital role in supporting the existence of biological parameters. This study was carried out for planktonic survey and in situ measurement of existing hydrological physico-chemical parameters from 9th July 2008 to 22nd June 2009 over the wet and dry seasons on seven sampling points of the Batticaloa lagoon. Comparatively, a higher number of species was recorded in the dry than in the wet season but there was little variation in species composition with respect to seasons. The compositions of phytoplankton were dominated by Bacillariophyceae which contributed about 55.81% to the total phytoplankton density. Nitzschia sp and Navicula sp lack seasonality and hence observed in both seasons. The 2 holoplankton groups of species increased in abundance during the wet season, while about 4 species lacked seasonality. Few phytoplankton species were reported to be potentially harmful/toxic bloom species. The information and observation in this study will be very useful in formulating management policies for the future of the Batticaloa lagoon.
Keywords
Geomorphology; Holoplankton; Lagoon; Nitzschia
Introduction
Planktons are small plants or animals that float, drift, or weakly swim in the water column. Phytoplankton comprises photosynthetic prokaryotes, which are major contributors of biomass and primary productivity in oligotrophic ecosystems [1]. These organisms are crucial to life in the aquatic environment because they make up the base of the marine food chain. The quantitative and qualitative abundance of the phytoplankton and zooplankton were significantly correlated by existing water quality conditions. Phytoplankton and zooplankton are good indicators for changes in nutrient pollution over time because they respond quickly to changes in nutrient input to the lagoon. The biological analysis of coastal waters, especially the phytoplankton analysis will describe clearly about the pollutant materials impact on the aquatic life and a decrease in biological diversity. Furthermore, the phytoplankton will reflect the condition of the waters, not only at the time of sampling, but also the condition at a previous time point [2]. Moreover, planktons are sensitive to many environmental conditions such as salinity, rainfall, temperature, dissolved oxygen levels, turbidity, and other factors [3]. On the other hand, zooplankton is the most important components of the aquatic ecosystem, playing a major role in energy transfer between the phytoplankton and the economically important fish populations. The abundance of zooplankton in lagoon depends on a great variety of abiotic and biotic factors, which collectively affect individual species of the zooplankton community. Generally zooplankton populations were poor compared to phytoplankton. Many environmental factors can affect zooplankton assemblages, including water temperature, nutrient concentrations, and salinity. Therefore, changes in primary production lead to changes in the distribution and abundance of zooplankton species [4]. Seasonal changes also affect the physico-chemical factors of surface water.
Objective
The aim of this study is to investigate the species composition of plankton community in relation to the existing water quality of the Batticaloa lagoon and their seasonal fluctuation. In addition this study examines whether planktons of the Batticaloa lagoon could be considered as bio-indicator of areas subjected to various anthropogenic and geomorphologic influences.
Study site
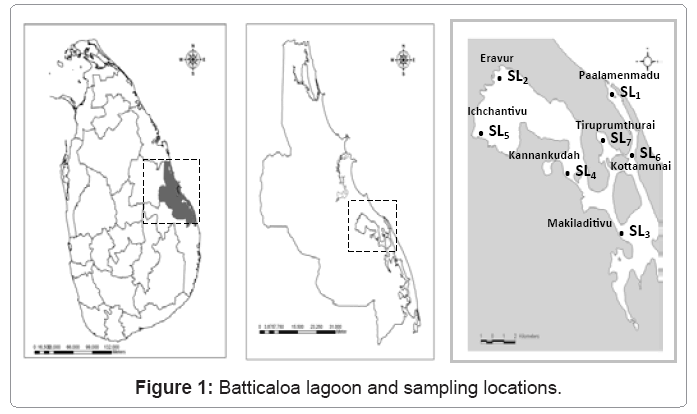
The Batticaloa lagoon is the third largest estuarine in Sri Lanka located between 7° 24’ - 7° 46’N and 81° 35’ - 81° 49’E. The lagoon is about 23 miles (56.8 km) long along meridian axis and 0.5 km to 4 km wide. It covers a total area of approximately 135.5 square kilometers, the Batticaloa lagoon extending from Kalmunai in the South to Eravur in the North. The lagoon is shallow with irregular bottom topography. It shows the lowest water depth (0.3 m) and the highest (6.5 m), recorded at the extreme northern region of the bar mouth. The average water depth of Batticaloa lagoon is about 1.5 m [5]. More than 90% of the lagoon is located in Batticaloa district where it reachs its maximum width. The lagoon communicates with the sea by two narrow canals, one at Palameenmadu and the other at Kallar. During the rainy season lagoon is in open communication with the ocean, but in dry season the bar mouth is closed by accumulation of sand due to wave action and ocean dynamics.
