Double-Blind Randomized Comparison of Carbetocin Versus Oxytocin for Decrease Blood Loss from Cesarean Section in Low-Risk Uterine Atony
Received: 24-Aug-2017 / Accepted Date: 04-Oct-2017 / Published Date: 11-Oct-2017 DOI: 10.4172/2376-127X.1000350
Abstract
Objective: The primary objective focuses on amount of blood loss from cesarean section in low-risk uterine atony patients in the carbetocin group comparing to that in the oxytocin group. The secondary objectives are to compare the incidences of postpartum haemorrhage (PPH) and blood transfusion, additional uterotonic use, hemoglobin and hematocrit change within 24 h after cesarean section and side effects between each group. Materials and methods: There were 122 pregnant women eligible in this non-inferior double-blind randomized controlled study comparing two uterotonic drugs (carbetocin and oxytocin) used in cesarean section at department of obstetrics and gynecology, faculty of medicine vajira hospital, navamindharathiraj university from July 2016 to January 2017. The data including amount of blood loss, incidences of PPH and blood transfusion, additional uterotonic use, hemoglobin and hematocrit change in 24 h after cesarean section and side effects were collected. Results: There were 61 pregnant women in each group. Amounts of blood loss in carbetocin group at immediate post opération and 24 h after operation were less than those in oxytocin group, but there was no statistical significance (P=0.097 and 0.1, respectively). In oxytocin group, there was significantly greater additional uterotonic use, but there was no statistic difference in incidences of PPH and blood transfusion, hemoglobin and hematocrit change within 24 h after cesarean section and side effects in both groups. Conclusion: Blood loss in low-risk uterine atony cesarean section in carbetocin was not different from oxytocin. Although additional uterotonic use was significantly lower in carbetocin group, incidences of PPH and blood transfusion, hemoglobin and hematocrit change within 24 h after cesarean section and side effects in both groups were not different.
Keywords: Carbetocin; Oxytocin; Cesarean section; Postpartum hemorrhage
Introduction
Nowadays, postpartum hemorrhage (PPH) has been still the common cause of maternal death after delivery with the incidence of 1:1,000, especially in developing countries [1]. The most common cause of PPH is uterine atony [1,2]. Cesarean section itself is one of PPH risk factors [3,4]. For this reason, World Health Organization (WHO 2012) and the Royal Thai College of Obstetrics and Gynecologists (RTCOG 2015) suggest the PPH prevention methods by administration of a uterotonic drug immediately after childbirth by cesarean section combined with controlled cord traction for placental delivery [5].
Carbetocin is an eight-amino acid long analogue of oxytocin and, in accordance, has a similar action with longer half-life [5]. Although, oxytocin is the drug of choice for PPH prevention, several trials from Italy, Canada, Philippines and United Kingdom and Cochrane Database of Systematic Reviews reported that the use of carbetocin could decrease incidences of PPH and blood transfusion, reduce the additional uterotonic drug use or uterine massage and lower side effects such as nausea and vomiting comparing to oxytocin use [6-10]. However, there was no significant difference in incidence of PPH reported in some researches; therefore the cost and benefit of its use should be considered [6-10].
In our Department of Obstetrics and Gynecology, cesarean section rate and carbetocin use trend to be rising while there has had no guideline or data about carbetocin use in Thailand yet. For these reasons, this research was conducted for studying the benefit of carbetocin comparing to oxytocin in low risk uterine atony cesarean section. The primary objective focuses on amount of blood loss from cesarean section in the carbetocin group comparing to that in the oxytocin group. The secondary objectives are to compare the incidences of PPH and blood transfusion, additional uterotonic use, hemoglobin and hematocrit change within 24 h after cesarean section and side effects between each group.
