Cardiac Ischemia and Angina Pectoris without Flow-Limiting Coronary Artery Disease (Coronary Syndrome X)
Received: 16-Nov-2015 / Accepted Date: 31-Dec-2015 / Published Date: 12-Jan-2016
Abstract
Coronary ischemic disease represents an important clinical problem worldwide. Each year millions of people (mostly women) with AP by visual analysis of biplane coronarography are found to have normal coronary arteries. However, as demonstrated from autoptic and ultrasound intravascular studies in vivo it would be more appropriate to say that coronarography did not detect obstructive or flow-limiting epicardial coronary artery disease. The pathology arises from a complex pathology in the microcoronary vessels and is usually called cardiac X syndrome. The paper describes the etiology and pathogenies of this pathology. Cardiac X syndrome is not a benign pathology because it induces angina pectoris and myocardial ischemia. Given the association between this pathology and consequent downstream morbidity, it seems logical to assess long-term cardiovascular outcomes by appropriate analysis. Drugs used in coronary artery disease are prescribed in cardiac X syndrome, but we lack data from double-blind randomized trials. Specific research to target interventions against this cardiac pathology CSX would be necessary.
Keywords: Cardiac Ischemia; cardiac X syndrome
77721Introduction
Coronary ischemic disease (CID) is a worldwide frequent pathology with increasing prevalence. Angina pectoris (AP) is a cardinal symptom of CID. Typical AP meets all of the following criteria: retrosternal chest discomfort of characteristic quality and duration; provoked by exertional or emotional stress; is relieved by rest and/or nitrates within minutes [1,2].
It was stated that ‘no patient with AP failed to show occlusion in at least one of the major coronary arteries’ [3] and flow-limiting coronary artery disease (CAD) causing ischemia was accepted as the cause of AP. However, this assumption is not unconditional because AP may also occur in other diseases, such as e.g. hypertrophic cardiomyopathy, severe aortic stenosis, profound anemia, and carboxy hemoglobin intoxication.
