Commentary Open Access
Is Body Weight Loss a Harbinger of Methamphetamine-induced Neurotoxicity?
Dean E Dluzen1*, Altaf S Darvesh2 and Werner J Geldenhuys31Department of Anatomy and Neurobiology, Department of Pharmaceutical Sciences, Northeast Ohio Medical University, USA
2Department of Pharmaceutical Sciences, Department of Psychiatry, Northeast Ohio Medical University, USA
3Department of Pharmaceutical Sciences, Northeast Ohio Medical University, USA
- *Corresponding Author:
- Dean Dluzen
Department of Anatomy and Neurobiology
Northeast Ohio Medical University
4209 State Route 44, P. O. Box 95
Rootstown, OH, USA
Tel: 330-325-6300
Fax: 330-325-5913
E-mail: ded@neomed.edu
Received October 17, 2012; Accepted November 22, 2012; Published November 29, 2012
Citation: Dluzen DE, Darvesh AS, Geldenhuys WJ (2012) Is Body Weight Loss a Harbinger of Methamphetamine-induced Neurotoxicity? J Addict Res Ther 3:138. doi:10.4172/2155-6105.1000138
Copyright: © 2012 Dluzen DE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Addiction Research & Therapy
Methamphetamine (MA) is a drug of abuse of societal concern. Euphoria, increased energy/alertness and enhanced libido all contribute to the appeal, and therefore, abuse of this psychostimulant. Weight loss represents still another attribute of MA as indicated in both human [1-4] and animal [5,6] studies. In fact, weight loss has been reported as a reason for MA use [7-9] and MA under the trademark name Desoxyn, has been approved for use clinically in the treatment of exogenous obesity, as well as for ADHD and narcolepsy [10].
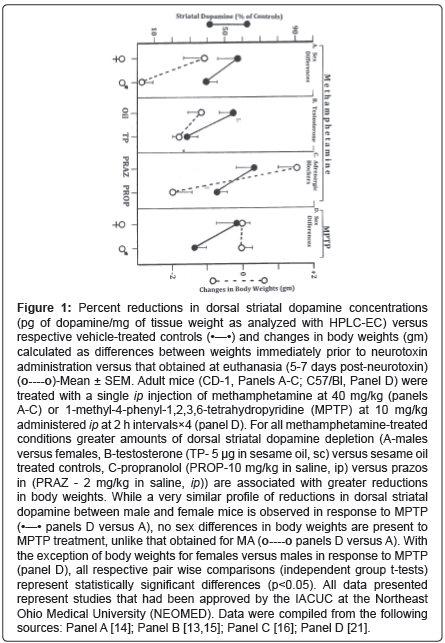
In animal studies, MA has served as an agent to induce neurotoxicity within the Nigrostriatal Dopaminergic (NSDA) system, and thus a means to study conditions that modulate this neurotoxicity. For example, sex, hormonal factors and adrenergic blockers all affect the degree of MAinduced neurotoxicity [11-16]. A summary of the pair wise differences in MA-induced neurotoxicity (dorsal striatal dopamine depletion) for these three different factors, along with the corresponding changes in body weight to MA is illustrated in panels A-C of the Figure 1. In all three situations, conditions showing greater amounts of dopamine depletion (A-males versus females, B-testosterone (TP) versus sesame oil vehicle treated controls, C-Propranolol (PROP) versus Prazosin (PRAZ)) are associated with the greater amounts of body weight reductions.
