Research Article Open Access
Comparison of Original and Internal Pathology Reports Referred for UrothelialCarcinoma to Determine Rate of Discrepancies and the Impact on Treatment Decisions
Luisa SC* and De Castro GJColumbia University Medical Center, New York, USA
- *Corresponding Author:
- Luisa SC
Columbia University Medical Center
Urology Herbert Irving Pavilion, New York, USA
Tel: 19145888133
E-mail: bibamillion@aol.com
Received date: January 21, 2017; Accepted date: February 03, 2017; Published date: February 06, 2017
Citation: Luisa SC, De Castro GJ (2017) Comparison of Original and Internal Pathology Reports Referred for Urothelial Carcinoma to Determine Rate of Discrepancies and the Impact on Treatment Decisions. J Clin Exp Pathol 7:302. doi: 10.4172/2161-0681.1000302
Copyright: © 2017 Luisa SC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Clinical & Experimental Pathology
Abstract
Objective: The purpose of the current study is to perform a standardized comparison of original and internal repeat pathology reviews of identical bladder specimens to identify discrepancies and characterize the impact of repeat review on treatment decisions as well as identify patients most likely to benefit from this practice.
Materials/Methods: Ninety-one patients with an outside diagnosis of urothelial cancer of the bladder were referred to our institution for repeat review of 91 bladder resection specimens and biopsies.
A discrepancy in either the presence or absence of muscularis propria and presence of invasive disease in the muscularis propria was deemed a “treatment-altering” characteristic, while presence of carcinoma in situ, lymphovascular invasion, or micropapillary features was deemed a “clinically-significant” characteristic.
Results: After repeat review at our institution, 29.7% (27) specimens had treatment altering discrepancies, and 61.5% (56) specimens had at least one clinically-significant discrepancy.
Conclusion: Repeat review of referred bladder specimens frequently impacts treatment decisions in patients with urothelial carcinoma.
Keywords
Comparison pathology; Urothelial carcinoma; Bladder tumors; Bladder biopsy; Transurethral resection
Introduction
Urothelial carcinoma is a significant cause of morbidity and mortality, with an estimated number of new cases reaching 76,960 in 2016 in the US. The number of new cases and deaths in men and women per 100,000 is estimated at 20.1 and 4.4 respectively [1]. The accurate diagnosis, histologic grading, staging and definitive diagnosis of bladder tumors depends heavily on the accurate interpretation of the specimen obtained from bladder biopsy and/or transurethral resection of bladder tumor (TURBT) [2].
Papillary urothelial neoplasms encompass a spectrum of morphologic findings, including tumors that behave aggressively and tumors that are biologically benign. Attempting to differentiate biologic behavior based solely on subtle histopathologic criteria is fraught with difficulties and perils, especially considering interobserver variability that has been documented in many studies with all classification schemes [3]. A study by Tosoni found significant interobserver differences in both the staging and the grading of tumors. From a total of 235 tumors initially diagnosed as T1, repeat review classified 35% as cTa, 56% as pT1 and 3% as pT2-4. In 39% of all biopsies there were interobserver differences in tumor grade [3].
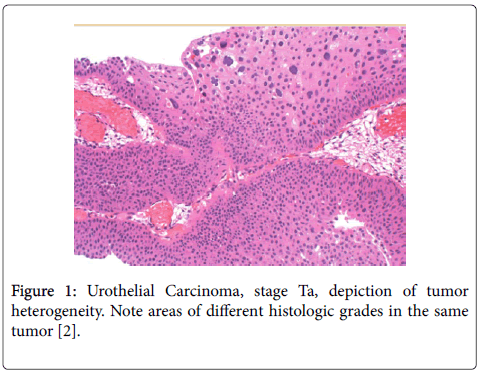
Abel et al. suggested that it might be advisable for urologists to discuss a patient’s treatment by reviewing the slides if a difficult treatment decision needs to be made. These discrepancies emphasize the importance of reviewing bladder tumor slides before radical treatment. While the 2004 WHO and ISUP system provides clearly defined histologic criteria for each of its diagnostic categories it fails to take into account tumor heterogeneity. The 1973 WHO classification system allows some flexibility in the diagnosis of these tumors (Figure 1) [2].