In 1967 two papers [4,5] described patients with typical AP and sometimes dyspnea and neurovegetative symptoms (e.g. perspiration, tachycardia and dizziness) without epicardial CAD (CAD). This cardiac pathology is usually called cardiac syndrome X (CSX) and is not rare [6-12].
Case Report
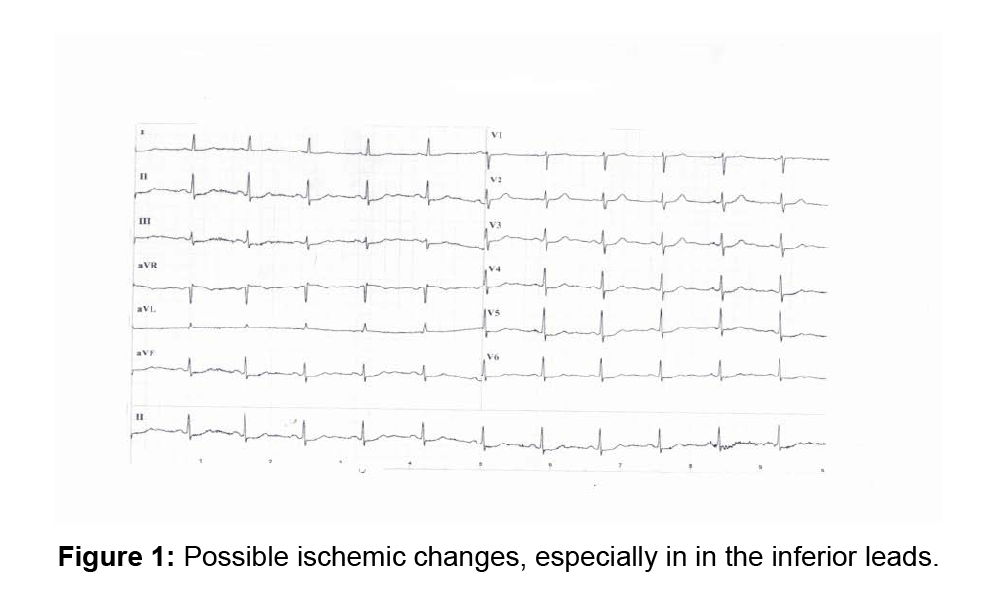
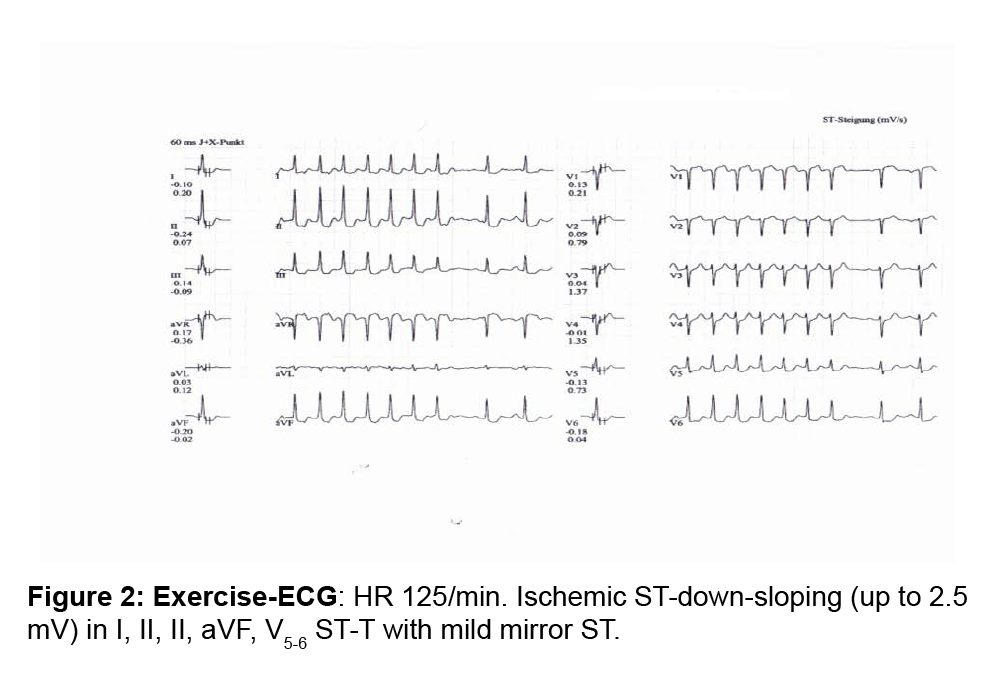
A 53-year old Caucasian woman had stable typical AP which was questionably relieved by sublingual nitroglycerin. Her father died at the age of 55 years because of acute myocardial infarction. The patient had no other cardiovascular risk factors. The resting ECG showed ischemic changes (Figure 1). Plasma levels of troponin, measured during and several hours after episodes of AP, were not increased. Echocardiography detected only a moderately impaired left ventricular relaxation. A physical stress-test was interrupted at 80 watt/min because of moderate AP with corresponding ischemic ECG changes. The ECG (Figure 2), showed a heart rate of 125/m with sometimes undetectable P waves, and ischemic ST-down-sloping (up to 2.5 mV) in I, II, II, aVF, V5-6 ST-T with mild mirror ST-up-sloping in aVR and V1. Stressechocardiography detected no dyskinesia, the left ventricular volume remained unchanged, the LVEF increased from 62 to 73%, the right ventricular function was normal. AP lasted up to 6 min and was not relieved by 0.8 mg sublingual nitroglycerin. The patient had typical AP with ischemic ECG changes, but troponin values were normal and exercise did not induce dyskinesia. We diagnosed a CSX but the referring physician required a ventriculo-coronarography: the left ventricle had normal volume and contraction and the coronary arteries (CA) were normal. The patient was treated with ranolazine 500 mg bid and rosuvastatin 5 mg/day. Follow-up: after 3 weeks the number of episodes and intensity of AP decreased by more than 50%, after 3 months the ST-segment of the resting ECG was normal; a stress-test under therapy with ranolazine was interrupted at 150 watt because of mild AP at a heart rate of 145/m, the ECG showed a -0.2 mV STdown- sloping in V4-5 with ascending morphology. In comparison to the first stress-test the patient could exercise 3 min longer before mild AP occurred and the work load increased by 50 watt.
