Review Article Open Access
Dental Complications of Sickle Cell Disease
Samir K. Ballas1, 2*, Clarisse Lopes de Castro Lobo1 and Wellington Espirito Cavalcanti1
1Clinical Hematology Division, Instituto de Hematologia Arthur de Siqueira Cavalcanti—HEMORIO, Rio de Janeiro, RJ, Brazil
2Cardeza Foundation for Hematologic Research, Department of Medicine, Jefferson Medical College, Thomas Jefferson University, Philadelphia, PA USA
- Corresponding Author:
- Samir K. Ballas
MD FACP, Cardeza Foundation, 1020 Locust Street
Philadelphia, PA 19107, USA
Tel: 856-745-6380
Fax: 856-795-0809
E-mail: samir.ballas@jefferson.edu
Received date August 12, 2014; Accepted date September 29, 2014; Published date October 6, 2014
Citation: Ballas SK, Lobo CLDC, Cavalcanti WE. (2014) Dental Complications of Sickle Cell Disease. J Interdiscipl Med Dent Sci 2:152. doi:10.4172/2376-032X.1000152
Copyright: © 2014 Ballas SK, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Visit for more related articles at JBR Journal of Interdisciplinary Medicine and Dental Science
Abstract
Sickle cell is a complex inherited molecular disorder of hemoglobin structure. Recurrent painful vaso-occlusive crises are its hallmark. In addition, it is associated with chronic hemolytic anemia and its consequences that often require blood transfusion. Moreover, infection and progressive organ and tissue damage result in several morbidities and increased mortality. Almost all body organs and tissues are affected including the nervous, cardiopulmonary, hepatobiliary, gastrointestinal, musculoskeletal, genitourinary, endocrine and dermatologic systems. Although most of these systems are relatively well described and studied, a few other systems have been neglected. Most common among these is the otolaryngological and dental systems despite the fact that patients and providers are aware that dental problems among patients with sickle cell disease are common and are often associated with other
complications of the disease. This review describes the known dental complications of sickle cell disease with the hope that it may generate more studies and clinical trials on this neglected aspect of sickle cell disease.
Keywords
Sickle cell disease; Dental complications; Caries; Malocclusion; Pulp necrosis; Gingivitis.
Introduction
Sickle cell disease (SCD) is an inherited disorder of hemoglobin (Hb) structure where the sickle gene is inherited homozygously from both parents in the case of sickle cell anemia (SS). In other types of SCD there is double heterozygosity where one parent has the sickle gene and the other parent has an abnormal gene for other types of abnormal Hb or β-thalassemia. The most common types of Hb SCD in decreasing order of prevalence include SS, Hb SC, sickle-β+-thalassemia, sickle- β0-thalassemia and Hb SX where X could be any other type of abnormal Hb such as Hb D. Hb E, Hb O-Arab, etc. The sickle mutation is the result of a single base change (GAG→GTG) in the sixth codon of exon 1 of the β-globin gene responsible for the synthesis of the β-globin polypeptide of the Hb molecule (α2β2). This change, in turn, results in replacement of the normal glutamic acid with valine at position 6 of the β-globin chain and the formation of sickle Hb (βsβs) [1-3]. In Hb SC disease the mutation results in replacement of the normal glutamic acid with lysine at position 6 of one of the β-globin polypeptide in addition to the replacement of the normal glutamic acid with valine at position 6 of the other allelic β-globin polypeptide resulting in Hb SC (βsβc).
Clinical Features of SCD
SCD is basically a triumvirate of pain syndromes, anemia & its sequelae and organ failure including infection. In addition, comorbid conditions often occur especially among the older adult patients. Pain, however, is the hallmark of SCD and dominates its clinical picture throughout the life of affected patients. All body organs are affected or are at risk to be affected by SCD. Thus, complications of the musculoskeletal, nervous, cardiopulmonary, hepatobiliary, genitourinary and the endocrine systems are commonly known and described in the literature [4]. Other systems, however, lack a similar attention to address their prevalence, clinical manifestations, management and association with the other complications of SCD. These include the otological, craniofacial, orofacial and dental complications of SCD [4]. The aim of this report is to describe the scanty studies about the dental complications and emphasize the need for controlled clinical trials on this neglected aspect of SCD. Table 1 lists the dental complications described in the literature. Most of these are case reports, observational, anecdotal or pilot studies in nature. Details on some of these are described below.