Figure 1: Urothelial Carcinoma, stage Ta, depiction of tumor heterogeneity. Note areas of different histologic grades in the same tumor [2].
This added flexibility may actually give a more accurate representation of tumor histology than attempting to force a lesion into a simple diagnostic category. Mikuz and colleagues demonstrated that interobserver agreement was higher using the 1973 WHO classification than when using either the 2004 WHO or 1999 WHO and ISUP systems [2].
While further studies are needed to address tumor heterogeneity on prognostic outcome, taking tumor heterogeneity into consideration improves prognostic accuracy [2]. Some in the European Association of Urology advocate the simultaneous use of both grading systems [2].
TURBT is the diagnostic procedure of choice for the diagnosis of urothelial carcinoma of the bladder. TURBT pathology is the primary data point that determines therapeutic options and ultimately influences treatment decisions. However, compared to other malignancies, the quantity and integrity of pathologic specimens obtained at the time of TURBT varies significantly and is dependent on several factors, including physical characteristics (e.g., size, location and multifocality) as well as surgical technique. TURBT specimens are excised in a piecemeal fashion, and vary in shape and size. Specimens are often twisted making orientation difficult. Papillary urothelial neoplasms are complex in nature and tangential sectioning further complicates accuracy of diagnosis [2]. The quality of the TURBT and the surgical pathologist’s interpretation of the TURBT is the cornerstone of bladder cancer staging. An ideal staging system should accurately reflect the natural history of cancer at this site, describing total cancer burden, assessing extent of spread at presentation and stratifying patients into prognostic groups for treatment planning [4]. At time of disease presentation, almost 70% of urothelial tumors are non-muscle invasive (pTa or T1) [3]. The remaining 20%-30% initially present as muscle invasive tumors (pT2-4). While some studies suggest a similar prognosis for stage pTa and stage pT1 there is increasing evidence for a greater risk and subsequent tumor progression of pT1 carcinomas than pTa carcinomas. Consequently, in those who do not respond satisfactorily to transurethral surgery or intravesical therapy, patients with pT1 tumors cystectomy is to be considered. Histological findings, and the quality of the pathology report dictates whether these patients should undergo cystectomy [3].
While staging is straightforward for low-grade non-invasive tumors, the staging for high-grade tumors is difficult because errors are common [4]. Upstaging at the time of radical cystectomy is reported in approximately 40% of clinically localized cases and downstaging at a rate of 20% [4]. Little, if any improvements have been made in the past 2 decades-indeed, there has been an increase in upstaging. The most challenging cases are T1 lesions and the accurate staging prior to radical cystectomy (RC) for advanced disease, especially if neoadjuvant chemotherapy is considered as treatment [4]. Adoption of a uniform staging system permits comparison of therapeutic interventions among different institutions. There have been multiple efforts made to standardize the reporting of TURBT pathology [2] but it is difficult to assess the diagnostic accuracy of a pathology report based on an outside pathologic interpretation of another surgeon’s resection. Because of this, it is common practice to obtain repeat review when a patient is referred with a previous diagnosis of urothelial carcinoma. The 2002, tumor, node, metastasis (TMN) classification approved by the Union International against Cancer has been widely accepted.
