Case Report Open Access
Effect of Indrayava (Holarrhena antidysenterica Seed) on Inpatient Uncomplicated Severe Hyperglycaemia: A Case Study
| Ashok Kumar Panda1*, Debajyoti Das1, Amit Kumar Dixit2 and Jayram Hazra3 | |
| 1Research Officers (Ayurveda), Department of Clinical Research, NRIADD, CCRAS, Kolkata, India | |
| 2Research Officer, Department of Biochemistry, NRIADD, CCRAS, Kolkata, India | |
| 3Director, National Research Institute of Ayurvedic Drug Development, CN-4, Sector-V, Bidhan Nagar, Kolkota-91, India | |
| Corresponding Author : | Ashok kumar Panda Research Officers (Ayurveda) Department of Clinical Research Tadong, Gangtok, Sikkim, India E-mail: ailtoakpanda_06@ yahoo.co.in |
| Received July 12, 2013; Accepted July 19, 2013; Published July 25, 2013 | |
| Citation: Panda AK, Das D, Dixit AK, Hazra J (2013) Effect of Indrayava (Holarrhena Antidysenterica Seed) on Inpatient Uncomplicated Severe Hyperglycaemia: A Case Study. J Homeop Ayurv Med 2:126. doi: 10.4172/2167-1206.1000126 | |
| Copyright: © 2013 Panda AK, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Traditional Medicine & Clinical Naturopathy
Abstract
Indrayava (Holarrhena antidysenterica seed) has been used in bleeding piles, diarrhea, eczema, fever and colic as per Ayurveda. The folklore use of Indrayava for curing diabetes is reported as taking10 grams of Indrayava soaked overnight in 80 ml of water and then drinking the liquid. The Pharmacological study for anti-diabetic activities and acute toxicological studies of the Indrayava are well documented and LD50 is greater than 3000 mg/kg body weight. We are presenting a case of newly diagnosed diabetes with severe hyperglycemia (Post Prandial blood sugar >500 mg/dl), administrated 10 gm of Indrayava soaked overnight in 80ml water and advised to take water for a period of 48 days in I.P.D. The primary treatment outcomes were reduction of fasting and post prandial blood sugar with glycosylated hemoglobin. There was a reduction of elevated triglyceride level with marked clinical improvement also. There was no change in the pre and post treatment biochemical and hematological safety profile of the patient. Our case study opens a modality of treatment in uncomplicated severe hyperglycemic patients. Further randomized control clinical trial is suggested.
| Keywords |
| Indrayava Holarrhena antidysenterica seed (HAS); Hypoglycaemia; fasting and post prandial blood; sugar |
| Introduction |
| Diabetes is rapidly emerging as a major health care problem in India and the prevalence of type 2 diabetes has increased greatly over the past decade. The average annual direct costs of hospitalized patients are more than double compared to those not hospitalized. Complications of the disease are also responsible for indirect costs in terms of productivity loss and absenteeism [1]. In case of common ailments 35% people prefer allopathic, 48% prefer Ayurveda and 17% prefer homeopathic medicines. 21.5% of the general public has first choice to treat diabetes with Ayurveda [2] and 73% of diabetic patients prefer effective Ayurveda medicine for their treatment either as food supplements or as herbal drugs. The focus of care for people with diabetes has shifted from specialists to General Practitioners (GPs) to alternative health care practitioners [3]. Information published by the DNA agency reported that Ayurvedic medicines were misleading for diabetic care; “Such medicines are neither effective nor tested by any drug controller”. Hence, it´s not advisable for patients to discontinue their regular medicines and rely on Ayurvedic ones” [4]. On the other hand there is Gymnemasylvestre and herbal formulas Ayush-82 and D-400 thatshowa glucose-lowering effect and deserve further study. Evidence of effectiveness of several other herbs is less extensive (C. tamala, Eugenia jambolana, and Momordicacharantia) [5]. Most studies that test herbal therapy indicate that heterogeneity exists in the herbs and formulas tested; more than 44 different interventions are identified with the method of preparation. |
