Research Article Open Access
Isolation, Molecular and Biochemical Identification of Paraoxon-Metabolizing Pseudomonas Species
Rupa Iyer*, Brian Iken and Tim TamezCenter for Life Sciences Technology, University of Houston, 385 Technology Building, Houston, Texas 77204, USA.
- *Corresponding Author:
- Dr. Rupa Iyer
Center for Life Sciences Technology
University of Houston
385 Technology Building, Houston, TX77204, USA
Tel: 713-743-0099
E-mail: riyer@uh.edu
Received Spetember 23, 2011; Accepted November 19, 2011; Published November 21, 2011
Citation:Iyer R, Iken B, Tamez T (2011) Isolation, Molecular and Biochemical Identification of Paraoxon-Metabolizing Pseudomonas Species. J Bioremed Biodegrad 2:132. doi: 10.4172/2155-6199.1000132
Copyright: © 2011 Iyer R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
Seven soil borne bacterial isolates were collected from locations throughout the Houston metropolitan area. All isolatesproved capable of degrading the pesticide paraoxon to p-nitrophenol in carbon limited media and showed a high degree of tolerance to an environment exposed to increasing concentrations of the pesticide. A combination of 16S rRNA sequencing and fatty acid methyl ester analysis (FAME) analysis was used to identify the unknown bacterial species. Universal bacterial primers were used for 16S rRNA analysis to tentatively identify all seven isolates as species belonging to the genus Pseudomonas. Sequencing results coupled with FAME analysis resolved the species identity of the seven isolates as either Pseudomonas aeruginosa or Pseudomonas putida. The results of this study demonstrate that a combination of molecular and biochemical analysis provide sufficient resolution to identify paraoxon degrading microbial populations.
Keywords
Paraoxon degradation; Pseudomonas; Pesticide; Soil bacteria
Abbreviations
FAME: Fatty Acid Methyl Ester, OP: organophosphorus, opd: organophosphorus degradation gene, OPH: organophosphorus hydrolase, CSM: Carbon-deficient media, GCSM: Glycerol carbon deficient media, MI: Max identity, SIM: Similarity index, PNP: P-Nitrophenol.
Introduction
Organophosphorus (OP) compounds are potent cholinesterase inhibitors widely used as insecticides and as chemical warfare agents [1- 3]. Cholinesterase inhibitors are potent neurotoxins that act upon the enzyme acetylcholinesterase preventing affected neurons from returning to their rest state upon activation and can result in paralysis, coma, or death upon exposure [4]. Self-poisoning through occupational exposure with OP pesticides occurs regularly in agricultural settings and continues to cause major health problems in developing countries [1,5- 7]. Accidental and purposeful exposure to the OP pesticides throughout the developing world is tallied in the millions with approximately 200,000 deaths worldwide annually [4,5,8-10]. In developed countries these compounds continue to threaten metropolitan water supplies and our agricultural centers through unintentional contamination [11,12]. The persistent use and health threat presented by these OP pesticides has resulted in a growing number of biodegradation research studies as a means to ascertain and control the spread of these compounds in contaminated environments.
OP pesticides while very dangerous do not typically remain in the environment for long periods of time [13]. Research has shown that reduced efficacy of these pesticides are the result of susceptibility to hydrolysis by microbial enzymes [13,14]. The degradation of these problematic compounds in both soil and aquatic environments has been tied to the presence of a rapidly growing community of bacterial microorganisms that metabolize OP compounds as sources of carbon through shared organphosphorus hydrolase (OPH) gene(s) [13,15-17]. Populations of microorganisms observed to efficiently break down a range of OP pesticides include, but are not limited to, members of the genera of Burkholderia [18], Pseudomonas [14,19,20], Serratia [14], Arthrobacter [18], Enterobacter [21], Flavobacterium [22,23], and Brevundimonas (including Brevundimonas diminuta, formerly Pseudomonas diminuta) [24]. Numerous lesser known genera of soil borne bacteria have shown some propensity for OP pesticide degradation as well suggesting that these OPH genes are mobile and rapidly dispersed through microbial agricultural populations [19,25]. Positive correlations between the concentrations of OP pesticides in the environments under study and the presence of OPH genes in rapidly growing microbial populations have been reported [18]. Therefore, microbial bioremediation technology can offer us a reliable and inexpensive way to safely remove these harmful compounds in both terrestrial and aquatic environments as well as from our food products [16].
Current biodegradation and bioremediation research on OP pesticides has focused primarily on microbial populations that can metabolize the pesticides methyl parathion, fenitrothion, or dianzon. Comparatively little is known about the breakdown of the pesticide paraoxon in the environment or the microbial species responsible for its degradation. The most common molecular technique used to identify microorganisms is 16S rRNA sequencing [26-29]. However, species of larger genera, notably Pseudomonas, have proven difficult to fully resolve through this method alone [26-28,30]. Secondary approaches to microbial population identification have included biochemical or physiological criteria that are then used to generate profiles that can be stored in databases for future reference. For instance, bacterial lipid concentrations are highly conserved and consistent to each individual species due to their importance in cell structure [31]. Physiological profiling methods may therefore try to quantify bacterial fatty acid composition to characterize species identities [31-33]. Bacterial fatty acids are extracted from grown bacterial cultures and chemically modified to form volatile fatty acid methyl esters [31]. These compounds are then loaded into a gas chromatograph to analyze the pattern of peaks unique to each organism [31]. Fatty acid methyl ester (FAME) analysis thus provides researchers with a simple framework for generating a large body of microbial profiles at relatively cheap cost [31,32].