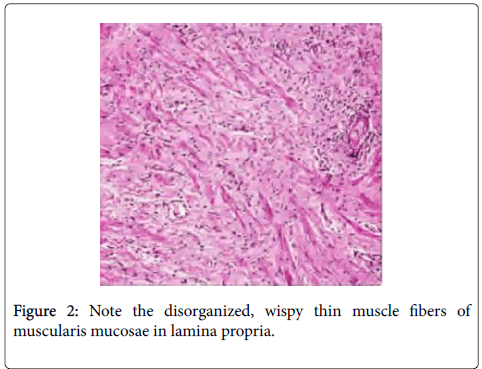
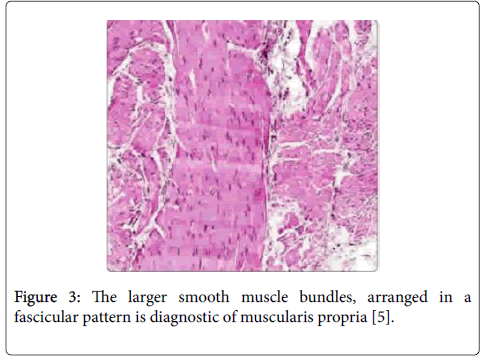
Several studies have confirmed benefits from repeat review when referred for urothelial carcinomas. Treatment of urothelial tumors depends upon the accurate identification of muscularis propria in the specimen and the presence or absence of muscle invasive disease. The identification of muscularis propria versus muscularis mucosae is a key pitfall in the pathologic evaluation of bladder specimens [2]. Angulo et al. were able to identify muscularis mucosae in 39% of patients and in an additional 26% when they used blood vessels as a landmark. In 5% of bladders, the thin, discontinuous wavy fibers of muscularis mucosae may become hypertrophied and mimic muscularis propria (Figures 2 and 3) [5].
Figure 3: The larger smooth muscle bundles, arranged in a fascicular pattern is diagnostic of muscularis propria [5].
Conversely, the misinterpretation of muscularis mucosae as muscularis propria can lead to overstaging and consequently unnecessary radical surgery. Use of immunohistochemical markers can help distinguish muscularis propria from muscularis mucosae. Smoothelin, identified as a marker for contractile smooth muscle and which has been suggested to be differentially expressed between the muscularis mucosae of the lamina propria and the muscularis propria [6]. Inflammatory responses to tumor, cautery artifact or thermal injury can cause severe morphologic distortion where grade and stage would become a challenge. Tumor retraction can mimic lymphovascular invasion; while adipose tissue in the lamina propria can be misdiagnosed as full-thickness invasion of the bladder wall with perivesical extension leading to overstaging; presence of von Brunn’s nest may also mimic lamina propria invasion, especially when they are prominent or when they have been distorted by inflammatory or cautery artifact. Another important consideration in urothelial cancer diagnosis that can also be challenging is secondary spread and metastasis. These factors all render the accurate diagnosis of invasion difficult. Diagnosis of invasion in some cases can be facilitated by immunohistochemical study with anticytokeratin antibodies. While immunohistochemistry may aid in the diagnosis of bladder cancer, it is not a panacea [6]. Given these pitfalls the importance of repeat review is evident and the potential for repeat review to spare the patient from unnecessary procedures warrants further review.
Methods and Materials
Using an Institutional Review Board-approved prospectively-acquired urologic oncology database, a query was performed to identify patients diagnosed with urothelial carcinoma at an outside facility and referred to a single urologist specializing in oncology at a tertiary care center from 2008-2012 with pathology slides for internal repeat review. The internal pathologists were given slides as well as the original pathology report; no gross specimens were reanalyzed. The Current WHO grading system was used. The query was limited to 2008 because that was the earliest year in which comprehensive outpatient data was available electronically.
A total of 102 internal pathology reports from TURBTs and biopsy specimens performed at outside institutions prior to referral were identified from 2008-2012. Eleven specimens were excluded from the original report, leaving 91 samples with both original outside and internal repeat review for analysis. Characteristics of both the original and internal reviews were collected by blinded reviewers (one reviewer for the original outside report and 2 additional reviewers for the internal reports). The distribution of each pathologic characteristic in the outside pathology reports and the internal repeat review were compared using chi-squared analyses to elicit underlying bias toward differing trends in pathologic diagnosis at our institution. Discrepancy rates between the two were compared. A discrepancy rate in presence or absence of muscularis propria in the lamina propria and presence of tumor involvement in the muscularis propria (clinical stage T2 or greater versus non-muscle invasive disease) was considered “treatment-altering.” Additionally, the presence or absence of secondary histology, carcinoma in situ, lymphovascular invasion, and micropapillary features was considered “clinically-significant discrepancy”. A scheme was developed to determine treatment options based on treatment altering discrepancies. Changes in treatment recommendations after internal repeat review were determined based on treatment altering discrepancies. All statistical analyses were performed using Stata SE version9.2.