Climate of the lagoon
The Batticaloa lagoon is exposed to two distinct seasons namely the wet and the dry. Rainy season lasts from September to December brought about by the North East monsoon and dry season lasts from April to July. The rainy season is accompanied by strong winds, precipitation and the low air temperatures. The dry months are characterized by the influence of high air temperatures and scarce rainfall promoting high rates of evaporation.
Materials and Methods
Planktonic composition and along with in situ measurement of existing hydrological physico-chemical parameters survey was conducted routinely at approximately week intervals for a year from 2nd July 2008 to 22nd December 2009 over the wet and dry seasons. In this context selection of sampling sites was vital. The locations were selected based on the entrance of waste materials and nutrients and geomorphological influences. Altogether seven sampling points were chosen, and marked as “SL1- SL7” (Sampling Location) as shown in the Figure 1.
Surface water samples for physico-chemical parameters were collected from the selected locations by dipping well labeled sterilized plastic containers of 250 ml to about 6-10 cm below the surface film. Salinity, density, pH and dissolve oxygen are measured in-situ. Rest of the water samples collected were transported to the laboratory and processed immediately. All physico-chemical parameters of water were measured by suitable instruments as shown in the Table 1.
| Parameter | Instrument | Unit |
|---|---|---|
| Turbidity | Hanna portable HI 93703 C microprocessor turbidity meter | Formazine Turbidity Unit (FTU). |
| Salinity | Portable ATAGO, S/MillE Hand Refractometer | Parts per thousand (ppt) or ‰ |
| Density | Portable ATAGO, S/MillE Hand Refractometer | g/cm3 |
| Temperature | Hanna portable HI 98128 water proof Temperature meter | Celsius (°C) |
| pH | Hanna portable HI 98128 water proof pH meter | pH units |
| Nitrate | Hanna portable HI 93728 Nitrate ISM (Low range) | Parts per million (ppm) or mg/L |
| Nitrite | Hanna portable HI 93708 Nitrite ISM (High range) | Parts per million (ppm) or mg/L |
| Phosphate | Hanna portable HI 93713 Phosphate ISM (Low range) | Parts per million (ppm) or mg/L |
| Dissolved oxygen | Hanna portable HI 8043 Dissolved Oxygen Meter | Parts per million (ppm) or mg/L |
Table 1: Water quality parameters and measuring instruments with unit.
Phytoplankton samples were collected by using sterilized, 500 ml wide mouth plastic container at each sampling station and immediately fixed in Lugol’s solution as ratio of 100:1 per sample and Lugol’s solution [6]. The samples were reduced to 10 ml by decanting the supernatant aliquot and centrifuged in Gallenkamp centrifuge for 20 minutes at 4 rpm. Identification and enumeration were done by using Olympus C011 (Japan) binocular microscope using key to genera of Algae by Edmonson [7] and species were identified to the lowest taxonomic level. Photographs were taken by using digital camera with universal attachment set. The same procedure was also used for the zooplankton studies and identified with aid of practical manual for students of costal marine Zooplankton by Todd et al. [8].
Results and Discussion
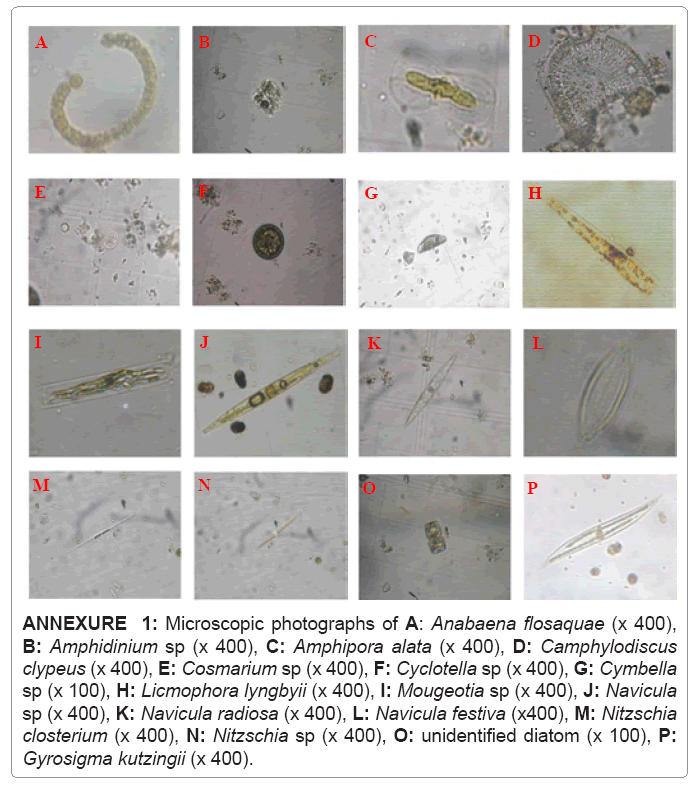
An important characteristic of lagoon is its biological diversity. Odiete et al. [9] reported that chemical measurements reflect water quality at a given time while biological assessment reflects conditions that have existed in a given environment over a long period of time. Main phytoplankton species available in the Batticaloa lagoon belong to five different groups with a total of 44 species (some of them shown in annexure 1), Bacillariophyceae formed most abundant group making up 24 species. This was followed by Cyanophyceae with 8 species from 8 genera. Chlorophyceae with 7 species, Dinophyceae with 3 different species while the Euglenophycea with only 2 species (Table 2).