Pathophysiology of ischemia and AP in CSX
Understanding the pathophysiology of CSX is particularly challenging because of heterogeneous etiology and prominent comorbidities [6-15] Multifactorial mechanisms interplay and cause myocardial ischemia and AP in CSX-patients with ‘normal’ CA. The most important mechanisms (Table 1) are endothelial dysfunction (ED), micro-vascular dysfunction (MVD), sometimes combined with coronary micro-vascular spams (CMVS), and epicardial CA spasm (CAS).
| Endothelial dysfunction is present in the overwhelming majority of cases. |
| Microvascular dysfunction is present in nearly 50% of cases, in some cases together with coronary microvascular spasms. |
| Epicardial coronary artery spam is present in about 4% of cases. |
| All these mechanisms interplay, contribute to microcoronary ischemia, reduce collateral perfusion and, in the occurrence of a severe coronary flow reduction, such after a thrombotic event, limit the reperfusion. |
Table 1: Mechanisms of ischemia and AP in CSX.
ED is a systemic disorder which is detected in patients with AP with/without CAD. ED results from vascular injury due to arteriosclerotic inducing factors, anticipates the angiographic documentation of anatomical lesions and has been linked with major adverse cardiovascular outcomes (MACE) in many conditions [15-23]. Known pathophysiologic mechanisms related to ED (Table 2).
| Oxidative stress has several negative effects: |
|---|
| • Increases intracellular superoxide; |
| • Inactivates nitric oxide and formation of peroxinitrite; |
| • Induces nitric oxide synthase uncoupling; |
| • Reduces nitric oxide signaling; |
| • Inhibits prostacyclin formation; |
| • Stimulates endothelin expression; |
| • Inhibits the activity of soluble guanylate cyclase. |
Table 2: Mechanisms involved in endothelial dysfunction.
MVD is defined by low coronary reserve, i.e. a dysregulation of coronary blood flow which is not attributable to flow-limiting epicardial CAD. MVD results from either structural or functional mechanisms in the microvasculature and induces an inappropriate vasodilatation of the coronary microcirculation [8,9,18,24]. MVD has also been linked with MACE in several pathologies [22-26]. CMVS is a dangerous pathophysiologic mechanism which has been detected in CSX-patients who also have MVD and it is linked with MACE [26]. In the WISE study CXS-women with AP were tested with adenosine and nearly 50% had MVD, which was an independent predictor of MACE [22-24]. ED is present in large majority of CSX-patients, and in nearly 50% of cases is combined with MVD. ED and MVD independently predict MACE such as, after a 5.4-years follow-up, death, acute coronary syndrome (ACS), myocardial infarction (MI), stroke, and hospitalization for heart failure [19,24,26]. These MACE are consistent with the pathophysiologic progression that is expected in disorder involving the microvasculature [15,19,22-26].
CAS is the 3rd etiologic mechanism in CSX [15]. It is a proven cause of myocardial ischemia and AP in CID [15,26-31], is linked with some genetic background and may be induced by many pathologic mechanisms [9,11-13,24-30]. In CSX- patients CAS has been induced with acetylcholine [29], adenosine [24], serotonin, ergonovine, histamine and many other, less frequently causative agents: when the endothelium is intact the tested agents cause vasodilation by releasing nitric oxide, but this not the case in CSX [30]. In the WISE study CXSwomen were tested with acetylcholine and a CAS could be detected in nearly 4% of women [15]. This is the same frequency that is found in patients with AP due to CAD [28-30]. CAS has been linked with an ACS in more than 50% of CSX-patients [26,27,30] and in some cases it was thought to be the cause of MI [26,31-34].