Results
Ninety-one patients who underwent bladder biopsy or TURBT at an outside institution with both original and internal pathology reports available were analysed. They included 69 men and 22 women (Table 1), with median age at the time of initial procedure was 68 years (range 31-93), and the median time from initial procedure to internal repeat review was 34 days (range 9-368 days).
| Characteristic | Patients |
|---|---|
| Age (years) | |
| Median | 68 |
| Range | 31-93 |
| Sex | |
| Male | 69 |
| Female | 22 |
| Time from initial procedure to repeat review (days) | |
| Median | 34 |
| Range | 9-368 |
Table 1: Patient characteristics.
Eighty-two of the 91 patients (90.1%) were diagnosed with primary urothelial carcinoma on original review, two were reclassified as non-urothelial carcinoma on repeat review (one small cell carcinoma and one lymphoepithelioma-like carcinoma). The overall distribution of pathological characteristics listed in the pathology reports and the internal reviews were compared in order to see if there were obvious biases toward certain pathological findings at our institution compared to outside institutions. (Tables 1 and 2).
| Characteristic | Original report | Internal report | P Value (chi-squared) |
|---|---|---|---|
| Primary histology | 0.86 | ||
| Urothelial Carcinoma | 82 | 83 | |
| Adenocarcinoma | 1 | 2 | |
| Squamous cell carcinoma | 2 | 2 | |
| Other | 6 | 4 | |
| Secondary histology identified | 0.27 | ||
| Yes | 9 | 14 | |
| No | 82 | 77 | |
| Muscularis identified in specimen | 0.44 | ||
| Yes | 62 | 57 | |
| No | 29 | 34 | |
| Muscularis invasion identified | 0.27 | ||
| Yes | 34 | 27 | |
| No | 57 | 64 | |
| Carcinoma in situ | 0.75 | ||
| Present | 29 | 31 | |
| Absent | 62 | 60 | |
| Lymphovascular invasion | 0.27 | ||
| Present | 9 | 14 | |
| Absent | 82 | 77 | |
| Micropapillary features | *0.03 | ||
| Present | 2 | 9 | |
| Absent | 89 | 82 | |
| Clinical stage | 0.88 | ||
| T0 | 2 | 1 | |
| Tis | 5 | 5 | |
| Ta | 16 | 19 | |
| T1 | 31 | 35 | |
| T2 or greater | 34 | 27 | |
| Unstageable | 3 | 4 |
Table 2: Pathological characteristics of original and repeat internal review. *Statistically significant difference (p<0.05).
The only finding that differed significantly in distribution was micropapillary features, diagnosed in 2 (2.2%) of the original outside reports compared to 9 (9.9%) in the internal reviews (p=0.029, chisquared).
The rates of all other findings were similar, implying that there was a lack of inherent bias towards toward any specific pathologic diagnosis upon internal review that would influence the overall rate of discrepancies.
