Research Article Open Access
Biodegradation of Natural Rubber Latex of Calotropis procera by Two Endophytic Fungal Species
Nadia H Mohamed1,2, Mady A Ismail1, Wael M Abdel-Mageed3,4 and Ahmed AM Shoreit1*
1Department of Botany and Microbiology, Faculty of Science, Assiut University, Assiut, Egypt
2Biology Department, Faculty of Science and Art, Samtah, Jazan University, Saudi Arabia
3Pharmacognosy Department, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia
4Pharmacognosy Department, Faculty of Pharmacy, Assiut University, Assiut 71526, Egypt
- *Corresponding Author:
- Ahmed Abdelfattah Mohamed Shoreit
Department of Botany and Microbiology
Faculty of Science, Assiut University, Assiut, Egypt
Tel: +201003406161
Fax: +20882342708
E-mail: ashoreit1968@yahoo.com
Received Date: December 31, 2016; Accepted Date: January 17, 2017; Published Date: January 19, 2017
Citation: Mohamed NH, Ismail MA, Abdel-Mageed WM, Shoreit AAM (2017) Biodegradation of Natural Rubber Latex of Calotropis procera by Two Endophytic Fungal Species. J Bioremediat Biodegrad 8:380. doi: 10.4172/2155-6199.1000380
Copyright: © 2017 Mohamed NH, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
In this study eight species related to Aspergillus (3 species), Fusarium (1), Penicillium (1), Emericella (1), Nigrospora (1) and Trichoderma (1) were isolated from leaves and latex of Calotropis procera. Only P. chrysogenium and A. niger were able to grow on natural rubber but other species were not. The degrading ability of (Penicillium chrysogenum and Aspergillus niger), isolated from latex of Calotropis procera was assessed. The degradation of rubber latex was determined by measuring the increase in protein content of the fungus (mg/g dry wt), reduction in molecular weight (g/mol) and inherent viscosity (dl/g) of the latex. Moreover, the degradation was also confirmed by observing the growth of these species strain using scanning electron microscopy.
Keywords
Calotropis procera; Penicillium chrysogenum; Aspergillus niger; Biodegradation; Natural rubber; Endophytic fungi
Introduction
Calotropis is a small genus belonging to family Asclepiadaceae, Calotropis gigantea and C. procera are the two most common species in this genus used in traditional medicine, and only these two species are usually reported in literature [1], and are distributed in tropical and subtropical Africa, Asia, and America [2-5].
Calotropis procera (Ait) WT Aiton is a small wild-growing tropical plant, known by different names like sodom apple, usher, dead sea apple, swallow wort, and milk weed [6,7]. All parts of this plant have the ability to produce large quantities of latex when cut or broken [7-9].
The plant latex could be separated after centrifugation into three layers: top layer containing Natural Rubber; middle layer of serum, and the bottom layer of lipids [9,10]. The chemical composition of latex is very complex, it contains (wt/wt) natural rubber poly (cis-1, 4-isoprene) 25 to 35%, which consider the main constituent of Natural rubber latex, and it is consisting of isoprene units C5H8 in the cisconfiguration, a highly unsaturated hydrocarbon, with an average molecular weight about 106 Da [11-13].
Natural Rubber is a basic material for manufacturing tires, latex gloves, condoms, seals, balloons, balls for sports and many other things [14]. Degradation potential of the microorganism is dependent upon colonization on natural rubber and accompanied by a loss in the weight of the rubber hydrocarbon and a decline in the relative viscosity of the polymer solution [13].
Many other reports have been published on the biodegradation of natural rubber hydrocarbon as a sole carbon source by bacteria [15-22], fungi [13,23,24]. Several microbes were used to biodegrade Calotropis procera latex [25,26].
The aim of this work is to assess the ability of mesophilic endophytic fungi isolated from Calotropis procera leaves and latex on sugar-free Czapek agar and to test their ability to metabolize and degrade rubber latex of C. procera as a sole carbon source.
Materials and Methods
Sampling, fractionation and preparation of natural rubber latex
The latex of Calotropis procera plant was obtained by taping method. Fresh latex was centrifuged at 17,000 rpm for 20 min at 4°C in SR4000 Prolabo centrifuge (France). latex was separated into three layers: a sticky top layer containing Natural Rubber; clear middle fraction of serum, and a small quantity of lipids at the bottom of the tube (Figure 1).
The natural rubber fraction was separated and washed three times by deionized water to remove impurities and dried at 30°C for 24 h, A known weight (20 mg) of the prepared natural rubber was dissolved in 2 ml of tetrahydrofuran (THF), then its molecular weight was determined by gel permeation chromatography (GPC) [9,13].
