B Cell Lymphoblastic Lymphoma Presenting as a Paravertebral Mass in a Child: A Case Report
Received: 22-Jun-2021 / Accepted Date: 06-Jul-2021 / Published Date: 13-Jul-2021 DOI: 10.4172/2476-2024.s3.008
Abstract
Lymphoblastic lymphomas are immature lymphoid malignancies accounting for approximately 30% of non Hodgkin lymphoma in children. Most of these neoplasms originate from T cell precursors while less than 25% of them originate from B lymphoblasts. On histology and immunohistochemistry, lymphoblasts in B lymphoblastic lymphoma (BLL) and B lymphoblastic leukaemia share the same features. However, they represent different diseases with distinct molecular alterations. Herein, we describe a case of BLL in a 2-year-old child presenting as a paravertebral mass, including the clinical and histopathological features with a review of the current literature.
Keywords: B lymphoblastic lymphoma; Childhood; Histopathology
Keywords
B lymphoblastic lymphoma; Childhood; Histopathology
Introduction
Lymphoblastic lymphomas are immature lymphoid malignancies accounting for approximately 30% of non Hodgkin lymphoma in children. Most of these neoplasms originate from T cell precursors while less than 25% of them originate from B lymphoblasts [1]. The most commonly involved sites are bone (26%) followed by skin or subcutaneous tissue (23%). On histology and immunohistochemistry, lymphoblasts in B lymphoblastic lymphoma (BLL) and B lymphoblastic leukaemia share the same features [2]. However, they represent different diseases with distinct molecular alterations [1]. Herein, we describe a case of BLL in a 2-year-old child presenting as a paravertebral mass, including the clinical and histopathological features with a review of the current literature.
Case Presentation
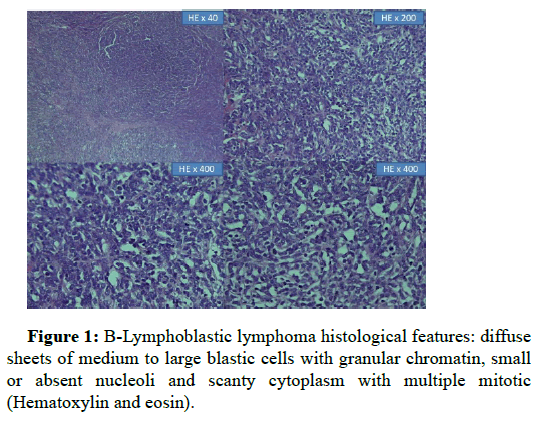
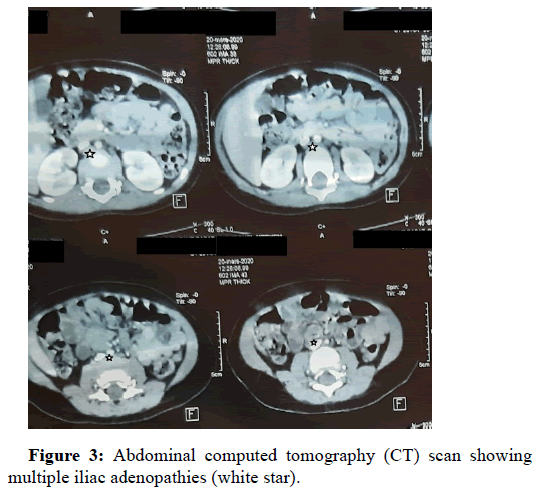
A 2-year old female was referred to our hospital for a history of a 6 months right lumbar paramedian mass. Upon physical examination, she was a healthy girl with a firm mass measuring 3 cm × 1 cm. Ultrasonography revealed a tissular subcutaneaous, hypoechogenic and heterogeneous mass with low vascularization index on Doppler. Surrounding soft tissues were unremarkable. Thoraco abdomino pelvic tomography confirmed these findings; and neither nodal involvement nor distant metastasis were detected. The patient’s blood cell counts were within normal range. Bone marrow aspirate was normal. The patient underwent surgical excision of the mass. On gross, it measured 2.7 × 1.8 cm, it was well circumscribed, lobulated, firm and graywhite. Hematoxylin-eosin stained sections revealed sheets of medium to large blastic cells with finely clumped nuclear chromatin, small or absent nucleoli and scanty cytoplasm with multiple mitotic figures. The tumor had a diffuse growth pattern with patchy fibrosis. There was no evidence of maturation (Figure 1). A large immunohistochemistry panel was performed (Figure 2) including: CD20, CD3, CD45, Desmin, myogenin, chromgranin, synaptophysin and CD99. Neoplastic cells were positive only for LCA CD45. Given the tumor’s localization, the age of the patient, the absence of lymph node involvement, lack of maturation signs, and positivity for LCA; with negative pan B and panT markers; the diagnosis of myeloid sarcoma was suggested. However, neoplastic cells stained negative with MPO, CD117, CD34 and CD68. A second look for immunohistochemical slides revealed focal positivity for CD20. Then, we performed another panel, cells were positive for CD19, CD43, CD45 and CD79a and Pax5. 