The discrepancy rates between each clinically-significant pathologic characteristic in the original and the internal repeat review are shown (Table 3).
| Characteristic | Discrepancy | No Discrepancy |
|---|---|---|
| Presence/absence of muscularis in specimen* | 25 (27.5%) | 66 (72.5%) |
| Presence/absence of muscularis invasion* | 11 (12.1%) | 80 (90.1%) |
| Presence/absence of secondary histology | 12 (13.2%) | 79 (86.8%) |
| Presence/absence of carcinoma in situ | 26 (28.6%) | 65 (71.4%) |
| Presence/absence of lymphovascular invasion | 9 (9.9%) | 82 (90.1%) |
| Presence/absence of micropapillary features | 9 (9.9%) | 82 (90.1%) |
| Any treatment altering charecteristic | 27 (29.7%) | 64 (70.3%) |
| Any clinically significant charecteristic** | 56 (61.5%) | 35 (38.5%) |
Table 3: Discrepancy rates between original review and repeat internal review in 91 patients (the presence or absence of each characteristic was compared between both pathology reports for each of 91 patients). *Treatment altering characteristics. **Clinically significant characteristics include all characteristics listed.
The two treatment-altering characteristics, presence or absence of muscularis propria in the specimen and presence or absence of tumor involvement in the muscularis propria showed discrepancies in 25 (27.5%) and 11 (12.1%) patients, respectively.
The characteristic with the highest discrepancy rate was the presence or absence of carcinoma in situ, which differed in 26 (28.6%) patients. In 27 (29.7%) of patients there was at least one treatment altering discrepancy. The overall discrepancy rate (including all patients who had at least one clinically-significant discrepancy) was 56 (61.5%). Of the 56 discrepant patients, 29 had only one discrepancy, 18 had two discrepancies and 9 had three discrepancies (Table 4).
| Number of clinically significant discrepancies per patient | Patients (n=91) |
|---|---|
| 0 | 35 (38.5%) |
| 1 | 29 (31.9%) |
| 2 | 18 (19.8%) |
| 3 | 9 (9.9%) |
Table 4: Frequency of multiple discrepancies.
The remainder of the analysis focused on the impact of repeat review on treatment decisions. Table 5 shows the simplified scheme used to select one of three recommended treatments based on the two treatment altering characteristics previously described in conjunction with the AUA guidelines and recommendations (Table 5).
| Muscularis invasion absent | Muscularis invasion present | |
|---|---|---|
| Muscularis present in specimen | Surveillance (+/- intravesical therapy) | Cystectomy |
| Muscularis absent from specimen | Repeat biopsy/TURBT | (Not applicable) |
Table 5: Simplified scheme for treatment recommendations based on treatment-altering pathological characteristics.
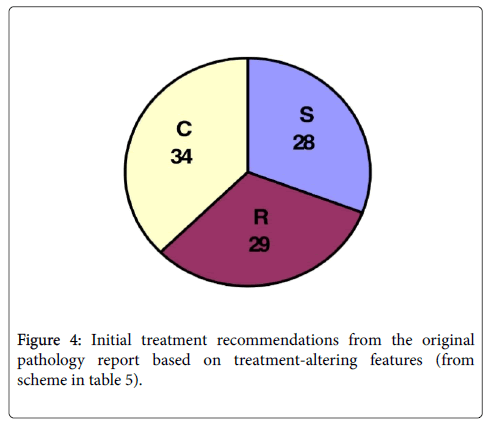
In summary, any specimen which had muscularis propria present but lacked tumor involvement in this layer would be recommended for surveillance (with or without intravesical therapy), while any specimen with muscularis propria involvement (muscle invasive disease) would be recommended for cystectomy (Figure 4).
All specimens with no muscularis propria identified would be considered inadequate for proper staging and would require a repeat transurethral resection (TUR). This scheme was used to classify treatment recommendations based on the original pathology report as well as revised recommendations based on the internal repeat review.
Figure 4 shows the initial treatment recommendations based on the pathology report from the original institution where each procedure was performed. Of the 91 patients, 28 (30.8%) would initially be recommended to undergo surveillance (S), 29 (31.9%) would require resection and 34 (37.4%) would need cystectomy(C) (Figure 4).
