Research Article Open Access
Field Management of Anthracnose (Colletotrichum lindemuthianum) in Common Bean through Fungicides and Bioagents
| Mohammed Amin*, Sileshi Fitsum, Thangavel Selvaraj and Negeri Mulugeta | ||
| Ambo University, Ethiopia | ||
| Corresponding Author : | Professor Mohammed Amin Department of Plant Science Ambo University, Ethiopia Tel: 0922811664 E-mail: yonias_1986@yahoo.com |
|
| Received December 04, 2013; Accepted March 18, 2014, 2014; Published March 20, 2014 | ||
| Citation: Amin M, Fitsum S, Selvaraj T, Mulugeta N (2014) Field Management of Anthracnose (Colletotrichum lindemuthianum) in Common Bean through Fungicides and Bioagents. Adv Crop Sci Tech 2:124. doi: 10.4172/2329-8863.1000124 | ||
| Copyright: © 2014 Amin M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | ||
Related article at Pubmed Pubmed  Scholar Google Scholar Google |
||
Visit for more related articles at Advances in Crop Science and Technology
Abstract
Common bean anthracnose is a major production constraint in bean growing regions of Ethiopia. This study aimed to determine whether foliar sprays of mancozeb, folpan and mancolaxyl or antagonistic bioagents; Trichoderma harzianum, Trichoderma viride and Pseudomonas fluorescens could reduce anthracnose symptoms and consequently, increase yield and yield components. A total of seven treatments were arranged in a randomized complete block design with three replications. Statistical analysis showed significant differences among treatments. Anthracnose incidence, severity, infected pods per plant and the area under disease progress curve were highest in the control plots compared to the fungicide sprayed and bioagent treated seed plots. The highest percentage of infected pods per plant of 78.9 and 55.0 recorded on the control and mancozeb sprayed plots, respectively. The highest AUDPC value resulted in the lowest yield of 1.01 t/ha in the control plots compared to a highest yield of 3.3 t/ha from the sprayed plots with folpan and 1.8 t/ha from plots treated with Pseudomonas fluorescens. Relative yield losses of 69.7, 46.3 and 22.8% were recorded from the control, seed treated plots with P. fluorescens and sprayed plots with mancolaxyl, respectively. Economic analysis revealed that the highest rate of return of 8,740 was obtained from Pseudomonas fluorescens seed treatment and the highest net benefit value of 43,154 calculated on folpan foliar spray treatment. The results of the present study support the novel possibility of using folpan foliar spray and Pseudomonas fluorescens seed treatments to decrease anthracnose symptoms in common bean and consequently, achieve greater yield.
| Keywords | |
| Bioagents, Folpan; Pseudomonas fluorescens; Mancolaxyl;Severity; Seed treatment | |
| Introduction | |
| Common bean (Phaseolus vulgaris L.) is a key grain legume crop and a vital source of nutrition worldwide. The FAO reports that half of the world’s common bean production occurs in low income, food deficit countries where this staple crop contributes to food security. The other half is produced in countries like the U.S., where common bean is an important economic crop with 769 thousand hectares of dry and snap beans planted in 2012, and with a farm gate value of $1.5 billion [1]. The value of the common bean crop exceeds that of all other legumes combined, including chickpea, lentil, pea, and cowpea, thus indicating the current and potential future economic role of this crop. | |
| In Ethiopia, common bean is mainly cultivated in the Eastern, Southern, South-western and Rift valley regions of the country [2]. Despite its economic significance and wide area of production, the national annual yield is low, ranging from 0.615-1.487 tons/ha between the years 2004 and 2010 [3]. The low national yield could be attributed to various constraints. But a recent study revealed that pests and diseases are ranked as the second important production constraints in the Central Rift Valley region, next to drought and third in the Southern parts of the country, next to drought and shortage of land [4]. Moreover, Yesuf [5] emphasized that diseases are known to be the major factors which threaten the productivity of common beans in general and common bean in particular. Anthracnose (Colletotrichum lindemuthianum), rust (Uromysis appendiculatus), angular leaf spot (Phaeoisariopsis