Research Article Open Access
Co-Substrating of Peanut Shells with Cornstalks Enhances Biodegradationby Pleurotus ostreatus
| Anike FN, Yusuf M and Isikhuemhen OS* | |
| Department of Natural Resources and Environmental Design, SAES, North Carolina A&T State University, Greensboro, North Carolina, USA | |
| Corresponding Author : | Isikhuemhen OS Department of Natural Resources and Environmental Design, SAES, North Carolina A&T State University, Greensboro, North Carolina, USA Tel: 336-558-8085 Fax: 336-334-7844 E-mail: omon@ncat.edu |
| Received November 01, 2015; Accepted January 22, 2016; Published January 26, 2016 | |
| Citation:Anike FN, Yusuf M, Isikhuemhen OS (2016) Co-Substrating of Peanut Shells with Cornstalks Enhances Biodegradation by Pleurotus ostreatus. J Bioremed Biodeg 7:327. doi: 10.4172/2155-6199.1000327 | |
| Copyright: © 2016 Anike FN, et al. This is an open-a ccess article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Bioremediation & Biodegradation
Abstract
World consumption of peanuts has increased tremendously, resulting in abundance of peanut shell waste. The high lignin content of peanut shells limits their bioconversion to useful products or recycling. Therefore, the synergy in co-substrating peanut shells (PS) and cornstalks (CS) to enhance biodegradation was evaluated. Various compositions of peanut shells and cornstalks (% dry weight) herein called co-substrate - 90PS:10CS, 75PS:25CS, 50PS:50CS, 25PS:75CS, 10PS:90CS, and two controls Cont1-100PS and Cont2-100CS were studied under solid state fermentation (SSF) with a white rot fungus, Pleurotus ostreatus for 120 days. A two-factorial experiment in a completely randomized design (CRD) was used. Results showed that substrate composition and fermentation time were important variables in substrate degradation. Lignin degradation, losses in organic matter, cellulose and hemicellulose increased with time. Increasing the composition of CS in co-substrates resulted in higher lignin loss. The most lignin (40.6%) was lost in co-substrates with 75-90% CS whereas more organic matter was lost in co-substrate containing 50% CS and above. Highest losses in organic matter (24.09%), cellulose (17.41%), and hemicellulose (52.07%) occurred in co-substrate with 50% CS, which is where the greatest reduction in C:N ratio (33%) also occurred. The macro- and micro-element content of co-substrates and the controls varied and increased significantly after fermentation. Co-substrating PS and CS appears to be a promising, environmentally-friendly approach for bioconversion of both agricultural wastes into bio-products with potential application in animal feed, biofuel, or for cultivation of mushrooms.
| Keywords |
| Biodegradation; Peanut shells; Cornstalks; Pleurotus ostreatus; White rot fungi; Lignocellulose; Solid state fermentation; Minerals; Heavy metals |
| Introduction |
| Peanut shells (PS) are abundant agro-industrial waste products that are recalcitrant to degradation under natural conditions [1]. The increasing expansion of peanut production has led to accumulation of large quantities of these shells all over the world. In the US alone, peanut production increased by 45% between 2011 and 2012, bringing production to 3.04 million metric tons [2]. Globally, 45.6 million metric tons of peanuts are produced annually [3]. It has been estimated that for every kg of peanuts produced, 230-300 g of peanut shells are generated [4]. Therefore, as much as 13.7 million metric tons of peanut shells are churned out every year, most of which are dumped into the environment or burned [5,6]. Peanut shells are a renewable resource that could be targeted for purposeful use in the food, feed, paper and bioenergy industries. However, as yet, few if any value-added uses exist for them [7]. |