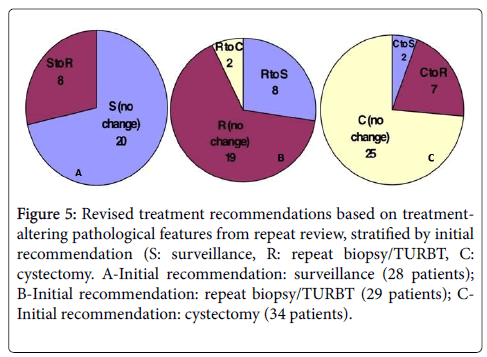
In Figure 5, each of the three initial treatment groups were further divided into revised recommendations based on treatment-altering pathologic characteristics from repeat internal review. In the initial surveillance group, 28.6% (8) patients had a revised recommendation of repeat resection while the remainder had no change. In the initial repeat resection group 8 (27.6%) patients had a revised recommendation of surveillance and 2 (6.9%) were recommended cystectomy. For the 34 patients initially recommended for cystectomy, 7 (20.6%) were recommended to undergo repeat resection while 2 (5.9%) were recommended surveillance (Figure 5).
Figure 5: Revised treatment recommendations based on treatment-altering pathological features from repeat review, stratified by initial recommendation (S: surveillance, R: repeat biopsy/TURBT, C: cystectomy. A-Initial recommendation: surveillance (28 patients); B-Initial recommendation: repeat biopsy/TURBT (29 patients); C-Initial recommendation: cystectomy (34 patients).
Discussion
In the past 2 decades, several studies have repeatedly shown the benefits of repeat review [7-12]. Discrepancy rates between original and repeat histopathologic diagnosis have been studied widely in a variety of pathologies, with rates ranging from 10 to 30%. A discrepancy is defined as any difference between the first and second interpretation. In urology, the greatest focus has been on discrepancy rates seen in prostate biopsies. Epstein et al. [13] found discrepancy rates in prostate biopsies were 1.3%. Werner found 13% discrepancy rates in prostate specimens [13]. Layfield found 8% discrepancy rate when reviewing cytologic material [13] Brimo and colleagues found 14.7% discrepancy rate when reviewing prostate needle biopsies before prostatectomy [7]. Coblentz, in a 2001 study on bladder tumors, cited an 18% discrepancy rate [8]. In the 2013 issue of Archives, Swapp and collegues examined an unprecedented 71,811 outside review cases from a 5 year period at the Mayo Clinic. Their study reinforces the value of accurate surgical pathology diagnosis and should prompt pathologists everywhere to consider repeat review [9].
When patients are referred with a diagnosis of urothelial carcinoma, it is common practice to perform a repeat transurethral resection for staging purposes [10]. However, many urologist will request an internal pathology review of original specimens prior to any procedure [11]. There are several potential benefits to this practice. Outside referrals come from a variety of settings, including community hospitals, private pathology laboratories, and other academic institutions; the experience with bladder specimens can vary significantly among these institutions and influence the accuracy and thoroughness of their pathology reported. In cases of borderline findings, an internal review can help sway treatment decisions in one direction or another. Perhaps more importantly, when a specimen is classified as nondiagnostic, repeat review has the potential to spare patients from an additional procedure if the specimen is deemed adequate on second look.
For the current study, we looked at consecutive patients referred to a single urologist who routinely refers all outside specimens for internal review prior to any procedure. This is intended to minimize selection bias that would be present in a setting where only certain specimens were referred for repeat examination (such as borderline reports or higher risk disease). There are 3 dedicated uropathologists who review the vast majority of the transurethral bladder biopsies and resections at our institution. Our concern would be that there could be institutional-specific trends towards diagnosing specific pathologic characteristics. To better assess this, we compared the original and repeat review rates of the different pathological features to see if there were any inherent biases that would impact the changes in treatment recommendations after repeat review (Table 2). Out of the eight factors initially examined, only the rate of micropapillary features were significantly different, appearing in only 2 of the original report and 9 of the internal review. All other characteristics, including those classified as ‘treatment-altering were similar, indicating that our internal pathologists do not differ in their overall distribution of diagnoses from the general community. This implies that the observed discrepancies in the original and the repeat review internal pathology reports are more likely to be a result of the act of repeat review itself and not institution specific [12].