Materials and Methods
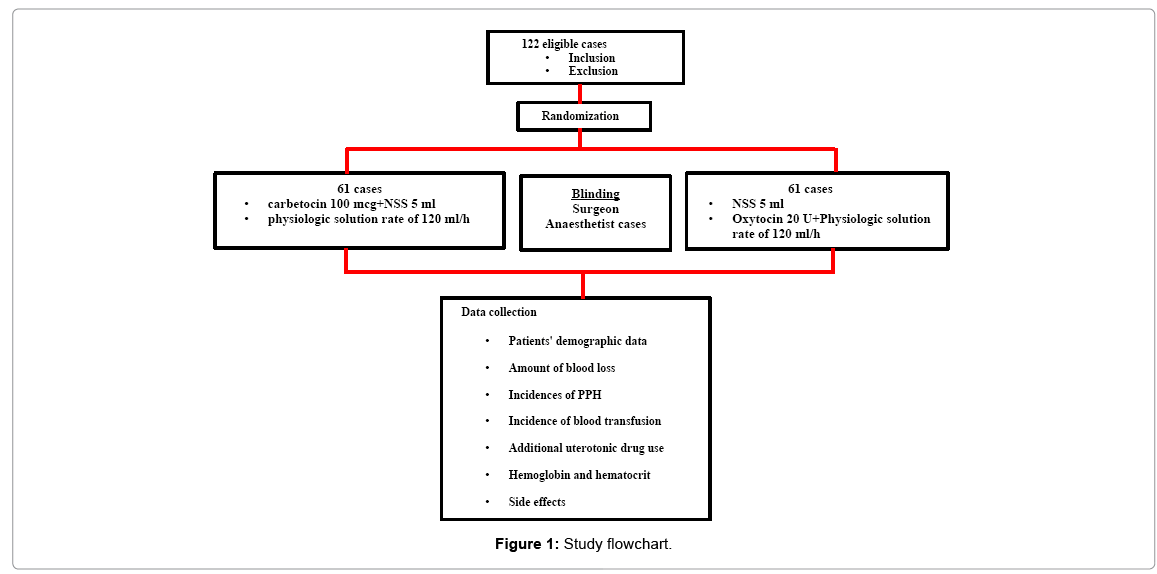
Non-inferiority double blind randomised controlled study comparing between carbetocin and oxytocin for PPH prevention in low risk uterine atony undergoing cesarean section was conducted at department of obstetrics and gynecology, faculty of medicine vajira hospital, navamindharathiraj university, Thailand from July 2016 to January 2017. This study was approved by Vajira Institutional Review Board (VIRB). From Uy et al., sample size was calculated by using a 100 ml estimated difference in intra-operative blood loss in carbetocin group comparing to oxytocin group [8]. The formula was shown below. With 80% of power and 95% of confidence, there were 61 participants needed in each group. The inclusion criteria were age 18 or greater, uncomplicated term singleton, having indication for cesarean section and no regional anesthetic limitation. The exclusion criteria were fetal distress, having uterotonic contraindication, acquired or drug induced coagulopathy, severe anemia (Hb ≤ 8 g/dl) and having risks of PPH such as prolonged latent phase of labor, having history of PPH, multiparity (more than 4 childbirths), antepartum massive blood loss, placenta previa, placenta abruption and placenta adherence [11,12].
Mathematical formula for sample size
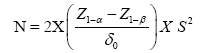
There were 122, aged 18 or greater, uncomplicated term singleton pregnancies who had indications for cesarean section with regional anesthesia recruited from ante-natal care unit and labor room. All participants were taken history, physical examination and ultrasound evaluation determining number of fetus, estimated fetal weight and amniotic fluid measurement. If there was no abnormality detected from the history, normal physical examination, no signs and symptoms of chorioamnionitis or pregnancy induced hypertension, no polyhydramios and estimated fetal weight less than 4500 g, the participants were defined to be uncomplicated pregnancy. No one was excluded from the study. Each patient received the information about the study. After answering their questions until clear understanding, the written informed consents were obtained.
All participants were randomized into 2 groups (carbetocin and oxytocin groups) by using computer generated. In carbetocin group, the patients received 100 μg carbetocin mixed in 5 ml normal saline slowly intravenous push and then 1,000 ml physiologic solution intravenous drip 120 ml/h after childbirth. In oxytocin group, the patients received 5 ml normal saline slowly intravenous push and then 20 units oxytocin mixed in 1,000 ml physiologic solution intravenous drip 120 ml/h after childbirth. All sets of drugs were contained in similar packaging by research nurse who did not associate with the surgical procedure or postpartum care. The ordinal numbers from 1 to 121 were labeled on each set before sending to the operative theater. The investigators, patients, surgeons, anesthetists and nurses at postpartum care unit did not know the random allocation sequence until the study finished.