Isolation of endophytic fungi from C. procera leaves and latex, calculation of colonizing frequency and Identification of endophytic fungi were done by the current authors according to these papers [13,21,22].
Methods used for measuring the capabilities of endophytic fungi to degrade pure natural rubber Growing of isolates on solid media coated with natural rubber film for 15 days of incubation
After preparation of pure natural rubber (NR), It was dissolved in tetrahydrofuran (THF) (20 mg/2 ml THF), and spotted on to agar media (each 50 μl/colony), then left in an oven at 30°C until THF is evaporated and the NR became white in appearance. Fungi were inoculated with tooth pick on the top of pure natural rubber (NR) spot, incubated for 15 days at 28°C. The diameters of colonies were measured, then fungi which grow well in solid media Growing of in liquid media for measuring biodegradation ability after 30 days. At the end of incubation period the rubber pieces and microorganisms were separated by centrifuging at 10000 rpm for 15 min, microorganisms (bottom layer) were subjected for protein determination while natural rubber pieces (top layer) were collected, washed with deionized water, and dried at 30°C and then subjected for analytical methods (molecular weight and viscosity) as well as SEM photography has been described before [13,22].
Protein determination
The total protein pellet was determined according to the method adopted by Lowry et al. [27].
Molecular weight determination by Gel Permeation Chromatography (GPC)
A known weight of natural rubber was dissolved in 2 ml THF and passed through a 0.45 μm-pore size filter. Gel permeation chromatogram of the the model Agilent technologies 1100, Germany (present in National Research Center, Doki, Cairo) was used. THF was used as the eluent with a flow rate 1 mL / min. Polymethylmethacrylate and polystyrene standards were used to calibrate the columns. The refractive index detector 104 A°, Colum PL gel Micrometer 100; 10000; 100000. The GPC apparatus was run under the following conditions: flow rate=2.000 ml/min., injection volume=100.000 μl, sample concentration=1.000 g/l. The values of molecular weight were calculated by means of a computer program [13,22].
Scanning Electron Microscopy (SEM)
The morphological changes during growth of the organism on natural rubber was also studied by scanning electron microscopy. The inoculated samples as well as the control were fixed overnight in glutaraldehyde 5%, then dried at 50°C. The samples were mounted on metal stubs and coated with gold and palladium (Jeol JFC1100E Iosputtering Device). Micrographs were taken with a Joel JSM- 4500 LV electron microscope operating at 15 KV in Assiut University, electron Microscopy unit [13,22].
Results and Discussion
Isolation and identification of fungi
Six hundred and seventy-five segments of leaf and 15 latex sample of C. procera were processed for the isolation of endophytic fungi. Eight species related to 6 genera were isolated and these were Aspergillus niger, A. fumigatus, A. flavus, Emericella nidulans, Fusarium oxysporum, Nigrospora oryzae, Penicillium chrysogenum and Trichoderma harzianum. Isolation of endophytic fungi from C. procera leaves and latex, calculation of colonizing frequency and Identification of endophytic fungi were done by the current authors (data not shown).
Growth of mesophilic endophytic fungi on solid media coated with natural rubber film after 15 days of incubation
Eight isolates (representative for the species isolated) were tested for their ability of growing on sugar-free Czapek agar coated with thin film of pure natural rubber (3 replicate agar plates for each isolate). Only 2 isolates related to Penicillium chrysogenum and Aspergillus niger could grow on pure natural rubber film (Table 1 and Figure 2), while Emericella nidulans, A. fumigatus, A. flavus, Nigrospora oryzae, Fusarium oxysporum and Trichoderma harzianum showed only growth on sugar-free Czapek agar supplemented with crude latex [28-32].
| Analysis parameters | Growth diameter (cm) | Protein content mg/g dry wt. | Viscosity measurements dl/g | Molecular weight g/mol× 104 | ||
|---|---|---|---|---|---|---|
| X ± SD | X ± SD | |||||
| Days of incubation | 15 | 15 | 30 | 15 | 30 | 30 |
| Control (natural rubber) | 0 | 6.3 ± 0.1 | 8.08 | |||
| P.chrysogenum | 2.83 ± 0.1 | 2.71* ± 0.1 | 5.23* ± 0.1 | 6.0 ± 0.10 | 5.5* ± 0.1 | 4.88*(40.0) |
| A.niger | 2.3 ± 0.1 | 1.71* ± 0.1 | 4.22* ± 0.1 | 6.2 ± 0.20 | 5.9* ± 0.2 | 5.82*(28.0) |
Table 1: Degradation of C. procera rubber fraction by P. chrysogenum and A. niger through determining protein content (mg/g mycelial dry wt); viscosity (dl/g) and molecular weight (g/mol) after shaking incubation for 15 and 30 days at 25°C respectively.