Left ventricular wall motion in CSX
In the WISE study ventriculography data showed that resting left ventricular wall motion was preserved in CSX-women [15-17]. Cardiac magnetic resonance imaging provides excellent resolution to evaluate perfusion and the technique has proven that in CSX-patients there a reduced sub-endocardial perfusion [15,16,29,30]. Many years ago our groups studied left ventricular motility by kinetocardiography and pharmacologic stress-testing (with either isoproterenol or dopamine) in patients with AP with and without flow-limiting CAD. Pharmacologic stress-testing induced left ventricular dyskinesia in nearly 30% of CSXpatients and the dyskinesia changes were indistinguishable from those detected in CAD-patients [32,33]. Isoproterenol and dobutamine have the potential to worsen ED and MVD, which are frequent pathologies in CSX. It is thus understandable that CSX-patients report AP with detectable ischemic ECG changes. If ischemia is important, dyskinesia may occur. Briefly, in CSX symptoms and signs of cardiac ischemia may be indistinguishable from those of CAD-patients. The difference lies in the fact that in CSX the mechanisms of ischemia are micro-vascular and flow-limiting epicardial CAD is absent.
Normal’ coronary arteries
CA are usually defined as normal by visual analysis of biplane coronarography. However, when using biplane coronarography, it would be wiser to speak of non-obstructive epicardial CAD. Indeed, autoptic studies have proven that visual analysis of angiographically normal CA segments underestimates the extent of atherosclerosis. Also, intravascular ultrasound allows high quality cross-sectional imaging of CA in vivo [13]. The technique was used to study angiographically normal CA reference segments in 884 patients with symptomatic native epicardial CAD who were evaluated for percutaneous stenting therapy [14]. Only 60 (6.8%) of these angiographically normal CA reference segments were normal. The reference segments contained less calcific and dense fibrotic plaque and proportionately more soft plaque elements. Independent predictors of reference segment percent crosssectional narrowing were male gender, patient age, diabetes mellitus, hypercholesterolemia and presence of multi-vessel disease. Independent predictors of reference segment calcification were patient age and serum creatinine levels. In 723 patients undergoing percutaneous stenting reference segment percent cross-sectional narrowing was similar to that in patients studied for diagnostic purposes, but calcium was greater in reference segments of treated patients. It is of note that reference segment disease was not an independent predictor of subsequent angiographic restenosis or clinical events within 12 months of follow-up.
In the WISE study 100 consecutive CSX-women were studied by intravascular ultrasound and about 80% had atherosclerotic plaques [14]. Extrapolating these findings, in the WISE study most of 48% of women who were classified as having normal CA, probably had some coronary plaques [15].
Epidemiology of CSX
Most CSX-patients are women [4,17]. Coronarography has shown that no flow-limiting CAD is detected in nearly 75% of women with typical AP [13,17]. Depending on different registry data this cardiac situation is also found in 5 to 15% of men [9-16,19-23]. It must thus be accepted that millions of people (mostly women) will probably be identified each year with AP without CAD.
Long-term outcomes in CSX
In 1940 it was stated that CSX-patients did not have an increased high cardiovascular risk, did not require cardiologic care [3] and to a certain extent that general medical care was unnecessary [15]. This is not true. Pathology progresses in CSX. CXS-patients are not only disabled because of persisting symptoms, especially AP, but also have frequent MACE [8-15,17,23,26,34-39]. It was suggested that in CSXpatients the prevalence of long-term MACE may be similar to that of asymptomatic patients with CAD [15,37]. However, in spite of extensive knowledge, we are still uncertain about the real occurrence of MACE in CSX-patients with stable AP. Present data on long-term MACE are probably inaccurate. The statistical method of composite outcome measures is often used to collect data on outcomes, but the findings may obfuscate data and lead to inaccurate conclusions [40]. To improve the validity of the analysis of long-term complications and poor outcomes in ACS the weighted composite endpoints method has been used [41]. Unfortunately, up to know this approach has not been used to assess MACE in either CSX or CAD-patients. Thus, at present we know that CSX is not a benign condition as previously believed, but we ignore the exact prevalence of long-term MACE in CSX and, also, we cannot accurately compare the prevalence of MACE between CSXand CAD-patients.
