Habilitation Outcomes in Paediatric Cochlear Implantation-A South Indian Experience
Received: 11-Sep-2018 / Accepted Date: 08-Oct-2018 / Published Date: 15-Oct-2018 DOI: 10.4172/2161-119X.1000354
Keywords: Habilitation; Cochlear implantation; Children; Patient
Abbreviations
CI: Cochlear Implants; AVH: Auditory Verbal Habilitation; CAP: Categories of Auditory Performance; SIR: Speech Intelligibility Rating; MAIS: Meaningful Auditory Integration Scale; MUSS: Meaningful Use of Speech Scale
Introduction
Cochlear Implants (CI) have become the standard of care for patients with bilateral severe to profound hearing loss who derive minimal or no benefit from conventional amplification with a hearing aid. CI is the recommended treatment for pre-lingual children who are hearing impaired either congenitally or very early in life and also for peri/postlingual patients who acquire profound hearing loss later in life. CI is an expensive treatment and cost effectiveness is therefore an essential factor when considering implantation [1,2]. A well documented foundation on which to base expectations of outcome is therefore very important. The efficacy of CI in the treatment of profound hearing loss has been documented by several CI centres [1,3,4]. However, due to advancements in implant technology, optimization of the implantation processes and expanding eligibility criteria, it is pertinent that results from centres performing the procedure are reported to the implant fraternity and both short and long-term outcomes are continuously evaluated to confirm consistency of service. The aim of this study was to assess the habilitation outcomes achieved at a large paediatric cochlear implant centre in South India-Madras ENT Research Foundation (MERF) in Chennai, based on the scores recorded for an age-matched cohort of children, at one year post-implantation. The outcome measures studied were-Categories of Auditory Performance (CAP), Speech Intelligibility Rating (SIR), Meaningful Auditory Integration Scale (MAIS) and Meaningful Use of Speech Scale (MUSS).
Study Methods
This was a cross-sectional retrospective study evaluating outcome scores for 115 profoundly hearing impaired but otherwise normal children who sequentially underwent uneventful cochlear implantation at our Institute between 2012 and 2014 and were habilitated over the next one year. Data was extracted from patient files, hospital charts and Auditory Verbal Habilitation records provided by the Medical Records Dept. of the hospital over the last year. Ethical approval from the Institutional Ethics Board was taken prior to data collection. The parents of implanted children were counselled about inclusion of their child’s data in this study and an appropriate written and informed consent was obtained with assurance to keep the data anonymous in literature. Children who had coexistent handicaps, inner ear anomalies, cognitive and behavioural issues, autism, dyslexia, difficult or incomplete implant insertion and those with significant residual hearing were all excluded to focus only on profoundly hearing impaired otherwise normal children. All the selected children had completed the in-house mandatory oneyear habilitation program with our professional team and their records were included in the study.
The pre-operative audiology test battery was scrutinized from medical files to confirm that these children had no residual hearing which could influence outcomes with CI. Thereby audiological assessment with Brainstem Evoked Response audiogram Audiometry (BERA), Auditory Steady-State Response (ASSR), Otoacoustic Emission (OAE), Pure Tone Audiometry (PTA), Speech Audiometry, Behavioural Observation Audiometry (BOA) and a hearing aid trial confirmed bilateral profound hearing loss in the cohort. In all participants bilateral sensorineural profound hearing loss (≥ 95 dB) across the speech frequency range was found. CT/MRI scans were also checked to confirm absence of any brain and inner ear abnormalities.
All children as part of the pre-operative work up had also been evaluated by a pediatrician, a child psychologist, an occupational therapist and a speech-language pathologist to confirm normal cognitive function, emotional fitness for implantation and to rule out co-morbidities, all of which could influence outcomes. Operative details were studied to confirm complete insertion of the electrode array uneventfully along with satisfactory intra-operative electrophysiological responses. The habilitation records were analyzed to check for satisfactory progress from the time of switch-on of device and over the one year and for any specific concerns with therapy recorded by the professionals. Since India is a multilingual country, habilitation was conducted in many of the major languages including English, Hindi and Tamil, but preferably in the child’s first language (mother-tongue). All the hospital files and habilitation records were maintained in English only.