In this study we describe the isolation and identification of microbial isolates present in soils collected from throughout the Houston metropolitan area capable of metabolizing paraoxon in carbon limiting media. Identification of these bacterial organisms was conducted through 16S rRNA analysis and fatty acid methyl ester (FAME) analysis. Degradation efficiency and tolerance to increasing levels of paraoxon were also investigated while PCR was used to screen isolates for the presence of a plasmid borne opd gene.
Materials and Methods
Chemicals, enzymes and oligonucleotides
Paraoxon (O,O-diethyl p-nitrophenyl phosphate) and all other chemicals were obtained from Sigma (St. Louis, MO). Egg white lysozyme, DNase-free ribonuclease A and ampicillin were also obtained from Sigma (St. Louis, MO). Synthetic deoxyoligonucleotides were purchased from Sigma (St. Louis, MO). Proteinase K was acquired from Promega (Madison, WI).
Media for bacterial growth
Luria-Bertani medium (LB) was prepared from the following components: 10 g/liter BactoTryptone (Difco Laboratories, Detroit, MI); 5 g/liter yeast extract (Difco Laboratories, Detroit, MI); 10 g/liter NaCl. The pH of the above media was adjusted to 7.5. LB media was used as an enriched medium to rapidly grow the cells of each sample isolate to an O.D. of 0.6-1.0 from glycerol stocks. For growth on a solid surface, the media were supplemented with 1.5 g/liter Bacto Agar (Difco Laboratories, Detroit, MI). Glycerol carbon-deficient minimal medium (GCSM) had the following composition: 0.2 g/liter MgSO4.7H2O; 0.08 g/liter Ca(NO3)24H2O; 0.005 g/liter FeSO4.7H2O; 4.8 g/liter K2HPO4; 1.2 g/ liter KH2PO4; 1% glycerol. The additional inoculation into GCSM was used to provide the cells with a transitory medium between an enriched medium (LB) and a final limited medium (CSM) to minimize the shock to the cells and limit cell death in each culture flask upon their inoculation into CSM. The carbon-deficient minimal medium (CSM) had the following composition: 0.2 g/liter MgSO4.7H2O; 0.08 g/liter Ca(NO3)2.4H2O; 0.005 g/liter FeSO4.7H2O; 4.8 g/liter K2HPO4; 1.2 g/ liter KH2PO4. Immediately prior to inoculation, CSM was supplemented with paraoxon (0.1 mg/ml). The final inoculation into CSM ensures that the only carbon source available to the bacterial isolates comes in the form of the pesticide paraoxon and therefore sustained growth in this media is possible only if the isolate can metabolize paraoxon.
Isolation of paraoxon-metabolizing microbial species from soil samples
A total of seven soil samples were collected from sites located throughout the Houston-Sugar Land-Baytown metropolitan area including Houston, Texas (29°45’46”N95°22’59”W), Alvin Texas (29°23’37”N95°16’18”W), League City, Texas (29°29’59”N95°05’23”W), Sealy, Texas (29°46’27”N96°9’27”W), and Katy, Texas (29°47’33”N95°49’21”W). Air-dried soil (1g) was suspended in 50 ml of the LB medium. The suspension was kept for 2 days at 30˚C on a shaker. Any insoluble materials were allowed to settle down and an aliquot (100μl) from the cleared supernatant was used to inoculate 3 ml of paraoxon-supplemented CSM. The culture was incubated for 1 week at 30˚C on a rotary shaker at 200 rpm. 100μl of the bacterial suspension was transferred into 3 ml of fresh CSM containing paraoxon and the incubation step was repeated. After five consecutive subcultivations, the seven isolates were plated on to CSM agar plates containing paraoxon (1ug/ml). After overnight incubation at 30˚C, the isolates formed on the plates were transferred to GCSM-glycerol stocks and frozen at -80˚C.
Isolation of total cellular DNA
Bacterial glycerol stocks were used to inoculate 3 mL LB medium and incubated overnight at 37ºC and 200 rpm for approximately 24 hours. 2 mL of the resulting bacterial suspension (OD=0.6) was pelleted at 13,000 rpm for 3 minutes and the total cellular DNA was extracted using the CTAB method outlined in Wilson et al. [34] with the following modifications: cells were resuspended in TE buffer containing lysozyme (50,000 units/ml) and ribonuclease A (300 Kunitz units/ml), and incubated for 1 hour at 37˚C. Immediately prior to lysis, proteinase K (> 30,000 units/g) was added to a final concentration 0.25 mg/ml. Cells were lysed with 0.5% (w/v) SDS for one hour at 37˚C. The subsequent removal of polysaccharides and residual proteins was performed as described in the original protocol.