| Species | Sampling location | |||||||
|---|---|---|---|---|---|---|---|---|
| SL1 | SL2 | SL3 | SL4 | SL5 | SL6 | SL7 | ||
| Bacillariophyceae (Diatoms) | Asterionella sp | ✓ | ✓ | ✓ | ||||
| Bacillaria sp | ✓ | ✓ | ||||||
| Camphylodiscus clypeus | ✓ | ✓ | ✓ | ✓ | ||||
| Closterium sp | ✓ | ✓ | ||||||
| Cocconeis placenta | ✓ | ✓ | ||||||
| Cocconies pediculus | ✓ | ✓ | ✓ | ✓ | ||||
| Coscinodiscus radiata | ✓ | ✓ | ✓ | |||||
| Cymbella turgidula | ✓ | ✓ | ✓ | ✓ | ||||
| Cymbella cistula | ✓ | ✓ | ✓ | ✓ | ||||
| Cyclotella sp | ✓ | ✓ | ✓ | |||||
| Cymatopleura sp | ✓ | ✓ | ✓ | ✓ | ||||
| Desmids sp | ✓ | ✓ | ✓ | ✓ | ||||
| Diatoma elongate | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Gryosigma kutzingii | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Licmophora lyngbyii | ✓ | |||||||
| Navicula elegensis | ✓ | ✓ | ✓ | ✓ | ||||
| Navicula radiosa | ✓ | ✓ | ✓ | ✓ | ||||
| Navicula festiva | ✓ | ✓ | ||||||
| Neidium minutissimus | ✓ | ✓ | ✓ | |||||
| Nitzschia inconspicula | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Nitzschia acicularia | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Tebellaria flucosa | ✓ | ✓ | ✓ | |||||
| Stephanodiscus rotula | ✓ | ✓ | ✓ | ✓ | ||||
| Synedra acus | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| SL1 | SL2 | SL3 | SL4 | SL5 | SL6 | SL7 | ||
| Cyanophyceae (Blue Green) | Anabaena flosaquae | ✓ | ✓ | ✓ | ✓ | |||
| Arthrospira sp | ✓ | ✓ | ✓ | |||||
| Coclosphaerium sp | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Chlorococcus sp | ✓ | ✓ | ✓ | |||||
| Gloethrichiae sp | ✓ | ✓ | ✓ | |||||
| Merismopedia sp | ✓ | ✓ | ✓ | ✓ | ||||
| Microcystis sp | ✓ | ✓ | ✓ | |||||
| Oscillatoria sp | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Chlorophycea (Green Algae) | Amphipora sp | ✓ | ✓ | ✓ | ✓ | |||
| Chlamydomonas | ✓ | ✓ | ✓ | |||||
| Chlorella sp | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||
| Cholorogonium sp | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Cosmarium sp | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||
| Mougeotia sp | ✓ | ✓ | ✓ | |||||
| Spirogyra | ✓ | ✓ | ✓ | |||||
| Dinophycea | Peridinium sp | ✓ | ✓ | ✓ | ||||
| Entzia acuta | ✓ | ✓ | ✓ | |||||
| Amphidinium sp | ✓ | ✓ | ✓ | |||||
| Euglenophycea | Phacus sp | ✓ | ✓ | ✓ | ||||
| Trachelomonas sp | ✓ | ✓ | ✓ | |||||
Table 2: Available composition of phytoplankton at sampling sites.