The habilitation program included two one-hour sessions a week with the same professional therapist throughout the program for one year, with sequential documentation of progress using the various standardized habilitation outcome scores. Four different outcome measures were used in this study namely; CAP, SIR, MAIS and MUSS. CAP and SIR scores were recorded for all the implantees who were habilitated as per the internationally standardized St. Gabriel’s curriculum. CAP is a global outcome measure applied to assess the auditory receptive abilities of hearing impaired children. It consists of a non-linear hierarchical scale with seven categories, the lowest being “no awareness of environmental sounds” and the highest “use of telephone with a known speaker”. SIR is a five-point hierarchical scale globally used to measure the speech intelligibility of cochlear implantees. It ranges from “unintelligible speech” to “speech that is intelligible to all listeners”. Inter-observer reliability for both outcome measures has been formally validated [1,5,6].
MAIS is designed to assess the ability to make meaningful use of sound in every-day situations in hearing-impaired children. This scoring system is based on a structured parent interview and consists of ten questions appraising the child’s bond to the CI device, alertness to sound and ability to derive meaning from auditory phenomena. MUSS is designed similar to MAIS, however, the questions included evaluate the child’s use of speech in everyday situations in the areas of vocal control, use of speech without gesture or sign and use of communication strategies. In both MAIS and MUSS, the parent responds to each question using a five-point scale (0-4) reflecting how often the child displays the behaviour in question. The highest score possible on both scales is 40 [7]. MAIS scores were acquired for all children between the age of three and eight and MUSS scores were acquired for all participants under the age of eight. Infant-Toddler MAIS (IT-MAIS) is designed for infants and toddlers and analogous to MAIS, however, it differs from MAIS in questions exploring the child’s vocal behaviour associated with use of the device and was applied instead of the MAIS score for children younger than three years of age [8]. For all implantees in this study, outcome data for the above mentioned measures of CAP, SIR, MAIS and MUSS had been obtained prospectively by the professional therapist responsible for the habilitation of the child and was entered into a central electronic database from where data collection for the present study was done. The therapists who had habilitated this study group were not involved in the current study in order to avoid bias in interpretation of outcomes. The data was anonymized and compiled in the SPSS software (IBM SPSS Statistics for Windows: version 20.0. Armonk, NY: IBM Corp) for statistical analysis with the help of a biostatistician.
Results
Study population
Demographic data and study population characteristics are presented in Table 1. Both sexes were equally represented in the study population. Most participants were pre-lingually deaf (93%), whereas post-lingual deafness occurred in only 7%. Eighty four (73%) patients received a Nucleus Freedom implant, of which 31 (37%) had a contour electrode, 21 (25%) had an advanced contour and 32 (38%) had a straight array electrode. Nineteen (17%) patients were implanted with a MEDEL Combi 40+ implant, four (3%) received a MEDEL Pulsar implant and seven (6%) were implanted with an Advanced Bionics Device. The coding strategies used varied between implants; Advanced Combination Encoder (ACE) for Nucleus implant systems, Continuous Interleaved Sampling (CIS) for the MEDEL Combi 40+ model and CISPRO for Pulsar and HiRes for Advanced Bionics.
| Median | Range (months) | |
|---|---|---|
| Age at onset of deafness, mo | 0 | 0-144 |
| Duration of deafness, mo | 43 | 4-199 |
| Age at diagnosis, mo | 13.5 | 1-156 |
| Age at implantation, mo | 47 | 9-199 |
| n (%) | ||
| Male | 62 (53.9) | |
| Female | 53 (46.1) | |
| Prelingual | 107 (93.0) | |
| Postlingual | 8 (7.0) | |
| Bilateral implantation | 3 (2.6) | |
| Unilateral implantation | 112 (97.4) | |
| Syndromic deafness | 9 (7.8) | |
| Inner ear abnormality | 21 (18.3) | |
| Additional disability | 28 (24.3) | |
| Comorbidities | 6 (5.2) | |
Table 1: Study characteristics of 115 cochlear implantees.