griseola) and common bacterial blight (Xanthomonas compestris pv. phaseoli) are common diseases of bean in Ethiopia [2]. Among these, anthracnose caused by the fungus Colletotrichum lindemuthianum (Sacc. and Magnus) Lams-Scrib. is the most wide spread and economically important seed borne disease, mainly in the tropical and sub-tropical bean growing regions of the world including Ethiopia [2,6]. It has been confirmed that infection of susceptible cultivars like Mexican-142 and Awash-1 in favorable environmental conditions leading to an epidemic could result in 100% yield loss [7]. A study by Tesfaye [8] stated that yield loss up to 62.8% due to anthracnose was recorded in Ethiopia on susceptible cultivars of common bean. | |
| The efficacies of seed dressing and foliar fungicides like benlate,difenoconazole, mancozeb and carbendazim have been carried out in Ethiopia [9,10]. However, due to the increased risk of developing resistance, there is a growing need for the evaluation of the efficacies of new alternative fungicides and other cost effective and ecofriendly management options. Folpan, a protective fungicide which is commonly applied for the control of a number of fungal diseases including anthracnose of cucumbers, melons, pumpkins, squash and tomatoes, has a multi-site activity, which provides excellent resistance management option [11]. Similarly, the co-formulation of metalaxyl and mancozeb (Mancolaxyl 72 WP), which resulted in satisfactory control of Colletotrichum coccodes on tomatoes, was found to be a good option to prevent fungicide resistance [12]. It has been reported that seed dressing or soil application of bioagents like Trichoderma viride, Trichoderma harzianum and Gliocladium virens caused significant inhibition of mycelial growth of C. lindemuthianum, thereby effectively controlled the seed borne infection and increased the seed germination of common bean [13]. Extracellular metabolites like siderophores, antibiotics, lytic enzymes and volatile compounds produced by rhizobacteria (Pseudomonas fluorescens and Bacillus cepacia) have also been reported to effectively reduce lesions and damages caused by C. lindemuthianum on bean plants [14]. Generally, recent innovations showed that biological control of crop diseases is getting increased attention as economic and environmentally sound approach. But in Ethiopia, the method has received comparatively little attention. Apparently, the management of common bean anthracnose through bioagents, particularly, P. fluorescens, T. viride and T. harzianum have not been studied so far in Ethiopia. Much work still needs to be done in the management of bean anthracnose through alternative fungicides and bio agents, since the disease is still causing devastation on the crop. Therefore, the present work was carried out with the objective of evaluating the efficacies of alternative foliar spray fungicides and seed treatment bioagents for the management of common bean anthracnose and also to determine the economics of fungicide sprays as well as bioagents seed treatments. | |
| Materials and Methods | |
| Description of the study area | |
| The field experiment for the management of common bean anthracnose through bioagents and fungicides was conducted at Ambo University’s research farm, during the main cropping season of 2013. Ambo is located 120 km west of Addis Ababa at 8°98’ South latitude and 37°83’ North longitude. It has a total geographical area of 83,598.69 sq. km., with elevation ranging from 1380-3300 meter above sea level. Annual rainfall ranged from 900-1100 mm and temperature ranged from 10-27°C, with an average of 18°C. The soil type of the study site is vertisol with a pH value of 6.8. | |
| Experimental materials used | |
| The highly susceptible common bean variety to bean anthracnose, Mexican-142 [15], was used in the field experiment. Discolored seeds with typical anthracnose lesions were obtained from the Ethiopian Seed Enterprise, Hawassa. Three fungicide schedules; mancozeb (Unizeb 80% WP), folpan 80 WDG and mancolaxyl 72 WP (Mancozeb with Metalaxyl) were evaluated separately against common bean anthracnose disease. Three antagonistic bioagents viz., Trichoderma harzianum, T. viride and Pseudomonas fluorescens were also evaluated separately against common bean anthracnose disease. All the three bioagents were obtained from the Department of Plant Sciences, Ambo University, Ethiopia. | |
