Case Report after Introducing a New Abutment Surface for Bone Anchored Hearing Implants: Hydroxiapatite Abutment Surfaces and Skin Reaction
Received: 13-Apr-2018 / Accepted Date: 08-May-2017 / Published Date: 14-May-2017
Abstract
In order to evaluate a new surface of an abutment and its skin reactions when introducing the bone anchored hearing system a clinical investigation was performed. Ten intended subjects were scheduled to receive a hydroxyapatitecoated abutment, using the tissue preserving surgery as a 1 step performance. Only 7 implants were installed due to peri-implant infections. The length of the abutments varied among the patients from 10-12 mm. Appointments were planned 1 week after surgery, at 7 weeks - 3 months, 6 and 12 months after surgery. Skin reactions were graded according to the Holgers´ scale. Unusual severe skin reactions were noted after implantation with the hydroxyapatite abutments, from 1 week to 7 months post-implantation, many of them classified as Holgers´. Some of the implanted persons suffered from pain. Positive bacterial cultures were found in 3 patients all showing Staphylococcus aureus. Good stability of all fixtures was achieved. Only 2 patients used after 1 year the primary implant and abutment.
Keywords: Bone anchored hearing implants; Peri-implant reactions; Hydroxyapatite
79176Introduction
Osseointegration of titanium implants has been helpful for anchoring hearing aids since the late 70’s. The fixture is still using the titanium metal of the same origin as when the method was firstly introduced. Refinements have been made concerning the width of the implant as well as the interior design aiming at better anchoring and stability. The abutment surface has been composed of titanium primarily but the roughness has been changed and other materials have been used for coating the titanium surface. The aim has been to get a better adherence of the skin to the abutment to reduce the imaginary space between skin and abutment surface. This in order to prevent and minimize bacterial invasion and avoid peri-implant infections [1,2]. The incidence of soft tissue skin reactions around adult bone anchored hearing implants (BAHIs) has been reported to be between 3.4–39.6 percent [3]. Cells pre-potential for allergic reactions has been shown to be present in the skin close to the installed titanium abutments [4,5].
There has been significant research to try and understand the pathophysiology of the tissues maround an abutment. Recently, the shape of the abutment has been the center of attention and debate, particularly the angle between the skin and abutment.
New surgical techniques have been developed over the years. At present, mostly tissue preserving interventions are in use [6,7] utilizing longer and individualized abutments. The expected risk for increased bacterial colonialization using this technique has not been fulfilled [8] on the contrary better outcome has been introduced.
Better adherence of the skin to a titanium surface is still an ongoing discussion. New ideas concerning the shape and surface of the abutment have been launched [9-11]. The present survey investigated hydroxiapatite surfaces of longer abutments and their reaction with the surrounding skin.
Case Presentation
Intentions were to implant 10 persons with a follow up time of 1 year. All patients were selected consecutively for a bone anchored hearing implant (BAHI). All interventions were made in local anesthesia using the tissue preserving technique, by the same surgeon [6], after tested with audiometry and given a bone-anchored test device on a soft-band for 3-4 weeks.
A Cochlear DermaLock Abutment (BA400, Cochlear) which is the only bone conduction abutment with a hydroxiapatite coated surface, were implanted in 7 patients. Unlike conventional titanium abutments, the DermaLock surface promotes soft tissue to bind to the abutment with the intention to seal the implant site. Inclusion criteria were: Eligible for the Cochlear Baha system (conductive or mixed hearing loss), Adults>18 years. The length of the abutments varied from 10-12 mm, all on a 4 mm fixture.