| Over the years, various attempts to utilize peanut shells have been made. Small fractions were incorporated into animal feed, especially for cattle [8], while some authors have experimented on their use as dietary fiber for humans [9]. Other non-food research trials include uses in pulp production [10], feedstock for bioethanol production [11,12], particle board component [13], and mulch and activated charcoal [7]. However, the major drawback to their use in large scale industrial operations is the high lignin content. |
| Much like other agricultural lignocellulolysic biomass, peanut shells are composed mainly of lignin, cellulose and hemicellulose. Lignin, a complex polymer, is the most recalcitrant component of plants, [14] and binds tightly to, and provides a physical seal around, cellulose and hemicellulose [11,15-18], thereby protecting them against attack by hydrolytic enzymes. The restricted bioavailability of these sugars and other components is thought to be largely physical, with lignin molecules reducing the surface area available to enzymatic penetration and activity [19]. In order to gain access to the sugars in cellulose and hemicellulose, lignin has to be broken down. Compositional analysis shows that lignin could account for up to 41.3% of the lignocellulose in peanut shells [11]. This value is 2 - 4 fold higher than most common agricultural wastes such as rice straw, wheat straw, sugar cane bagasse and corn cob/stover [11,12]. The high lignin content of peanut shells is largely responsible for their recalcitrance to biodegradation under normal environmental conditions; for their reportedly low digestibility in ruminants [6,20-23], and for the cost-intensive hydrolysis that is required to obtain fermentable sugars from them for bioenergy and pulp industries [12]. White rot fungi are nature’s reservoir of inexpensive, environmentally- friendly, complex enzymes that can selectively degrade lignin in situ, and their use for these purposes is27 supported in literature [24,25]. Pleurotus ostreatus, a white rot fungus, is primarily a lignocellulosic biomass decomposer. Several reports show that P. ostreatus can effectively colonize many lignocellulosic substrates [5,26-28], preferentially degrade lignin and leave the bulk of cellulose and/or hemicellulose behind [29,30]. Tests on the potential use of 3 agricultural wastes for cultivating Pleurotus species [5] indicated that the colonization rate of peanut shell substrates was significantly lower than cotton waste or wheat straw. Reports have shown that P. ostreatus could potentially degrade single or combined lignocellulosic substrates [31,32], however degradation was enhanced in combined substrates [27,33,34]. |
| To date, there are no reports on how degradation of peanut shells (PS) could be improved by white rot fungi during solid state fermentation, nor is there information on the effect of co-substrating on delignification of peanut shells. Cornstalks (CS) are widely used for the cultivation of P. ostreatus [5], and are relatively low in lignin (15.1%) [11]. Combining both substrates (PS and CS) could provide the needed synergy to improve degradation and bioconversion of peanut shells to more useful products. The aim of this study was to investigate the degradation of peanut shells alone and in combination with cornstalks by P. ostreatus, and to determine substrate combination level(s) that promote optimum delignification. |
| Materials and Methods |
| Strain and inoculum preparation |
| Pleurotus ostreatus (MBFBL 400) was selected from the Mushroom Biology and Fungal Biotechnology Laboratory (MBFBL) culture collection at North Carolina A&T State University Greensboro, NC, and maintained on potato dextrose agar (PDA) media. The media (39 g of PDA per 1 L deionized water) was prepared according to the manufacturer’s instruction (DIFCO, USA). Media plates (100 × 15 mm) were inoculated with 4 mm agar plugs from actively growing mycelia of MBFBL 400 and incubated at 25°C. For the liquid inoculum, actively growing mycelia (7 days old) was homogenized with 200 ml of sterile water in a Waring laboratory blender. |