16S rRNA sequencing
Fragments of the 16S rRNA gene were amplified by PCR from cellular DNA using universal 16S rRNA-specific primers (Forward: AGAGTTTGATCMTGGCTCAG, Reverse: GGTTACCTTGTTACGACTT) [35,36] and PCR 2x Master Mix (Promega, Madison, WI). PCR conditions were set as follows: initial denaturation at 94˚C for 4.5 min, 32 cycles consisting of denaturation at 94˚C for 0.5 min, annealing at 52˚C for 0.5 min, and extension at 72˚C for 1 min, and final elongation at 72˚C for 4 min. PCR products were purified using a QIA quick PCR Purification Kit (Qiagen) and cloned into the pJET1.2/blunt cloning using the CloneJet PCR Cloning Kit and a sticky-end cloning protocol supplied by the manufacturer (Fermentas). Successful insertion of a gene into the plasmid disrupts a lethal gene product ensuring that only cells that receive an insert grow on selective antibiotic media. Approximately 5 uL of the ligation mixture was used directly to transform chemically competent DH5alpha cells at 42˚C for 40 seconds. Following a 1 hour incubation period in LB media 125 uL aliquots cells were plated onto LB-ampicillin-agar plates and grown overnight. Transformants were grown overnight in liquid LB media and harvested for plasmid DNA using a Qiaprep Spin Miniprep Kit (Qiagen). Minipreps for each isolate were sequenced bi-directionally by the dye-terminator method with plasmid specific primers supplied with the kit from the manufacturer (Fermentas) by SeqWright, Inc. (Houston, TX). DNA alignments were made using ClustalX 2.0.12, and sequences were manually edited when necessary to remove gaps and undetermined bases pairs. Corrected sequences were then compared to highly similar sequences in the NCBI database to determine the genus and species of each bacterial isolate.
Phylogenetic analysis of gene sequences
Sequences were aligned with MUSCLE (v3.7) configured for the highest accuracy (MUSCLE with default settings) and phylogenetic affiliations were evaluated using the maximum likelihood method implemented in the PhyML program (v3.0 aLRT). The HKY85 substitution model was selected assuming an estimated proportion of invariant sites (of 0.417) and 4 gamma-distributed rate categories to account for rate heterogeneity across sites. The gamma shape parameter was estimated directly from the data. Reliability for internal branches was assessed using the aLRT test (SH-Like).Graphical representation of the phylogenetic tree was performed with TreeDyn (v198.3) [37-42].
Detection of an organophosphorus degradation gene byPCR amplification
Total cellular DNA from each bacterial isolate was used as a template to probe for the presence of a plasmid-based organophosphorus degradation (opd) gene first isolated in Flavobacterium [15]. PCR amplification of the opd gene region was conducted using series of two forward primers (F196 – CGCGGTCCTATCACAATCTC and F450 - CGCCACTTTCGATGCGAT) and two reverse primers (R757 – TCAGTATCATCGCTGTGACC and R840 – CTTCTAGACCAATCGCACTG) with PCR 2x Master Mix (Promega, Madison, WI) [23]. PCR conditions were set as follows: initial denaturation at 94˚C for 5 min, 35 cycles consisting of denaturation at 94˚C for 1 min, annealing at 55˚C for 1 min, and extension at 72˚C for 1 min, and final elongation at 72˚C for 5 min [23]. PCR products were purified using a QIAquick PCR Purification Kit (Qiagen) and checked through electrophoresis on a 1.2% agarose gel.
Fatty Acid Content Analysis
Each isolate was streaked out onto separate LB agar plates, incubated overnight at 30˚C and shipped to MIDI Labs (Newark, DE) to carry outfatty acid extraction and identification through gas chromatography.
Growth of bacterial isolates and degradation of paraoxon in supplemented media
Isolates taken from glycerol stock were inoculated in 100 mL LB media and placed in an incubator at 30˚C, 200 rpm for approx 24 hours. Cells were then pelleted at 2500 rpm for 10 minutes at 4˚C. The supernatant was poured off and the pellet placed on ice. 50 mL of GCSM media was added and the mixture while on ice was added to a rotary shaker to resuspend the pellet over a period of 1 hour. The cell suspension was transferred to a clean 250 mL flask and incubated at 30˚C, 200 rpm for 48 hours. Following the 48 hour period incubation in GCSM each sample culture was divided into 25 mL aliquots and spun down at 2500 rpm for 10 minutes at 4˚C. Both 25 mL pellets were resuspended in 15 mL of CSM medium on a rotary shaker for 1 hour. 35 mL of CSM was added to the resuspended pellets to a total volume of 50 mL that was then transferred to a 250 mL foil covered flask to prevent photo-hydrolysis in which paraoxon was inoculated to a final volume of 100 ug/mL. The breakdown of paraoxon was then measured by the absorbance of p-nitrophenol in solution. Each mixture was incubated at 30˚C, 200 rpm for 72 hours and duplicate absorbance readings of each sample were taken at 405 nm (p-nitrophenol). Aliquots of 200 uL non-inoculated CSM media were used as a blank and a non-opd containing Pseudomonas aeruginosa strain ATCC10145 was used as a negative control.