| Experimental design and treatments | |
| The treatments were subjected to RCBD with three replications. Three fungicides, three antagonistic bioagents and one control, total 7 treatments were evaluated against common bean anthracnose. Spacing between plants was maintained at 10 cm and between rows 40 cm. There were 21 plots, each consisting of 6 rows. Plots had a width of 2.4 m and length 2m. Each row had 20 plants. In general, there were 120 plants per plot in which thirteen of them were pre-tagged from the central four rows. The total experimental area landed on 182.4 m2 of land. Seeds were planted at the rate of two seeds per hole and thinned to one plant, 15 days after sowing (DAS) to insure 120 plants per plot. All agronomic practices were kept uniform for all plots of each treatment. Mancozeb (Unizeb 80% WP) was applied at the rate of 2 kg/ha, folpan 80 WDG at the rate of 2.6 kg/ha and mancolaxy 72 WP (mancozeb + metalaxyl) at the rate of 3.5 Kg/ha. All the fungicides were applied as of the disease onset at 7 days interval for 4 times using a Knap-sack sprayer. Bioagents were applied as seed treatments by the method of [16]. Talc based formulations (28x10-6 cfu/g product) of T. viride and T. harzianum were used at the rate of 40 g/Kg of seeds, soaked in 1L of water for 24 hrs [17]. Similarly, a talc based formulation of P. fluorescens by the method of [18] was used at the rate of 10 g/Kg to soak the seeds in 1L of water for 24 hrs. The treated seeds were dried overnight before sowing. | |
| Anthracnose disease assessment | |
| Thirteen plants were selected and pre-tagged from each plot using W-shaped sampling after the plants emerged. Disease epidemic data were collected from pre-tagged plants starting from the onset of the first anthracnose symptoms at 14-days intervals. Plants that showed symptoms of anthracnose were counted from the pre-tagged plants and the percentage of disease incidence was calculated according to the formula by Wheeler [19]. | |
| The severity of anthracnose on the leaves of pre-tagged common bean plants were graded using standard disease scales of: 1-9, where, 1= no visible disease symptoms; 3= presence of very small lesions, mostly on the primary vein of leaf’s lower side or on the pod, that covers approximately 1% of surface area; 5= presence of several small lesions on the petiole or on the primary and secondary veins of the leaf’s lower side or small round lesions on the pods, with or without reduced sporulation, that covers approximately 5% of the pods surface area; 7= presence of enlarged lesions on the lower side of the leaf. Necrotic lesions can also be observed on the upper leaf surface and on the petioles. On the pods, the presence of medium lesions are evident but also some small and large lesions generally with sporulation and that cover approximately 10% of the pod’s surface area may be found and 9= more than 25% of the leaf surface area covered with large coalescing and generally necrotic lesions resulting in defoliation [20]. | |
| Assessment of crop growth, seed yield and yield components | |
| The Plant height, number of pods per plant, infected pods per plant and seeds per pod were recorded from the 13 pre-tagged plants. The harvested pods were sun dried and the respective seed yields of the different treatments were measured. Bean yield data was adjusted at 10% moisture content after measuring using a moisture tester. Seed yield per plot was converted into tons per hectare. The weight of 100 seeds was taken randomly from harvested seed lots. | |
| The area under the disease progressive curve and disease progress rates | |
| The area under the disease progressive curve (AUDPC) was computed from the PSI data recorded at each date of assessment as described by Campbell and Madden [21]. | |
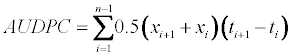 |
|
| Where n is the total number of assessments, ti is the time of the ith assessment in days from the first assessment date, xi is percentage of disease severity at ith assessment. AUDPC was expressed in percentdays because the severity (x) was expressed in percent and time (t) in days. The rates of disease progress were obtained from the regression of the PSI data fit to the Gompertz model [-ln (lnY)] with dates of assessments. | |
| Statistical analysis | |