Figure 1: Percent reductions in dorsal striatal dopamine concentrations (pg of dopamine/mg of tissue weight as analyzed with HPLC-EC) versus respective vehicle-treated controls (•—•) and changes in body weights (gm) calculated as differences between weights immediately prior to neurotoxin administration versus that obtained at euthanasia (5-7 days post-neurotoxin) (o----o)-Mean ± SEM. Adult mice (CD-1, Panels A-C; C57/Bl, Panel D) were treated with a single ip injection of methamphetamine at 40 mg/kg (panels A-C) or 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) at 10 mg/kg administered ip at 2 h intervals×4 (panel D). For all methamphetamine-treated conditions greater amounts of dorsal striatal dopamine depletion (A-males versus females, B-testosterone (TP- 5 μg in sesame oil, sc) versus sesame oil treated controls, C-propranolol (PROP-10 mg/kg in saline, ip) versus prazos in (PRAZ - 2 mg/kg in saline, ip)) are associated with greater reductions in body weights. While a very similar profile of reductions in dorsal striatal dopamine between male and female mice is observed in response to MPTP (•—• panels D versus A), no sex differences in body weights are present to MPTP treatment, unlike that obtained for MA (o----o panels D versus A). With the exception of body weights for females versus males in response to MPTP (panel D), all respective pair wise comparisons (independent group t-tests) represent statistically significant differences (p<0.05). All data presented represent studies that had been approved by the IACUC at the Northeast Ohio Medical University (NEOMED). Data were compiled from the following sources: Panel A [14]; Panel B [13,15]; Panel C [16]; Panel D [21].
A cursory and seemingly obvious conclusion would be that the greater extent of neurotoxicity produces a corresponding greater degree of generalized malaise, as indicated by body weights. The data presented in panel D of the Figure 1 suggest that this may not necessarily be the case. Here are presented results obtained using a similar sex-differences protocol as that presented in panel A, however, with NSDA neurotoxicity induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). While nearly identical dorsal striatal dopamine reductions are obtained with MA and MPTP (A versus D), the corresponding sex differences in body weight present to MA, are completely absent with MPTP (A versus D). Such results suggest that sex differences in response to neurotoxins targeting the NSDA system do not inescapably generate identical changes in body weights. Rather, body weight reductions following MA treatment appear to be closely associated with the degree of NSDA loss and are specific for MA-induced NSDA neurotoxicity.
Confirmation or refutation of this intriguing relationship awaits further investigation involving formal experiments on dose-response, time-course, cross-species analyses and food restriction/palatability. Moreover, differences in MA regimes between the mouse models as described above and the clinical/street use of this drug also warrant consideration in evaluating this relationship. While remaining cognizant of these limitations, data derived from these animal experiments have served as relatively valid and reliable models for indices of effects in humans. Women show less toxicity [17] and diminished dopamine responses [18] to MA/amphetamine as compared with men, which is similar to that of results obtained with mice [19,20]. Of particular relevance to the present report, are the findings indicating that weight loss is a pervasive characteristic of MA, as seen in mice and humans [1-6]. Given that one of the presumptive “benefits” of MA, both as a street drug and for clinical application is its capacity for weight reduction, an important implication for this relationship between body weight and neurotoxicity as described above is the potential for unwitting damage to the NSDA system as associated with the weight reduction resulting from MA. Rephrased as a question: Is Body Weight Loss a Harbinger of Methamphetamine-induced Neurotoxicity?
References
- Kramer JC, Fischman VS, Littlefield DC (1967) Amphetamine abuse. Pattern and effects of high doses taken intravenously. JAMA 201: 305-309.
- Albertson TE, Derlet RW, Van Hoozen BE (1999) Methamphetamine and the expanding complications of amphetamines. West J Med 170: 214-219.
- Neale A, Abraham S, Russell J (2009) “Ice” use and eating disorders: a report of three cases. Int J Eat Disord 42: 188-191.
- Krasnova IN, Justinova Z, Ladenheim B, Jayanthi S, McCoy MT, et al. (2010) Methamphetamine self-administration is associated with persistent biochemical alterations in striatal and cortical dopaminergic terminals in the rat. PLoS ONE 5: e8790.
- Bittner SE, Wagner GC, Aigner TG, Seiden LS (1981) Effects of a high-dose treatment of methamphetamine on caudate dopamine and anorexia in rats. Pharmacol Biochem Behav 14: 481-486.