Causes of MACE in CSX
By definition in CSX myocardial ischemia and AP are not due to CAD but are caused by complex micro-coronary pathophysiologic substrates (ED, MVD, MVCS and CAS). These pathophysiologic entities are linked with long-term MACE [8-12,15,19,22,26,34-39]. Table 3 summarizes the most important causes of MACE in CSX.
| In nearly 95% of cases the pathophysiologic mechanisms are ED and MVD (sometimes with CMVS) while epicardial CAS occurs in nearly 4% of cases. These mechanisms interplay with the plaque erosion or rupture and induce MACE. As in other arteriosclerotic conditions, plaque rupture or erosion, coagulation dysfunction, several products of inflammation etc. may cause acute thrombosis and MACE. |
| ACS and MI result from coronary thrombosis. ACs and MI are usually associated with rupture or erosion of vulnerable plaques, usually not detected by conventional coronarography. |
| In patients with sudden coronary death the most frequent cause (in a third of cases) is plaque rupture due to erosion. In the rest of cases several causes were detected, such as calcified nodules. |
| Patient gender and age are important. • In men age does not play any role in the occurrence of plaque rupture. • Plaque rupture is the cause of death in about one third of women, but in less than one sixth of men. • Acute cardiac thrombi are the predominant cause of MI in women <50-year; whereas plaque rupture is the predominant cause in women >50-year old women. |
Table 3: Causes of MACE in CSX.
ACS, MI, and sudden death result from coronary thrombosis, which is usually associated with rupture or erosion of vulnerable plaques (usually not detected by conventional coronarography) [13-15]. In CXS-patients with sudden death plaque rupture was the most frequent cause (in more than 30% of cases in a third of cases erosion and in the rest several causes were detected, such as calcified nodules [15,42]. Gender and age are important in CSX-patients with ACS/MI. Independent predictors for ACS/MI are female sex and younger age. CAS/MI are detected in 10-25 of women and only in 6-10% of men [15,37-39]. There is a gender difference, because plaque rupture was the cause of death in about one third of women, but in less than one sixth of men [15,36,37,39,42]. In men age did not play any role in the occurrence of plaque rupture.
Patient age is also relevant. Indeed, in 50-year-old women plaque rupture was the predominant cause. Interestingly, age did not play any role in the occurrence of plaque rupture in men [15,36,37,39,42].
Of course, ED, MVD and sometimes CAS interplay with the plaque erosion or rupture and worsen the outcomes.
Therapy of CSX
Obviously, cardiac revascularization is not a therapeutic option in CSX. There are no double-blind randomized trials on the effects of medical therapy in this pathology. Drugs used in CID are empirically used in CSX.
