H. pylori Associated Spruelike Duodenitis Presenting with Chronic Diarrhea, Hypoalbuminemia and Edema – A Case Report
Received: 12-Jan-2014 / Accepted Date: 06-Apr-2015 / Published Date: 10-Apr-2015 DOI: 10.4172/2161-0681.1000223
Abstract
Helicobacter pylori (H.pylori) infection is known to cause varied clinical presentations and histological changes in the gastrointestinal tract. It may be one of the several mimickers of celiac disease. We present a case of a 48 year old female who presented with intermittent episodes of diarrhea, loss of appetite and weight loss. On evaluation she had Iron deficiency, megaloblastic anemia and hypoalbuminemia causing pedal edema. Histopathological examination of gastric and duodenal mucosa revealed H.pylori associated gastritis sprue like morphological changes in the duodenal mucosa. She was put on anti H.pylori regimen leading to rapid clinical improvement within two months of initiating treatment. Hence, the possibility of H.pylori induced duodenitis should also be considered in the differential diagnosis of chronic diarrhoeal diseases.
Keywords: Helicobacter pylori; Intraepithelial lymphocytosis; Duodenum
312407Introduction
Helicobacter pylori (H. pylori), first described as “unidentified curved bacilli on gastric epithelium” by Warren and Marshall was briefly called Campylobacter pyloridis and then Campylobacter pyloris to acquire its present name in the year 1989. H. pylori is a spiral shaped gram negative bacterium which can be easily detected in histologic sections [1]. Infection by this organism can have varied clinical and pathological presentations including histological gastritis, peptic ulcer disease, Vitamin B12 deficiency, gastric mucosa- associated lymphoid tissue lymphoma and gastric adenocarcinoma [2]. Histologic evidence of duodenal intraepithelial lymphocytosis has been demonstrated in patients with H. pylori gastritis and may also mimic celiac disease (celiac sprue, gluten- sensitive enteropathy) histologically [3,4]. We report a case of 48 year old lady presenting clinically and histologically with sprue like disease associated with H. pylori induced active gastritis.
Case Report
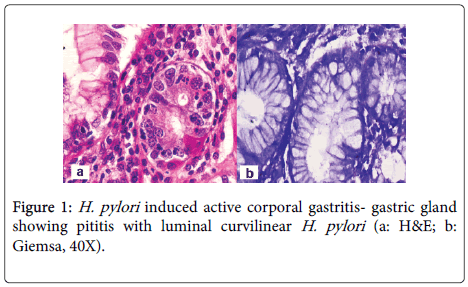
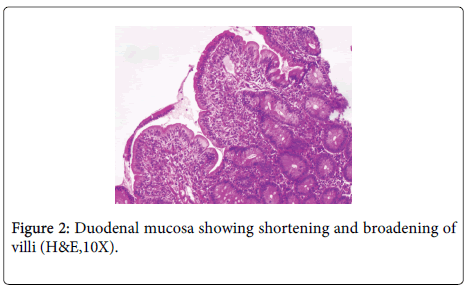
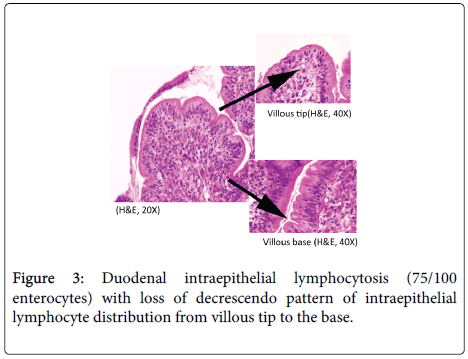
A 48 year old lady, resident of Karnataka, India presented with intermittent episodes of diarrhea, abdominal pain and loss of appetite since 1 ½ years. Each episode lasted for 5 to 6 days. She also gave a history of loss of weight. She had no history of food allergy. Routine haematological investigations revealed haemoglobin (Hb)- 9.2 gms/dl and haematocrit(Hct)- 28.4 gm/dl. Peripheral smear showed a normocytic normochromic to microcytic hypochromic RBC picture. Her platelet counts, total leucocyte counts (TLC), red blood cell (RBC) indices, urine and stool examination were normal. She was started empirically on oral iron and pancreatic enzymes supplements. Six months later, she revisited the hospital with similar episodes of diarrhea. Investigations revealed normal haemoglobin levels (13.5 gms/dl). Sigmoidoscopy was done and colonic biopsy obtained was histologically normal. She was treated symptomatically. She was not relieved of her symptoms for the next 6 months and continued to have diarrhea and further weight loss. She also developed pitting type of pedal edema. On investigations, her Hb had dropped to 10.4 gms/dl, Peripheral smear revealed macrocytic RBCs, hypersegmented neutrophils and reduced platelets suggestive of megaloblastic anemia. Biochemistry revealed hypoalbuminemia (2.7 gms/dl), reduced Vitamin B12 levels (104 pg/ml) and normal folate levels (5.5 ng/ml). No parasites or fat globules were detected on stool examination. Gastrodeodenoscopy was normal. Endoscopic biopsy from the second part of duodenum, antrum and pylorus of stomach were sent for histopathological examination. Gastric corpus showed glands lined by chief and parietal cells surrounded by lamina propria containing a diffuse dense lymphoplasmacytic and neutrophilic infiltrate. Surface mucus and few glandular lumina showed few curvilinear organisms suggestive of H.pylori which stained with Giemsa stain (Figure 1). However, these organisms were not identified in the gastric antrum which had a diffuse plasma cell infiltrate. Biopsy from the second part of duodenum showed distortion