Biodegradation abilities of Penicillium chrysogenum and Aspergillus niger grown in liquid media after 15 and 30 days of incubation
Fungal protein, viscosity and molecular weight of P. chrysogenum: Protein content of P. chrysogenum grown on natural rubber increased significantly from 2.71 mg/g mycelial dry weight after 15 days to 5.23 mg/g after 30 days of incubation in liquid Cz medium. On the other hand rubber viscosity decreased significantly from 6.0 dl/g after 15 days to 5.5 dl/g after 30 days of incubation.
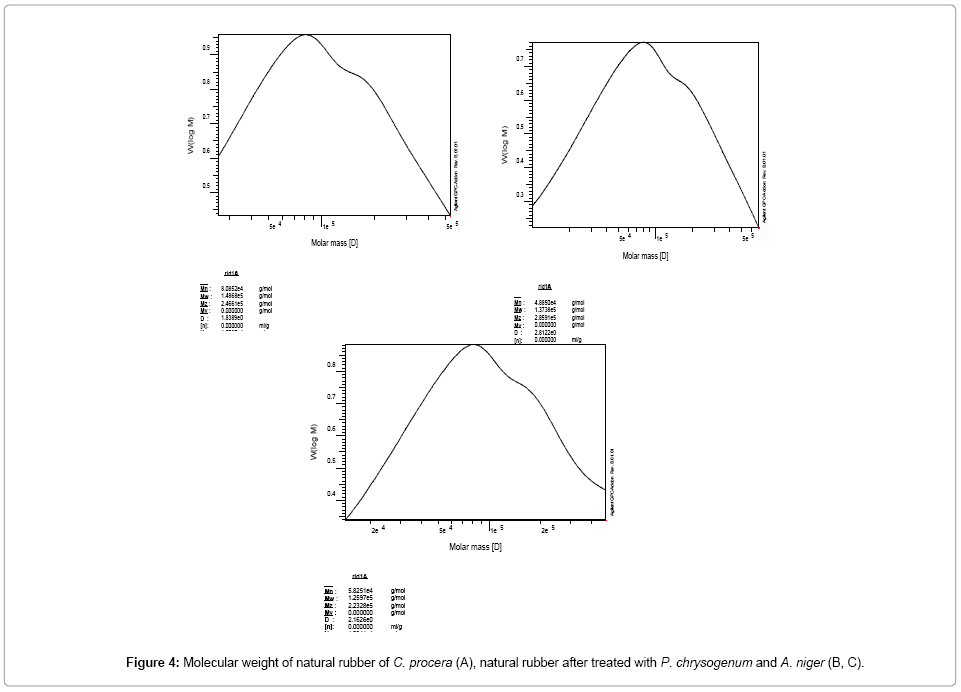
Molecular weight of natural rubber (that was previously inoculated with P. chrysogenum), decreased from 8.08 × 104 (in control medium) to 4.88 × 104 g/mol after 30 days (degradation rate of P. chrysogenum after 30 days=40.0% comparable with the control (Table 1; Figure 3).
Figure 3: Degradation of Calotropis latex rubber (20 mg/25 ml Cz small medium under shaking 150 rpm at 25°C for the endophytic fungi, A. niger and P. chrysogenum as determined by Scanning Electron Microscope (SEM): uninoculated rubber (A), inoculated rubber latex after 15 days (B) and 30 days (C) with A. niger or P. chrysogenum.
Fungal protein, rubber viscosity and molecular weight of A. niger: Protein content for A. niger grown on natural rubber increased significantly from 1.71 mg/g mycelial dry weight after 15 days to 4.22 mg/g after 30 days. On the other hand rubber viscosity decreased significantly from 6.2 dl/g after 15 days to 5.9 dl/g after 30 days of incubation.
Molecular weight of separated natural rubber inoculated with A. niger, decreased from 8.08 × 104 to 5.82 × 104 g/mol after 30 days (degradation rate of A. niger after 30 days=28.0% comparable with the control (Table 1, Figure 3).
1. Values in brackets represent percentage of degradation rate of natural rubber by the endophytic fungus in case of viscosity and molecular weight after 30 days.