Short-acting sublingual or spray nitroglycerin and spray iso-sorbide nitrates are largely used to treat exertional and spontaneous AP in CSX, but this therapy is less effective than in patients with CAD. Low-dose aspirin is largely used, even if there is no evidence that long-term aspirin should be given to patients, even with known cardiovascular disease. Theoretical arguments that aspirin can prevent MACE by reducing the propagation of thrombus are countered by evidence that plaque hemorrhage from vasa vasorum may also cause plaque growth instability; aspirin may also detract from the benefits of angiotensinconverting enzyme inhibitors [43]. Statins (HMG-CoA reductase inhibitors) are used to treat endothelial dysfunction and it is postulated that these drugs may improve endothelial dysfunction in CSX. A study has proven that, via a nitric oxide-dependent mechanism, pravastatin reverses obesity-induced dysfunction of pluripotent stem-cells derived from endothelial cells [44]. β-blockers, calcium antagonists, and long-acting nitrates or nicorandil are often used to treat stable AP in CSX-patients. However, β-blockers are usually less effective than in CAD. Nitrates and nicorandil are less effective and induce more adverse effects than in CAD-patients. Of note, a study in patients with stable vasospastic AP has shown that, when compared with calcium antagonists, nitrates did not improve prognosis; on the contrary, the combined therapy increased the risk for MACE, especially when transdermal nitroglycerin and nicorandil were used [45]. Ranolazine is frequently off-label used in CSX [46,47]. There are no controlled studies in CSX but many cardiologists report a better efficacy in reducing AP than with β-blockers, calcium antagonists, and long-acting nitrates or nicorandil.
Conclusion
CID represents an important clinical problem worldwide. The number of percutaneous dilation and stenting is expected to increase over the coming decades. It has been recognized that the prevalence of CID in women is at least the same as in men. However, we have learned that CSX is frequent because each year millions of people (mostly women) with AP are found to have normal CA. Indeed normal CA are found in nearly 75%
of women and in up to 15% of men with typical AP. Also, data from large MI registries suggest a 5 to 25% prevalence of cases without CAD [24]. By visual analysis of biplane coronarography it is often said that these people have normal CA. However, as demonstrated from autoptic and ultrasound intravascular studies in vivo it would be more appropriate to say that coronarography did not detect obstructive or flow-limiting epicardial CAD.
In CSX ED and MVD (sometimes also MVCS and epicardial CAS) are established coronary pathophysiologic substrates. Symptoms and signs of cardiac pathology are the same as those of patients with CAD. The difference lies in the fact that in CSX the mechanisms of cardiac ischemia are micro-vascular and flow-limiting epicardial CAD is absent.
The pathologic entities work by interplay in CSX and are linked with long-term MACE, specifically hospitalization, ACS, MI, coronary revascularization and sudden death. The prevalence of MACE outcomes in CXS is probably smaller than in CAD, but we lack accurate data. It is nonetheless unjustified to dismiss the CSX as a benign condition. Given the association between CSX and consequent downstream morbidity, it seems logical to assess long-term MACE by appropriate analysis, e.g. by the ‘weighted composite endpoints’ approach.
Drugs used in CAD are also prescribed in CSX, but we lack data from specific double blind randomized trials. It seems that statins and ranolazine might exert positive effects. However, specific research to target interventions against CSX would be necessary.
References
- Felker GM, Shaw LK, O'Connor CM (2002)A standardized definition of ischemic cardiomyopathy for use in clinical research. J Am Coll Cardio 39:210-218.
- Zellweger MJ (2015) Management of stable coronary artery disease. Cardiovasc Medicine 18:16-19.
- Blumgart HL, Schlessinger MJ, Davis D(1940) Studies on the relation of the clinical manifestations of angina pectoris, coronary thrombosis and myocardial infarction to the pathologic findings. Am Heart J 19:1-90.
- Kemp HG, Elliott WC, Gorlin R (1967) The anginal syndrome with normal coronary arteriography. Trans Assoc Am Phys 17:1471-1822.
- Likoff W, Segal B, Kasparian H (1967) Paradox of normal selective coronary arteriograms in patients considered to have unmistakable coronary heart disease. N Engl J Med 276:1063-1066.
- Crea F (1995) Syndrome X: can the puzzle be unraveled? Eur Heart J 16:1455-1456.
- Crea F (2003) Prevalence, pathogenesis, diagnosis and treatment of cardiac syndrome X. e-journal Cardiol Practice 1:1-3.
- Klimusina J, Porretta AP, Segatto JM,Facchinis M, Bomio F,et al. (2013)Cardiac X syndrome: an overview of the literature and the local experience in Southern Switzerland. Cardiov Med 16: 20-28.
