Research Article Open Access
Biosorption Studies and Kinetics on Textile Effluent Treatment Using Packed Bed Reactor
| Bharath Ganesan*, Karthikeyan Mayakrishnan, Venkatesaprabhu and Ponmurugan Ponnusamy | |
| Department of Biotechnology, K.S. Rangasamy College of Technology, Tiruchengode, Tamil Nadu, India | |
| Corresponding Author : | Bharath Ganesan Department of Biotechnology K.S. Rangasamy College of Technology Tiruchengode, Tamil Nadu, India Tel: +91 9047976171 E-mail: bharathganesh.m@gmail.com |
| Received May 23, 2014; Accepted June 28, 2014; Published July 02, 2014 | |
| Citation: Ganesan B, Mayakrishnan K, Venkatesaprabhu, Ponnusamy P (2014) Biosorption Studies and Kinetics on Textile Effluent Treatment Using Packed Bed Reactor. J Bioremed Biodeg 5:233. doi:10.4172/2155-6199.1000233 | |
| Copyright: © 2014 Ganesan B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
|
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
The utilization of Aspergillus niger biomass for the treatment of effluent collected from the dying units provides an insight into the potential of fungal biomass in treatment of textile effluent. The decolorization capability of the biomass was observed under shake flask studies using UV spectrophotometry and packed bed column studies were carried out for analyzing biosorption efficacy in a pilot scale. The process parameters maintained were, retention time varying from 80 to 26 min, flow rate from 2 ml/min to 6 ml/min and porosity of the column as 0.425. The dye decolorization by the fungal biomass and the metal adsorption was found to be maximum at 2 ml/min of flow rate and 80 minutes of retention time. The adsorption isotherms Langmuir and Freundlich were evaluated for the significance of the adsorption process. Specific metal uptake rate of the biomass has been studied to identify the rate of metal uptake by the biomass.
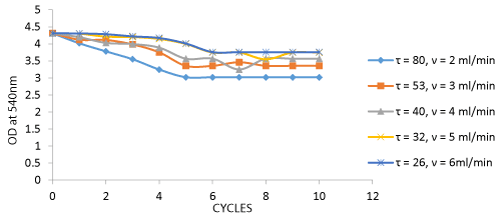
The above graph represents data for the rate of decolorization by coir (control). During the process bed saturation was found at 6 cycles at a flow rate of 2 ml/min. The number of cycles for decolorization increased as the flow rate increased. The analysis showed that coir also influences the process of adsorption (Figure 2).
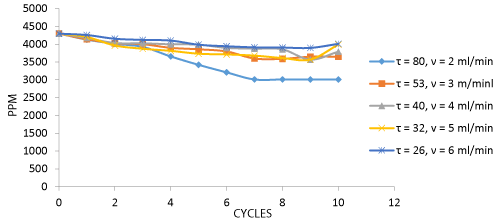
The graph above represents data for the rate of biosorption by coir (control). Cr uptake was maximum at 2 ml/min of flow rate at the end of 8 cycles and the efficacy decreased in biosorption of total Cr due to the increase in flow rate (Figure 3).
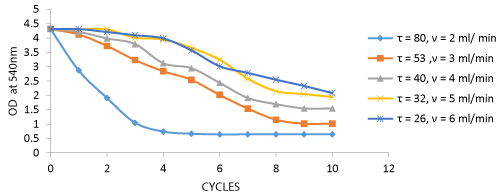
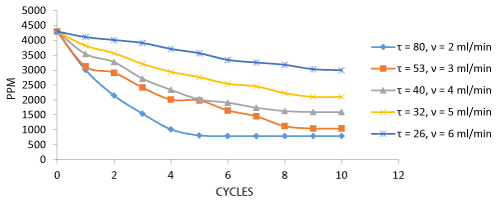
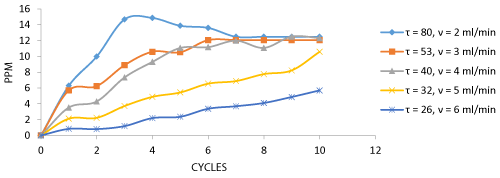
The Decolorization of dyes and the number of cycles for decolorization at various flow rates are depicted in the graph. The dye decolorization was found to be maximum at a flow rate of 2 ml/min providing maximum retention time for the decolorization process which provides maximum dye Decolorization at 5 cycles and the number of cycles increased due to increase in flow rate (Figure 4). The biosorption of total Cr and number of cycles for biosorption process at various flow rates are depicted in the graph.