| Substrates and experimental design |
| Milled peanut shells were obtained from the Good Peanut Company in Aulander, North Carolina. Cornstalks were obtained from the North Carolina A&T State University Farm. Both substrates were oven-dried at 80°C for 24 hours and further milled separately to 2 mm sieve size in a Thomas Willey Mill. Five grams of peanut shell and corn stalk in 5 replicates were dried separately to constant weight and used to determine the moisture content of starting material. Milled peanut shells (PS) and cornstalks (CS) were mixed in five different compositions by percentage as follows: 90PS:10CS, 75PS:25CS, 50PS:50CS, 25PS:75CS, 10PS:90CS and the controls Cont1-100PS and Cont2-100CS. Substrates composed of PS and CS are subsequently referred to as co-substrates. |
| 15 g of each substrate was brought to 70% moisture content, loaded into a 500 ml Erlenmeyer flask, covered with a foam stopper, sealed with aluminum foil and sterilized at 121°C for 1 hr. Co-substrates and the controls were inoculated by pipetting 8 ml liquid inoculum through an indented hole at the center of the substrates and incubated for 0, 30, 60, 90, and 120 days. Immediately after inoculation, the “0 day” flasks in each treatment were oven-dried at 90°C to constant weight. The experimental design was a 7 × 5 factorial experiment in a completely randomized design (CRD) with 4 replications in each treatment (140 experimental units). |
| At 30 day intervals, selected flasks from each treatment were sampled and oven dried as above. The resulting biomass (fungus+substrate) were weighed and recorded. Loss of organic matter (LOM) was calculated as the percentage difference in weight between the experimental and 0 day flasks in each substrate combination. The samples were then milled to fine particle sizes with a coffee grinder and used for analysis. |
| Sample analysis |
| Milled samples were analyzed for lignin, cellulose, and hemicellulose at the Rumen Fermentation Profiling Lab, West Virginia University (WV) USA, using the procedures for NDF and ADF analysis [35-37]. The carbon and nitrogen (C, N) were analyzed with the Perkin Elmer 2400 Series CHNS/O Elemental Analyzer. Tin capsules (8 × 5 mm) containing 5 mg of samples were loaded into the analyzer. The basis for determining C and N is the “dynamic flash combustion” that converts organic or inorganic samples into combustible products. The released gases from the combusted products were passed through a reduction furnace, separated into individual gases, analyzed in a chromatographic column, and separated and detected by a thermal conductivity detector that gave an output signal proportional to the concentration of the individual components of the mixture. |
| containing 5 mg of samples were loaded into the analyzer. The basis for determining C and N is the “dynamic flash combustion” that converts organic or inorganic samples into combustible products. The released gases from the combusted products were passed through a reduction furnace, separated into individual gases, analyzed in a chromatographic column, and separated and detected by a thermal conductivity detector that gave an output signal proportional to the concentration of the individual components of the mixture. |
| The mineral concentrations (Ca, P, Fe, Pb, Mg, Mn, Zn, Cu and K) were determined through an inductively coupled plasma-atomic emission spectrophotometer (ICP-AES) at the David H. Murdock Research Institute in Kannapolis, NC. Solid samples 0.1 g were predigested with 7.5 ml of concentrated nitric acid (HNO3) and 2.5 ml of concentrated hydrochloric acid (HCl) in unsealed omni vessels for 15 minutes. The vessels were loaded into a high performance Microwave Accelerated Reaction System (MARS 5) and heated for digestion at 200°C for 15 minutes. The resulting liquid samples were diluted with deionized water to bring the volume up to 50 ml for ICP-AES analysis. |