Literature Review of Dental Complications in SCD
Dental caries
Caries are the most common dental diseases globally and in patients with SCD as well. Caries are disorders of teeth characterized by progressive demineralization and destruction of the enamel, dentin and cementum of the teeth. Acidification of the oral environment is the major cause of caries. Acidification is caused by the fermentation of residual food particles (mostly sugars or carbohydrates) on the surface of teeth. Fermentation, in turn, is catalyzed by resident bacterial micro-organisms on dental plaques (also called “bio film” in Brazil). Untreated, caries cause gradual destruction and fractures of teeth and are associated with infection of the surrounding soft tissues and, in rare cases, thrombosis of the cavernous sinus that could be fatal. Moreover, caries often cause bad taste and halitosis that could be socially embarrassing. Dental caries are also referred to as tooth decay, dental decay or dental cavities. Cariology is a branch of dentistry devoted to the study of dental caries [5-7].
| Dental Complicationsof Sickle Cell Disease |
|---|
| Dental Caries |
| Dental Erosions |
| Infractions |
| Hypodontia |
| Malocclusions |
| Pulp Necrosis |
| Abnormal Trabecular spacing |
| Infection |
Table 1: Reported dental complications of sickle cell disease.
Patients with SCD seem to be more susceptible to dental caries than control patients without SCD [8,9]. The frequent use of soft drinks containing sugar for hydration and inadequate oral hygiene are predisposing factors. Moreover, the prevalence of caries in children and adults with SCD seem to be associated with the socioeconomic status of affected families. Thus, Adult African Americans with SCD and lowincome (< $15,000) and Brazilian children with SS whose families have low-income (< the Brazilian Minimum Wage) seem to be at high risk for dental caries and are less likely to receive adequate oral hygiene and preventative therapy [8,9]. One older report, however, found lower risk for caries in patients with SCD [10].
Recently, Passos et al. [11] reported that SCD does not specifically predispose to caries or periodontal disease. Risk factors known to cause caries and periodontal disease such as older age, daily smoking and lack of daily dental flossing had more influence on oral complications than the direct impact of SCD [11]. Moreover, de Matos et al. evaluated the oral microbial colonization of pediatric patients with SS after longterm prophylaxis with penicillin [12]. They found significantly higher oral level of yeasts and uncommon fungal species in the SS group compared to controls but the prevalence of caries was similar in both SS and controls [12]. Whether the utilization of oral opioids is associated with increased prevalence of dental caries among patients with SCD is unknown. Severe carious lesions have been reported in patients without SCD who used methadone syrup in a drug rehabilitation program [13]. In addition, there are anecdotes of multiple dental problems among patients with SCD including edentulous patients who lost all their decayed teeth.
Dental erosion
These are erosions of enamel due to acidity that is not caused by bacteria but by food especially acidic fruit juices. Intrinsic causes of acidity include certain disorders such as gastroesophageal reflux where gastric acid is in contact with teeth. In severe cases erosion may spread to involve dentin beneath enamel. Dental erosion is also known as acid erosion. Dental erosions have not been described in patients with SCD [14,15].
Infractions
These are micro-cracks within the enamel of teeth. Although they have been observed anecdotally by providers and patients with SCD they have not been reported in this patient population.
Hypodontia
This refers to missing teeth due to failure for them to develop. It is also known as tooth agenesis. It has been described in one patient with Hb SC disease [16].
Malocclusion
In dentistry occlusion refers to the way opposing teeth meet. Edward Angle, the father of modern orthodontics introduced the term malocclusion which is defined as a misalignment of the teeth between the two corresponding dental arches [17].