2. Figures in the table are means of three replicates ± standard deviation.
3. *Values Significant at PC 0.05 level.
In this respect, Ismail et al. [13], showed high degradation rate of poly (cis-1, 4-isoprene) rubber (Figure 4) by Aspergillus terreus 31.04% and A. flavus 28.73% after 30 days and comparable with the control. The ability was also determined by measuring the increase in protein content of each fungus, reduction in molecular weight and inherent viscosity and observing the growth using scanning electron microscopy (SEM).
Roy et al. [17] stated also that significant decrease in the molecular weight of the natural rubber was treated with Aspergillus sp. after 20 weeks (MW 2.70 × 105 in the control to 1.90 × 104). Indicating that Aspergillus sp. was able to degrade the natural rubber.
Also Nayanashree and Thippeswamy [24] found that Aspergillus niger and Penicillium species effectively degraded Hevea brasiliensis rubber (Figure 4).
Scanning Electron Microscopy (SEM)
The 15 and 30 days old inoculated natural rubber samples with P. chrysogenum and A. niger were photographed using scanning electron microscopy (SEM). These SEM microphotographs clearly indicate the colonization of the fungus on the natural rubber surface (Figure 3). In this respect, the colonization and/or growth on the rubber surface using by SEM micrographs have been observed for Aspergillus terreus, Aspergillus flavus and Myceliophthora thermophila [13], Aspergillus sp. and Pseudomonas sp. [17].
Conclusion
The degradation process of natural rubber of C. procera by P. chrysogenum and A. niger were proven by the increase in protein content, decrease in both molecular weight and inherent viscosity, and by observing the fungal growth on the surface of natural rubber using scanning electron microscopy (SEM). The current results demonstrate that the degrading fungal isolates could be used as promising tools for the removal of disposal of rubber products.
Conflict of Interest Statement
We declare that we have no conflict of interest.
Acknowledgements
Authors thanks Assiut University for funding and technical support.
References
- Singh U, Wadhwani AM, Johri BM (1983) Dictionary of economic plants in India. ICAR, New Delhi, India.
- Rahman MA, Wilcock CC (1991) A taxonomic revision of Calotropis (Asclepiadaceae). Nordic Journal of Botany 11: 301-308.
- Begum N, Sharma B, Pandey RS (2011)Evaluation of insecticidal efficacy of Calotropis procera and Annona squamosa ethanol extracts against Musca domestica. J Biofertil Biopestici 1: 101-106.
- Doshi H, Satodiya H, Thakur MC, Parabia F, Khan A(2011) Phytochemical screening and biological activity of Calotropis procera (Ait). R. Br. (Asclepiadaceae) against selected bacteria and Anopheles stephansi larvae. International Journal of Plant Research1: 29-33.
- Ibrahim SR, Mohamed GA, Shaala LA, Banuls LM, Kiss R, et al. (2015) Calotroposides H–N, new cytotoxic oxypregnane oligoglycosides from the root bark of Calotropis procera. Steroids 96: 63-72.
- TourN, TaleleG (2011) Anti-inflammatory and gastromucosal protective effects of Calotropis procera (Asclepiadaceae) stem bark. J Nat Med 65: 598-605.
- Mohamed NH, Liu M, Abdel-Mageed WM, Alwahibi LH, Dai H, et al. (2016) Cytotoxic cardenolides from the latex of Calotropis procera. Bioorganic & Medicinal Chemistry Letters 25: 4615-4620.
- Ramos MV, Aguiar VC, MeloVM, Mesquita RO, Silvestre PP, et al. (2007) Immunological and allergenic responses induced by latex fractions of Calotropis procera (Ait). R Br Journal of Ethnopharmacology 111: 115-122.
- Mohamed NH, Ismail MA, Abdel-Mageed WM, Shoreit AAM (2014) Antimicrobial activity of latex silver nanoparticles using Calotropis procera. Asian Pac J Trop Biomed 4: 864-871.
- Kang H, Kang MY, Han KH (2000) Identification of Natural Rubber and Characterization of Rubber Biosynthetic Activity in Fig Tree. Plant Physiology 123:1133-1142.
- Rifaat HM,Yosery MA (2004) Identification and characterization of rubber degrading Actinobacteria. Applied Ecology and Environmental Research 2: 63-70.
- Rose K, Steinbüchel A (2005) Biodegradation of natural rubber and related compounds: recent insights into a hardly understood catabolic capability of microorganisms. Applied Environmental Microbiology 71: 2803-2812.