| Statistical analysis |
| The study was conducted as a 2 factorial experiment in a completely randomized design (CRD). Experimental data, which are means of 5 replications, were subjected to Analysis of Variance (ANOVA) with Statistical Analysis Software (SAS) version 9.3 at 5% level of significance. Duncan’s multiple range test was used for multiple comparisons on sample means. Significant differences between means were indicated by the p-value (P<0.05). |
| Results and Discussion |
| Pleurotus ostreatus mediated degradation of substrates composed of various proportions of peanut shells (PS) and cornstalks (CS) were studied over the course of 120 days. P. ostreatus possess enzymes that synergistically degrade lignocellulosic materials in many plant species [38-42]. Little has been done to break the recalcitrance of PS to degradation and generate usable or recyclable bio-products. Discussions on degradation, mineral profiles and lead content of resulting substrates are presented. |
| Loss of organic matter and C:N ratio |
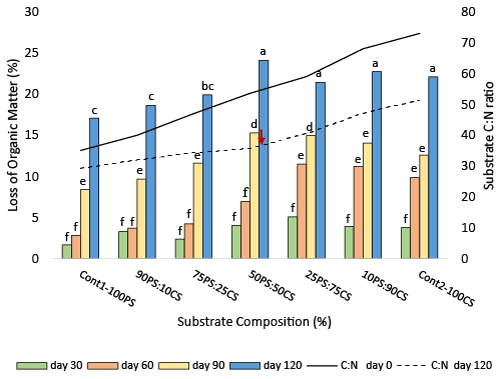
| Loss of organic matter (LOM) resulting from degradation of substrates composed of PS and CS is presented in Figure 1. The time course of organic matter degradation showed similar trend in all substrates. LOM varied among the substrates studied. More organic matter (18.6 – 24.1%) was lost in co-substrates composed of PS and CS, than peanut shell alone (17%) after 120 days. Increasing CS composition of co-substrates beyond 10% increased LOM, although the rate of increase did not differ when substrate CS composition reached 75-100%. The highest organic matter loss (24.1%) was in co-substrate composed of equal amounts of PS and CS (50PS:50CS). At this composition, degradation increased by 2.4 fold over the control-peanut shell (Cont1-PS100). The 22.8% LOM recorded in cornstalks substrate (Cont2-100CS) is similar to 24% reported by others [26,38,43], screened 4 different white rot fungi on two substrate compositions, and found that P. ostreatus had the fastest colonization rate at 50:50 mixtures. Our results show that regardless of substrate composition, degradation increased over time, and LOM ranged from 1.7 - 4% in 30 days, 2.8 - 11.5% in 60 days and 8.4 - 15.3% in 90 days. The highest losses in organic matter were recorded at 120 days, which was also the longest incubation time for the substrates. Siqueira et al. [44] reported similar trends and showed that P. ostreatus colonization of mixed substrates composed of banana and cotton waste and/or bahiagrass varied over time. It is thought that organic matter lost from substrates during degradation were utilized for growth of the fungus and partly lost as CO2 and H2O during respiration [40,45]. The 24.1% loss in organic matter orchestrated by co-substrating PS with CS represents a substantial reduction in mass from the standpoint of peanut shell disposal. |
| Substrate carbon-nitrogen (C:N) balance is a critical factor in degradation of organic compounds present in lignocellulosic residues [46,47] of PS and CS [11]. Carbon, which is needed for energy, is usually present in excess, while nitrogen, which is useful in protein synthesis and other cellular activities, is present in limited amounts. The ratio of carbon to nitrogen in initial substrates (prior to degradation) is higher in CS (73:1) than PS (35:1) [48,49] with in between values in cosubstrates (Figure 1). P. ostreatus degradation of organic matter released essential soluble substances required for fungal metabolism (50), and caused a 16.5 - 33.3% reduction in C:N ratio across the co-substrate and the controls (Figure 1). |