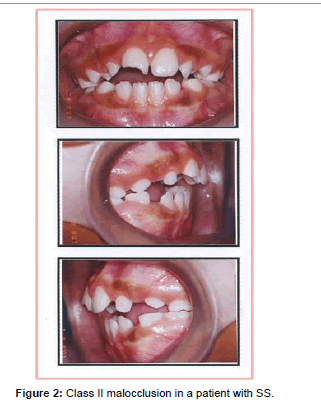
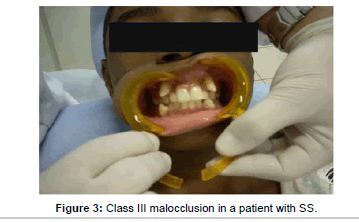
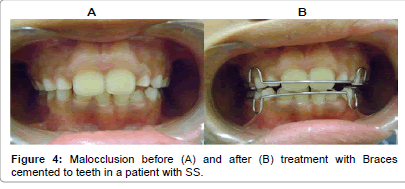
Angle divided malocclusion into three classes depending on the severity and the relation between the teeth involved. In class I the occlusion in a patient with SS (Figure 1) is normal for the first maxillary molar but abnormal for the other teeth with crowding, rotation, and spacing and over or under eruption. In Class II (Figure 2) there is discrepancy in jaw growth in a patient with SS where the lower jaw is deficient in forward growth causing an overjet (often referred to as an overbite) > 3 mm. In Class III (Figure 3) malocclusion in a patient with SS the lower jaw grows too far forward ahead of the upper jaw causing an under-bite also referred to as negative overjet [18]. Management of malocclusion includes the application of braces to the protruding teeth as shown in a patient with SS (Figure 4). The braces are ceramic or metallic sets of wire that are cemented to the teeth for 2 – 3 years. They are adjusted and tightened periodically by applying gradually increasing force to move the teeth into alignment.
Malocclusion has been described in sickle cell disease. Most of the reports, however, are case reports and retrospective or prospective observational studies [10,19-22]. Table 2 lists the prevalence of the three Classes of malocclusion in patients with SS in Lagos, Nigeria [22] and in Rio de Janeiro at HEMORIO. These rep orts showed that Class II malocclusion was most common in patients with SCD. The pathophysiology of malocclusion in SCD is not well known. It is thought that hypoxia secondary to sickling is associated with osteonecrosis of the jaw especially in the mandible causing mental nerve neuropathy [4,23]. Bone marrow hyperplasia in facial bones is also associated with depression of nasal bridge, midfacial overgrowth and malocclusion. Moreover, facial growth alterations which are common in SCD might influence malocclusion due to protrusive maxilla and more forward growth tendency of the mandible [24,25]. In some studies dental abnormalities were also relatively common in control subjects without SCD and in one study the prevalence was higher in controls [10]. Thus the pathophysiology of the association between SCD and dental complications is not clear and the scientific proof that SCD causes malocclusion is insufficient [26,27].
| Malocclusion Class | Lagos Patients*(n) % | HEMORIO patients(n) % |
|---|---|---|
| Class I | (90) 77 | (87) 43 |
| Class II | (25) 21 | (107) 52 |
| Class III | (2) 2 | (10) 5 |
| Total patients studied | (117) 100 | (204) 100 |
Numbers indicate the number and the % of patients with sickle cell disease for each class of malocclusion. The prevalence of all classes of malocclusion in Lagos was significantly higher (p < 0.05) than in normal matched children who did not have sickle cell disease. The data from HEMORIO is Institutional data that has not been reported before.
Table 2: Prevalence of the Classes of malocclusion in patients with sickle cell disease in Lagos and in HEMORIO.
Pulp necrosis
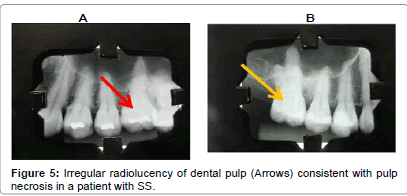
Pulp necrosis (also referred to as Queratinosis in Brazil) or dead pulp (Figure 5) is defined as necrotic dental pulp due to trauma, infection or chemical reaction characterized by no response to thermal stimulation. A necrotic dental pulp causes toothache, discoloration of the tooth, acute apical periodontitis, or dental abscess [28]. Two observational reports suggest that SS is a potentially risk factor for pulp necrosis in intact permanent teeth [29,30].
Infection
Poor dental hygiene leads to the formation of plaque on teeth when food particles interact with bacteria normally present in the mouth, Plaque that remains on the teeth for a long time hardens and forms tartar also called calculus. Persistent plaque and tartar cause gingivitis. If untreated, gingivitis progresses to more serious complications such as chronic periodontitis, dental abscess and bone destruction culminating in tooth loss [31]. Periodontal infection may precipitate painful vaso-occlusive crises and increase the frequency of hospital admissions among adult patients with SCD [32,33].