Tolerance inoculation of bacterial isolates in paraoxon-supplemented media
10 mL of CSM media was aliquoted into three 15 mL falcon tubes covered in foil to prevent photo-hydrolysis. The pesticide paraoxon was added to each tube at concentrations of 200 ug/mL CSM, 300 ug/mL CSM and 400 ug/mL CSM. A total of 200 uL of previously grown sample culture was then added to the CSM+ paraoxon tubes and incubated at 30˚C, 200 rpm, for eight days. Once a day duplicate absorbance readings were taken for 200 uL of each sample at 600 nm for each respective paraoxon concentration. Aliquots of 200 uL non-inoculated CSM media were used as a blank, E.coli cells transformed with the Puc19 plasmid were used as a negative control and a strain of P.putida carrying the opd (organophosphorus degradation) gene was used as a positive control.
Results and Discussion
We have isolated seven bacterial strains collected from locations throughout the Houston metropolitan area (Table 1) that were able to grow in paraoxon-supplemented media. To identify the species collected in the study the 16S rRNA gene of each isolate was probed by PCR using universal bacterial primers as described in Baker et al. and Frank et al. [35,36]. Sequencing results (Table 2) show that all seven of the bacterial isolates appear to belong to the genus Pseudomonas with six of the seven isolates identified as Pseudomonas aeruginosa with max identity (MI) values ranging from 88% to 99%. The last isolate labeled in the study as CBF10 was tentatively identified as Pseudomonas putida (80% MI). Results of a phylogenetic analysis of the seven collected isolates conducted by aligning 16S rRNA data from our 7 soil isolates with closely related strains of Pseudomonas aeruginosa and Pseudomonas putida along with representatives of both species’ intrageneric groups can be seen in Figure 2.
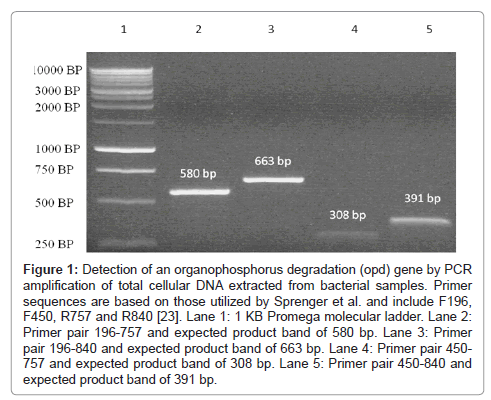
Figure 1: Detection of an organophosphorus degradation (opd) gene by PCR amplification of total cellular DNA extracted from bacterial samples. Primer sequences are based on those utilized by Sprenger et al. and include F196, F450, R757 and R840 [23]. Lane 1: 1 KB Promega molecular ladder. Lane 2: Primer pair 196-757 and expected product band of 580 bp. Lane 3: Primer pair 196-840 and expected product band of 663 bp. Lane 4: Primer pair 450- 757 and expected product band of 308 bp. Lane 5: Primer pair 450-840 and expected product band of 391 bp.
| Sequence Designation | Classification | Location | Purpose of Location |
|---|---|---|---|
| ADF10 | Gram-negative | Sealy, TX, | Field adjacent to hotel. |
| CBF10 | Gram-negative | Fairchilds, TX | Farmland |
| CMF10 | Gram-negative | Houston, TX | Field |
| KNF10 | Gram-negative | Houston, TX | FamilyGarden |
| MLF10 | Gram-negative | Alvin, TX | Small Family Farm |
| NAF10 | Gram-negative | League City, TX | FamilyGarden |
| TTF10 | Gram-negative | Katy, TX | Field |
Table 1: Location of each soil isolate collected for this study.
| Sequence Designation | Max Identity % | Sample Identity |
|---|---|---|
| ADF10 | 99% | Pseudomonas aeruginosa |
| CBF10 | 99% | Pseudomonas putida |
| CMF10 | 99% | Pseudomonas aeruginosa |
| KNF10 | 99% | Pseudomonas aeruginosa |
| MLF10 | 90% | Pseudomonas aeruginosa |
| NAF10 | 99% | Pseudomonas aeruginosa |
| TTF10 | 88% | Pseudomonas aeruginosa |
Table 2: Results of 16S rRNA analysis using the NCBI BLAST tool for highly similar sequences. The max identity details the highest percent identity for a set of aligned segments to the subject sequence.