15% to 20% of cells expressed TdT CD10. Proliferation’s index with Ki 67 was approximately 80%. Thus the diagnosis of B LBL was retained. Two months later a thoracoabdomino pelvic tomography revealied multiple susclavicular, para-aortic and iliac right adenopathies (Figure 3). Unfortunately genetic analysis is not available in our institution. The child was referred to oncology department for chemotherapy. After a follow-up period of 6 months, complete remission was obtained with ALLregimen treatment.
Discussion
Most of B immature cells neoplasms present as B-lymphoblastic leukaemia, while only 10%-20% of cases present as BLL [1]. In the current WHO classification of tumors of hematopoietic and lymphoid tissues, lymphoblastic lymphoma refers to cases presenting as confined process without peripheral blood or significant bone marrow involvement (less than 25%) [1-3]. Cortelazzo, S et al. reported that 64% of cases are aged <18 years [4]. The largest series in the English literature of B LBL in children encountered 53 patients, the median age of diagnosis was 8 years (ranging from 0,25 to 16,7 years) with no difference in gender distribution [2]. Our patient was younger than reported median age (2 years). At diagnosis, presenting symptoms are related to the involvement’s site; lymph nodes, skin, soft tissue, and bone represent the most common localizations, while gonads, head, neck and kidney are unusual ones [3]. Presence of CNS disease at diagnosis was reported in 6% of patients [1]. Our patient had a soft tissue mass and metachronous nodal disease without BM neither CNS’s involvment. Molecular genetic analysis shows hyperdipoidy in 48% of the cases, BCR-ABL1 like phenotype (found in 10% of paediatric B-ALL) was not reported in B-LBL [1].
The diagnosis requires identifying blasts; in BM or tissue biopsy [5]. It combines (i) characteristic morphology, (ii) precursor cells positive markers, (iii) with lineage definition and (vi) subtyping by additional stainings and/or genetic analysis [6]. On cytology, lymphoblasts vary from small cells with (1)scant gray-blue cytoplasm, (2) round or convoluted nuclei, (3) with condensed nuclear chromatin without nucleoli; to large cells with a prominent nucleoli, a moderate and occasionaly vacuolated cytoplasm [4,5] . Uncommon features include: coarse myeloblasts-like and hand mirror appearance [4]. On morphology, B lymphoblasts can’t be distinguished from their T counterparts [4]. Thus, the diagnosis requires immunophenotypic analysis [1]. B lymphoblasts are positif for CD19, CD79a (which might be positive in many T ALL) CD22, PAX5 (the most specific B lineage marker), TdT and CD10 [7]. Variable expression for CD 34, myeloperoxidase and CD20 has been reported [1,7]. CD3 should be negative [1,8]. In our case due to the unusual clinical context the differential diagnosis was broad. It included the spectrum of pediatric small round blue cell malignancies. The major entities to consider were [6,9] :
• T LBL: frequent mediastinum’s involvment and at least one positive pan T-cell marker.