Additional disabilities included autism spectrum disorders, cognitive impairment, behavioural difficulties including ADHD, oro-facial motor disability, oro-facial anatomical anomalies, visual impairment and physical difficulties. Cognitive impairment was most frequent and accounted for 11 (39%) of 28 cases. Four patients had more than one additional disability. Co-morbidity occurred in six patients; epilepsy (1), inflammatory bowel disease (1), hypothyroidism (1), hydrocephalus (1), cardiac anomaly (1) and peri-ventricular leukomalacia (1). Twenty one (18%) patients were diagnosed with inner ear abnormalities, the most common being Mondini dysplasia (7) followed by cochlear ossification (5), common cavity deformity (3), abnormally rotated cochlea (2), enlarged vestibular aqueduct syndrome (2), cochlear nerve hypoplasia (1) and anomalous course of the internal carotid artery (1). Prior to implantation 83 (72%) patients had been treated with a hearing aid. Cause of deafness for the study population is presented in Table 2. In 99 (86%) patients deafness was congenital, whereas, meningitis, mumps and typhoid fever accounted for nine cases and the cause remained idiopathic for seven patients.
| Etiology | n (%) |
|---|---|
| Congenital | 99 (86.1) |
| -Consanguinity | -19 (16.5) |
| -Congenital Rubella syndrome | -3 (2.6) |
| -Pendred syndrome | -1 (0.9) |
| -Usher syndrome | -1 (0.9) |
| -Waardenberg syndrome | -1 (0.9) |
| -Branchio-oto-renal syndrome | -1 (0.9) |
| -Goldenhar syndrome | -1 (0.9) |
| -Chudley McCullough syndrome | -1 (0.9) |
| -Premature | -3 (2.6) |
| -Unspecified maternal fever during pregnancy | -2 (1.7) |
| Meningitis | 7 (6.1) |
| Mumps | 1 (0.9) |
| Thypoid fever | 1 (0.9) |
| Unknown | 7 (6.1) |
Table 2: Aetiology of Hearing Loss for 115 cochlear implantees.
Outcome measures
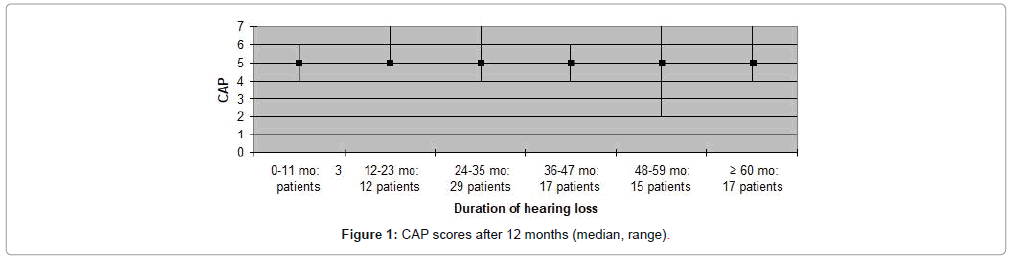
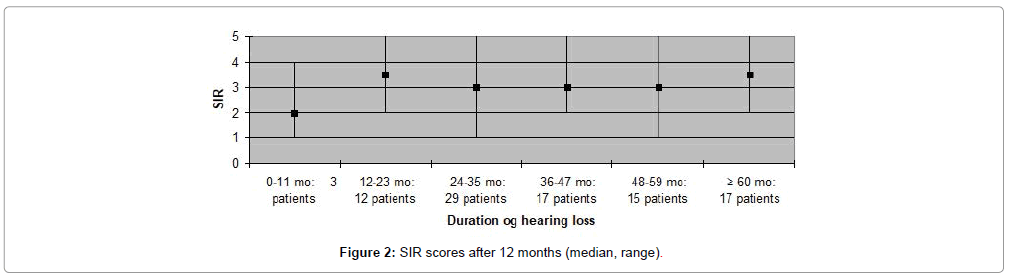
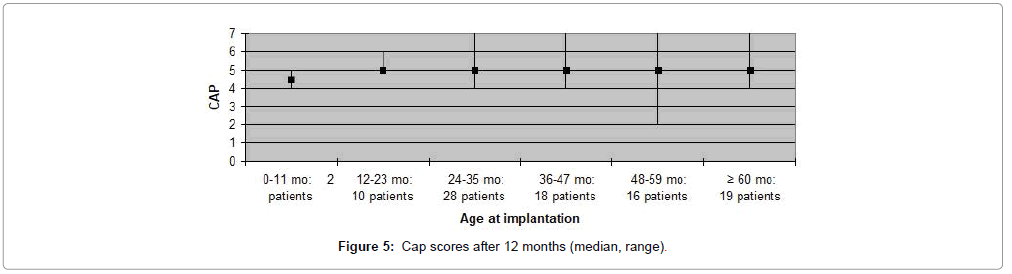
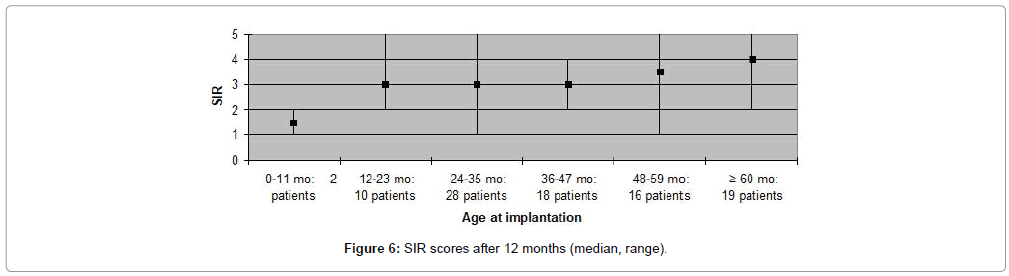