Intra-operative blood loss was the sum of amount of blood in suction bag and estimated blood volume from all blood-stained swabs while postoperative blood loss was calculated by weighing all sanitary pads used within 24 h. Total amount of blood loss was confirmed by hemoglobin and hematocrit change at 24 h post cesarean section. Blood transfusion depended on acceptable blood loss that was calculated by anesthetists. During the operation, the additional uterotonic drugs could be administered if the surgeons perceived inadequate uterine contraction routine pre- and post-operative care was performed.
Patients’ demographic data, amount of blood loss, incidences of PPH and blood transfusion, additional uterotonic use, hemoglobin and hematocrit change in 24 h after cesarean section and side effects were collected.
The data were analyzed by intention to treat using the SPSS software version 22. Categorical data were analyzed by using chi-square tests and continuous variables were analyzed by using Student’s t tests. Mann-Whitney U tests were used for variables that were not normally distributed. Statistical significance was considered at P<0.05.
Results
All 122 eligible participants were randomized into 2 groups. Patients’ demographic data were similar in both groups (Table 1 and Figure 1).
| Characteristics | Carbetocin group | Oxytocin group |
|---|---|---|
| (n=61) | (n=61) | |
| Mean age (years)(SD) | 29.20 (5.856) | 29.46 (6.508) |
| BMI (kg/m2) (SD) | 27.72 (3.66) | 28.69 (5.55) |
| Parity (n) (%) | ||
| 0 | 16 (26.2) | 23 (37.7) |
| 1 | 34 (55.7) | 26 (42.6) |
| ≥ 2 | 11 (18) | 12 (19.7) |
| Geatitional age (days) (SD) | 269.85 (4.58) | 271.61 (5.62) |
| Hb (g/dl) (SD) | 11.75 (1.12) | 11.74 (1.24) |
| Hct (%) (SD) | 35.43 (3.03) | 35.32 (3.28) |
| Indication for cesarean section (%) and previous cesarean section (n) (%) | 37 (60.7) | 31 (50.8) |
| Malpresentation | 5 (8.2) | 1 (1.65) |
| Cephalo-pelvic disproportion | 16 (26.2) | 17 (27.87) |
| Failed induction | 3 (4.9) | 4 (6.56) |
| Fetal none reassuring | 0 | 8 (13.12) |
| Operation time (min) (SD) | 77.61 (23.00) | 76.57 (23.26) |
| Surgeon level | ||
| Staff | 16(26.2) | 20(32.8) |
| Resident | 45 (73.8) | 41 (68.2) |
| Fetal weight (g) (SD) | 3109.03 (370.61) | 3279.7 (431.95) |
Table 1: Patients' demographic data.
From Table 2, although, amount of blood loss both at immediate post-operation and 24 h after cesarean section in carbetocin group were less than those in oxytocin group, there was no statistic significance. Moreover, the other operative outcomes including incidences of PPH and blood transfusion were not difference. However, there was significantly lower additional uterotonic use in carbetocin group (9.8%) compared to oxytocin group (36.1%) (p=0.001). Only 1 additional uterotonic drug (methylergometrine) was administered in most cases. There was only one case in carbetocin group using 2 additional drugs while in oxytocin group, there were 5 cases using 2 additional drugs and 2 cases using 3 or more drugs. There was no statistic difference in side effects. Nausea and vomiting were the most common side effects in both groups (Table 2).