| Analysis of variance was performed for disease parameters (incidence, PSI, AUDPC, disease progress rates and infected pods per plant) and yield related parameters (seed yield, pods per plant and 100 seed weight) using the Statistical Analysis System version 9.1.3 software [22]. Least significance difference was used to separate treatment means (P<0.05). Correlation analysis was used to examine the relationship between epidemic data and seed yield. | |
| Cost and benefit analysis | |
| The price of common bean seeds (Birr/Kg) was assessed from the local market and the total price of the commodity obtained from each treatment was computed on hectare basis. Input costs like fungicides, bioagents and labor were converted into hectare basis according to their frequencies used. Since there were significant differences between mean yields of treatments, the obtained data were analyzed using the partial budget analysis method [23]. | |
| Results and Discussion | |
| Bean anthracnose incidence and severity | |
| The severity and incidence of anthracnose increased with time from the 39 DAS onwards. Disease incidence data showed highly significant differences (P<0.01) among treatments at 39 and 67 DAS and significant differences (P<0.05) at 53 and 81 DAS. There were no significant differences in disease incidences among treatments at the final (95 DAS) date of disease assessment (Table 1). Maximum disease incidences were recorded from the untreated control, 74.4% at the initial (39 DAS) and 100% at the final (95 DAS) date of disease assessment. The least disease incidences were recorded from plots treated with folpan foliar spray fungicide, 5.1% and 87.2%, at the initial (39 DAS) and final (95 DAS) respectively, dates of disease assessment. Among the bioagents, plots treated with T. viride and P. fluorescens showed the least disease incidence (38.5%) at the initial date (39 DAS) of disease assessment. P. fluorescens showed the least disease incidence at 39, 53, 67 and 81 DAS. Disease incidence reached its maximum at the podding stage on the control plots, which could be due to the accumulation of secondary inoculums, susceptibility of the crop’s stage and or the occurrence of favorable environmental condition. As stated by [24] when no heavy seedling infection is observed, another phase of marked susceptibility will be encountered at the early stage of pod formation. Mean severity of 59.3 at the podding stage was recorded at Ambo by Tesfaye [8]. | |
| The percent of severity index (PSI) data revealed that the severity of anthracnose on the control plot was higher than the protected plots. Highly significant differences (P<0.001) among treatments were recorded at all dates of assessment. Maximum disease severity, 32.2% at the initial (39 DAS) and 96.0% at the final (95 DAS) dates of disease assessment were recorded from the untreated control. The least disease severities were recorded from plots spayed with folpan foliar spray fungicide, 1.1% and 34.2%, at the initial (39 DAS) and final (95 DAS) respectively, followed by plots treated with mancozeb foliar spray fungicide, 7.7% at the initial (39 DAS) dates of disease assessment (Table 1). Spraying foliage at flowering initiation, late flowering, and pod fill to achieve satisfactory bean anthracnose disease control [25]. Among the bioagents, the least disease severities were recorded from plots treated with P. fluorescens, 5.4% and 55.6%, followed by plots treated with T. viride, 18.52% and 56.98% at the initial (39 DAS) and final (95 DAS) respectively, dates of disease assessment. High disease severity on the control plots indicated that all the treatments significantly reduced the severity of anthracnose at both dates of assessment. The fungicide sprays particularly permitted the crop to reach physiological maturity without being under severe anthracnose infection. | |
| Area under diseases progress curve (AUDPC) | |
| For evaluating practical disease management strategies, the two most commonly used tools are comparing disease progress curves and AUDPC between treatments [26]. The AUDPC analysis showed the overall disease development was significantly affected by the management program applied. The increase in incidence throughout the assessment days indicated the spread of the disease in space. The data showed highly significant differences (P<0.01) among treatments. Maximum AUDPC value (3197.5 %-days) was computed from the untreated control, whereas the minimum value (835.8 %-days) from plots treated with folpan spray fungicide followed by mancozeb (1552.3 %-days). Among the bioagents, the least AUDPC value was recorded on plots treated with P. fluorescens seed treatment (Table 1). | |