Iron overload
Scrapings obtained from the buccal mucosa of multiply transfused 40 patients with β-thalassemia major and 20 patients with SS and stained with Prussian blue stain showed the presence of iron in 72.5% of the thalassemia patients and 35% of the SS patients. None of the control normal subjects had iron present in scrapings from their buccal mucosa [34]. Moreover, the prevalence of dental caries and periodontal diseases was significantly more in β-thalassemia patients followed by SS patients than control group [35]. In addition, the presence of decayed missing filled teeth was significantly (p < 0.001) higher in the thalassemia group than in patients with SS [35]. Patients with β-thalassemia usually receive more blood transfusion than patients with SS. Together; these findings suggest that dental complications of patients with β-thalassemia and to lesser extent patients with SS are associated with iron overload. Further studies are needed to confirm these findings.
Conclusion
The strength of this mini review is the emphasis on the importance of dental complications in SCD which often precipitate or worsen other clinical features of the disease including vaso-occlusive crises and systemic infections. The weakness of the review is the lack of evidence that certain dental complications such as caries are specific to SCD bur are as frequent as in other patient populations. Specifically, patents with SCD often experience orofacial pain which seems to be of two types. The first type occurs in the absence of dental pathology and is probably associated with vaso-occlusive painful crises [29,36]. The pathophysiology of this type of pain seems to be due to vasoocclusion within the microcirculation of the facial bones and dental pulp with small microinfarcts in the bone marrow [36]. The second type of orofacial pain is due to specific oral and dental pathology including dental caries, pulpal necrosis, enamel hypomineralization and infections [37,38]. Dental care and dental hygiene among patients with SCD is a neglected area that requires more studies. Moreover, some dentists in Urban USA refuse to see patients receiving medical assistance or Medicare [8,38,39].
References
- Ingram VM (1956) A specific chemical difference between the globins of normal human and sickle-cell anaemiahaemoglobin.Nature 178: 792-794.
- INGRAM VM (1959) Abnormal human haemoglobins. III. The chemical difference between normal and sickle cell haemoglobins.BiochimBiophysActa 36: 402-411.
- INGRAM VM (1957) Gene mutations in human haemoglobin: the chemical difference between normal and sickle cell haemoglobin.Nature 180: 326-328.
- Ballas SK (2014) Sickle cell pain, 2nd edition, International Association for the Study of Pain (2ndedn), IASP, Washington DC, USA.
- Rogers AH (2008) Molecular oral microbiology,Caister Academic Press, Norfolk, UK.
- Johnson C (2007) Biology of the human dentition II.
- Ratini M (2014) Cavernous sinus thrombosis.
- Luna AC, Rodrigues MJ, Menezes VA, Marques KM, Santos FA (2012) Caries prevalence and socioeconomic factors in children with sickle cell anemia.Braz Oral Res 26: 43-49.
- Laurence B, George D, Woods D, Shosanya A, Katz RV, et al. (2006) The association between sickle cell disease and dental caries in African Americans.Spec Care Dentist 26: 95-100.
- Okafor LA, Nonnoo DC, Ojehanon PI, Aikhionbare O (1986) Oral and dental complications of sickle cell disease in Nigerians.Angiology 37: 672-675.
- Passos CP, Santos PR, Aguiar MC, Cangussu MC, Toralles MB, et al. (2012) Sickle cell disease does not predispose to caries or periodontal disease.Spec Care Dentist 32: 55-60.
- de Matos BM, Ribeiro ZE, Balducci I, Figueiredo MS, Back-Brito GN, et al. (2014) Oral microbial colonization in children with sickle cell anaemia under long-term prophylaxis with penicillin.Arch Oral Biol 59: 1042-1047.
- Sheedy JJ (1996) Methadone and caries. Case reports.Aust Dent J 41: 367-369.
- Dugmore CR, Rock WP (2004) A multifactorial analysis of factors associated with dental erosion.Br Dent J 196: 283-286.
- Gandara BK, Truelove EL (1999) Diagnosis and management of dental erosion.J Contemp Dent Pract 1: 16-23.
- Oredugba FA (2005) Hypodontia in an adolescent with the HbSC genotype: a case report.Int J Paediatr Dent 15: 455-458.
- Gruenbaum T (2010) Famous figures in dentistry. Mouth-JASDA 30: 18.
- Oltramari-Navarro PV, de Almeida RR, Conti AC, Navarro Rde L, de Almeida MR, et al. (2013) Early treatment protocol for skeletal Class III malocclusion.Braz Dent J 24: 167-173.
- Alves e Luna AC, Godoy F, de Menezes VA (2014) Malocclusion and treatment need in children and adolescents with sickle cell disease.Angle Orthod 84: 467-472.