| Outcomes | Carbetocin group (n=61) |
Oxytocin group (n=61) |
p-value |
|---|---|---|---|
| Blood loss (ml) | |||
| Immediate post cesarean section | 400 | 500 | 0.097 |
| 24 h after cesarean section | 560 | 600 | 0.1 |
| Additional uterotonic use (n) (%) | 6 (9.8) | 22 (36.1) | 0.001 |
| Post operation | |||
| Hb(%)(SD) | 10.88 (1.70) | 10.96 (1.38) | 0.771 |
| Hct (%)(SD) | 33.48 (3.26) | 33.08 (3.74) | 0.527 |
| Median Hct change | 0.7 | 0.8 | 0.127 |
| Incidence of PPH (n) (%) | 4 (6.6) | 3 (5) | 1.00 |
| Blood transfusion (n) (%) | 2(3.3) | 2 (3.3) | 1.00 |
| Side effects (n) (%) | |||
| Nausea | 19 (31.13) | 13 (21.31) | 0.363 |
| Vomiting | 12 (19.67) | 22 (36.06) | |
| Headache | 0 | 1 (1.63) | |
Table 2: Operative outcomes (amount of blood loss, additional uterotonic use, incidences of PPH and blood transfusion), side effects.
There were no significant differences in pre-operative hemoglobin and hematocrit in both groups (as shown in Table 1). Postoperative hemoglobin was 10.8 g/dl in carbetocin group nearly similar to the level of 10.9 g/dl in oxytocin group. Postoperative hematocrit was 33.48% in carbetocin group, whereas in oxytocin group, it was 33.08% (p=0.527). Median Hb change in carbetocin group was 0.7 g/dl not different from the level of 0.8 g/dl in oxytocin group (p=0.127).
Discussion
Cabetocin is an eight-amino acid long analogue of oxytocin. Its action is similar to oxytocin with longer half-life. Although oxytocin is the first line uterotonic drug using for PPH prevention and treatment, carbetocin has been approved for promoting uterine contraction in order to prevent PPH in cesarean section. For the study in high risk PPH from Italy shows blood loss post cesarean section, the drop of hematocrit level and side effects in carbetocin group and oxytocin group are not different while additional uterotonic use is significantly greater in oxytocin group [6]. For the study from Canada reports that blood loss after cesarean section is significantly less in carbetocin group whereas side effects are not different [7].
In Philippine, the study in elective cesarean section report that blood loss, additional uterotonic use and uterine massage are significantly lower in carbetocin group but there is no significant difference in blood transfusion and side effects [8]. The study from United Kingdom informs that blood loss after cesarean section, blood transfusion and incidence of PPH in both groups are not different. Additional uterotonic use and side effects are significantly less in carbetocin group [9].
The study in emergency cases from Malaysia reports that additional uterotonic use is lower in carbetocin group but PPH, blood transfusion and operative time are not different in both groups [13]. The study in severe pre-eclampsia patients from Panama shows that carbetocin is as effective as oxytocin in prevention of PPH. Carbetocin has a safety profile similar to that of oxytocin [14]. Cochrane Database of Systematic Reviews concludes that additional uterotonic use and blood loss are significantly lower in carbetocin group but PPH, blood transfusion and side effects are not different [10].
This study showed that blood loss in low-risk uterine atony cesarean section in carbetocin was not different from oxytocin. Although additional uterotonic use was significantly lower in carbetocin group, incidences of PPH and blood transfusion, hemoglobin and hematocrit change within 24 h after cesarean section and side effects in both groups were not different. These results were compatible with previous studies.
However, it has been wondering whether the advantage of carbetocin can overcome its high price, especially in patients with low PPH risks. Furthermore, there has had no guideline or data about carbetocin use in Thailand yet. This study has been the first trial in Thailand focusing on the effects of carbetocin compared with oxytocin administrating in low-risk uterine atony patients.
From the results, not only the use of additional uterotonic, such as methyergometrine, oxytocin, misoprostol and prostaglandin F2-alpha, was significantly greater in oxytocin group but also combination of 2 or more additional uterotonic drugs was administered more frequent. Use of additional uterotonic depended on surgeons’ decision. It based on their perception about the intensity. However, there was no difference in amount of blood loss both at immediate post-operation and 24 h after cesarean. In addition, there was no statistically significant difference in the other outcomes such as incidences of PPH and blood transfusion and side effects.
Conclusion
In conclusion, carbetocin could not significantly reduce blood loss in low-risk uterine atony cesarean section. Although additional uterotonic use was significantly lower in carbetocin group, incidences of PPH and blood transfusion, hemoglobin and hematocrit change within 24 h after cesarean section and side effects in both groups were not different. Although carbetocin has been approved for PPH prevention in cesarean section, its advantage over oxytocin is not obvious, especially in low risk uterine atony cesarean section.