| Hyperglycaemia complicates diabetes ranges from the asymptomatic and benign in patients with mild to moderate uncomplicated hyperglycemia to life-threatening (i.e. Diabetic ketoacidosis (DKA) or Hyperosmolar Hyperglycemic State (HHS).Severe hyperglycemia is a condition when the blood glucose level is more than 350mg/dl [6]. Implementing a standardized insulin order set promoting the use of scheduled basal and nutritional insulin therapy is a key intervention in the inpatient management of severe diabetes [7,8]. However, a potent herbal drug can be administered by monitoring the risk and benefit ratio. |
| Practitioners of Ayurveda medicine believe that Kutaja is a divine herb bestowed by the Almighty. Indrayava is the seed of Kutaja (HAS) of the family –Apocynaceae. The meaning of Indrayava is Indra´s seed; Indra is the lord of divine realm. The traditional use of Indrayava for curing diabetes is reported as taking 10 gm of Indrayava soaked overnight in 80 ml water and then drinking the remaining liquid [9]. There is no potent marketed product for severe hyperglycaemia [10]. A pilot study was conducted earlier that found promising antihypoglycaemic effect of Indrayava (HAS) in mild to moderate Type- II diabetics. The administrated dose was 10 mg of Indrayava (HAS) soaked overnight in 80ml of water and it was advised to drink the infusion in the early morning on an empty stomach for severe Type-II diabetic patients, following standard guidelines [11]. |
| Case Report |
| A 65-year-old poor, rural and illiterate woman came to our O.P.D (Registration no- 4331/12-13) on 26-11-2012 with a complaint of frequent urination, weight loss, lethargy and bilateral knee pain for the past 3-4 months, with no family history of diabetes. She has a history of depression and had taken tricyclic anti-depressant drug for one year with no more depression or any psychiatric problem remaining. |
| General examination reveals VataKaphaPrakruti (physical constitution), Alpaaharashakti (anorexia), Krurakosta (constipation), amalipta Jivha (coated tongue) and alpanidra (sleeplessness). |
| Physical examination reveals an anxious woman with blood pressure 130/70 mmHg, pulse 84per minute and regular, respiratory rate 22 per minute, weight 59 kg, and 148 cm height. BMI is 26.94. No central obesity, no fungal/skin acanthuses. Lungs are clear to percussion, heart sound is normal. No decreased or absent sensory stimuli to light touch or temperature, no loss of deep tendon reflexes in ankles. No dry feet, muscle atrophy, claw toes or ulcer. Swelling and tender in both knee joints with restricted movement and cracking sound. The patient was provisionally diagnosed as VyadhiSankar i.e. having many diseases at once (Sandhigatavata; osteoarthritis and Madhumeha; diabetes). |
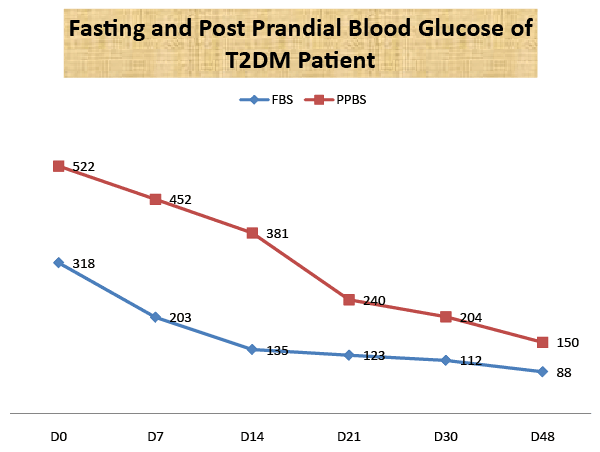
| The patient was sent to the Biochemistry department for Fasting and Post Prandial blood sugar testing. The Fasting blood sugar was 318 mg/dl with Post Prandial Blood sugar was 522 mg/dl. The Patient was admitted in the IPD (Reg. no-150, Bed no-F11) for Ayurvedic treatment. |
| Laboratory evaluation |
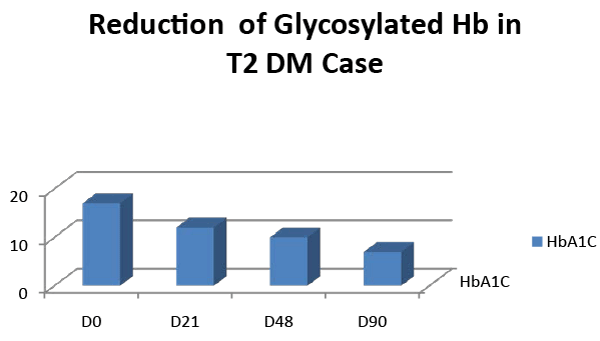
| Hemoglobin concentration; Hb% was 12.5 gm/dl, ESR was 20 (1st hour),TG-195 mg/dl, serum calcium-9 mg/dl, HbA1c- 17.00, RA factor, CRP and ASO titre were negative with normal haematological, liver and renal function test. Knee X-ray shows osteoarthritic changes in both knee joints. |
| Assessment |
| Based on medical history, records, physical exam, and lab results. She was assessed as newly diagnosed with uncomplicated severe hyperglycaemia with osteoarthritis in both knees. The primary assessment found diminished Agni (digestive fire) and predominance of Vatadosa. The patient’s self-care management and lifestyle deficits are observed with poor understanding of diabetes. |
| Management |