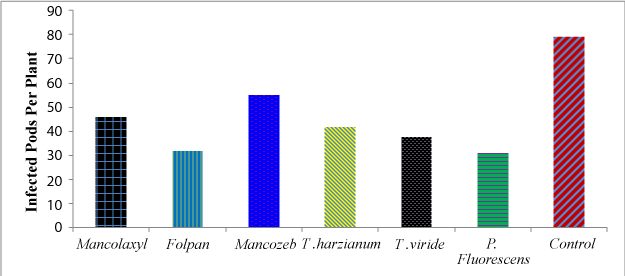
| Infected pods per plant due to bean anthracnose | |
| Data on the percentage of infected pods per plant showed highly significant differences (P<0.01) among treatments. The highest percentage of pod infection (78.9%) was recorded from the control plots, whereas the least (31.0%) from plots treated with P. fluorescens, followed by (31.8%), from folpan treated plots (Figure 1). Frequent rainfall with optimum temperature encountered in August and September of the growing season might have contributed much for the observed severe pod infection. As reported by [27] if the pod filling stage of beans coincides with frequent rainfall and moderate temperature in areas like Bako and Ambo, the risk of severe pod and seed infection must be anticipated. | |
| Field management of bean anthracnose using fungicidal and bioagents on yield components and seed yield | |
| Data on yield parameters showed highly significant differences (P<0.01) among treatments in the number of pods per plant, seeds per pod and seed yield, whereas, no significant differences were observed in the 100 seed weight. Plots treated with mancolaxyl foliar spray fungicide gave the highest number of pods per plant (29.4) followed by folpan (28.9) and mancozeb (25.6) foliar spray fungicides. Among the bioagents, P. fluorescens gave the highest number of pods per plant (22.4). The least number of pods per plant was recorded from the untreated control (13.1). The highest number of seeds per pod was obtained from plots treated with folpan foliar spray (5.1) followed by plots treated with the bioagents, T. harzianum (4.9) and P. fluorescens (4.7). The untreated control gave the least number of seeds per pod (3.2). The seed yield data showed highly significant differences among treatments. The maximum yield (3.3t ha-1) was obtained from plots treated with folpan foliar fungicide, followed by 2.6 t ha-1 and 2.4t ha-1, from plots treated with the foliar fungicides, mancolaxyl and mancozeb respectively. Among the bioagents, P. fluorescens gave the highest yield (1.8 t ha-1) (Table 2). | |
| Common bean yield loss due to anthracnose | |
| The computed relative yield losses showed notable differences among treatments. Yield losses were highly reduced by fungicide chemicals; mancolaxyl (22.8%) and mancozeb (27.3%), compared to the untreated control and the bioagents. The highest yield loss was calculated from the untreated control (69.7%). Among the bioagents, plots treated with P. fluorescens gave the least relative yield loss (46.3%), whereas both T. viride and T .harzianum showed a relative yield loss of (54.1%). The results are in agreement with the findings of [8], which evaluated the severity of bean anthracnose and its effect on yield and found that high disease severity between 17.2%-76.6% resulted in mean yield loss of 67.2%. Similarly, Sharma et al. [28] reported that pod infection had direct effect on seed yield and stressed that the pod development stage is the most vulnerable stage of common beans for quick disease progress. Apparently, the severe pod infection (78.9%) which was recorded on the untreated control plots, could have contributed much for the estimated yield loss. As reported by Conner et al. [29], application of the foliar fungicide headline could reduce losses in seed yield and quality from bean anthracnose disease. | |
| Disease progress rate (r) | |
| Comparisons of the rates of development of disease among the treatments were subsequently made based on the Gompertz model by fitting the PSI data with dates of assessment (Table 3). The highest disease progress rate (0.98 unit-days), was computed from the untreated control, whereas the least (0.91 unit-days), from plots treated with mancozeb foliar spray. Among the bioagents, the least disease progress rate was attained in plots treated with T. viride (0.93 unit-days). But generally, high disease progress rates were observed in all the treatments. This could be due to high density of initial inoculum from the infected seeds. | |