There was a high rate of discrepancies between the original and internal reviews for the 91 patients samples analyzed (Table 3). The treatment-altering discrepancy rate seen in this study was 29.7%. This is similar to the discrepancy rates previously reported in the few prior studies looking at this topic. Lee et al reported 25.3% of patients who underwent repeat internal review after outside TURBT had sufficient discrepancies to alter their management. Another study by Coblentz cited an 18% discrepancy rate. Our overall clinically- significant discrepancy rate of 61.5% is much higher than either of the previously published studies, and many specimens in our study had multiple discrepancies (Table 4). However, compared to prior studies, the current study included a more thorough and inclusive list of characteristics frequently found on pathology reports and known to frequently impact decision-making. The overall clinical relevance of this number carries less significance than the treatment altering discrepancy rate, but it does help to demonstrate the extreme variability between different pathologic reviews which should be considered each time a repeat review is obtained.
Although there are few consensus statements to be made in the management of urothelial carcinoma, we created a treatment scheme based on 2 core principles outlined in the American Urologic Association clinical practice guidelines for urothelial carcinoma: 1) sampling of the muscularis propria is necessary for adequate staging, and 2) muscle-invasive disease should be managed with cystectomy when possible. Table 5 outlines treatment recommendations based on the findings in the pathological specimens. Of course, this is overly simplified and does not account for other significant yet more subjective considerations such as prior treatments and recurrences as well as other pathological characteristics (i.e., micropapillary features, lymphovascular invasion, etc.) that might influence a patient to undergo cystectomy with non-muscle invasive disease. Additionally, many urologists will perform a repeat resection regardless of initial findings or ultimate plans for radical surgery. Our simplified treatment scheme does provide a generalized classification system that is consistent and reproducible and allows us to estimate the impact of repeat review on treatment decisions in a way that would otherwise be difficult outside of a randomized, controlled study.
The treatment recommendations based on the original reports were relatively evenly distributed (Figure 4). The three initial recommendation groups represent the treatments these patients would have undergone if a repeat review was not performed. The easiest way to analyze the impact of internal repeat review is to look at each of the three initial recommendation groups individually (Figure 5). In the first group which was initially recommended surveillance 8 out of 28 patients where changed to repeat biopsy after internal repeat review. More importantly, no patients were changed to cystectomy. Given the low likelihood of missing muscle-invasive disease on original review, this group of patients is unlikely to be positively impacted by undergoing internal repeat review and would be reasonably managed with follow-up surveillance cystoscopy in 3 months based on the original outside report.
The second group of 29 patients was recommended to undergo repeat resection due to lack of muscularis propria seen in the original pathology report. However, muscle was identified in 10/29 patients on repeat internal review (including 8 with no muscle involvement and 2 with muscle-invasive disease), saving about one third of patients from an unnecessary diagnostic procedure. This group of patients would appear to benefit significantly from internal repeat review. However, there are specific exceptions that may not call for repeat analysis, such as cases when there is no muscle in the specimen but a massive unresectable tumor burden that cannot be managed with a transurethral approach.
For the final group of patients who were originally recommended to undergo cystectomy only 2 patients had revised recommendations to undergo surveillance. Another 7 patients would be advised to have repeat resections; however, repeat resection prior to cystectomy is a common practice for all patients for the accurate staging and surgical planning and may even improve outcomes after cystectomy [13]. Given the relatively small percentage of patients who would be spared from repeat resection, internal repeat review for this purpose is unlikely to be effective. There are specific scenarios in which the repeat review could help make other treatment decisions such as choice of neoadjuvant treatment or choice of diversion.