ANNEXURE 1: Microscopic photographs of A: Anabaena flosaquae (x 400), B: Amphidinium sp (x 400), C: Amphipora alata (x 400), D: Camphylodiscus clypeus (x 400), E: Cosmarium sp (x 400), F: Cyclotella sp (x 400), G: Cymbella sp (x 100), H: Licmophora lyngbyii (x 400), I: Mougeotia sp (x 400), J: Navicula sp (x 400), K: Navicula radiosa (x 400), L: Navicula festiva (x400), M: Nitzschia closterium (x 400), N: Nitzschia sp (x 400), O: unidentified diatom (x 100), P: Gyrosigma kutzingii (x 400).
During the study, SL7 was frequently found to be with dense algal blooms, at times it was also noticed in SL5. Due to the shallowness in this area, there was an increase in temperature, also effluents from the shrimp farms and slaughter house increased the input of nutrient. These conditions created a favorable environment for algal blooming. Matsuoka et al. [10] found that phytoplankton type diatoms have been associated with more eutrophic conditions. Diatoms dominating in SL7 were evidence that the area was in eutrophic condition. Dinoflagellates are more in an environment with high temperature [11], this condition supports the domination of dinoflagellates (Amphidinium sp) in SL6. Therefore, phytoplankton abundance was low in SL6. Dinoflagellates are more prevalent not only in an environment with high temperature but also in circulating water with lack of nutrients. The above conditions were present in SL1 hence, dinoflagellates were found to be prevailing in this location.
Most of the phytoplankton possesses the ability to carry out photosynthesis. Increase in turbidity leads to lowering of photosynthetic depth [12], and loss of suitable substrates for benthic fauna declines the biodiversity. Due to the existence of high turbidity in SL7, the phytoplankton related to photosynthesis were low whereas diatoms and cyanobacteria related to algal blooming were found be in abundance. There was no direct and serious effect of pH on the phytoplankton. However, when the turbidity increases there was a corresponding shift in the hydrogen ion concentration towards the alkalinity and increase in the level of dissolved salts, which promote the coagulation of fine particulate matter [13]. From this it can be understood that pH levels have indirect impact on the phytoplankton community. Salinity has a pronounced influence on phytoplankton growth. One might expect growth to slow as salinity increases [14]. Majority of the recorded species were found more frequently at low salinities (< 15%) than at high salinities (< 29%). Cyanobacteria might adapt to a change in salinity although they grow slowly at higher salinities [15]. During the present study, only once salinity was recorded 29% at SL6. Anabaenasp, Oscillatoria sp, Chlorococcus sp, and Merismopedia sp were also detected in this location. Skeletonema sp and other forms, primarily diatoms, had a high frequency of occurrence at low salinities (< 13%) but was also twice recorded at salinities greater than 22%. Nitzschia sp was found at all observed salinities. Chlorophycea and Cyanobacteria abundance decreased with the increasing salinities. Salinity has been known to have a positive impact on the growth of algal blooms and also high salinity has been known to stunt algae growth [16].
As an additional proof of evidence, Chlorophycea were found to be at low quantity at the location SL7. Diatom (Navicula radiosa) and flagellate phytoplankton (Peridinium sp) and the presence of Chlorophycea could indicate slightly higher salinity and possibly some eutrophication [17] at the locations of SL6 and SL7.
Freshwater phytoplankton do not survive at these higher salinities. Only one fresh water phytoplankton (Licmophora lyngbyii) was found in (SL2) when the salinity was lower (8%) during the month of December. In addition, diatoms species that have a high salinity tolerance were found everywhere.
Occurrence of species such as Nitzschia, Microcystis (blue-green algae) is a very good indicator of pollution. According to Gerloff et al. [18], Microcystis has a relatively low phosphorus requirement and has the ability to utilize sulphur as an alternative to phosphorus in its metabolism. Furthermore, the prevalence of Euglenoids may be a further indication of organic contamination. Most important nutrients here are phosphates and nitrites, which favour the phytoplankton growth mainly in surface light layers. Appreciable quantity of Nitzschia and nitrogen fixing phytoplankton (Anabaena) were observed in SL5 and SL7 as these locations were subjected to more nitrate pollution. However, Mishra et al. [19] pointed out that the stable hydrographic conditions coupled with increased nutrient concentrations have triggered the blooming of this diatom species. The Puthupaalam Bridge and its associated road is a barrier for the flow of water at locations SL6 and SL7. This causes water to be stagnant. This condition favours the algal blooming in these locations frequently.
