Research Article Open Access
Electrolyte Materials for Solid Oxide Fuel Cell
Mridula Biswas*
High Tmperature Energy Materials Centre, Korea Institute of Science and Technology, 39-1 Hawolgok, Seongbuk, Seoul 136-791, South Korea
- *Corresponding Author:
- Mridula Biswas
High Temperature Energy Materials Centre
Korea Institute of Science and Technology
39-1 Hawolgok, Seongbuk, Seoul 136-791, Korea
Tel: +919836050800
E-mail: luckymridula@gmail.com
Received Date: September 05, 2013; Accepted Date: October 04, 2013; Published Date: October 11, 2013
Citation: Biswas M (2013) Electrolyte Materials for Solid Oxide Fuel Cell. J Powder Metall Min 2:117. doi: 10.4172/2168-9806.1000117
Copyright: © 2013 Biswas M. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Powder Metallurgy & Mining
Abstract
The first criterion for the electrolyte of solid oxide fuel cells (SOFC) is to have sufficient ionic conductivity without any significant electronic conductivity. Since the cell operates at high temperature, it must withstand that operating temperature along with stability in both reducing and oxidizing environments. It must be impermeable to gas to prevent mixing between fuel and oxidizer before they reach to the reaction sites. There are two groups of electrolytes based on their ion conducting phenomenon. They are oxide ion conducting and proton conducting. Oxide ion conducting materials are widely used. Therefore, the primary focus is given on these materials.
Keywords
Oxygen ion conductors; Zirconia; Ceria; Proton conductors
Introduction
The electrolyte is the central part of a Solid Oxide Fuel Cell (SOFC), which is sandwitched between cathode and anode. The oxygen ions (O2-) reduced on cathode or proton (H+) oxidized on anode sides is transported through the electrolyte materials and reacts with hydrogen or oxyegen, respectively, to form water on either side of the cell. The electrons (e-) generated due to the reaction pass in the opposite direction through the outer circuit [1]. There are two types of materials: oxide ion and proton conducting electrolytes.
Its electronic conductivity must be kept as low as possible to prevent losses from leakage currents. The high operating temperatures of SOFCs allow the kinetics of oxygen ion transport to be sufficient for good performance. However, as the operating temperature approaches the lower limit for SOFCs at around 600°C, the electrolyte begins to have large ionic transport resistances and affect the performance. Popular electrolyte materials include yttrium stabilized zirconia (YSZ) (often the 8% form Y8SZ), scandia stabilized zirconia (ScSZ) and gadolinium doped ceria (GDC) [2]. Materials choice for this purpose is difficult because attention must be paid to many parameters such as gas tightness, electronically nonconducting, chemical stability over a wide oxygen partial pressure (1-10-21 atm) and temperature range, good bending strength etc [3].
Materials for electrolyte of solid oxide fuel cell
The basic reactions for the generation of electricity directly from electrochemical reaction are given below:
For oxide ion generating reaction:
1/2 O2 + 2e- = O2-
H2 + O2- = H2O + 2e-
2H2 + O2 = 2H2O (1)
The reaction of hydrogen and oxygen is an exothermic one that produces heat about 57 kcal per mole of reactant.
For proton generating reaction:
H2 = 2H+ + 2e-
1/2 O2 + 2e- = O2-
2H+ + O2- = H2O (2)
Electrons generated from the above reactions flows through the outer circuit so as to provide electricity [4].
Oxide ion conducting electrolytes
Oxide ion conducting electrolytes cover a wide range of materials which basically include fluorite-, perovskite-, brownmillerite- structured materials.
Fluorite-structured electrolytes
This structure is adopted by ceria at room temperature and zirconia at high temperature. Basically zirconia and ceria based materials are widely investigated for its application as electrolyte in solid oxide fuel cell (SOFC) [2]. Zirconia is stabilized to its cubic structure at room temperature by the addition of divalent and trivalent cations [5,6]. Ceria forms solid solutions with many divalent and trivalent cations which show fluorite structure at room temperature and takes part in ionic conduction [7]. Yittria stabilized zirconia is not the best ionic conductor [8]. Bismuth oxide-based materials, ceria-based materials and strontium doped lanthanum oxide show better conductivity than showed by zirconia-based materials. T. Takahashi et al reported the same about bismuth oxide-based materials [9]. Among all these materials δ-Bi2O3 has an oxygen deficient fluorite structure where 1/4th the normal fluorite anion sites are vacant. This is the reason for its very high conductivity. But the drawback of this material is that it suffers from chemical instability. It is of monoclinic structure at room temperature, but cubic at high temperature. It gets readily reduced at low oxygen partial pressure and decomposes into bismuth metal at oxygen partial pressure of about 10-13 atm [10,11]. Therefore, the practical use of bismuth oxide as electrolyte of SOFC is under serious question. So it is better to go for ceria and zirconia-based materials for the application as electrolyte in SOFC.