| The highest reduction in C:N ratio (33.3%) occurred in the cosubstrate with equal amounts of PS and CS (50PS:50CS) where C:N ratio is 55:1, and it was also the co-substrate where quantitative loss in organic matter was highest. The results support the finding that maximum colonization and degradation by white rot fungi occur when there is a good balance between substrate carbon and nitrogen, and that optimum C:N ratio is strain [50,51] and substrate dependent. Similar to our finding, variability in C:N ratio in combined substrates were also recorded in cotton seed hulls and wheat bran or cotton seed hull and rice straw [31]. The reduction in C:N ratios may have resulted from reductions in carbon, since carbon is used for cellular metabolism by P. ostreatus during growth, and some are lost as CO2. Future experiments involving PS and CS will utilize a C:N ratio of 55:1 for bioconversion of peanut shells by P. ostreatus in mixed substrate conditions. |
| Degradation of lignin, cellulose and hemicellulose |
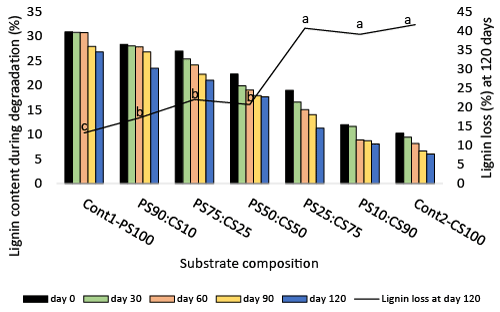
| Among the 3 biopolymers present in non-degraded single and cosubstrates, cellulose was the most abundant (Tables 1 and 2; Figure 2). Peanut shells-Cont1-100PS had higher lignin content (30.82%) than cornstalks -Cont2-100CS (10.24%) (Figure 2), while cornstalks were higher in cellulose (44.41% vs 37.32%) (Table 1) and hemicellulose (27.01% vs 9.12%) (Table 2). Therefore, it is not surprising that cosubstrates with more PS had more lignin, while those with more CS contained more cellulose and hemicellulose. The analysis results indicate that non-degraded peanut shells contain far more lignin (30.82%) than other common agricultural wastes- corn cobs 5.6%; rice straw 9.9%; corn stover 15.1%, and wheat straw 14.5% [11]. Lignin binds with hemicellulose through covalent linkages and physical bindings, and prevents accessibility and biodegradation of plant carbohydrates by microorganisms [52]. Therefore, the recalcitrance of peanut shells to degradation could be attributed mostly to the high lignin content. |
| Lignin |
| The trend of lignin degradation was invariably similar across cosubstrates and increased with time (Figure 2). Co-substrating resulted in higher loss of lignin (17.17-32.75%) compared to control – Cont1- 100PS (13.2%). However in the first 60 days there was no significant degradation of lignin in co-substrates with high (75-90%) PS content compared to control. Lignin degradation was highest at 120 days in all substrates irrespective of composition. More lignin was lost in high (75-90%) vs low (25-50%) CS containing co-substrates. The average lignin degraded at low CS co-substrate (19.94%) differed significantly from high CS co-substrate (40.37%). In both cases losses were higher than peanut shell alone (13.2%). It could not be determined whether increased degradation of lignin in co-substrates resulted from degradation of more accessible lignin in CS or the synergy brought by co-substrating PS and CS. P. ostreatus is widely reported to be a selective degrader of lignin [30,53]. A systematic screening of ligninolytic fungi [54] reported that lignin was preferentially degraded from wheat straw by P. ostreatus. White rot fungi, of which P. ostreatus is an example, possess unique ligninolytic enzymes that completely degrade lignin to CO2 and H2O [55]. These extracellular enzymes depolymerize lignin through the C-C bond and ether bond cleavage reactions [56], and prevent repolymerization between fragmented units [57]. Studies have shown that they selectively degrade lignin while partially decomposing cellulose and hemicellulose [52]. Lignin degradation releases soluble substances and other byproducts of lignolysis, which in turn enhance fungal activities and increase the rate of degradation. |
| Cellulose and hemicellulose |