None of the included persons had an ongoing ear or skin infection in the area when implanted. Information concerning clinical signs and symptoms, gender, concomitant medication, skin diseases, peri-implant infections (the extent of the inflammatory reaction classified according to the Holgers´ scale of 1-4) [12], numbness around the implant, change of abutment length, abutment loss and skin overgrowth were recorded (Table 1). Stability was tested with the Ostell instrument [13] and loading of the processor took place >4 weeks after surgery [14].
| Age Mean | Side N | Gender N | Ab length N, mm | Peri-implant infection N | Holger scale | Ab change | Ab removal | Finished study | |
|---|---|---|---|---|---|---|---|---|---|
| Patients # A-G | 52 | 6 L, 1 R | 7 F | 6 10 mm, 12 mm | 7 | 3-4 | 4 | 5 | 2 28% |
Note: N: number; L: left; R: right; M: male; F: female; Ab: Abutment
Table 1: Demographics from 7 patients implanted and followed during a 1 year period.
Ethical permissions (2012/1987-31/1) were achieved and all patients gave their written consent for participation and to photos taken at every appointment. Follow up were set to 1 week and 7 weeks -3 months post-surgery, at 6 and 12 months after surgery.
Mean age of the patients were 52 years (range 24- 80 years of age), all 7 were females. Table 1 reveals background information for the patients. Six persons used a hydroxiapatite abutment of 10 mm and 1 used a 12 mm from start. All patients were fitted with a BP 100 processor (Cochlear, Gothenburg, Sweden).
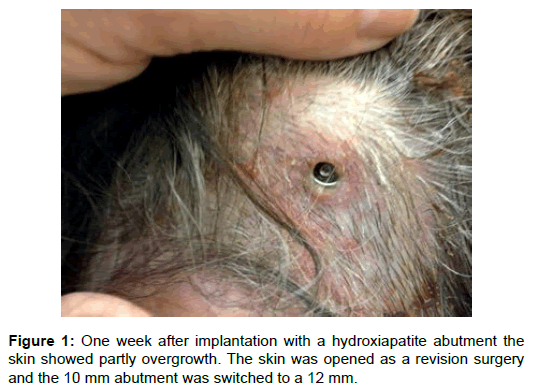
Patient A, with a pituitary adenoma and a mixed hearing loss (MHL), had already at time for the post-operative appointment an infection with a skin overgrowth and pus (Figure 1). The 10 mm abutment was directly changed to a 12 mm, after opening the skin with a small incision in an out-patient procedure. The patient was given antibiotics post-operation. The skin around this coated abutment was continuously infected and painful even though intensive skin care and antibiotics systemically and locally were given and stayed in place for 3 months, before it had to be removed. She had to use her processor on a headband during the treatment. Samples showed growth of Staphylococcus aureus. Another alternative with a transcutaneous system was introduced.
Patient B did not show up for the 1 month appointment and said when seen at 3 month, that she had had so much trouble and pain and was afraid to come to the hospital. She could not use her processor. She was implanted due to a MHL. At this time the whole abutment was covered with crusts, coagulated blood, pus and granulation tissue. After several appointments with a lot of pain, intensive cleaning, tissue surgery, etching of granulation tissue and antibiotics both systemically and locally it was still impossible to reduce the severe peri-implant infection and pain. The abutment had to be removed after 10 months. She has now been provided with other hearing solutions.
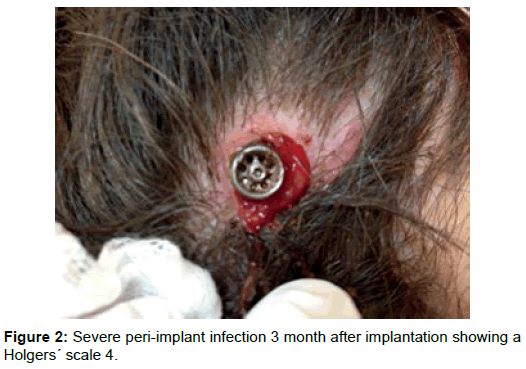
Patient C, who suffers from Turner syndrome and had a conductive hearing loss, had after 3 month a severe and aching peri-implant infection. She had a lot of pain and could not touch the area nor cleans it and could not sleep on the implanted side (Figure 2). She had a massive amount of hair and the hair was constantly gluing to the infected skin area with pus. After both revision surgery, removing of hair around the area and conservative treatment, the clinical signs of infection grew worse and the abutment had to be removed after 5 months. Patient C had another system introduced, but after 1 year she wanted to have also this implant removed, because she did not feel any hearing gain by the processor in her study situation.