Zirconia-based oxide ion conductors
The solid electrolytes to be used for high temperature fuel cell should have a good number of properties such as high oxide ion conduction with negligible electronic conductivity, phase stability, non-porous, good mechanical strength, thermal shock resistance, chemical inertness to the reactive gas, compatibility with electrode in the view of thermal expansion coefficient and chemical inertness towards the electrodes. These requirements are sufficiently fulfilled by doped zirconia. Addition of some rare earth, alkaline earth and lanthanide oxides stabilizes monoclinic zirconia to cubic fluorite phase at room temperature; moreover, this addition enhances ionic conductivity to an extended oxygen partial pressure range of SOFC operation. This range covers 1 to 10-18 atm pressure which an SOFC electrolyte has to face at the anode side during operation at high temperature while other renowned electrolyte, doped ceria, fails due to its electronic conduction and poor mechanical strength. The oxides which have come to the forefront for their use as dopants for zirconia are calcia (CaO), magnesia (MgO), scandia (Sc2O3) and yittria (Y2O3). Stabilized zirconia is stable over wide temperature range, in this range ionic conductivity is independent of oxygen partial pressure over several orders of magnitude [12,13]. Yittria stabilized zirconia (YSZ) shows electronic conductivity at oxygen partial pressure of 10-30 atm which is far below the pressure range of 0.21 - 10-20 atm during the operation of SOFC [14]. Its oxygen transference number is one over a wide range of temperature and oxygen partial pressure [15].
From Table 1, considering the resistivity data it can be concluded that calcium is not a better dopant than yittria. It has been established that clusters comprising of two oxygen vacancies at the opposite ends of a body diagonal in one of the anion cubes of the fluorite structure exist throughout the body. It is presumed that this vacancy dimmer is stabilized by trivalent ions [7]. The association is called cluster. It is assumed that these clusters exist in a group, not in isolation. In some cases ordered phases (Ca2Zr4O9) get precipitated out, but still there is a controversy [15-17]. There are also evidences for short range order. As for example, Yittria doped zirconia shows short range order [18]. The site exclusion principle states that the presence of a cluster at a site prevents other clusters from occupying adjacent sites in the lattice [7].
| Dopant | Mol% | Resistivity at 800 �?°C (Ocm) |
Resistivity at 1000 �?°C (Ocm) |
|---|---|---|---|
| CaO | 15 | 250 | 50 |
| YO1.5 | 20 | 45 | 10 |
| (YO1.5)0.08(YbO1.5)0.08 | 16 | 20 | 5 |
Table 1: Resistivity data of doped ZrO2.
Literature reported that conductivity increases with dopant concentration upto a certain amount which is required to fully stabilize monoclinic zirconia to its cubic fluorite structure [13]. Conductivity decreases with further addition of dopant. This decrease can be explained in terms of dopant-vacancy interactions. For low dopant concentration, the aliovalent ions will be isolated in the host cation sites and very few of them will share a common oxygen site. Therefore, the interaction between dopant ions and oxygen vacancies leads to the formation of shor range clusters which can be described as dopant-vacancy complex, while the other dopants will bear effective negative charge compared to zirconium ion. As for example, one half of the dopant ions will be in cluster with oxygen vacancies since two yttrium ions produce one oxygen ion vacancy. Since this cluster formation process is reversible, dissociation of this complex also occurs. When concentration of dopant increases the probability of formation of a cluster containing two dopants with a single oxygen vacancy gets enhanced. This cluster acts as trap due to coulombic interaction. So, the mobility of oxygen vacancies gets reduced [19,20]. Yittria is widely used as the stabilizer for zirconia because of its abundance and cost effectiveness compared to other dopants [3]. Scandia and ytterbia have also found application due to their better ionic conductivity; but yttria doping is widely used due to the above mentioned reasons. Literatures report that 8 - 10.5 mol% yittria stabilizes zirconia to its cubic structure [21]. Literature also reported the doping of zirconia with more than one cations instead of only one cation [22,23]. As for example, such compositions are zirconia doped with yittria (Y2O3), scandia and ytterbia (Yb2O3) (composition 1), zirconia (ZrO2) doped with yittria (Y2O3), dysprosia (Dy2O3) and ytterbia (Yb2O3) (composition 2). All the dopants were chosen on the basis of their radius according to the Hume Rothery rule [24]. To achieve maximum solid solubility, the atomic radii of the solute and solvent atoms must differ by no more than 15%. The maximum Electrical conductivities were 0.18 and 0.16 S/cm for compositions 1 and 2, respectively, with the combined dopant concentration of 8.3 mol% at 1000°C [25]. Bending strength is another important factor to maintain mechanical integrity and to withstand thermal stress with other components of the cell. It was about 100 MPa for those compositions, which does not suffice the need at the operating temperature of SOFC. On the other hand, yittria stabilized zirconia is a promising candidate, in this regard too, with the bending strength of 400 MPa [26]. Thermal expansion is another factor to be considered. Zirconia stabilized with 8 mol% yttria exhibits a very consistent coefficient of thermal expansion of 10.5x10-6/K from 25°C to 1000°C [27].