| Cellulose content in non-degraded substrates varied from 37.32% (Cont1) to 43.93% (Cont2) and in between values in co-substrates (Table 1). There were delayed but significant losses in cellulose in substrates and co-substrates over time, and most losses occurred at 120 days. In general, cellulose degradation was not detected in the first 60 days of fermentation. The observed delay could be related to nonsignificant lignin degradation in the substrate during same period. |
| Significant degradation of cellulose was first recorded at 90 days except in co-substrates 75PS:25CS and 50PS:50C. Further utilization of cellulose did not occur between 90 and 120 days in high PS (75- 100%) containing substrates. However co-substrates with lower amounts of PS (<75%) showed significant losses except 25PS:75CS. It is not clear at this time why cellulose loss in co-substrate 25PS:75CS differed among low PS containing co-substrates. Highest amount of cellulose (17.41%) was lost in co-substrate 50PS:50CS at 120 days compared to day 0 treatment. The results showed that, in contrast to high lignin degradation (40.37%), less cellulose (17.41%) was degraded (Table 1), which are in agreement with Tsang et al. [58], who obtained a 20% average loss in cellulose from degradation of wheat straw by Pleurotus sajor-caju (Pleurotus pulmonarius var. sajor-caju), P. sapidus, P. cornucopiae, and P. ostreatus. |
| Hemicellulose degradation in some co-substrates (containing 50% CS and above) started within the first 30 days of fermentation, making it the first of the 3 biopolymers to be significantly degraded (Table 2). However, hemicellulose remained relatively intact in the control- Peanut shell (Cont1-100PS) throughout the fermentation time. Co-substrating led to significant loss in hemicellulose (20.07-52.07%) compared to 13.38% in the control (Cont1-100PS). Co-substrate composed of equal amount of PS and CS (50PS:50CS) had the highest percentage loss of hemicellulose (52.07%) at 120 days. In contrast less cellulose (17:41%) was degraded at this co-substrate during same period of fermentation. This implies that hemicellulose was utilized more than cellulose by P. ostreatus. It has been reported that hemicellulose is preferentially utilized over cellulose during solid state fermentation by P. ostreatus [27,59,60]. Wang et al. [61] also reported low degradation of cellulose by P. ostreatus cultivated on spent beer grains. |
| Elemental composition of substrates |
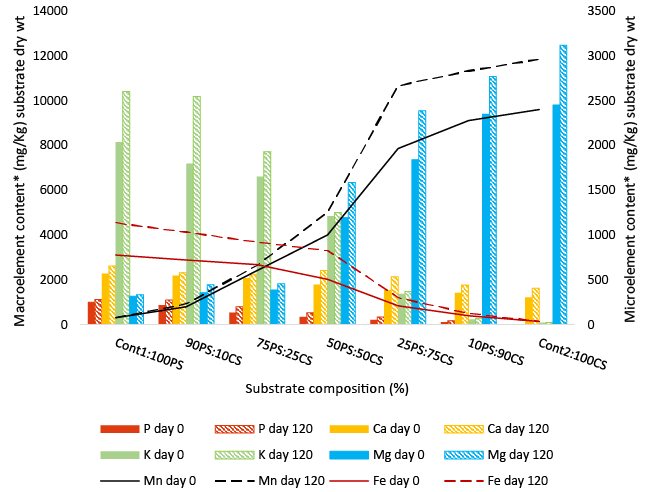
| Plant biomass is a complex heterogeneous mixture composed of organic and inorganic materials, including mineral elements [62]. Plant species vary in the type and quantity of these elements, which could be categorized as macro (large amounts) and micro (trace amounts) depending on concentration generally present. Results show that peanut shells are richer than cornstalks in 6 of 8 tested elements (mg/ kg dry weight); macro elements - potassium (8157 vs. 91), calcium (2279 vs. 1223), and phosphorus (873 vs. 9); micro elements - iron (777 vs. 35), copper (37 vs. 5) and zinc (28 vs. not detected), but lower in magnesium (1268 vs. 9805) and manganese (74 vs. 2402) (Table 3). Zinc was not detected in cornstalks (Cont2). At the end of 120 days fermentation, both co-substrates and the controls contained increased amounts of macro and micro elements compared to non-degraded substrates (Figure 3). Greater increases were recorded in co-substrates with 50-90% CS and the control- 100% CS). The greatest increases, and the co-substrates where they occurred, were phosphorus 40% (100CS:0PS), calcium 28% (75CS:25PS), potassium 29% (75CS:25PS), magnesium 27% (50CS:50PS), manganese 28% (75CS:25PS) and iron 38% (50CS:50PS). Kasuya et al. [63] reported an increase in mineral content of coffee Husks for use in animal feed after degradation by Pleurotus ostreatus. Mineral contents may have increased as a result of lowering of organic matter during degradation. Minerals contributed by PS and CS to the co-substrates could have influence colonization and degradation by P. ostreatus. |