For clinical application, the guideline for carbetocin use can be developed by weighing its effectiveness and its high cost. It may not be necessary for cesarean section in low risk uterine atony, especially in developing countries where most people have been in middle to low socioeconomic status. It may be more suitable for cesarean section in high risk uterine atony. Therefore, the Further study is to study the benefit of carbetocin versus oxytocin in high risk uterine atony group.
Acknowledgement
This study was supported by our institute’s fund for academic purposes. No other external fund was involved in the study.
References
- Belfort MA, Lockwood CJ, Levine D, Barss VA (2017) Overview of postpartum hemorrhage.
- (2006) ACOG practice bulletin: Clinical management guidelines for obstetrician-gynecologists number 76, October 2006: Postpartum hemorrhage. Obstet Gynecol 108: 1039-1047.
- Cunningham FG, Leveno KJ, Bloom SL, Spong CY, Dashe JS, et al. (2014) Williams obstetrics. 24th Edition. Mcgraw-Hill Education.
- Liu S, Liston RM, Joseph KS, Heaman M, Sauve R, et al. (2007) Maternal mortality and severe morbidity associated with low-risk planned cesarean delivery versus planned vaginal delivery at term. CMAJ 176: 455-460.
- WHO (2012) WHO recommendations for the prevention and treatment of postpartum haemorrhage.
- Larciprete G, Montagnoli C, Frigo M, Panetta V, Todde C, et al (2013) Carbetocin versus oxytocin in caesarean section with high risk of post-partum haemorrhage. J Prenat Med 7: 12-18.
- Boucher M, Horbay GL, Griffin P, Deschamps Y, Desjardins C, et al. (1998) Double-blind, randomized comparison of the effect of carbetocin and oxytocin on intraoperative blood loss and uterine tone of patients undergoing cesarean section. J Perinatol 18: 202-207.
- Uy DL, Pangilinan NCP, Ricero-Cabingue C (2013) Carbetocin versus oxytocin for the prevention of postpartum hemorrhage following elective cesarean section: Rizal medical center experience. POGS 37: 71-79.
- Attilakos G, Psaroudakis D, Ash J, Buchanan R, Winter C, et al. (2010) Carbetocin versus oxytocin for the prevention of postpartum haemorrhage following caesarean section: The results of a double-blind randomised trial. BJOG: 117: 929-936.
- Su LL, Chong YS, Samuel M (2012) Carbetocin for preventing postpartum hemorrhage. Cochrane Database Syst Rev.
- Macones GA, Hankins GD, Spong CY, Hauth J, Moore T (2008) The 2008 National Institute of Child Health and Human Development workshop report on electronic fetal monitoring: Update on definitions, interpretation and research guidelines. Obstet Gynecol 112: 661-666.
- Breathnach F, Geary M (2009) Uterine Atony: Definition, prevention, non-surgical management and uterine temponade. Semin Perinatol 33: 82-87.
- Razali N, Md latar IL, Chan YK, Omar SZ, Tan Pc (2016) Carbetocin compared to oxytocin in emergency cesarean section: A randomized trial. Eur J Obstet Gynecol Reprod Biol 198: 35-39.
- Reyes OA, Gonzalez GM (2011) Carbetocin versus oxytocin for prevention of postpartum hemorrhage in patients with severe preeclampsia: A double-blind randomized controlled trial. J obstet Gynaecol can 33: 1099-1104.
Citation: Jenkumwong P, Ratchanon S, Ouitrakul S (2017) Double-Blind Randomized Comparison of Carbetocin Versus Oxytocin for Decrease Blood Loss from Cesarean Section in Low-Risk Uterine Atony. J Preg Child Health 4: 350. DOI: 10.4172/2376-127X.1000350
Copyright: © 2017 Jenkumwong P, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 6651
- [From(publication date): 0-2017 - Nov 25, 2025]
- Breakdown by view type
- HTML page views: 5460
- PDF downloads: 1191