Ceria-based oxide ion conductors
Ceria-based materials have found potential application as electrolyte materials for the intermediate temperature (500-750°C) solid oxide fuel cell (ITSOFC) [28]. ITSOFC is favourable compared to high temperature SOFC. Several advantages are obtained with the reduction in operating temperature; they are: less prone to thermal and mechanical stress, wide range of materials selection, short startup time, easy maintenance, better thermal management, much more economic, reduced effect of thermally activated processes [29]. A descriptive study was carried out on a series of ceria compositions; CaO, SrO, MgO and BaO have been used as dopants [30]. The result shows that CaO and SrO enhances electrical conductivity to a great extent but MgO and BaO do not enhance so far. Literature reported the effects of rare earth oxides (such as Sm2O3, Gd2O3 and Y2O3) as dopant in ceria on electrical conductivity [30]. The observation led to the conclusions that doping with Sm3+ among rare earth oxides and Ca2+ among alkaline earth oxides produces maximum conductivity. The problems associated with ceria based materials are that they are prone to reduction at low oxygen partial pressure, which is prevalent on the anode side of SOFC, that leads to electronic conductivity [28] and possible large mechanical disintegration under large oxygen chemical potential gradients typical of SOFC operation [31,32]. This problem can be solved by decreasing operating temperature to below 650° to 700°C. Enumerable work has been carried out by different research groups to get rid of this problem. But there was no solution without yittria stabilized zirconia. Ceria based materials are mechanically very weak. Yahiro et al and Mehta et al deposited a thin film of yittria stabilized zirconia (YSZ) on ceria-based materials on anode side to suppress electronic conduction [33,34]. But the problem occurs with the operating temperature; ceria based electrolytes are used in SOFC operated at 550°C and lower whereas YSZ thin film can work only at higher temperature to suppress electronic conduction [34]. So the problem has to be addressed further to have proper solution. Another solution is that doped ceria can be used with doped lanthanum gallate in multilayer cells [31,32]. But this multilayer electrolyte shows very poor performance due to formation of new layer caused by inter-diffusion at the boundary of YSZ and ceria, which, in turn, causes low ionic conduction [35]. This layer may introduce thermal expansion mismatch at the operating temperature, resulting into microcracks [33,36]. At reduced operating temperature, the overall cell voltage will be lower due to critical polarization resistance of electrodes, rendering poor cell performance, non-uniform current and/or distribution of reaction rate throughout the electrolytes may cause local heating which can lead irreversible decomposition of ceria based materials [37].
Perovskite-structured oxide ion conductors
According to the literature reports, some doped perovskite (ABO3) materials can serve as solid electrolyte materials for SOFC. Al or Mg doped calcium titanate (CaTiO3) exhibits highest oxide ion conductivity [38]. CaTi0.95Mg0.05O3 and Ca-doped Al2O3 show very high transport number (0.9) with no electronic conductivity in reducing atmospheres. However, they are not suitable for SOFC application due to their lower oxide ion conductivity than that shown by YSZ. This lower conductivity results from limited solid solubility of dopants in these oxides less open crystal structure than fluorite structured YSZ [39,40]. But surprisingly lanthanum strontium gallium magnesium oxide (La0.9Sr0.1Ga0.8Mg0.2O3) showed oxide ion conductivity higher than those of ZrO2-based and CeO2-based ion conductors and lower than that of Bi2O3-based ion conductors [39,40]. This material exhibited oxide ion conduction with limited whole and electronic conduction upto 10-23 atm oxygen partial pressure [41]. But this material does not possess good mechanical strength [42,43]. Bending strengths of La0.9Sr0.1Ga0.8Mg0.2O3 at room temperature and 900°C were 162 ± 14 and 55 ± 11 MPa, respectively, which is not suitable for SOFC application [41]. This material does not show good creep behavior [44]. It showed thermal expansion coefficient of 11.5 x 10-6/K down from the room temperature to 1000°C, which is compatible with other components of SOFC [45].
Brownmillerite-structured oxide ion conductors
This material has a general formula of A2B´B”O5 or A2B2O5. This is a perovskite structured material with one sixth of oxygen sites vacant. The most popular material in this group for SOFC application is Ba2Ln2O5 [35]. Brownmillerites exhibit exhibition conductivity higher than that of fluorite structured oxides [46]. Hole is the major carrier in electrical conduction beyond oxygen partial pressure of 10-3 atm. This material shows a large increase in conductivity at around 900°C. This may be attributed to the transition from oxygen vacancy ordered brownmillerite to oxygen disordered pseudoperovskite structure. This material also shows proton conduction below the temperature of 400°C. Ba2Ln2O5.H2O is responsible for proton conduction [47,48].