- Pithon MM (2011) Orthodontic treatment in a patient with sickle cell anemia.Am J OrthodDentofacialOrthop 140: 713-719.
- daCosta OO, Kehinde MO, Ibidapo MO (2005) Occlusal features of sickle cell anaemia patients in Lagos, Nigeria.Niger Postgrad Med J 12: 121-124.
- Oredugba FA, Savage KO (2002) Anthropometric finding in Nigerian children with sickle cell disease.Pediatr Dent 24: 321-325.
- Konotey-Ahulu FI (1972) Mental-nerve neuropathy: a complication of sickle-cell crisis.Lancet 2: 388.
- Shnorhokian HI, Chapman DC, Nazif MM, Zullo TG (1984) Cephalometric study of American black children with sickle-cell disease.ASDC J Dent Child 51: 431-433.
- Lawson W, Patel ZM, Lin FY (2008) The development and pathologic processes that influence maxillary sinus pneumatization.Anat Rec (Hoboken) 291: 1554-1563.
- Javed F, Correa FO, Nooh N, Almas K, Romanos GE, et al. (2013) Orofacial manifestations in patients with sickle cell disease.Am J Med Sci 345: 234-237.
- Costa CP, de Carvalho HL, Thomaz EB, Sousa Sde F (2012) Craniofacial bone abnormalities and malocclusion in individuals with sickle cell anemia: a critical review of the literature.Rev Bras HematolHemoter 34: 60-63.
- Kontakiotis EG, Filippatos CG, Stefopoulos S, Tzanetakis GN (2014) A prospective study of the incidence of asymptomatic pulp necrosis following crown preparation.IntEndodJ .
- DemirbaÅŸ Kaya A, Aktener BO, Unsal C (2004) Pulpal necrosis with sickle cell anaemia.IntEndod J 37: 602-606.
- Costa CP, Thomaz EB, Souza Sde F (2013) Association between Sickle Cell Anemia and Pulp Necrosis.J Endod 39: 177-181.
- [No authors listed] (2000) Parameter on plaque-induced gingivitis. American Academy of Periodontology.J Periodontol 71: 851-852.
- Rada RE, Bronny AT, Hasiakos PS (1987) Sickle cell crisis precipitated by periodontal infection: report of two cases.J Am Dent Assoc 114: 799-801.
- Laurence B, Haywood C Jr, Lanzkron S (2013) Dental infections increase the likelihood of hospital admissions among adult patients with sickle cell disease.Community Dent Health 30: 168-172.
- Chittamsetty H, Sekhar MS, Ahmed SA, Suri C, Palla S, et al. (2013) A Non-Invasive Technique which Demonstrates the Iron in the Buccal Mucosa of Sickle Cell Anaemia and Thalassaemia Patients who Undergo Repeated Blood Transfusions.J ClinDiagn Res 7: 1219-1222.
- Singh J, Singh N, Kumar A, Kedia NB, Agarwal A (2013) Dental and periodontal health status of Beta thalassemia major and sickle cell anemic patients: a comparative study.J Int Oral Health 5: 53-58.
- O'Rourke CA, Hawley GM (1998) Sickle cell disorder and orofacial pain in Jamaican patients.Br Dent J 185: 90-92.
- da Fonseca M, Oueis HS, Casamassimo PS (2007) Sickle cell anemia: a review for the pediatric dentist.Pediatr Dent 29: 159-169.
- Laurence B, Reid BC, Katz RV (2002) Sickle cell anemia and dental caries: a literature review and pilot study.Spec Care Dentist 22: 70-74.
- Titsas A, Ferguson MM (2002) Impact of opioid use on dentistry.Aust Dent J 47: 94-98.
Relevant Topics
- Cementogenesis
- Coronal Fractures
- Dental Debonding
- Dental Fear
- Dental Implant
- Dental Malocclusion
- Dental Pulp Capping
- Dental Radiography
- Dental Science
- Dental Surgery
- Dental Trauma
- Dentistry
- Emergency Dental Care
- Forensic Dentistry
- Laser Dentistry
- Leukoplakia
- Occlusion
- Oral Cancer
- Oral Precancer
- Osseointegration
- Pulpotomy
- Tooth Replantation
Recommended Journals
Article Tools
Article Usage
- Total views: 27276
- [From(publication date):
December-2014 - Aug 19, 2025] - Breakdown by view type
- HTML page views : 22302
- PDF downloads : 4974