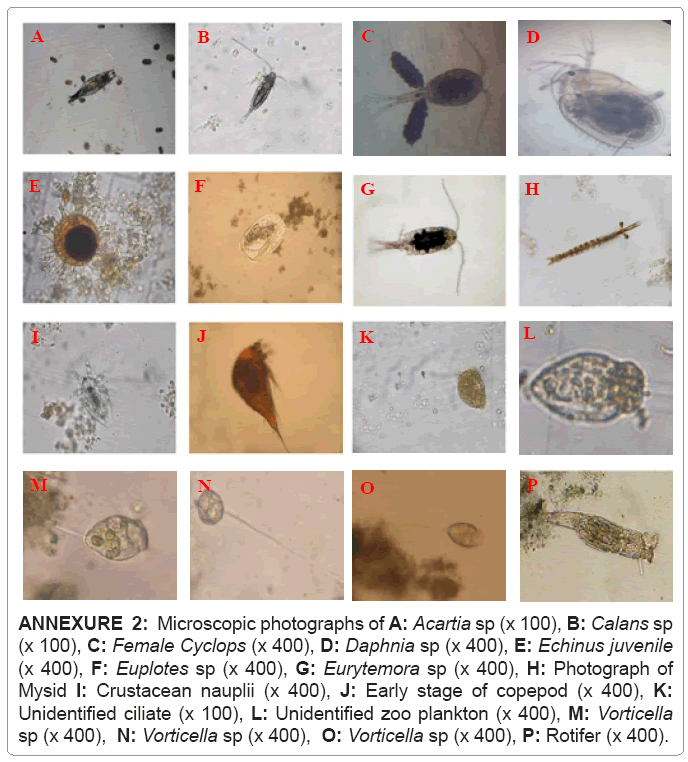
The holoplankton groups of Crustacean Copepoda, Protozoa, Coelenterata and Rotifera, contributed about 81% to the total zooplankton density (Annexure 2) while rest represented only about 19% to the total zooplankton. The latter were composed of the larvae of Mollusca, Mysidacea, euphausids and other groups of Ostracoda and Daphnia as shown in Table 3.
| Phylum | Class | Zooplankton species |
|---|---|---|
| Arthropoda | Barnchiopoda | Daphnia sp |
| Calanoid | Calanus sp | |
| Acartia sp | ||
| Copepod | Tigriopus sp | |
| Crustacea | Crab zoea, Nauplii | |
| Mysis, Shrimp larvae | ||
| Malacostraca | Mysid shrimp | |
| Maxillopoda | Cyclops sp | |
| Maxillopoda | Barnacles larvae | |
| Maxillopoda | Eurytemora sp | |
| Monogononta | Euphausids | |
| Ostracoda | Ostracoda sp | |
| Coelenterata | Schypozoa | Coelenterate medusae |
| Protozoan | Ciliates | Euplotes sp |
| Schypidia sp | ||
| Unidentified ciliate | ||
| Vorticella collserola | ||
| Vorticella sp | ||
| Rotifera | Heterotrichea | Unidentified rotifer |
| Mollusc | Gastropoda | Mollusc larvae |
| Echinodermata | Astroidea | Echinus Juvenile stage |
Table 3: Available composition of zooplankton at sampling sites.
ANNEXURE 2: Microscopic photographs of A: Acartia sp (x 100), B: Calans sp (x 100), C: Female Cyclops (x 400), D: Daphnia sp (x 400), E: Echinus juvenile (x 400), F: Euplotes sp (x 400), G: Eurytemora sp (x 400), H: Photograph of Mysid I: Crustacean nauplii (x 400), J: Early stage of copepod (x 400), K: Unidentified ciliate (x 100), L: Unidentified zoo plankton (x 400), M: Vorticella sp (x 400), N: Vorticella sp (x 400), O: Vorticella sp (x 400), P: Rotifer (x 400).
The majority of zooplankton species of Batticaloa lagoon were typical of strongly brackish water but the northern part of the lagoon (SL1) shows a mixture of marine species and brackish water. The present study revealed that species composition of zooplankton was completely dominated by Ciliates (Euplotes sp) and other protozoan throughout the study period. In addition, Crustacean copepoda, Rotifera and Coelenterata medusae also contributed to zooplankton community of the Batticaloa lagoon and various invertebrate and fish larvae were counted. The frequency of rotifers alongside other organisms could be an additional indicator to the level of contamination in SL5 and SL6. Further, more larvae of Mollusca, Mysidacea and other groups of Ostracoda sp, Daphnia sp were recorded while Cyclops were recorded only when salinity was below 9%. Copepods dominated at the location SL2 and were much lower in abundance at the shallow area. In addition, crab zoea were the most dominant and abundant zooplanktons in SL4.