The CAP, SIR, MAIS and MUSS scores obtained one year after cochlear implantation, are shown in Figures 1-8. Median scores have been used for the description since they reflect the outcomes better in comparison to mean average values in these graphs. Patients are categorized according to duration of deafness in Figures 1-4 and according to age at implantation in Figures 5-8. Median CAP score was 5.0. Seventy four (64%) patients achieved a CAP score of five and 32 (28%) patients scored either six or seven. Only nine (8%) patients received a score lower than five, implying that 106 (92%) patients, as a minimum, were able to understand common phrases without lip-reading. Median SIR score was 3.0. Fifty six (49%) patients achieved a SIR score of four or five. Thirty four (30%) patients scored three and 25 (21%) scored two or lower. Ninety (79%) patients, thus, could produce some degree of connected intelligible speech after one year.
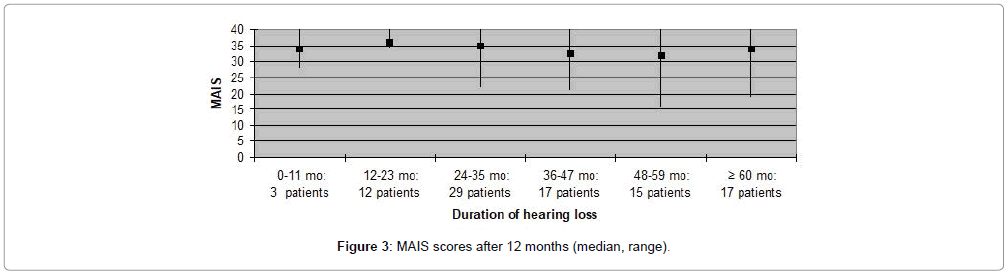
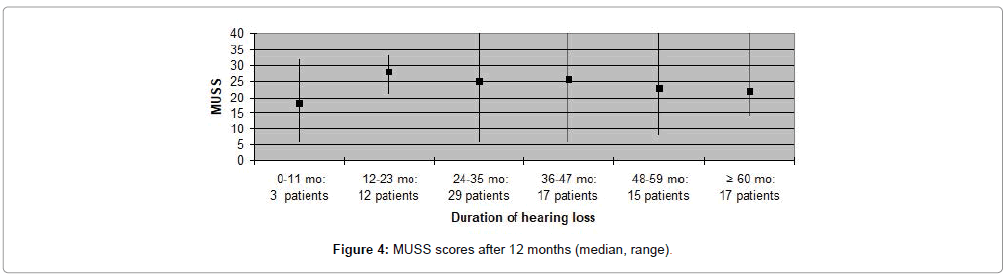
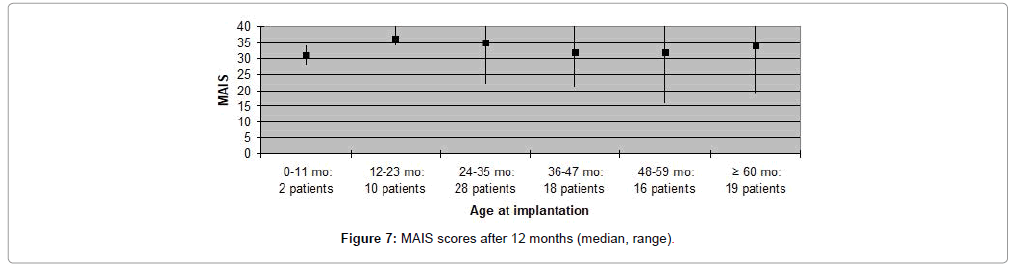
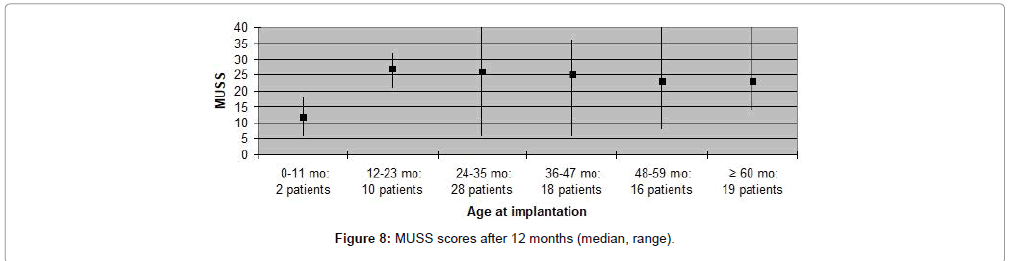
MAIS and MUSS scores were obtained for ninety one (79%) patients, only, as the remaining patients were older than eight years of age. Fifty nine (65%) patients received a MAIS score higher than 30, resulting in a median score above 30 both when evaluating participants according to duration of deafness, as well as age at implantation. The variation in MUSS scores was greater. Twenty (22%) patients scored higher than 30, while 43 (47%) patients received a score between 21 and 30. The median score in all patient categories was between 20 and 30, except for patients who had been deaf for less than one year where median score was 18 and for patients implanted at the age 0-11 months in which the median score was 12. Complications observed during the first year after CI are presented in Table 3. Five (4%) patients experienced minor complications that either resolved spontaneously or could be treated conservatively. Major complications, requiring surgical intervention and explantation of the implant followed by subsequent re-implantation, occurred in five (4%) patients (Figures 1-8).
| n (%) | |
|---|---|
| Minor complications¹ | 5 (4.3) |
| -Facial nerve twitching on stimulation | -2 (1.7) |
| -Temporary facial paresis | -1 (0.9) |