of normal villous architecture characterized by alteration of crypt villous ratio (1.5-2:1; normal is 3-5:1) (Figure 2), increase in intraepithelial lymphocytes (75/100 enterocytes) with loss of normal decrescendo pattern (Figure 3). Lamina contained diffuse dense plasma cell infiltrate. No parasites were seen. A histopathological diagnosis of H.pylori associated corporal active gastritis with sprue like duodenitis was rendered. The patient was started on anti H. pylori regimen with Pantacid, levofloxacin and amoxicillin along with parenteral Vitamin B12. Three months following initiation of therapy, patient improved symptomatically and gained 2 kgs weight. Her peripheral blood picture showed normal RBC morphology with absence of hypersegmented neutrophils. Her haemoglobin level was 10.5 gm/dl, hamatocrit was 32.4 gm/dl while her serum albumin levels rose to 3.7 gms/dl. Two years since then, she has been asymptomatic.
Discussion
Normal intraepithelial lymphocytes (IELs), in duodenal biopsy, is considered as 5]. However, various studies have cited the upper limit of normal duodenal IEL as <40/100 enterocytes [3,6,7]. Celiac disease(CD)/gluten sensitive enteropathy (GSE)/celiac sprue is characterized morphologically by villous atrophy, intraepithelial lymphocytosis and chronic duodenal inflammation [3]. However, similar “sprue like architectural changes” in the duodenal mucosa has been described in various other clinical conditions (Table 1).
| Celiac disease/Celiac sprue/Gluten sensitive enteropathy | Clinical features, characteristic IEL distribution (villous tip heavy) serological screening (antibodies to tissue transglutaminase, gliadin ), response to gluten free diet (GFD), age >2years,exclusion of other possible cause |
| Refractory sprue | Unresponsiveness to GFD, exclusion of other possible causes |
| Tropical sprue (TS) | Mucosal changes in entire length of small intestine, Vitamin B12 and folate deficiency, response to tetracycline |
| Viral gastroenteritis | Unresponsiveness to GFD, complete recovery with normal morphology |
| Parasitic infections like microsporidiosis, cyclosporidiosis, isosporiasis | Usually seen in immunocompromised patients, demonstration of microorganism on the surface or within the enterocytes lining the villi |
| Autoimmune enteropathy | Unresponsiveness to GFD, anti-enterocyte antibodies, response to steroids and immunosuppressive therapy |
| Crohn disease | Focal acute inflammation flanked by normal appearing mucosa |
| Zollinger Ellison syndrome | Concomitant gastric parietal cell hyperplasia |
| Enteropathy-type intestinal T cell lymphoma | Multifocal monoclonal IELs affecting the entire small intestine. |
Table 1: Differential diagnosis and salient features of conditions with intraepithelial lymphocytosis with partial/total villous architectural changes [4,5,8,9].
Duodenal intraepithelial lymphocytosis of uniform distribution and villous tip heavy with loss of decrescendo pattern has also been described as a feature of celiac sprue [8]. H. pylori infection, a potential cause of raised duodenal IELs, can also lead to sprue like changes in the duodenum [5]. The present case showed H.pylori associated active corporal gastritis along with similar histologic features. In the study by Hayat et al, 4 of the 13 (31%) patients with H. pylori associated lymphocytic gastritis had raised intraepithelial lymphocytes in the second part of the duodenum; 2 of these 4 patients also had associated villous atrophy [7]. Further, our patient was on normal diet without gluten restriction prior to onset of symptoms, throughout her symptomatic period and following anti-microbial therapy. The absence of symptoms in the two year follow up period, excludes the possibility of celiac sprue in the present case. Hence, serum immune-electrophoresis and other serological tests for celiac disease were not performed. Memeo et al. also observed a high rate of duodenal intraepithelial lymphocytosis in patients with H. pylori gastritis. Majority of these cases had patchy and villous base predominant pattern of intraepithelial lymphocyte distribution. However, no associated villous architectural abnormalities were seen [3]. Some workers have opined that H. pylori colonization of the duodenum occurs only in areas of diffuse gastric metaplasia (DGM) [9]. We did not identify foci of DGM in the current case.