Distributions of zooplankton in SL1 were determined by its salinity requirement rather than geography. However, salinity variation has an impact on zooplankton density and species richness. A noticeable decrease in density and species richness of zooplankton was noticed with high salinity in the location of SL6 and SL7. Salinity was the most important factor influencing zooplankton assemblages and has an effect on physiological processes, as a result, species have adapted for certain salinities. Ostracoda and Daphnia are freshwater zooplanktons but can tolerate a large range of salinities [20]. Ciliates (Euplotes sp) dominated in higher salinity condition, and also in locations where temperature and organic matter levels are high (SL7). Euplotes sp has not been previously reported from this lagoon. Copepods were restricted to this shallow area and cyclopoid copepods (Eurytemora sp) predominated in terms of numerical abundance.
The degree to which turbidity will persist in a lagoon is largely dependent upon the velocity and turbulence of the lagoon. Clear evidences, indicating high water turbidity and significant contents of organic and nutrient matter were created by the shrimp farm effluents at SL7 with corresponding changes in zooplankton following the worsening water quality. This in turn increased the turbidity, resulting in reduction in the relative abundance of the zooplankton. However there was no significant water flow existing in SL6 and SL7, therefore suspended particles stagnated. When compared to other locations, SL6 and SL7 were found to possess low quantity of zooplanktons.
Sudden fluctuations in air temperature usually do not have any immediate effects on the water temperature of the lagoon, but there is an establishment of thermocline due to the lack of water movement [21]. The presence of thermocline resulted in the difference of zooplankton community on SL4. Furthermore, temperature impacts zooplankton on both species physiology and primary productivity. This in turn, impacts feeding rates of zooplankton [22].
Zooplankton density was not only influenced by the increment of water temperature but also by the decrease of nitrate concentration. The high temperature and low nitrate concentration at SL4 caused more zooplankton availability on comparative basis. Though, different species were not observed, copepod was the most numerous and widespread.
Predation also affects the abundance of zooplankton in the Batticaloa Lagoon because of constitute significant food source for fish larvae [23]. Young fish were potentially important regulators of zooplankton communities. Because of the favourable conditions prevailing at SL3 for breeding, planktivouros fishes and grazing juvenile fishes were found in higher abundance [24], resulting in lower zooplankton density. There was low existence of zooplankton in SL7 due to the feeding of zooplankton by the mollusk larvae [23]. Therefore, there was a decrease in their density with richness of these groups in these locations.
Seasonal Variation of Planktons
The distributions of plankton flora in lagoons are mostly related to prevailing ecological conditions, besides the nature of bottom sediments and fertility of water [25]. The flushing of planktonic algal forms towards the sea during the rains by flood waters, could also account for the reduced phytoplankton diversity in the wet season. Similarly, reduced phytoplankton diversity in the wet season may be linked to the low water clarity which reduces the amount of light available to the planktonic algal component for photosynthesis. During the sampling period, the water temperature data strongly evidenced seasonality, higher temperatures in dry season (mean temperature 33.34 ± 0.23°C) and a decrease immediately from the beginning of the wet season (mean temperature 29.24 ± 0.31°C) caused by air temperature variation. Thus results in lowering of phytoplankton.
In the dry season between the May and the first half of July, phytoplankton composed primarily of diatoms and members of the Chlorophyta, while the Cyanophyta were not abundant. During the early part of October this latter division was dominated by Oscillatoria sp. However, this organism again became prevalent during late November. During the middle of August, conditions became optimal for the growth of the blue-greens and a very rapid development occurred for three weeks with the population reaching its peak during the latter part of the month. Nitzschia and Navicula lack seasonality and hence observed in both seasons.
Seasonal Variation of Zooplankton
The seasonal diversity of different zooplankton components is regulated by several factors. The factors like temperature, transparency, turbidity, conductivity and dissolved oxygen play an important role in regulating diversity and seasonal population densities [26]. Gasiûnaitë and Razinkovas [27] found that both qualitative and quantitative characteristics of crustacean zooplankton exhibited strong dependence on mean salinity. Seasonal distribution of the zooplankton of the sampling station indicated considerable variation in zooplankton population during the dry season, while about 4 species lacked a seasonal preference. When the salinity declined during wet season, the fresh water zooplankton namely Ostracoda, Daphnia, Cyclops were observed in SL4. The runoff during wet season results in the increase in turbidity which in turn reduces the relative abundance of the zooplankton, particularly in the area between slaughter house and shrimp farm (SL7). Similarly, the high intensity of sunlight reduces the zooplankton. However zoolplanktons were observed comparatively low in both seasons, this in turn creates difficulty in observing seasonality of zooplanktons.