Layered aurivillius-structured compounds
Aurivillius compound is a complex oxide system of bismuth and vanadium; the formula of this layer structure is Bi4V2O11-δ [49]. This material shows phase conversion within three phases of α, β and γ over a wide temperature range. This type of material shows conductivity decay upon prolonged heating due to phase separation [50]. This oxide has been modified with the addition of rare earth oxides ( L2O3, where L includes Gd, Er, Dy, Y, Ho, Nd, Sm) [51]. The doped BIMEVOX (BI-Bismuth, ME- dopant metal, V- vanadium, OX-oxygen) gets reduced at low oxygen partial pressure and thereby producing metallic bismuth [52,53] in the following manner:
(Bi2O3)1-x(Me2O3)x → (Bi2O3)1-x-α(Me2O3)x + 2αBi + (3α/2)O2
Therefore, this material does not find application as electrolyte of SOFC.
LAMOX Family
La2Mo2O9 (LAMOX) is a fast oxygen ion conductor compared to yittria stabilized zirconia materials (YSZ). LAMOX undergoes a reversible phase transition from the nonconductive monoclinic form (α) to the highly conductive cubic (β) form at around 580°C. This phase transition is the primary impediment for this material to find application as a component of SOFC. This material may undergo mechanical breakdown due to repeated cycle of phase transition [54,55]. Praseodymium (Pr) has been used as dopant in LAMOX to suppress its phase transformation, but no suppression occurred; Pr significantly lowers down the phase transition temperature [55]. La2Mo2O9 exhibits high coefficient of thermal expansion of 16.8x10-6 and 13.5x10-6/K at low temperature phase and high temperature phase [56], respectively, which are not compatible with those of other components [27]. Its ionic conductivity is 2.6 x 10-2 S/cm, which is higher than YSZ materials [57,58]. Molybdenum is prone to reduction in low oxygen pressure [57]. This is the drawback of this material. The ionic transference number of La2Mo2O9 is high (around 0.98) in moderate reducing atmosphere, but it showed phase conversion at oxygen partial pressure of 10-8 Pa at 800°C [59].
Apatite oxides
Apatite structured oxides, such as La10Si6O27, La10Ge6O27, etc., exhibits oxide ion conductivity [60,61]. It shows consistent oxide ion conductivity over a wide range of oxygen partial pressure from 1 to 10-21 atm. These apatite’s exhibit hexagonal symmetry with very open structure which may help in oxide ion conduction while its activation energy is very low. This is the reason for investigating this material. Conductivity of this oxide is not higher than that of 8 mol% YSZ at the temperature higher than 600°C [60]; this may be the reason for slow research activity on this materials.
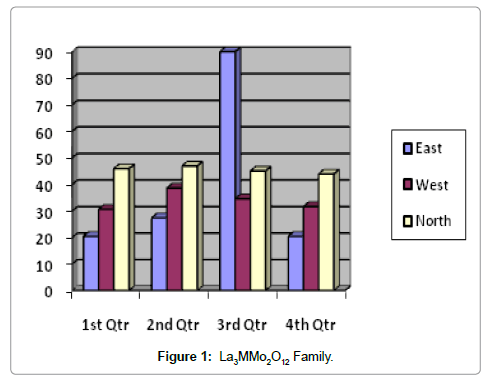
La3MMo2O12 family
This is very recent invention based on LAMOX family [62,54]. General formula of this material is La3MMo2O12, where M indicates In, Ga and Al. La3GaMo2O12 showed highest conductivity among the three different compositions. Their conductivities at 800°C are summarized in Table 2 [63]. The conductivity value of La3GaMo2O12 is higher than that of 8 mol% YSZ [58]. The main drawback of LAMOX material was its very high CTE which is not suitable for SOFC. LAMOX doped with In showed the lowest CTE of average 14.7x10-6/K and 12.7x10-6/K for high temperature and low temperature phases, respectively [56] and higher than that of 8 mol% YSZ [27]. All the three materials (doped with In, Ga and Al) showed phase transformation at high temperatures leading to change in unit cell volume [63] which is detrimental for their use with other components of SOFC at the temperature of its operation (Figure 1).
La2W2O9 family
| Composition | Conductivity(S/cm) |
|---|---|
| La3GaMo2O12 | 3.6 x 10-2 |
| La3AlMo2O12 | 2.4 x 10-2 |
| La3InMo2O12 | 3.6 x 10-3 |
Table 2: Conductivity Data of La2MMo2O12 Family.