- Davidson C, Lee TH, Ellinwood EH (2005) Acute and chronic continuous methamphetamine have different long-term behavioral and neurochemical consequences. Neurochem Int 46: 189-203.
- Rawson RA, Gonzales R, Brethen P (2002) Treatment of methamphetamine use disorders: an update. J Subst Abuse Treat 23: 145-150.
- Cretzmeyer M, Sarrazin MV, Huber DL, Block RI, Hall JA (2003) Treatment of methamphetamine abuse: research findings and clinical directions. J Subst Abuse Treat 24: 267-277.
- Brecht ML, O'Brien A, von Mayrhauser C, Anglin MD (2004) Methamphetamine use behaviors and gender differences. Addict Behav 29: 89-106.
- http://dailymed.nlm.nih.gov/dailymed/archives/fdaDrugInfo.cfm?archiveid=73929#CP
- Gao X, Dluzen DE (2001) The effect of testosterone upon methamphetamine neurotoxicity of the nigrostriatal dopaminergic system. Brain Res 892: 63-69.
- Dluzen DE, McDermott JL (2006) Estrogen, testosterone, and methamphetamine toxicity. Ann NY Acad Sci 1074: 282-294.
- Lewis C, Dluzen DE (2008) Testosterone enhances dopamine depletion by methamphetamine in male, but not female, mice. Neurosci Lett 448: 130-133.
- Dluzen DE, McDermott JL, Bourque M, Di Paolo T, Darvesh AS, et al. (2011) Markers associated with sex differences in methamphetamine-induced striatal dopamine neurotoxicity. Curr Neuropharmacol 9: 40-44.
- Buletko AB, Dluzen DE, McDermott JL, Darvesh AS, Geldenhuys WJ (2012) Markers associated with testosterone enhancement of methamphetamine-induced striatal dopaminergic neurotoxicity. Neurotoxicol Teratol 34: 338-343.
- Darvesh AS, Geldenhuys WJ, Jones TJ, Carroll RT, Van der Schyf CJ, et al. (2012) Adrenergic receptors modulate methamphetamine-induced hyperthermia and dopaminergic neurotoxicity in mice. Soc Neurosci Ann Mtg, New Orleans.
- Hall JN, Broderick PM (1991) Community networks for response to abuse outbreaks of methamphetamine and its analogs. NIDA Res Monogr 115: 109-120.
- Munro CA, McCaul ME, Wong DF, Oswald LM, Zhou Y, et al. (2006) Sex differences in striatal dopamine release in healthy adults. Biol Psychiatry 59: 966-974.
- Wagner GC, Tekirian TL, Cheo CT (1993) Sexual differences in sensitivity to methamphetamine toxicity. J Neural Transm Gen Sect 93: 67-70.
- Dluzen DE, Salvaterra TJ (2005) Sex differences in methamphetamine-evoked striatal dopamine output are abolished following gonadectomy: comparisons with potassium-evoked output and responses in prepubertal mice. Neuroendocrinology 82: 78-86.
Relevant Topics
- Addiction Recovery
- Alcohol Addiction Treatment
- Alcohol Rehabilitation
- Amphetamine Addiction
- Amphetamine-Related Disorders
- Cocaine Addiction
- Cocaine-Related Disorders
- Computer Addiction Research
- Drug Addiction Treatment
- Drug Rehabilitation
- Facts About Alcoholism
- Food Addiction Research
- Heroin Addiction Treatment
- Holistic Addiction Treatment
- Hospital-Addiction Syndrome
- Morphine Addiction
- Munchausen Syndrome
- Neonatal Abstinence Syndrome
- Nutritional Suitability
- Opioid-Related Disorders
- Relapse prevention
- Substance-Related Disorders
Recommended Journals
Article Tools
Article Usage
- Total views: 14851
- [From(publication date):
December-2012 - Nov 25, 2025] - Breakdown by view type
- HTML page views : 10086
- PDF downloads : 4765