Barnacle, polychaete, gastropod and crab larvae occurred during most of the year, while larvaceans and medusae were more common in the dry season. There was little variation in numbers or species composition during wet and dry seasons, despite variations in river discharge, salinity and nutrient content. This was attributed to the relatively high nutrient levels.
Findings
The particular hydrological physico-chemical conditions of lagoon were the main factors determining seasonal variations and the association structure of phytoplankton assemblages. In dry season phytoplankton were higher than the wet season due to the low temperature and also this might be attributed to the higher turbidity and nitrate levels. An inverse relationship existed with the salinity as phytoplankton ‘blooms’ followed by the low salinity periods and the phytoplankton concentration fell as the salinity increased. It appears that the general availability of nutrients and the adaptation of the plankton to fluctuating salinity were the factors responsible for the observations of the plankton population. Zooplankton do not show marked seasonal variation in species composition or numbers.
The compositions of phytoplankton was dominated by Bacillariophyceae which contributed about 55.81% to the total phytoplankton density; while Cyanophycea (18.60%), Chlrophycea (13.95%), Dinophycea (6.97%) and Euglenophycea (4.65%) were altogether represented rest of the total phytoplankton. Chlorella, Oscillatoria, Nitzschia and Cymbella were found to be good indicators of water pollution. This is the evidence that places where the above mentioned phytoplanktons were prevalent at higher values were polluted.
Cyclotella (Bacillariophyceae) was observed in both wet and dry season as a tolerant species. The possible reason for this observation might be its resilient ability to withstand the varied environmental factors while Nitzschia was least in the wet season. This might be attributed to nutrients status. Since stable, stressful lagoon conditions favour Cyanophyte diversity and dominance. The stable hydrographic conditions in SL7 coupled with increased nutrient concentrations had triggered the blooming of these diatoms (Asterionella, Anabeana) species. The sites influenced by marine waters (SL1) generally exhibited slightly lower numbers of species but which generally demonstrate cosmopolitan distributions. Despite the heavy constraint imposed by the salinity, it is evident that other environmental factors, e.g. temperature, play a major role in the regulation of the planktonic communities. Navicula sp, Nitzschia sp were more abundant at the end of dry season. Chlorella, Microcystis, Cymbella and Nitszchia were pollution indicators.
The majority of zooplankton species of Batticaloa lagoon are typical of strongly brackish water but the northern part of the lagoon shows a mixture of marine species and brackish water. However Ostracoda, Daphnia and Cyclops were recorded when the salinity was below 10% it was expected that salinity is the major variable in species distribution. Ciliates (Euloptes) dominated higher salinity condition and where temperature and organic matter levels are high. At SL2, the greater abundance of zooplankton in these deeper waters was evidence of an environment more suitable to promote growth and reproduction of the zooplanktonic species. A deeper water column, lack of intensive predation and better food conditions were found. There was no evidence that liberation of larvae was confined to any one season of the year. The 2 holoplankton groups of species increased in abundance during the wet season, while about 4 species, Barnacle, Polycheate, gastropoda and crab larvae lacked seasonality. Rotifers were found act as pollution indicators while Euplotes have not been previous recorded.
Future Consideration
Most of the dominant species of phytoplankton were not considered as harmful and dangerous for human health. However, certain species of Anabaena, Microcystis, Oscillatoriya are known to produce certain neurotoxin, hepatotoxin and skin damages. In addition Amphidinium sp also observed in the lagoon produce biologically active haemolytic compounds and may be implicated in ciguatera (phytotoxin). These have to be viewed as a threat to lagoon food safety. Monitoring of toxic microalgae especially bloom forming ones in the waters of Batticaloa lagoon would help to have better understanding of the problem of phytotoxin.
References
- Partensky F, Blanchot J, Vaulot D (1999) Differential distribution and ecology of Prochlorococcus and Synechococcusin oceanic waters: A Review. Bulletin Institute of Oceanography, Monaco 19: 457-475.
- Choi JK, Shim JH (1986) The ecological study of phytoplankton in Kyeonggi Bay, Yellow Sea. Journal of the Oceanological Societyof Korea 21: 156-170.
- Satsmadjis J (1985) Comparison of indicators of pollution in the Mediterranean. Mar Pollut Bull 16: 395-400.