LAMOX family suffers from phase transition especially due to molybdenum (Mo) [57]; it has been tried to overcome by introducing tungsten(W) in place of Mo. Reducibility of W is less compared to that of Mo [64,65]. W was chosen in place of Mo on the basis of their similar ionic radii (0.59Å for Mo6+ and 0.6Å for W6+) anticipating that 80% solid solubility can be achieved [66]. However, triclinic La2W2O9 (room temperature phase) undergoes reversible phase transition at 1070°C [67]; therefore, the problem has not been solved with this replacement. However, the problem was tried to be solved with the partial substitution of La and W by Ba and V [68]. The maximum conductivity of 0.011 and 0.006 S/cm were obtained for Ba0.2 substitution in place of La and for V0.3 substitution in place of W while it is 0.032 S/cm for LAMOX at 650°C [69]. Therefore, the result obtained was not so satisfactory.
Proton conducting oxides
Some perovskite oxides exhibit proton conduction in hydrogen containing environment at high temperature [70]. The primary compounds such as BaZrO3, BaCeO3, SrCeO3, SrZrO3 and CaZrO3, are not good conductor; they show good conduction property when doped with rare earth ions such as yittrium (Y) or ytterbium (Yb) due to the presence of electron hole. The reaction takes place in the following manner [35]:
H2 + 2h. = 2H+
where h. symbolizes hole.
SrCeO3 doped with Yb (5 mol%) was studied and the obtained conductivity was around 0.002 S/cm in a humidified atmosphere, which is far below the conductivity of 8 mol% YSZ [70-72]. Proton conduction increases in the following manner: BaCeO3>SrCeO3>SrZrO3>CaZrO3. This trend gets reversed upto SrZrO3 when chemical compatibility comes into consideration. Other proton conducting materials are BaThO3 and BaTbO3 doped with gadolia [73,74], BaTh0.9Ga0.1O3, Sr2Gd2O5 and Sr2Dy2O5 [75]. Maximum proton conductivity obtained was 8.7x10-2 S/ cm at 550°C. The main advantage of this material is that fuel cell does not require water removing unit because proton conductor generate steam in oxidant electrode [3]. Chemical stability of the materials has to be improved. They disintegrate in carbon dioxide and moisture containing environment. Therefore, researchers are trying to make solid solution (as for example, BaZrO3- BaCeO3) to extract good properties of both the materials as well as to avoid their detrimental properties. However, these materials also have the problem of phase separation after a long term use. To address this problem, amount of dopant level is being tried to adjust. BaO evaporation at high temperature is another problem; this is dealt with reduction in sintering temperature. These materials are very hard to sinter, therefore high temperature is necessary. In these materials grain boundaries act as the trap for proton; therefore, reduction of grain boundaries is another technical aspects, which requires very high temperature. Therefore, the problems associated with these materials will take a long time to be properly solved.
Conclusion
Presently research work is being carried out for YSZ, doped CeO2, doped BaZrO3, BaCeO3 and doped BaZrO3- BaCeO3 solid solution. Researchers are putting their effort in developing thin electrolytes. They are developing many techniques like pulse laser deposition, chemical solution deposition technique, etc. for the ordered arrangement of electrolyte molecules in a thin layer without leaving any void in the layer to maintain gas tightness.
Acknowledgement
The authors’ sincere thanks go to the senior scientists and junior students of High Temperature Energy Materials Centre, Korea Institute of Science and Technology, Seoul, South Korea.
References
- http://www2.fz-juelich.de/ief/ief-1//index.php?index=70
- Hibino T, Tsunekawa H, Tanimoto S, Sano N (1990) Improvement of a single-chamber solid-oxide fuel cell and evaluation of new cell designs. Journal of the Electrochemical Society 147: 1338-1343.
- Minh NQ (1993) Ceramic fuel cells. Journal of the American Ceramic Society 76: 563-588.
- Larminie J, Dicks A (2003) Fuel cell systems explained’, III; John Wiley & Sons, Inc., Chichester, West Sussex, UK.
- Garay JE, Glade SC, Kumar PA, Anselmi-Tamburini U, Munir ZA (2006) Characterization of densified fully stabilized nanometric zirconia by positron annihilation spectroscopy. Journal of Applied Physics 99: 1-7.
- Anselmi-Tamburini U, Garay JE, Munir ZA, Tacca A, Maglia F, et al. (2004) Spark plasma sintering and characterization of bulk nanostructured fully-stabilized zirconia (FSZ): I. Densification studies. Journal of Materials Research19: 3255-3262.
- Toft Sørensen O (1981) Nonstoichiometric Oxides’, Academic Press, New York.