All seven isolates were prepped and shipped to Midi Labs to compare the fatty acid content of the isolates to that of archived library species. SIM (Similarity) index values over 0.7 are considered to be strong identity matches for unknown samples. Isolates ADF10, CMF10, MLF10, NAF10, and KNF10, and TTF10 were strongly identified as Pseudomonas aeurginosa GC subgroup A with SIM values of 0.915, 0.911, 0.905, 0.843 and 0.903 and 0.920 respectively (Table 3). FAME analysis originally determined that CBF10 had been a mixed sample and upon further isolation and submission reanalysis confirmed this sample as Pseudomonas putida biotype A with a SIM value of 0.709. Further 16S rRNA sequencing of the purified CBF10 isolate confirmed its identity as P.putida with 99% MI. A pure isolate of Stenotrophomonas maltophiliawas also obtained from the mixed culture (CBF10), but has not been studied in detail and was not included in this study.
| Sequence Designation | SIM Value Index | Sample Identity |
|---|---|---|
| ADF10 | 0.915 | Pseudomonas aeruginosa |
| CBF10 | 0.709 | Pseudomonas putida |
| CMF10 | 0.911 | Pseudomonas aeruginosa |
| KNF10 | 0.903 | Pseudomonas aeruginosa |
| MLF10 | 0.905 | Pseudomonas aeruginosa |
| NAF10 | 0.843 | Pseudomonas aeruginosa |
| TTF10 | 0.920 | Pseudomonas aeruginosa |
Table 3: A color coded (green = high, yellow = moderate, red = poor) similarity index of likely sample identities based on the organism’s fatty acid content. The number in the similarity index value column expresses how closely the organism’s fatty acid content matched that of a library strain used as its match. The closer the number is to 1 the more likely the species indicated is the correct species for the samples in the study.
Phylogenetic analysis of 16S rRNA taken from all isolates suggests that all strains of Pseudomonas aeruginosa collected for the study are virtually identical to each other genotypically. While the lone Pseudomonas putida isolate is closely related to archive species, it is the only strain to demonstrate the ability to degrade paraoxon. These results suggest that Pseudomonas species are ubiquitous to environments exposed to OP pesticides and nearly identical strains harboring the opd gene are widely dispersed throughout the Houston area. The relative lack of additional genera possessing an opd gene in our samples may be due to the relatively small sampling area from which isolates were drawn and/or additional selective pressures that have conferred a decisive advantage to an opportunistic pathogen like Pseudomonas aeruginosa. A new study to investigate opd harboring microbial populations in contaminated aquatic environments may yield a better assortment of different organisms. Based on these above results we suggest that 16S rRNA sequencing coupled with FAME analysis represents the most reproducible and accurate method of identifying species of soil borne bacteria that metabolize paraoxon currently available.
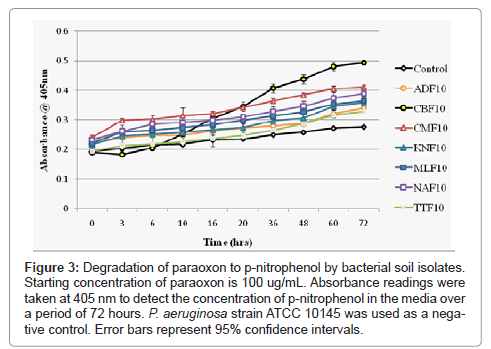
The presence of a Flavobacterium-likeopd gene was resolved through PCR amplification of four unique fragments of the gene from total cellular DNA extracted from each isolate. Each of the four primer combinations F196-R757, F196-R840, F450-R757 and R450-R840 used in the study resulted in the expected product size, 580 bp, 663 bp, 308 bp, and 391 bp respectively (Figure 1). Each Pseudomonas isolate was further tested to determine their efficiency in metabolizing paraoxon and their level of growth tolerance in limited media supplemented with increasing concentrations of the pesticide. The time frame for paraoxon degradation to p-nitrophenol was 72 hours while the time frames for the three studies on paraoxon tolerance of soil isolates were spread out over a period of 8 days. Each day represented a 24 hour period with Day 1 designating a point 24 hours post inoculation of the CSM culture flask with paraoxon. All seven samples were found to be capable of degrading paraoxon to p-nitrophenol (Figure 3) with varying degrees of efficacy. The Pseudomonas putida (CBF10) isolate begins to rapidly degrade paraoxon after 10 hours and continued to do so steadily for the duration of the experiment. The rates at which the six Pseudomonas aeruginosa isolates degrade paraoxon are slower, but remain very consistent throughout the course of the study without the early spike in p-nitrophenol production that characterized the P.putida isolate.
Figure 3: Degradation of paraoxon to p-nitrophenol by bacterial soil isolates. Starting concentration of paraoxon is 100 ug/mL. Absorbance readings were taken at 405 nm to detect the concentration of p-nitrophenol in the media over a period of 72 hours. P. aeruginosa strain ATCC 10145 was used as a negative control. Error bars represent 95% confidence intervals.
The production of colored metabolites such as pyocyanin and pyoverdine by Pseudomonas aeruginosa species can potentially interfere with absorbance readings at 405 nm. To ensure that the degradation kinetics were not skewed, a non-opd containing strain of P.aeruginosa was used as a negative control to take into account any increases in absorbance that may be result from the presence of these compounds. In general the degradation curve exhibited by these isolates demonstrates consistent, steady rate of p-nitrophenol production and renders virtually all Pseudomonas aeruginosa isolates virtually indistinguishable from one another. Furthermore, while the degradative rate of paraoxon to p-nitrophenol by Pseudomonas putida was notably faster than the other six isolates, it also increased at a near constant rate following the initial jump seen 10 hours into the study. These results suggest that the opd gene is constitutively active in all isolates, but the organophosphorus hydrolase found in Pseudomonas putida may have a stronger affinity for the substrate paraoxon. None of the tested isolates showed any propensity to degrade p-nitrophenol further even with observation beyond 72 hours. Determination of the full opd sequence of each isolate and their associated upstream and downstream elements is under current investigation.