| After the assessment of the patient, the line of treatment was Deepana (carminative), Pachana (digestive), Anulomana (Laxative) and Tiktakasaya dravya (drugs with bitter and astringent taste) with normal diet. She was advisedto take Sanjeevani Vati (250mg twice daily before food), Vaisanar churna (6gm at bed time for two weeks) and Indrayava churna -10gm as cold effusion in 80ml water, morning and evening on empty stomach) with Maha Narayana taila as external application on the knee joint. Sanjeevanivati, Vaisanar churna were stopped after 7 days of treatment as the patient achieved niram Jhiva (Uncoated tongue).Yogaraj gugulu (1000 mg, twice daily, after breakfast and dinner) was administrated after 14days of admission for two weeks as patient got substantial relief from knee pain. The other medications were continued with estimation of blood sugar (fasting and post prandial) with a seven day interval. The patient required long duration of treatment to reduce the clinical findings and normalise the blood sugar level. |
| Discharge summary |
| The patient was discharged after 48days of above treatment withfasting blood sugar 108 mg/dl, post prandial blood sugar of 150 mg/ dl, and with normal biochemical and haematological profile (Figures 1,2). Her body weight increased from 59 kg to 61 kg and urination was regularised with good vigour and vitality. The patient was discharged and advised to continue with medications and report after 15days. Her HbA1C was assessed after three months; 7.00 with PPBS 140 mg/dl. |
| Discussion |
| There is a reduction of fasting and post prandial blood sugar with glycosylated hemoglobin after 48 days administration of HAS (Table 1). There is a reduction of elevated triglyceride level with marked clinical improvement also. There is no change in the pre and post treatment biochemical and hematological safety profile of the patient. The folklore use of Indrayava in diabetics was reported from Sikkim, West Bengal, Orissa and Jharkhand. Bhavprakash narrated that Indrayava is astringent, cold in potency, a febrifuge, ant helminthic and alleviates of all three dosas (Tridoshgna), cures bleeding piles, diarrhea, eczema, fever and colic. The Pharmacological study for anti-diabetic activities and acute toxicological studies of Indrayavais well documented [12-14]. The acute toxicity study for the ethanol extract of Holarrhena antidysenterica seed was carried out and LD50 is greater than 3000 mg/ kg body weight. The blood glucose level was significantly reduced by the administration of 300 mg/kg ethanol extract of HAS. Holarrhena antidysenterica seedshows promising action on mild to moderate Type-II diabetes subjects in the preliminary study. Indrayava contains amino acids in free state, mainly aspartic acid and arginine. An enzyme serine protease was isolated with three other known pentacyclic triterpenoids, namely lupeol, betulinaldehyde, and betulinic acid and a steroidal compound stigma sterol were isolated from this seeds.Other new compounds dihydrocanaric acid, â�?�? amyrin, betulin, and olenolic acid are also recently isolated [15-16]. HAS may have potentiating action on β cells of islets or stimulation of blood glucose uptake by peripheral tissue or/and inhibition of endogenous glucose production or activation of gluconeogenesis in liver and muscles [13]. The isolation of active compounds from HAS and pharmacological and clinical studies may initiate new drug development. Our case study opens the way for new treatment modalities in uncomplicated severe hyperglycemic patients. A further randomized controlled clinical trial is suggested. |
References
|
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
 |
| Figure 1 | Figure 2 |
Relevant Topics
- Acupuncture Therapy
- Advances in Naturopathic Treatment
- African Traditional Medicine
- Australian Traditional Medicine
- Chinese Acupuncture
- Chinese Medicine
- Clinical Naturopathic Medicine
- Clinical Naturopathy
- Herbal Medicines
- Holistic Cancer Treatment
- Holistic health
- Holistic Nutrition
- Homeopathic Medicine
- Homeopathic Remedies
- Japanese Traditional Medicine
- Korean Traditional Medicine
- Natural Remedies
- Naturopathic Medicine
- Naturopathic Practioner Communications
- Naturopathy
- Naturopathy Clinic Management
- Traditional Asian Medicine
- Traditional medicine
- Traditional Plant Medicine
- UK naturopathy
Recommended Journals
Article Tools
Article Usage
- Total views: 33403
- [From(publication date):
September-2013 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 28406
- PDF downloads : 4997