Patients infected with H. pylori present with upper gastrointestinal symptoms like dyspepsia, epigastric or abdominal pain and gastro esophageal reflux. A small, but significant group of patients can however present with symptoms similar to celiac disease such as chronic diarrhea and nutritional deficiency anemia with some of them showing duodenal intraepithelial lymphocytosis [3,5,10]. Detection of H. pylori in patients with Vitamin B12 deficiency and correction of the same after H. pylori eradication has prompted few authors to suggest a causal relationship between H. pylori and Vitamin B12 deficiency [11]. We noticed a similar therapeutic response in the present case. However, follow up gastric and duodenal biopsies to evaluate therapeutic response couldn’t be performed since our patient refused further scopy examination citing symptomatic improvement and better sense of wellbeing. An added feature of our case was the malabsorption associated hypoalbuminemia resulting in pedal edema which resolved after anti H.pylori therapy. Various authors have commented on H. pylori infection associated hypoalbuminemia and giant gastric folds [12,13]. The latter feature, however, was not identified in the current case.
Duodenal intraepithelial lymphocytosis with villous atrophy is also described as a feature of TS with similar clinical manifestations; however these patients show prompt response to tetracycline [9,14]. In the present case, normal folate levels, absence of steatorrhea and prompt response to anti H. pylori therapy ruled out TS and viral gastroenteritis. Normal stool examination and absence of any other microorganisms in the duodenal biopsy ruled out concurrent parasitic infestation.
To conclude, H. pylori gastritis may co-exist with duodenal villous alterations and increased IELs. If the latter findings are taken in isolation, this may be labelled as “celiac disease”. This case serves to highlight the clinical and morphologic overlap in these diseases. Hence, patients with “sprue like changes” should also be evaluated for the presence of H. pylori.
References
- Peterson WL, Fendrick AM, Cave DR, Peura DA, Garabedian-Ruffalo SM, et al. (2000) Helicobacter pylori-related disease: guidelines for testing and treatment. Arch Intern Med160:1285-1291.
- Memeo L, Jhang J, Hibshoosh H, Green PH, Rotterdam H, et al.(2005) Duodenal intraepithelial lymphocytosis with normal villous architecture: common occurrence in H.pylori gastritis. Mod Pathol18:1134-1144.
- Dickson BC, Streutker CJ, Chetty R (2006) Coeliac disease: an update for pathologists. J ClinPathol59:1008-1016.
- Owens SR, Greenson JK (2007) The pathology of malabsorption: current concepts. Histopathology 50:64-82.
- Mahadeva S,Wyatt JI, Howdle PD (2002) Is a raised intraepithelial lymphocyte count with normal duodenal villous architecture clinically relevant? J ClinPathol55:424-428.
- Hayat M, Cairns A, DixonMF, O'Mahony S (2002) Quantitation of intraepitheliallymphocytes in humanduodenum: what is normal? J ClinPathol55:393-394
- Goldstein NS, Underhill J (2001) Morphologic features suggestive of gluten sensitivity in architecturally normal duodenal biopsy specimens. Am J ClinPathol116:63-71.
- Serra S, Jani PA (2006) An approach to duodenal biopsies. J ClinPathol 59:1133-1150.
- Carmel R, Aurangzeb I, Qian D (2001) Associations of food-cobalamin absorption with ethnic origin, age, Helicobacter pylori infection, and serum markers of gastritis.Am J Gastroenterol96:63-70.
- Walker MM (2003)What is tropical sprue? J GastroenterolHepatol. 18:887-890.
- Shimoyama T, Fukuda S, Mikami T, Munakata A, Yoshida Y, et al. (1995) A case of giant gastric folds with hypoalbuminemia healed by Helicobacter pylori eradication. Digestive Endoscopy7:185-189.
- Salmeron M, Desplaces N, Lavergen A, Houdart R (1989) Campylobacter pylori hypertrophic erosive gastritis and hypoalbuminemia healed by cephalexin therapy. GastroenterolClinBiol13: 109-110.
- Fauci AS, Braunwald E, Kasper DL, Hauser SL, Longo DL, et al. (2008) Chapter 288:Disorders of absorption. In: Harrison’s principles of internal medicine. McGraw Hill,USA.
Citation: Vasudevan G, Nayal B, Rao L, Pai G (2015) H. pylori Associated Spruelike Duodenitis Presenting with Chronic Diarrhea, Hypoalbuminemia and Edema – A Case Report. J Clin Exp Pathol 5:223. DOI: 10.4172/2161-0681.1000223
Copyright: ©2015 Vasudevan G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 19497
- [From(publication date): 6-2015 - Aug 22, 2025]
- Breakdown by view type
- HTML page views: 14733
- PDF downloads: 4764