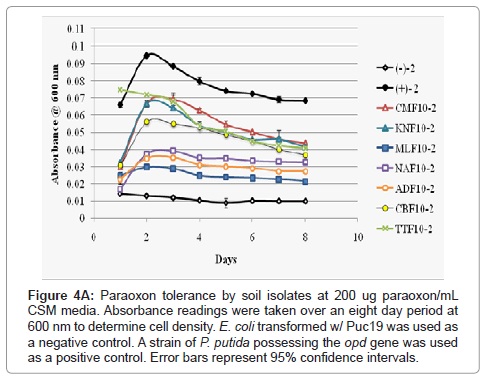
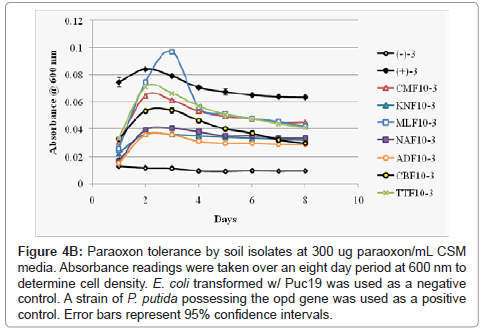
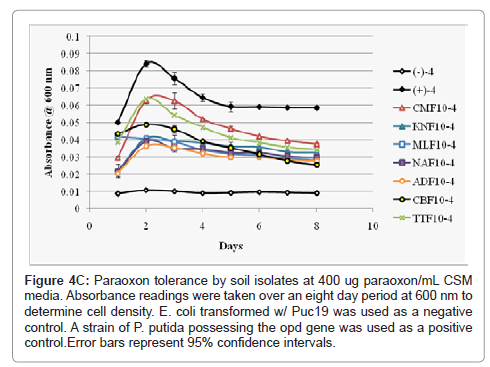
As p-nitrophenol is quite toxic, though not as toxic as paraoxon, high concentrations of the metabolic product would likely severely reduce an isolate’s ability to further grow and continue metabolizing paraoxon in its environment. To derive a clearer picture of the relationship between the growth of the soil isolates and paraoxon/p-nitropheol tolerance each isolate was inoculated into CSM media at a lower volume with increasing concentrations (200 ug paraoxon/mL (low) CSM, 300 ug paraoxon/mL CSM (intermediate), and 400 ug paraoxon/mL CSM (high) of the pesticide. Bacterial growth was measured over an extended period of eight days (Figures 4A-4C). At all paraoxon concentrations isolates CMF10 and TTF10 showed strong growth, but also the sharpest declines after 48 hours. KNF10 grew well at a low concentration but was overtaken by the CBF10 isolate at higher concentrations. Isolates ADF10 and NAF10 grew slowly at all concentrations though did not undergo as steep of a decline later in the study. MLF10 did poorly at the lowest concentration of paraoxon inoculation, but improved noticeably at higher concentrations. Overall, growth for each isolate generally peaked within 24-48 hours with two notable exceptions. Rapid growth was seen within 24 hours in the TTF10 isolate at the lowest paraoxon concentration and an extended growth phase was recorded for the MLF10 isolate that went substantially beyond 48 hours when grown in an intermediate concentration of 300 ug paraoxon/mL CSM. The rate of growth of the MLF10 isolate dropped again when exposed to a high concentration of the pesticide suggesting that a threshold response may have been responsible for this sudden burst of growth followed by an abbreviated stationary phase and steep lag phase as p-nitrophenol concentrations began to peak. Beyond the two exceptions noted above, slight reductions in the peak of the growth curve were followed by steeper lag phases in all isolates as the concentration of paraoxon was increased. Unsurprisingly, the CBF10 isolate’s tendency to rapidly degrade paraoxon manifested as the sharpest drops in bacterial growth.
Figure 4a: Paraoxon tolerance by soil isolates at 200 ug paraoxon/mL CSM media. Absorbance readings were taken over an eight day period at 600 nm to determine cell density. E. coli transformed w/ Puc19 was used as a negative control. A strain of P. putida possessing the opd gene was used as a positive control. Error bars represent 95% confidence intervals.
Figure 4b: Paraoxon tolerance by soil isolates at 300 ug paraoxon/mL CSM media. Absorbance readings were taken over an eight day period at 600 nm to determine cell density. E. coli transformed w/ Puc19 was used as a negative control. A strain of P. putida possessing the opd gene was used as a positive control. Error bars represent 95% confidence intervals.
Figure 4c: Paraoxon tolerance by soil isolates at 400 ug paraoxon/mL CSM media. Absorbance readings were taken over an eight day period at 600 nm to determine cell density. E. coli transformed w/ Puc19 was used as a negative control. A strain of P. putida possessing the opd gene was used as a positive control.Error bars represent 95% confidence intervals.