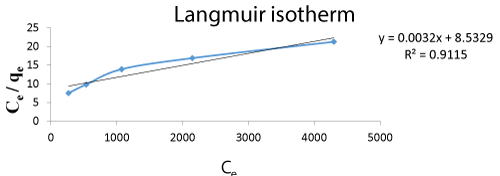
The Langmuir isotherm was analysed and showed the process of Cr biosorption occurred in a monolayer. The Qmax value was obtained from the process showing that the interaction between the sorbent and the sorbate at 4 ml/min of flow rate was 8.5329. The R2 value 0.9115 shows the isotherm model fits to the reaction.
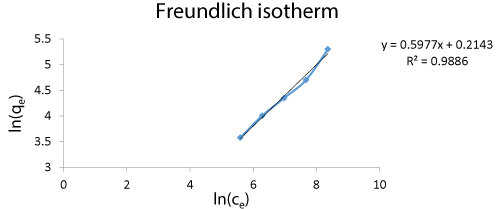
Freundlich isotherm was analyzed for the determination of heterogeneity of the process and to determine degree of non-linearity. The process of Cr biosorption was nonlinear. The R2 value 0.9886 shows the isotherm model for the pilot scale process thus depicting the overall process to be non-linear. The Langmuir isotherm and the Freundlich isotherm have a significant fit and shows that the interaction between the biomass and Cr occurs at a more effective rate.
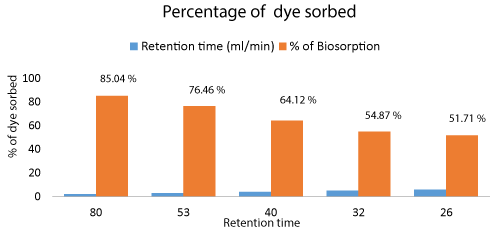
The dye decolorization by Aspergillus niger biomass was analyzed from samples collected after each cycle. The results show that maximum of 85.01% of the effluent was decolorized at 80 minutes of retention time in the column. There is a gradual decrease in the decolorization as retention time decreases due to increased flow rate. This shows that flow rate plays an important role in the biosorption process (Figure 9).
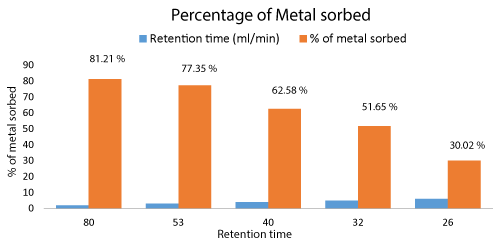
The Cr metal biosorption by Aspergillus niger biomass was analyzed from the collected samples after each cycle. 81.21% of Cr metal was biosorbed by treating of effluent in the packed bed column which was maximum during 2 ml/min of flow rate. The efficiency of the metal removal depended on retention time. The higher the retention time of effluent in the column the higher is the metal biosorption.
References
- Ahmad I, Ansari MI, Aqil F (2006) Biosorption of Ni, Cr and Cd by metal tolerant Aspergillus niger and Penicillium sp. using single and multi-metal solution. Indian J Exp Biol 44: 73-76.
- Saratale RG, Palmiere G, Liu WX (2009) Outlook of bacterial of azo dyes: a review. International Journal of Environmental Research and Public Health 179: 790-243.
- Eltaief Khelif, Nigam P, Marchant R, Robinson T (2008) ‘‘Aerobic Decolorization of the indigo dye-containing textile wastewater using continuous combined bioreactors’’. J Hazardous Material 152: 683-689.
- Kikani BA, Shukla RJ, Singh SP (2006) Biosorption of heavy metal chromium polluted effluent: Influence of sediment properties. Eng Life Science.
- Marrot B, Zheng XH, Hardin I (2001) Decolorization of the reconstituted textile effluent by different process treatments: Enzymatic catalysis, coagulation/flocculation and nanofiltration processes. Desalination 268: 27-37.
- Mukhopadhyay M, Couto SR, Maximo CS (2008) Copper biosorption in a column of pretreated Aspergillus niger biomass. Desalination 269: 29-37.
- Sivakumar K, Swaminathan K, Shetty K (2011) Assessment Studies On Wastewater Pollution By Textile Dyeing And Bleaching Industries At Karur, Tamil Nadu. J Chem 4: 264-269.
- Upadhye VB, Tezsos M, Spinti M (2004) Kinetics and mechanism of the bacterial and ferric sulphate oxidation of effluent. Hydrometallurgy 75: 99-110.
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 | Figure 5 |
 |
 |
 |
 |
| Figure 6 | Figure 7 | Figure 8 | Figure 9 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 13931
- [From(publication date):
September-2014 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 9290
- PDF downloads : 4641