- Cocco G, Laub D (1993)Ungeklärter thorakaler Schmerz. DIA14:995-998.
- Cocco G, Chu D (2007) Stress induced cardiomyopathy, a review. Eur J Int Med 18:369-379.
- Cocco G, Jerie P (2015) Angina pectoris in patients without flow-limiting coronary artery disease (cardiac syndrome X). A forest of a variety of trees. Cardiology J.
- Mintz GS, Painter JA, Pichard AD (1995)Atherosclerosis in angiographically "normal" coronary artery reference segments: an intravascular ultrasound study with clinical correlations. J Am Coll Cardiol 25:1479-1485.
- Khuddus MA, Pepine CJ, Handberg EM (2010) An intravascular ultrasound analysis in women experiencing chest pain in the absence of obstructive coronary artery disease: a substudy from the National Heart, Lung and Blood Institute-Sponsored Women's Ischemia Syndrome Evaluation (WISE). J Inter Cardiol 23:511-519.
- Della Rocca D, Pepine CJ(2014)Some thought on the continuing dilemma of angina pectoris.Eur Heart J 35:1361-1364.
- Bairey MCN, Porretta AP, Kelsey SF, Recichek N, RogersWJ, et al. (1999) The Women’s Ischemia Syndrome Evaluation (WISE) Study: protocol design, methodology and feasibility report. J Am Coll Cardiol 33:1453-1461.
- Gulati M, Cooper-DeHoff RM, McClure C (2009)Adverse cardiovascular outcomes in women with non-obstructive coronary artery disease: a report from the Women's Ischemia Syndrome Evaluation Study and the St James Women Take Heart Project. Arch Intern Med 169:843-850.
- Cannon RO 3rd, Watson RM, Rosing DR, Epstein SE (1983) Angina caused by reduced vasodilator reserve of the small coronary arteries. J Am Coll Cardiol 1:1359-1373.
- Suwaidi JA, Hamasaki S, Higano ST (2000) Long-term follow-up of patients with mild coronary artery disease and endothelial dysfunction. Circulation 101:948-954.
- Endemann DH, Schiffrin El(2004) Endothelial Dysfunction. JASN15:1983-1992.
- Félétou M, Vanhoutte PM (2006) Endothelial dysfunction: a multifaceted disorder (The Wiggers Award Lecture).Am J Physiology - Heart and Circulatory Physiology.
- Von Mering GO, Arant CB, Wessel TR (2004) National Heart, Lung, Blood Institute. Abnormal coronary vasomotion as a prognostic indicator of cardiovascular events in women: results from the National Heart, Lung, and Blood Institute-Sponsored Women's Ischemia SynÂdrome Evaluation (WISE). Circulation 109:722-523.
- Camici PG, Crea F(2007) Coronary microvascular dysfunction. N Engl J Med 356:830-840.
- Pepine CJ, Anderson RD, Sharaf BL (2010) Coronary microvascular reactivity to adenosine predicts adverse outcome in women evaluated for suspected ischemia. J Am Coll Cardiol 55:2825-2832.
- Shaw LJ, Shaw RE, Merz CN (2008) American College of Cardiology-National Cardiovascular Data Registry Investigators. Impact of ethnicity and gender differences on angiographic coronary artery disease prevalence and in hospital mortality in the American College of Cardiology-National Cardiovascular Data Registry.Circulation 117:1787-1701.
- Bugiardini R, Women (2006)non-specific chest pain, and normal or near-normal coronary angiograms are not synonymous with favourable outcomes. Eur Heart J 27:1387-1389.
- Frederick A. Heupler Jr (1980) Syndrome of symptomatic coronary arterial spasm with nearly normal coronary arteriograms. Am J Cardiol 45:873–881.
- Bertrand ME, LaBlanche JM, Tilmant (1982)Frequency of provoked coronary arterial spasm in 1089 consecutive patients undergoing coronary arteriography.Circulation65:1299-1206.