- http://inanoschool.au.dk/fileadmin/inano/iNANOSchool/PhD_theses_PDF/ PhDA fhandling 090810 Michael Brammer Sillassen.pdf
- Takahashi T, Esaka T, Iwahara H (1977) Conduction in Bi2O3-based oxide ion conductors under low oxygen pressure I. Current blackening of the Bi2O3-Y2O3electrolyte. Journal of Applied Electrochemistry 7: 299-302.
- Verkerk MJ, Burggraff AJ (1981) High oxygen ion conduction in sintered oxides of the Bi2O3-Ln2O3 system. Solid State Ionics 3/4: 463-467.
- http://www.chimica.unipd.it/antonella.glisenti/privata/ documentazione/insegnamenti/ Fuel%20Cells/Bi2O3.pdf
- Patterson JW, Borgen EC, Rapp RA (1967) Mixed conduction in Zr0.85Ca0.15O1.85and Th0.85Y0.15O1.925solid electrolytes. Journal of Electrochemical Society 114: 752-758.
- Subbarao EC (1981) Advances in ceramics’, III; 1981, The American Ceramic Society, Coloumbus, OH.
- Eeppner W (1977) Electronic transport properties and electrically inducedp-n junction in ZrO2+ 10 m/o Y2O3. Journal of Solid State Chemistry 20: 305-314.
- Allpress JG, Rossel HJ (1975) A microdomain description of defective fluorite-type phasesCaxM1-xO2-x(M=Zr, Hf;x= 0.1–0.2). Journal of Solid State Chemistry 15: 68-78.
- Morinaga M, Cohen JB (1980) X-ray diffraction study of Zr(Ca,Y)O2-x. II. Local ionic arrangements. Acta Crystallographia A 36: 520-530.
- Stubican VS, Ray SP (1977) Phase equilibria and ordering in the system ZrO2-CaO. Journal of the American Ceramic Society 60: 534–537.
- Kondoh J, Kawashima T, Kikuchi S, Tomii Y, Yasuhiko (1998) Effect of aging on ytria-stabilized zirconia I. A study of its electrochemical properties. Journal of Electrochemical Society 145: 1527-1536.
- Wang DY, Nowick AS (1981) Dielectric relaxation from a network of charged defects in dilute CeO2:Y2O3solid solutions. Solid State Ionics 5: 551-554.
- Anderson MP, Nowick AS (1981)Relaxation produced by defect complexes in cerium dioxide doped with trivalent cations. Journal of Physics 42: 823-828.
- Wu M, Chen A, Xu R, Li Y (2003) Nanocrystalline LiNi1-xCoxO2 cathode materials prepared by a gel-combustion process. Materials Science Engineering B 99: 336-339.
- Qi X, Zhou J, Yue Z, Gui Z, Li L (2003) Auto-combustion synthesis of nanocrystalline LaFeO3. Materials Chemistry and Physics 78: 25-29.
- Fraigi L, Lamas DG, Walsoe de Reca NE (1999) Novel method to prepare nanocrystalline SnO2powders by a gel-combustion process. Nanostructured Materials 11: 311-318.
- Mizutani U (2010) Hume-Rothery Rules for Structurally Complex Alloy Phases. CRC Press.
- Lv ZG, Yao P, Guo RS, Dai FY (2007) Study on zirconia solid electrolytes doped by complex additives. Materials Science and Enginering A 458: 355-360.
- Sammes NM, Tompsett G, Zhang Y, Cartner A, Torrens R, et al. (1996) The structural and mechanical properties of (CeO2)1-x(GdO1.5)x electrolytes. Journal of the Japanese Electrochemical Society 64: 674-680.
- Hayashi H, Saitou T, Maruyama N, Inada H, Kawamura K, et al. (2005) Thermal expansion coefficient of yttria stabilized zirconia for various yttria contents. Solid State Ionics 176: 613-619.
- Sammes NM, Cai Z (1997) Ionic conductivity of ceria/yttria stabilized zirconia electrolyte materials. Solid State Ionics 100: 39-44.
- Fuel Cell Handbook, fifth Edition, EG & G Services, Parsons, Inc., Science Application International Corporation, U.S.Department of Energy, WestVirginia-26507-0880, October 2000.
- Yahiro H, Ohuchi T, Eguchi K, Arai H (1988) Electrical properties and microstructure in the system ceria-alkaline earth oxides. Journal of Materials Science 23: 1036-1041.
- Yasuda I, Hishinuma M (1998) Ionic and Mixed Conducting Ceramics’, III; The Electrochemical Society, Pennington, NJ.
- Atkinson A (1997) Chemically-induced stresses in gadolinium-doped ceria solid oxide fuel cell electrolytes. Solid State Ionics 95: 249-258.
- Yahiro H, Baba Y, Eguchi K, Arai H (1988) High temperature fuel cell with ceria-yttria solid electrolyte. Journal of the Electrochemical Society 135: 2077-2080.