Patient D, with a bilateral sensorineural hearing loss and no possibility to use conventional hearing aids, showed at the first follow up 1 week post-surgery signs of infection around the 10 mm long abutment. The area was cleaned and ointment with antibiotics prescribed. After 1 month the skin had a tendency to overgrow the abutment and pus was present, a minor revision surgery was performed and the abutment was changed to a similar one, but of 12 mm length. She came frequently for cleaning and treatment and suffered from pain and after 5 months the abutment had to be removed.
Patient E suffers from Mb Down and had since earlier an abutment on the right side due to bilateral MHL, and was now implanted on the left side. She suffered after surgery from periods with peri-implant infections up to Holgers´ 3 with pus, but they have all been possible to cure conservatively with extra cleaning, etching and ointment. Staphylococcus aureus were grown in samples from the surrounding skin. She uses still after 1 year the same abutment. Patient E is very satisfied with her hearing solution.
Patient F suffered from Wegener’s disease and had no problems with her implant until 7 months post-implantation when she suffered from a Holgers´ 3 peri-implant infection with pus. Intensive cleaning, ointment, antibiotics and etching cured the infection and she is still a user of the hydroxiapatite abutment. Growth of Staphylococcus aureus was collected. Patient F had a conductive hearing loss and is satisfied with the hearing gain.
Patient G had after 1 month severe itching and granulation tissue around the abutment. Patient G was implanted due to bilateral MHL. An area of 20 mm around the abutment was irritated and red with small papules and with granulation tissue and pus close to the abutment. Many appointments occurred when trying to cure the ongoing periimplant infection conservatively, but it was not successful and the abutment was removed after 6 months.
Five patients had their abutments removed due to severe periimplant infections, grading 3-4 on the Holges´scale, after meticulous conservative work with the skin and were directed to other hearing solutions. The other two patients had primarily and repeatedly, severe infections but the skin was possible to cure. Change of abutment was performed in 3 patients before total removal. Out of the 7 implanted only 2 are using the coated abutment today. No patient complained of numbness around the implant site. Ostell measurement showed good stability in all cases and no spontaneous implant losses were recorded. When removing and changing the abutment it was noted that the skin was not totally fixed to the abutment surface but patches of threads containing tissue and blood vessels were adhering to the surface with some bleeding during the changing procedure.
Discussion
The case report studied 7 patients with a hydroxiapatite surface. All suffered from postoperative, severe peri-implant infections and only 2 of them are using their DermaLock coated abutment after 1 year while the other 5 had to change the concept due to peri-implant infections.
It is know from earlier studies that the non-skin thinning surgical technique is beneficial when implanting BAHIs in both children and adults [6,15,16]. Many of the earlier known complications related to the skin were reduced. The tissue preservation technique requires longer abutments due to the individually varying thickness of the skin and 10 and 12 mm hydroxiapatite abutments were used in the present study. Even though the strategy and set up were identical and the patients were operated by the same surgeon in the same operating rooms, with exactly the same technique, assisted by the same nurses all patients studied suffered from severe peri-implant infections with Holgers´ 3-4. All patients had the same instructions for skin care after surgery. During inclusion period for the present 7 patients, other patients were implanted with other techniques and abutments in the same setting without any outcome of peri-implant infections. When a comparison was made with another study including 10 patients from the same hospital from the same time period, using a flat surface, only minor problems (Holgers´1-2) were recorded and all of them used their abutments after 1 year [6].