- Yasue H, Horio Y, Nakamura N(1986)Induction of coronary artery spasm by acetylcholine in patients with variant angina: possible role of the parasympathetic nervous system in the pathogenesis of coronary artery spasm. Circulation 74:955-963.
- Kugiyama K, Yasue H, Okumura K (1996) Nitric Oxide Activity Is Deficient in Spasm Arteries of Patients With Coronary Spastic Angina. Circulation 94:266-272.
- Pepine CJ (2008)Provoked coronary spasm and acute coronary syndromes. J Am Coll Cardiol 52:528-530.
- Strozzi C, Cocco G (1977) The kinetocardiogram during the isoproterenol test for the assessment of coronary heart disease. Cardiology 62:277-290.
- Strozzi C, Cocco G, Padovan GC (1979) Modificazioni dei movimenti precordiali in cardiopatie non ischemiÂche durante l’infusione intravenosa di isoproterenolo e di dopamina. G Ital Cardiol 9:635-639.
- Cheng TO, Bashour T, Keiser GA (1972)Myocardial infarction in the absence of coronary arteriosclerosis. Results of coronary spasm. Am J Cardiol 30:680-682.
- Niccoli G, Scalone G, Crea F (2015)Acute myocardial infarction with no obstructive coronary atherosclerosis: mechanisms and management. Eur Heart j36:475-481.
- Kaski JC(2004) Pathophysiology and management of patients with chest pain and normal coronary arteriograms (cardiac syndrome X). Circulation109:568-572.
- Roger VL, Go AS, Lloyd-Jones DM(2011) American Heart Association Statistics Committee Stroke Statistics Subcommittee. Heart disease and stroke statistics update: a report from the American Heart Association. Circulation 123:18-209.
- Jespersen L, Hvelplund A, Abildstrøm SZ (2012) Stable angina pectoris with no obstructive coronary artery disease is associated with increased risks of major adverse cardiovascular events. Eur Heart J 33:734-744.
- Pepine CJ, Douglas PS (2012)Rethinking stable ischemic heart disease: is this the beginning of a new era? J Am Coll Cardiol60:957-959.
- Ciolino JD, Carter RE(2015)Reanalysis or redefinition of the hypothesis? Eur Heart H 36:340-341.
- Bakal JA, Roe MT, Ohman(2015)Applying novel methods to assess clinical outcomes: insights from the TRILOGY ACS trial. Eur Heart J 36:385-392.
- Finn AV, Nakano M, Narula J (2010)Concept of vulnerable/unstable plaque. Arterioscler Thromb Vasc 30:1282-1292.
- Cleland JGF,(2013)Is aspirin useful in primary prevention? Eur Heart J 34:3412-3418.
- Gu M, Mordwinkin NM, Kooreman NG (2015) Pravastatin reverses obesity-induced dysfunction of induced pluÂripotent stem-cells derived endothelial cells via a nitric oxide-dependent mechanism. Eur Heart J 36:806-816.
- Takahashi JT, Nihei T, Takagi Y (2015)Prognostic impact of chronic nitrate therapy in patients with vasospastic angina multicenter registry study of the Japanese coronary spasm association. Eur Heart J 36:228-237.
- Cocco G (2012). Management of myocardial ischemia. Is ranolazine needed? For all or some patients with myocardial ischemia? Expert Opin Pharmacother.
- Cocco G. Indicated and off-label use of Ranolazine. e-journal of the ESC Council for Cardiology Practice.
Citation: Cocco G, Jerie P, Amiet P (2016) Cardiac Ischemia and Angina Pectoris without Flow-Limiting Coronary Artery Disease (Coronary Syndrome X). Cardiovasc Ther 1: 101.
Copyright: © 2016 Cocco G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Open Access Journals
Article Usage
- Total views: 13360
- [From(publication date): 2-2016 - Aug 30, 2025]
- Breakdown by view type
- HTML page views: 12394
- PDF downloads: 966