| Presence of lead (Pb) in substrates |
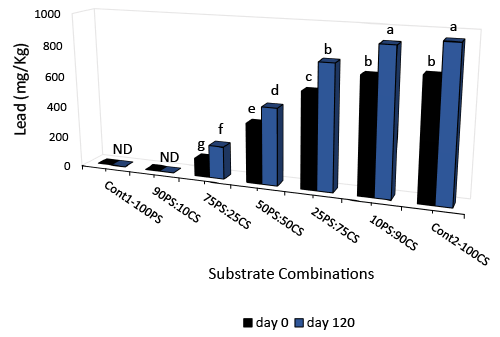
| The presence of lead in the substrates and co-substrates is shown in Figure 4. Lead (mg/kg dry weight) was detected CS (813.6 mg), but not in PS. In co-substrates, lead was only detected when the proportion of CS ranged between 75%-100%. In this co-substrate, lead content increased with incubation time and was highest at 120 days. The increase in lead from day 0 to day 120 is significant, and ranged from 194.43 mg/kg in CS-only substrates, to 94.69-185.87 mg/kg in cosubstrates where detected. |
| Lead is toxic to humans and animals, and the quantities found in CS containing substrates before (115.41 – 757.92 mg/kg dry weight) and after (210.10-952.35) degradation are very high when compared to the limit allowed to be dumped into soil − 420 mg/kg [64,65], or the permissible limit for human ingestion (0.1 mg/kg) [66]. There has been increasing concern in recent years over lead (Pb) transfer in soilplant systems. Li et al. [67] reported that soil factors such as pH and organic matter increase bioaccumulation of lead in corn grain. Also, corn seedlings have been used to bio-accumulate Pb in contaminated soil [68]. Significant levels of Pb get to plants and the food chain through atmospheric deposition from industrial waste and smokestack emissions, vehicle exhaust and degradation, and agricultural uses of fertilizers and pesticides [69]. It has been shown that vegetative mycelia and mushroom fruit bodies accumulate heavy metals from their substratum [70], and in high proportions too [71]. Since our result and those of others show that CS used in the cultivation of P. ostreatus bio-accumulate Pb [67], we recommend that CS used for cultivation of mushrooms, organic composts and other biomaterials of economic importance should be tested for levels of Pb before use. This will prevent unintended contamination of human food, animal feed, agricultural soils and dwellings with Pb [71]. |
| Conclusion |
| Peanut shells (PS) are high in lignin, resulting in recalcitrance to degradation. Biological degradation of PS by P. ostreatus and, by extension, other white rot fungi, could be enhanced by co-substrating with cornstalks. Increasing the composition of CS in co-substrates resulted in higher lignin loss. When both substrates were mixed at equal proportions (percent composition), a 2.4 fold loss in organic material occurred; substrate carbon:nitrogen ratio reduced significantly; more lignin was degraded and hemicellulose was utilized more than cellulose. PS was richer than CS in 6 of the 8 minerals tested and degradation resulted in higher mineral content in all substrates/co-substrates. The increased removal of lignin in co-substrates of PS and CS suggests that the combination of the two substrates could be a useful means for biodegrading peanut shells to obtain commercially-useful bio-products with potential application in animal feed, biofuel, or for cultivation of mushrooms. |
References
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
Relevant Topics
- Anaerobic Biodegradation
- Biodegradable Balloons
- Biodegradable Confetti
- Biodegradable Diapers
- Biodegradable Plastics
- Biodegradable Sunscreen
- Biodegradation
- Bioremediation Bacteria
- Bioremediation Oil Spills
- Bioremediation Plants
- Bioremediation Products
- Ex Situ Bioremediation
- Heavy Metal Bioremediation
- In Situ Bioremediation
- Mycoremediation
- Non Biodegradable
- Phytoremediation
- Sewage Water Treatment
- Soil Bioremediation
- Types of Upwelling
- Waste Degredation
- Xenobiotics
Recommended Journals
Article Tools
Article Usage
- Total views: 13880
- [From(publication date):
January-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 12659
- PDF downloads : 1221