- Tsai T, Barnett SA (1997) Solid Oxides Fuel Cells V’, The Electrochemical Society Proceedings, Pennington, NJ.
- Singhal SC, Kendall K (2003) High-Temperature Solid Oxide Fuel Cells: Fundamentals, Design and Applications. Elsevier Science.
- Tsoda A, Gupta A, Naoumoidis A, Skarmoutsos D, Nikolopoulos P (1998) Performance of a double-layer CGO/YSZ electrolyte for solid oxide fuel cells. Ionics 4: 234-240.
- Kharton VV, Figueiredo FM, Navarro L, Naumovich EN, Kovalevsky AV, et al. (2001) Ceria-based materials for solid oxide fuel cells. Journal of Materials Science 36: 1105-1117.
- Takahashi T, Iwahara H (1971) Ionic conduction in perovskite-type oxide solid solution and its application to the solid electrolyte fuel cell. Energy Conversion 11: 105-111.
- Ishihara T, Matsuda H, Takita Y (1994) Doped LaGaO3 perovskite type oxide as a new oxide ionic conductor. Journal of American Chemical Society 116: 3801-3803.
- Feng M, Goodenough JB (1994) A superior oxide-ion electrolyte. European Journal of Solid State Inorganic Chemistry 31: 663-672.
- Kim JH, Yoo HI (2001) Partial electronic conductivity and electrolytic domain of La0.9Sr0.1Ga0.8Mg0.2O3-d. Solid State Ionics 140: 105-113.
- Drennan J, Zelizko V, Hay D, Ciacchi FT, Rajendran S, et al. (1997) Characterisation, conductivity and mechanical properties of the oxygen-ion conductor La0.9Sr0.1Ga0.8Mg0.2O3-x. Journal of Materials Chemistry 7: 79-83.
- Du Y, Sammes NM (2001) Fabrication of tubular electrolytes for solid oxide fuel cells using strontium- and magnesium-doped LaGaO3materials. Journal of European Ceramic Society 21: 727-735.
- Wolfenstine J (1999) Intrinsic creep resistance of two solid oxygen fuel cell electrolytes cubic zirconia vs. Doped lanthanum gallate. Electrochemical and Solid-State Letters 2: 210-211.
- Hayashi H, Suzuki M, Inaba H (2000) Thermal expansion of Sr- and Mg-doped LaGaO3. Solid State Ionics 128: 131-139.
- Kendall KR, Navas C, Thomas JK, Loye HCZ (1995) Recent developments in perovskite-based oxide ion conductors. Solid State Ionics 82: 215-223.
- Zhang GB, Smyth DM (1995) Protonic conduction in Ba2In2O5. Solid State Ionics 82: 153-160.
- Schober T, Friedrich J, Krug F (1997) Phase transition in the oxygen and proton conductor Ba2In2O5in humid atmospheres below 300°C. Solid State Ionics 99: 9-13.
- Abraham F, Debreville-Gresse MF, Mairesse G, Novogrocki G (1988) Phase transitions and ionic conductivity in Bi4V2O11an oxide with a layered structure. Solid State Ionics 28-30: 529-532.
- Krok F, Abrahams I, Wrobel W, Kozanecka-Szmigiel A, Dygas JR (2006) Oxide-ion conductors for fuel cells. Materials Science (Poland) 24: 13-22.
- Takahashi T, Iwara H, Esaka T (1977) High oxide ion conduction in sintered oxide of the system Bi2O3-M2O5. Journal of the Electrochemical Society 124: 1563-1569.
- Yaremchenko AA, Kharton VV, Naumovich EN, Tonoyan AA (2000) Stability of d-Bi2O3-based solid electrolytes. Materials Research Bulletin 35: 515-520.
- Verkerk MJ, Burggraaf AJ (1981) High oxygen ion conduction in sintered oxides of the Bi2O3-Dy2O3System. Journal of the Electrochemical Society 128: 75-82.
- Goutenoire F, Isnard O, Retoux R, Lacorre P (2000) Crystal structure of La2Mo2O9, a new fast oxide-ion conductor. Chemistry of Materials 12: 2575-2580.
- Subramania A, Saradha T, Muzhumathi S (2007) Synthesis, sinterability and ionic conductivity of nanocrystalline Pr-doped La2Mo2O9fast oxide-ion conductors. Journal of Power Sources 167: 319-324.
- Marrero-López D, Canales-Vázquez J, Ruiz-Morales JC, Irving JTS, Núñez P (2005) Electrical conductivity and redox stability of La2Mo2-xWxO9materials. Electrochimica Acta 50: 4385-4395.
- Georges S, Goutenoire F, Bohnke O, Steil MC, Skinner SJ, et al. (2004) The LAMOX family of fast oxide-ion conductors: Overview and recent results. Journal of New Materials for Electrochemical Systems 7: 51-57.