The isolation of these paraoxon-degrading bacteria effectively demonstrates sites exposed to OP compounds around Houston. As the organophosphorus hydrolase encoded by the opd gene has a very broad spectrum of viable substrates it cannot be stated for sure that these areas were explicitly exposed to the pesticide paraoxon itself. However, since the pesticide is still in widespread use throughout Asia and Africa, any organisms capable of breaking it down are critically important to global bioremediation efforts. Taking into account the individual rates of degradation and tolerance to different concentrations of paraoxon, the Pseudomonas putida (CBF10) isolate emerges as the best short term choice for bioremediation of contaminated environments in which immediate relief is required such as in the event of a biochemical attack or large scale accidental exposure near residential areas. If the environment is repeatedly exposed to moderate to high concentrations of the pesticide over an extended period of time, as happens in agricultural settings, the action of the Pseudomonas aeruginosa isolates would ultimately prevail as they would be able to tolerate the incrementally smaller rises in p-nitrophenol production for a far greater duration.
Acknowledgements
Partial funding for this project was provided by the Texas Workforce Commission (Meeting Industries’ Critical Workforce Initiative) grant. The authors wish to thank Kevin Smith, biotechnology lab manager, and the Fall 2010 BTEC 3100 undergraduate class for assistance with sample preparation. The authors also wish to thank Kinjal Gandhi and Taha Salim undergraduate biotechnology majors, for their help in compiling degradation and tolerance data and Ashish Damania for his help in compiling bioinformatics data. Special thanks to Dr. Donna Pattison for review of the manuscript.
References
- Eddleston M, Karalliedde L, Buckley N, Fernando R, Hutchinson G, et al. (2002) Pesticide poisoning in the developing world - A minimum pesticides list. Lancet 360: 1163-1167.
- U. S. Congress, Office of Technology Assessment (1992) Disposal of Chemi- cal Weapons: Alternative Technologies-Background Paper. U. S. Government Printing Office.
- World Health Organization in collaboration with the United Nations Environmental Programme (1990) Public Impact of Pesticides Used In Agriculture. WHO, Geneva: 128.
- Aardema H, Meertens JH, Ligtenberg JJ, Peters-Pollman OM, Tulleken JE, et al. (2008) Organophosphorus pesticide poisonings: cases and developments. Neth J Med 66: 149-153.
- Eddleston M, Buckley NA, Eyer P, Dawson AH (2008) Management of acute organophosphorus poisoning. Lancet 371: 597-607.
- Eddleston M, Eyer P, Worek F, Mohamed F, Senarathna L, von Meyer L, et al. (2005) Differences between organophosphorus insecticides in human selfpoisoning: a prospective cohort study. Lancet 366: 1452-1459.
- Karalliedde L, Senanayake N (1989) Organophosphorus insecticide poisoning. Br J Anaesth 63: 736-750.
- Buckley NA, Karalliedde L, Dawson A, Senanayake N, Eddleston M (2004) Where is the evidence for treatments used in pesticide poisoning? Is clinical toxicology fiddling while the developing world burns. J Toxicol Clin Toxicol 42: 113-116.
- van der Hoek W, Konradsen F (2006) Analysis of 8000 hospital admissions for acute poisoning in a rural area of Sri Lanka. Clin Toxicol (Phila) 44: 225-231.
- van der Hoek W, Konradsen F, Athukorala K, Wanigadewa T (1998) Pesticide poisoning: a major health problem in Sri Lanka. Soc Sci Med 46: 495-504.
- Coupe RH, Manning MA, Foreman WT, Goolsby DA, Majewski MS (2000) Occurrence of pesticides in rain and air in urban and agricultural areas of Mississippi. April-September 1995. Sci Total Environ 248: 227-240.
- Schipper PN, Vissers MJ, van der Linden AM (2008) Pesticides in groundwater and drinking water wells: overview of the situation in the Netherlands. Water Sci Technol 57: 1277-1286.
- Kumar S, Mukerji K G, Lal R (1996) Molecular aspects of pesticide degradation by microorganisms. Crit Rev Microbiol. 22: 1-26.
- Cycon M, W�?³jcik M, Piotrowska-Seget Z (2009) Biodegradation of the organophosphorus insecticide diazinon by Serratia sp. and Pseudomonas sp. and their use in bioremediation of contaminated soil. Chemosphere 76: 494-501.
- Harper LL, McDaniel CS, Miller CE, Wild JR (1988) Dissimilar plasmids isolated from Pseudomonas diminuta MG and Flavobacterium sp. (ATCC 27551) contain identical opd genes. Appl Environ Microbiol 54: 2586-2589.
- Islam SM, Math RK, Cho KM, Lim WJ, Hong SY, et al. (2010) Organophosphorus hydrolase (OpdB) of Lactobacillus brevis WCP902 from kimchi is able to degrade organophosphorus pesticides. J Agric Food Chem 58: 5380-5386.