| Experimental studies have shown that the rates of disease increase were considerably influenced by the number of initial disease foci [26,30]. In an experiment with southern blight of processing carrot, the rate of disease increase generally increased as the number of initial foci increased [31]. | |
| Correlation between yield and disease parameters | |
| Correlations among the disease parameters and with the yield parameters revealed highly significant (P<0.01) positive correlations between PSI and AUDPC and also between PSI and the percentage of pod infection. Significant (P<0.05) positive correlations were observed between AUDPC and the percentage pod infection and also with the disease progressive rate. Disease parameters, AUDPC and the terminal disease severity (PSI) showed significant (P<0.05) negative correlations with the seed yield. As reported by Sharma et al. [28], highly significant correlations between anthracnose severity and percentage reductions in the number of seeds per pod and seed weight. Marcinkowska and Borucka [32] found significant positive correlation between the incidences of C. lindemuthianum in P. vulgaris seeds and leaf, pod and stem infection by the pathogen under natural field conditions. The disease progressive rates and percentage of pod infection showed nonsignificant negative correlations with seed yield. Similarly, the disease progressive rates showed non-significant positive correlations with the percentage of pod infection and terminal disease severity. The results suggest that reliable yield loss estimates could be made on the basis of the severity level by employing regression equations. Especially, for crops growing under epidemic conditions, PSI value recorded during the podding stage of common bean could be a good indication of the expected yield (Table 4). | |
| Cost benefit analysis | |
| Cost benefit analysis was computed for all the treatments using the partial budget analysis method (Table 5). Due to the seasonal pattern of production and marketing of common bean, fluctuations in prices are very common. Meanwhile, the price of common bean from November to December was assessed and an average of 17 Birr/kg was used to compute the total sale revenue and net benefit of the total produce obtained. | |
| From the data analyzed, the highest variable cost (input and labor cost) was computed for mancolaxyl (13,939 Birr/ha). The highest net benefit (43,154 Birr/ha) was obtained from folpan and the least (7,503 Birr/ha) from the control. Folpan also gave the highest marginal benefit and cost benefit ratio. Input cost of the bioagents was found to be cheaper than the fungicidal treatments. Among them, P. fluorescens gave the highest marginal benefit. This bioagent also gave the highest marginal rate of return (8,740%) among all the treatments which indicated its economic advantage, especially for the resource poor farmers. | |
| Conclusion | |
| Common bean anthracnose is serious threat to bean production in the major common bean growing regions of Ethiopia, especially in areas like Ambo, where frequent rainfall and moderate temperature that prevail during the main cropping season, predispose the crop to attack by various pathogens including C. lindemuthianum. An alternative fungicide, folpan, with multi-site activities could be an important option for the management of the anthracnose disease. Similarly, the bioagents evaluated in the study were found to be economically important options that need to be further investigated, where P. fluorescens showed promising results in the control of common bean anthracnose. | |
| References | |
References
|
|
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 | Table 4 |
Figures at a glance
 |
| Figure 1 |
Relevant Topics
- Agricultural science
- Agronomy
- Climate impact on crops
- Crop Productivity
- Crop Sciences
- Crop Technology
- Field Crops Research
- Hybrid Seed Technology
- Irrigation Technology
- Organic Cover Crops
- Organic Crops
- Pest Management
- Plant Genetics
- Plant Breeding
- Plant Nutrition
- Seed Production
- Seed Science and Technology
- Soil Fertility
- Weed Control
Recommended Journals
Article Tools
Article Usage
- Total views: 19237
- [From(publication date):
April-2014 - Aug 18, 2025] - Breakdown by view type
- HTML page views : 14019
- PDF downloads : 5218