- Ismail MA, Mohamed NH, Shoreit AAM (2013) Degradation of Ficus elastica rubber latex by Aspergillus terreus, Aspergillus flavus and Myceliophthora thermophila. International Biodeterioration & Biodegradation 78: 82-88.
- Bode HB, Zeeck A, Hahn KP, Jendrossek D (2000) Physiological and chemical investigation into microbial degradation of Synthetic poly (cis-1, 4-isoprene). Applied and Environmental Microbiology 66: 3680-3685.
- Rose K, Tenberge KB, Steinbüchel A(2005) Identification and characterization of genes from Streptomyces sp. strain K30 responsible for clear zone formation on natural rubber latex and poly(cis-1, 4-isoprene) rubber degradation. Biomacromolecules 6: 180-188.
- Ibrahim EMA, Arenskötter M, Luftmann H, Steinbüchel A (2006) Identification of poly (cis-1, 4-isoprene) degradation intermediates during growth of moderately thermophilic Actinomycetes on rubber and cloning of a functional lcp homologue from Nocardia farcinica strain E1. Appl Environ Microbiol 72: 3375-3382.
- Roy RV, Das M, Banerjee R, Bhowmick AK (2006) Comparative studies on rubber biodegradation through solid-state and submerged fermentation. Journal of Process Biochemistry 41: 181-186.
- Warneke S, Arenskötter M, Tenberge KB, Steinbüchel A (2007) Bacterial degradation of poly (trans-1, 4-isoprene) (gutta percha). Microbiology 153: 347-356.
- Bröker D, Dietz D, Arenskötter M, Steinbüchel A (2008) The genomes of non-clearing-zone-forming and natural rubber-degrading species Gordonia polyisoprenivorans and Gordonia westfalica harbor genes expressing LCP activity in Streptomyces strains. Applied and Environmental Microbiology 74: 2288-2297.
- YikmisM, Arenskötter M, Rose K, Lange N, Wernsmann H (2008) Secretion and Transcriptional Regulation of the Latex-Clearing Protein, Lcp, by the Rubber Degrading Bacterium Streptomyces sp. Strain K30. Applied and Environmental Microbiology 74: 5373-5382.
- Hesham AE, Mohamed NH, Ismail MA, Shoreit AAM (2012)16S rRNA gene sequences analysis of Ficus elastic rubber latex degrading thermophilic Bacillusstrain ASU7 isolated from Egypt.Biodegradation 23: 717-724.
- Hesham AE, Mohamed NH, Ismail MA, Shoreit AAM(2015) Degradation of Natural Rubber Latex by New Streptomyces labedae Strain ASU03 Isolated from Egyptian Soil. Microbiology 84: 351-358.
- Borel M, Kergomard A, Renard MF (1982) Degradation of natural rubber by fungi imperfecti. Agricultural Biology and Chemistry 46: 877-881.
- Nayanashree G, Thippeswamy B(2013) Natural Rubber Degradation byAspergillus Niger and Penicillium Sp. International Journal of Recent Scientific Research 4: 1337-1341.
- Sharma DK, Tiwari M, Arora M, Behera BK (1997) Microbial transformation and biodegradation of Calotropis procera latex towards obtaining value added chemicals, pharmaceuticals, and fuels. Petroleum Science and Technology 15: 137-169.
- Behera BK, Arora M, Sharma DK (2000) Studies on Biotransformation of Calotropis Procera Latex - A Renewable Source of Petroleum, Value-Added Chemicals, and Products. Energy Sources 22: 781-807.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein Measurement with the Folin Phenol Reagent. Journal of Biological Chemistry 193: 265-275.
- Zyska BJ (1981) Rubber. Microbial Biodegradation, Academic Press, New York, USA, pp: 323-385.
- Aly KI, Wahdan H, Hussein AM (2009) New polymer syntheses.Part 43: Novel polyamides- Based Diarylidenecyclopentanone: synthesis, characterization, and corrosion inhibition behavior. Journal of Applied Polymer Science 112: 513-523.
- Pandy BP (2012) Plant Anatomy. 6th edn. S Chand and Company Ltd., New Delhi, India, pp: 57-58.
- Shibuya H, Zhang R, Park JD, Baek NI, Takeda Y, et al. (1992) Indonesian medicinal plants. Chem Pharm Bull 40: 2647-2653.
- Strobel G, Daisy B (2003) Bioprospecting for microbial endophytes and their natural products. Microbiology and Molecular Biology Reviews 67: 491-502.
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 6598
- [From(publication date):
January-2017 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 5484
- PDF downloads : 1114