- Mulbry WW, Kearney PC, Nelson JO, Karns JS (1987) Physical comparison of parathion hydrolase plasmids from Pseudomonas diminuta and Flavobacterium sp. Plasmid 18: 173-177.
- Choi MK, Kim KD, Ahn KM, Shin DH, Hwang JH, et al. (2009) Genetic and phenotypic diversity of parathion-degrading bacteria isolated from rice paddy soils. J Microbiol Biotechnol 9: 1679-1687.
- Chanika E, Georgiadou D, Soueref E, Karas P, Karanasios E, et al. (2011) Isolation of soil bacteria able to hydrolyze both organophosphate and carbamate pesticides. Bioresour Technol 102: 3184-3192.
- Rani NL, Lalithakumari D (1994) Degradation of methyl parathion by Pseudomonas putida. Can J of Microbiol 40: 1000-1006.
- Singh B, Walker KA, Morgan AW, Wright DJ (2004) Biodegradation of chlorpyrifos by Enterobacter strain B-14 and its use in bioremediation of contaminated soils. Appl Environ Microbiol 70: 4855-4863.
- Sethunathan N, Yoshida T (1973) A Flavobacterium sp. that degrades diazinon and parathion. Can J of Microbiol 19: 873-875.
- Sprenger WW, Dijkstra A, Zwart GJ, Agterveld MP, Noort PC, et al. (2003) Competition of a parathion-hydrolyzing Flavobacterium with bacteria from ditch water in carbon-, nitrate- and phosphate-limited continuous cultures. FEMS Microbiol Ecol 43: 45-53.
- Serdar CM, Gibson DT, Munnecke DM, Lancaster JH (1982) Plasmid involvement in parathion hydrolysis by Pseudomonas diminuta. Appl Environ Microbiol 44: 246-249.
- Zhang R, Cui Z, Jiang J, He J, Gu X, et al. (2005) Diversity of organophosphorus pesticide-degrading bacteria in a polluted soil and conservation of their organophosphorus hydrolase genes. Can J Microbiol 51: 337-43.
- Anzai Y, Kim H, Park JY, Wakabayashi H, Oyaizu H (2000) Phylogenetic affiliation of the pseudomonads based on 16S rRNA sequence. Int J Syst Evol Microbiol 50: 1563-1568.
- Mulet M, Lalucat J, Garc�?a-Vald�?©s E (2010) DNA sequence-based analysis of the Pseudomonas species. Environ Microbiol 12: 1513-1530.
- Peix A, Ram�?rez-Bahena MH, Vel�?¡zquez E (2009) Historical evolution and current status of the taxonomy of genus Pseudomonas. Infect Genet Evol 9: 1132-1147.
- Speigelman D, Whissell G, Greer CW (2005) A survey of the methods for the characterization of microbial consortia and communities. Can J Microbiol 51: 355-386.
- Yamamoto S, Kasai H, Arnold DL, Jackson RW, Vivian A, et al. (2000) Phylogeny of the genus Pseudomonas: intrageneric structure reconstructed from the nucleotide sequences of gyrB and rpoD genes. Microbiol 146: 2385-2394.
- Haack SK, Garchow H, Odelson DA, Forney LJ, Klug MJ (1994) Accuracy, reproducibility, and interpretation of Fatty Acid methyl ester profiles of model bacterial communities. Appl Environ Microbiol 60: 2483-2493.
- Nelson KY, Razban B, McMartin DW, Cullimore DR, Ono T, et al. (2010) A rapid methodology using fatty acid methyl esters to profile bacterial community structures in microbial fuel cells. Bioelectrochemistry. 78: 80-86.
- Qasem JA, Al-Zenki S, Al-Marzouk A (2010) Identification and characterization of Streptococcus agalactiae isolates using 16S rRNA sequencing and cellular fatty acid composition analysis. Pak J Biol Sci 13: 9-15.
- Wilson K (1990) Preparation of genomic DNA from bacteria. In Current Protocols in Molecular Biology 2: 1-2.
- Baker GC, Smith JJ, Cowan DA (2003) Review and re-analysis of domainspecific 16S primers. J Microbiol Methods 55: 541-555.
- Frank JA, Reich CI, Sharma S, Weisbaum JS, Wilson BA, et al. (2008) Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl Environ Microbiol 74: 2461-2470.
- Dereeper A, Audic S, Claverie JM, Blanc G (2010) BLAST-EXPLORER helps you building datasets for phylogenetic analysis. BMC Evol Biol 10: 8.
- Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, et al. (2008) Phylogeny. fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36: 465-469.
- Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32: 1792-1797.
- Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52: 696-704.
- Anisimova M, Gascuel O (2006) Approximate likelihood ratio test for branchs: A fast, accurate and powerful alternative. Syst Biol 55: 539-552.
- Chevenet F, Brun C, Banuls AL, Jacq B, Chisten R (2006) Tree Dyn: towards dynamic graphics and annotations for analyses of trees. BMC Bioinformatics 7: 439.
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 15625
- [From(publication date):
November-2011 - Dec 20, 2025] - Breakdown by view type
- HTML page views : 10783
- PDF downloads : 4842