- Cong L, He T, Yuan J, Guan P, Huang Y, et al. (2003) Synthesis and characterization of IT-electrolyte with perovskite structure La 0.8Sr 0.2Ga 0.85Mg 0.15O3- dby glycine–nitrate combustion method. Journal of Alloys and Compounds 348: 325-331.
- Marrero-López D, Ruiz-Morales JC, Pérez-Coll D, Núñez P, Abrantes JCC, et al. (2004) Stability and transport properties of La2Mo2O9. Journal of Solid State Electrochemistry 8: 638-643.
- Nakayama S, Sakamoto M (1998) Electrical properties of new type high oxide ionic conductor RE10Si6O27 (RE = La, Pr, Nd, Sm, Gd, Dy). Journal of European Ceramic Society 18: 1413-1418.
- Arikawa H, Nishiguchi H, Ishihara T, Takita Y (2000) Oxide ion conductivity in Sr-doped La10Ge6O27apatite oxide. Solid State Ionics 136-137: 31-37.
- Lacorre P, Goutenoire F, Bohnke O, Retoux R, Laligant Y (2000) Designing fast oxide-ion conductors based on La2Mo2O9. Nature 404: 856-858.
- Xia T, Li J, Li Q, Liu X, Meng J, et al. (2006) Synthesis, structural characterization and electrical property of new oxide ion conductors: La3MMo2O12(M=In, Ga and Al). Solid State Ionics 177: 3267-3273.
- Jin TY, Rao MVM, Cheng CL, Tsai DS, Hung MH (2007) Structural stability and ion conductivity of the Dy and W substituted La2Mo2O9. Solid State Ionics 178: 367-374.
- Marrero-Lopez D, Pena-Martinez J, Ruiz-Morales JC, Perez-Coll D, Martin-Sedeno MC, et al. (2007) Applicability of La2Mo2-yWyO9materials as solid electrolyte for SOFCs. Solid State Ionics 178: 1366-1378.
- Collado J, Aranda MAG, Cabeza A, Olivera-Pastor P, Bruque S (2002) Synthesis, structures, and thermal expansion of the La2W2-xMoxO9series. Journal of Solid State Chemistry 167: 80-85.
- Yoshimura M, Rouanet A (1976) High temperature phase relation in the system La2O3.WO3. Materials Research Bulletin 11: 151-158.
- Marrero-Lopez D, Pena-Martinez J, Ruiz-Morales JC, Nunez P (2008) Phase stability and ionic conductivity in substituted La2W2O9. Journal of Solid State Chemistry 181: 253-262.
- Iwahara H, Uchida H, Maeda N (1982) High temperature fuel and steam electrolysis cells using proton conductive solid electrolytes. Journal of Power Sources 7: 293-301.
- Iwahara H, Uchida H, Ono K, Ogaki K (1988) Proton conduction in sintered oxides based on BaCeO. Journal of Electrochemical Society 135: 529-533.
- Hishinuma K, Kumaki T, Nakai Z, Yushimura M, Somiya S (1986) Characterization of Y2O3-ZrO2 powder Synthesis under Hydrothermal Conditions: in Advances in Ceramics. American Ceramic Society, Westerville, OH.
- Kriechbaum GW, Kleinschmit P, Peuckert D (1988) Wet chemical Synthesis of Zirconia powders Ceramic Transactions. American Ceramic Society, Westerville, OH.
- Stokes SJ, Islam MS (2010) Defect chemistry and proton dopant association in BaZrO3 and BaPrO3. Journal of Materials Chemistry 20: 6258-6264.
- Furøy KA, Haugsrud R, Hänsel M, Magrasó A, Norby T (2007) Role of protons in the electrical conductivity of acceptor-doped BaPrO3, BaTbO3, and BaThO3. Solid State Ionics 178: 461-467.
- Xin X, Lü Z, Huang X, Sha X, Zhang Y, et al. (2006) Influence of synthesis routes on the performance from weakly agglomerated yttria-stabilized zirconia nanomaterials. Materials Research Bulletin 41: 1319-1329.
Relevant Topics
- Additive Manufacturing
- Coal Mining
- Colloid Chemistry
- Composite Materials Fabrication
- Compressive Strength
- Extractive Metallurgy
- Fracture Toughness
- Geological Materials
- Hydrometallurgy
- Industrial Engineering
- Materials Chemistry
- Materials Processing and Manufacturing
- Metal Casting Technology
- Metallic Materials
- Metallurgical Engineering
- Metallurgy
- Mineral Processing
- Nanomaterial
- Resource Extraction
- Rock Mechanics
- Surface Mining
Recommended Journals
Article Tools
Article Usage
- Total views: 24756
- [From(publication date):
November-2013 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 19423
- PDF downloads : 5333