- Francis RC, Hare SR, Hallowed AB, Wooster WS (1998) Effects of interdecadal climate variability on the oceanic ecosystems of the NE Pac. Fish Oceanography 7: 1-21.
- Scot AD (1989) A directory of Asian wetlands (2nd edn), Cambridge University Press, Cambridge, UK.
- APHA Standard Methods for the Examination of Water and Waste water (1992) American Public Health Association (20th edn), Pleneum Press, Washington, DC.
- Edmondson WT (1992) Algae, key to genera in fresh water Biology.New Delhi, India: International Books & periodicals supply services 118-189.
- Todd CD, Laverack MS, Boxshall GA (1996) A practical manual for students. Coastal Marine Zooplankton (2nd edn),UK: Cambridge University Press.
- Odiete WO, Nwokoro RC, Daramola T (2003) Biological assessment of four courses in Lagos metropolis receiving industrial and domestic waste discharge. Nigeria Environmental Society 1: 1-14.
- Matsouka K, Joyce LB, Kotani Y, Matsuyama Y (2003) Modern dinoflagellate cysts in hypertrophic coastal waters of Tokyo Bay, Japan. J Plankton Res 25: 1461-1470.
- Oviatt CA (2004) The changing ecology of temperate coastal waters during a warming trend. Estuaries and Coasts 27: 895-904.
- Nwankwo DI, Akinsoji A (1992) Epiphyte community on water hyacinth Eichhornia crassipes in Coastal waters of south western Nigeria. Arch Hydrobiol 124: 501-511.
- Phillips J (1972) Chemical processes in estuaries (3rd edn), Applied science publishers, London.
- Marcarelli A, Wurtsbaugh W, Olivia G (2006) Salinity controls phytoplankton response to nutrient enrichment in the Great Salt Lake, Utah, USA. Can J Fish Aquat Sci 63: 2236-2248.
- Barron S, Weber C, Marino R, Davidson E, Tomasky G, et al. (2002) Effects of varying salinity on phytoplankton growth in a low-salinity coastal pond under two nutrient conditions. Biol Bull 203: 260-261.
- Abed R, Kohls K, Dirk B (2007) Effect of salinity changes on the bacterial diversity, photosynthesis and oxygen consumption of cyanobacterial mats from an intertidal flat of the Arabian Gulf. Environmental Microbiology 9: 1384-1392.
- Frey DG (1986) Cladocera Analysis (5th edn), Wiley Press, Chichester, UK.
- Gerloff GC, Fitzgerald GP, Skoog F (1952) The mineral nutrition of microcystis aeruginosa.Am J Bot 39: 26-32.
- Mishra S, Sahu G, Mohanty AK, Singh SK, Panigrahy RC (2006) Impact of the diatom Asterionella glacialis bloom on the water quality and phytoplankton community structure in coastal waters of Gopalpur Sea, Bay of Bengal. Asian Journal of Water Environment Pollution 3: 71-77.
- Athersuch J, Horne DJ, Whittaker JE (1990) Marine and brackish water Ostracods (5th edn), Prentice Hall, New York.
- Hutchinson GE (1967) In Introduction to Lake Biology and Limnoplankton (2nd edn), John Wiley Press, New York, USA.
- Nybakken JW (2003) An Ecological Approach (6th edn), Addison Wesley Longman, California, USA.
- Damodara W (1983) Tintinnida (Protozoa- Ciliata). A vital link in the estuarine food web. Bulletin of the National Institute of Oceanography and Fisheries 16: 403-407.
- Turner JT, Anderson DM (1983) Zooplankton grazing during dinoflagellate blooms in a Cape Cod Embayment, with observations of the predation upon tintinnids by copepods. Marine Ecology 4: 359-374.
- Konsowa A (2007) Spatial and Temporal Variation of Phytoplankton Abundance and Composition in the Hypersaline Bardawil Lagoon, North Sinai, Egyptian. Journal of Applied Sciences Research 3: 1240-1250.
- Patil SS, Auti RG (2005) Seasonal variations of zooplankton from Salim Ali Lake of Aurangabad. Journal of Bio Infonet 2: 81-85.
- Gasiûnaitë Z, Razinkovas A (2002) The salinity tolerance of two cladoceran species from the Curonian Lagoon: an experimental study. Sea and Environment 2: 28-32.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 19552
- [From(publication date):
September-2012 - Oct 16, 2025] - Breakdown by view type
- HTML page views : 14235
- PDF downloads : 5317