Already after 7 days the abutment was overgrown in patient A and after 1 month in patient D. Their abutments were changed to longer ones. No explanation could be identified. No solution to the following repeated skin infections, starting at different time period after surgery, was found among all patients using the hydroxiapatite abutments. Bacterial samples were taken in 6 of the implanted persons, and in 3 of them Staphylococcus Aures were growing while the rest showed no growth. These bacteria are commonly reported in skin infections surrounding implants [17]. A lot of explanations for the increase in peri- implant infections surrounding the hydroxiapatite abutment surface came forward during discussions, among them different care or contamination during the surgical procedure which both could be removed as arguments. The gender with 7 females can be discussed as well as a skin related underlying disease (Wegener’s disease) which was present in one woman. However, this patient was among the 2 who was still using the same abutment after 1 year.
Pocket formation around the abutment and epidermal down growth between the skin and the abutment has been interpreted as complications in relation to forming infections. In a study in sheep using the hydroxiapatite coated abutment and compared to noncoated titanium abutments, it could be shown that a significantly reduced pocket depth was achieved [1]. The connective interface of hydroxiapatite has also been tested in humans with an increased skin connection, a reported healing of the skin 7 days after tissue-preserving surgery, with an 18.75% peri-implant infection rate and with a Holgers´ scale of 2 [18,19]. This is not in accordance with the present report where all patients suffered from severe peri-implant infections.
When removing the hydroxiapatite abutments in the present study, either for changing the length or to finally remove the implant, it could be noted that there was an uneven growth of skin tissue on the hydroxiapatite surface. Threads of tissue could be seen to adhere to the surface, and they were ripped off when unscrewing and changing the abutment. This occurrence is not seen when changing abutments with a smooth titanium surface. When changing abutments with a smooth surface it could be seen that there is a small/thin face covering the tissue in the exposed hole, often called epidermal down-growth, embracing the abutment but not adhering to it but also giving a possibility for biofilms to grew on the implant surface [15]. One hypothetical explanation to the increased tissue infections could be that when the patient moves the head a more smooth and glossy surface permits the skin to move slightly around the abutment, while this is prohibited with the rougher surface where the skin is patchy fixed to it. When the head and skin is moving the small attached threads is ripped off repeatedly and might give small traumatic bleedings with following exposure to the surrounding microbiology. Another hypothetical explanation could be that the reported cells pre-potential for allergic reactions has been activated due to constant irritation and prepared the skin for infection [4,5].
These severe peri-implant infections caused a lot of inconvenience for all surgically treated patients, with pain, several extra appointments, loss of hearing when not able to use the intended processor and new surgeries and strategies in 5 cases.
These problems seem to exist among also other patients than those presented here and the explanation for the massive peri-implant infections is still unsolved.
Conclusion
The present investigation implanted 7 bone anchored abutments with a hydroxiapatite covering with the intention to tightly adhere the skin to the abutment surface. In the present report the benefit of this design could not be proven. Patients having had this type of abutment, all suffered from severe peri-implant infections and most of them had to terminate their installation before 1 year had passed. The users were, however, content with the hearing outcome.
Disclosure Statement
A grant was supported from Cochlear for a full study of 10 patients. The study was terminated in advance.
References
- Faber HT, Wolf MJF, Rooy WJ (2009) Bone-anchored hearing aid implant location in relation to skin reactions. Arch Otolaryngol Head Neck Surg 135: 742-746.
- Larsson AL, Wigren S, Andersson M, Ekeroth G, Flynn M, et al. (2012) Histologic evaluation of soft tissue integration of experimental abutments for bone anchored hearing implants using surgery without soft tissue reduction. Otol Neurotol 33: 1445-1451.
- Wazen JJ, Wycherly B, Daugherty J (2011) Complications of bone-anchored hearing devices. Adv Otorhinolaryngol 71: 63-72.