• Granulocytic sarcoma is positive for MPO and CD34.
• Rhabdomyosarcoma: presence of rhabdomyoblasts and/ or positivity for desmin, myogenin and myoD1 are diagnostic key features.
• Undifferentiated neuroblastoma: negativity for neuron‐specific enolase (NSE) CD56 with LCA positivity can rule out the diagnosis.
• Ewing’s Sarcoma: consisting of sheets of monomorphus cohesive cells and positivity for CD99 (may also be expressed in immature lymphoid neoplasms) [1,6,9].
Prognostic factors include only stage of disease [1]. In the European Intergroup EURO-LB02 trial; stage III disease was associated with a significant reduction in event free survival (EFS) [10]. ALL-type treatment regimens represent the current Frontline treatment in pB-LBL [1]. Autologous or allogenic hematopoietic stem cell transplant (HSCT) are indicated in patients with refractory B LBL [1]. CD19 and/or CD22 might be novel therapeutic targets [10].
Conclusion
In conclusion, we have reported the clinical and histopathological features of this rare hematopoietic malignancy with unusual presentation. Positive diagnosis requires careful morphological analysis and a large immunohistochemistry panel to rule out other round blue cell tumors.
References
- Burkhardt B, Hermiston ML (2019) Lymphoblastic lymphoma inchildren and adolescents: Review of current challenges and futureopportunities. Br J Haematol 185: 1158-1170.
- Ducassou S, Ferlay C, Bergeron C, Girard S, Laureys G, et al. (2011)Â Clinical presentation, evolution, and prognosis of precursor B-cell lymphoblastic lymphoma in trials LMT96, EORTC 58881, and EORTC58951. Br J Haematol 152: 441-451.
- Schmidt E, Burkhardt B (2013) Lymphoblastic lymphoma in childhood and adolescence. J Pediatr Hematol Oncol 30: 484-508.
- Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. (2017) WHOÂ Classification of tumours of haematopoietic and lymphoid tissues, Revised 4th ed. International Agency for Research on Cancer.
- Cortelazzo S, Ponzoni M, Ferreri AJ, Hoelzer D (2011) Lymphoblastic lymphoma. Crit Rev Oncol Hematol 79: 330-343.
- Oschlies I, Burkhardt B, Chassagne-Clement C, d'Amore ES, Hansson U, et al. (2011) Diagnosis and immunophenotype of 188 pediatric lymphoblastic lymphomas treated within a randomized prospective trial: Experiences and preliminary recommendations from the European childhood lymphoma pathology panel. Am J Surg Pathol 35: 836-844.
- Wenzinger C, Williams E, Gru AA (2018) Updates in the pathology of precursor lymphoid neoplasms in the revised fourth edition of the who classification of tumors of hematopoietic and lymphoid tissues. Curr Hematol Malig Rep 13:275-288.
- Chaudhary N, Borker A (2013) Pediatric precursor B-cell lymphoblasticlymphoma presenting with extensive skeletal lesions. Ann Med Health Sci Res 3: 262-264.
- Parham DM (2010) Modern diagnosis of small cell malignancies of children. Surg Pathol Clin 3: 515-551.
- Landmann E, Burkhardt B, Zimmermann M, Meyer U, Woessmann W, etal. (2017) Results and conclusions of the European Intergroup EURO-LB02 trial in children and adolescents with lymphoblastic lymphoma.Haematologica 102: 2086-2096.
Citation: Sabrine D, Ismail B, Lamia R, Najat L (2021) B cell Lymphoblastic Lymphoma Presenting as a Paravertebral Mass in a Child: A Case Report. Diagn Pathol Open S3: 008. DOI: 10.4172/2476-2024.s3.008
Copyright: © 2021 Derqaoui S. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Tools
Article Usage
- Total views: 2576
- [From(publication date): 0-2021 - Dec 09, 2025]
- Breakdown by view type
- HTML page views: 1742
- PDF downloads: 834