There are a few apparent limitations in this study. The most obvious is the fact the outcomes are based on theoretical treatment decisions rather than the actual treatments these patients received. An alternative approach would have been to take this group of patients and compare their actual treatments to another group of patients referred with outside pathology that was not internally reviewed. However, the previously-discussed selection biases would preclude a retrospective study of this nature; a randomized controlled study would be necessary. Although we felt our bias was minimized with the current design of consecutive patients from a single urologist, the fact remains that all patients in the current study were referred to one urologist at a tertiary care center who specializes in difficult urothelial carcinoma cases. This undoubtedly influenced the baseline characteristics of the patients who populated this study, making the results less applicable to the general population of patients with urothelial carcinoma and more specific to patients referred to specialists.
Conclusion
Two decades of studies have consistently shown that repeat internal review of pathology reports can be considered a cost-effective form of preventive medicine. Restaging and grading may prevent unnecessary surgery, chemotherapy, and radiotherapy. The patients who seem to benefit the most from repeat review are those with absence of muscularis propria on the original pathology report, but where it was identified on repeat review. Potentially one third of these patients would be spared form the morbidity and expense of a repeat transurethral resection. While Tsung et al. found that repeat reviews should be standard practice and that pathologic societies should adopt strong positions on second opinion [7,14]. Further studies are needed to better determine the actual impact of repeat review and better identify patients most likely to benefit from this practice.
References
- SEER Stat fact sheets: “Bladder Cancer.” National Institute of Health, USA.
- BostwickDG, Cheng L (2008) Urologic surgical pathology. Elsevier Health Sciences, USA.
- Toson I, Wagner U, Sauter G, Egloff M, knonagel H (2000) Clinical significance of interobserver differences in staging and grading of superficial bladder cancer. BJU Int 85: 48-53.
- Bostrom PJ, Bas WG van Rhijn, Fleshner N, Finelli A, Jewett M, et al. (2010) Staging and staging errors in bladder cancer. European Urology Supplements 9: 2-9.
- Cheng L, Montironi R, Davidson D, Lopez-Beltran A (2009) Staging and reporting Urothelial carcinoma of the urinary bladder. Modern Pathology 22: S70-S95.
- Paner GP, Shen SS, Lapetino S, Venkataraman G, Barkan GA (2009) Diagnostic utility of antibody to smoothelin in the distinction of muscularis propria from muscularis mucosae of the urinary bladder. Am J SurgPathol 33: 91-98.
- Allen TC (2013) Second Opinions, Pathologists’ Preventive Medicine. Arch Pathol Lab Med 137: 310-311.
- Coblentz TR, Mills SE, Theodorescu D (2001) Impact of second opinion pathology in the definitive management of patients with bladder cancer. Cancer 91: 1284-1290.
- Swapp RE, Aubry MC, Salomao DR, Cheville JC (2013) Outside case review of surgical pathology for referred patients: the impact on patient care. Arch Pathol Lab Med 137: 233-240.
- Mazzucchelli R, Scarpelli M, Lopez-Beltran A, Cheng L, Di Primio R, et al. (2012) A contemporary update on pathology reporting for urinary bladder cancer. Int J ImmunopatholPharmacol 25: 565-571.
- Gupta D, Layfield LJ (2000) Prevalence of inter-institutional anatomic pathology slide review: a survey of current practice. Am J SurgPathol 24: 280-284.
- Pons F, Orsola A, Morote J, Bellmunt J (2011) Variant forms of bladder cancer: basic considerations of treatment approaches. CurrOncol Rep 13: 216-221.
- Hall MC, Chang SS, Dalbagni G, Pruthi RS, Seigne JD (2007) Guideline for the management of nonmuscle invasive bladder cancer (stages Ta,T1, and Tis): 2007 update. J Urol 178: 2314-2330.
- Epstein JI, Walsh PC, Sanfilippo F (1996) Clinical and cost impact of second pathology: review of prostate biopsies prior to radical prostatectomy. Am J Surg Pathol 20: 851-857.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 5245
- [From(publication date):
February-2017 - Aug 20, 2025] - Breakdown by view type
- HTML page views : 4264
- PDF downloads : 981