- Holgers KM, Thomsen P, Tjellström (1995) Immunohistochemical study of the soft tissue around long-term skin-penetrating titanium implants. Biomaterials 16: 611-616.
- Thewes M, Kretschmer R, Gfesser Mb, Rakoski J, Nerlich M, et al. (2001) Immunohistochemical characterization of the perivascular infiltrates cells in tissues adjacent to stainless steel implants compared with titanium implants. Arch Orthop Trauma Surg 121: 223-226.
- Hultcrantz M (2011) Outcome of the bone-anchored hearing aid procedure without skin thinning: A prospective clinical trial. Otol & Neurotol 32: 1134-1139.
- Dumon T, Medina M, Sperling NM (2015) Punch and drill: Implantation of bone anchored hearing device through a minimal skin punch incision versus implantation with dermatome and soft tissue reduction. Ann Otol Rhinol Laryngol 125: 199-206.
- Hultcrantz M, Lanis A (2014) A five year follow-up on the osseointegration of bone-anchored hearing aid implantation without tissue reduction. Otol Neurotol 35: 1480-1485.
- Nelissen RC, den Besten CA, Mylanus EA, Myrthe KSH Â (2016) Stability, survival, and tolerability of a 4.5-mm-wide bone-anchored hearing implant: 6-month data from a randomized controlled clinical trial. Eur Arch Otorhinolaryngol 273: 105-111.
- Allis TJ, Owen BD, Chen B, Jones DT, Moore GF (2014) Longer length Bahaâ„¢ abutments decrease wound complications and revision surgery. Laryngoscope 124: 989-992.
- Westerkull P (2011) The ponto bone-anchored hearing system. In: Kompis M, Caversaccio MD (eds.), Implantable Bone Conduction Hearing Aids. pp: 32-40.
- Holgers KM (2000) Characteristics of the inflammatory process around skin-penetrating titanium implants for aural rehabilitation. Audiology 39: 253-259.
- Hultcrantz M, Lanis A (2014) Stability testing after osseointegrated implant surgery without skin thinning in children: Case reports after abutment loss. Otol Neurotol 35: 1102-1104.
- Faber HT, Dun CA, Nelissen RC, Mylanus EA, Myrthe KSH, et al. (2013) Bone-anchored hearing implant loading at 3 weeks: stability and tolerability after 6 months. Otol Neurotol 34: 104-110.
- Liu CC, Chadha NK, Bance M, Hong P (2013) The current practice trends in pediatric bone-anchored hearing aids in Canada: a national clinical and surgical practice survey. J Otolaryngol Head Neck Surg 1: 42-43.
- Caruso A, Giannuzzi AL, Sozzi V, Sanna M (2017) Bone anchored hearing implants without skin thinning: the Gruppo Otologico surgical and audiological experience. Eur Arch Otorhinolaryngol 274: 695-700.
- Monksfield P, Chapple IL, Matthews JB, Grant MM, Addison O, et al. (2011) Biofilm formation on bone-anchored hearing aids. J Laryngol Otol 125: 1125-1130.
- Iseri M, Orhan KS, YarıktaÅ? MH, Kara A, Durgut M et al. (2015) Surgical and audiological evaluation of the Baha BA400. J Laryngol Otol 129: 32-37.
- van Hoof M, Wigren S, Dumiel H, Savelkoul PH, Flynn M, et al. (2015) Can the hydroxyapatite-coated skin-penetrating abutment for bone conduction hearing implants integrate with the surrounding skin? Front Surg 14: 2-45.
Citation: Hultcrantz M (2017) Case Report after Introducing a New Abutment Surface for Bone Anchored Hearing Implants: Hydroxiapatite Abutment Surfaces and Skin Reaction. J Med Imp Surg 2: 115.
Copyright: © 2017 Hultcrantz M. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Usage
- Total views: 8601
- [From(publication date): 0-2017 - Aug 29, 2025]
- Breakdown by view type
- HTML page views: 7633
- PDF downloads: 968