| -Wound seroma | -2 (1.7) |
| Major complications² | 5 (4.3) |
| -Wound infection | -2 (1.7) |
| -Wound infection due to trauma | -1 (0.9) |
| -Device failure due to trauma | -1 (0.9) |
| -Electrode extrusion due to CSOM | -1 (0.9) |
| Total | 10 (8.6) |
Table 3: Complications due to CI one year postoperatively for 115 patients.
Discussion
All outcome measures assessed in the present study showed high scores for both auditory and communication skills after implantation. One year post-operatively, more than 90% of the participants could manage without lip reading and more than one-third were able to speak in short sentences. One year is a relatively short follow-up period when evaluating the functional outcome following cochlear implantation. Communication skills continue to improve for several years after implantation and, therefore, the present results only reflect short-term outcome of CI [1,9,10]. However, most patients achieved high scores for both CAP and SIR one year postoperatively in the present study, probably indicating that they, in the future, will master communication skills at a higher level.
Currently an ongoing study based on this cohort is looking at their long-term habilitation outcomes, the results of which will be available by 2020. This will be of immense interest since it will reflect the first long term benefits documented in a South Indian population. In the western world, tangible benefits have already been documented well over the past decade as shown in the study done by Beadle et al. [1] in 2005 in the UK, where 30 CI children were followed up for 10 years to assess their long-term functional outcomes and academic-occupational status. These authors report that after 10 years of implant experience, 26 subjects (87%) always wore their device; 2 subjects (7%) frequently; and 1 subject (3%), occasionally. Only one child had discontinued use of his device. 26 (87%) of these children understood a conversation without lip reading and 18 (60%) used the telephone with a familiar speaker. 23 (77%) of these subjects also used speech intelligible to an average listener or a listener with little experience of a deaf person’s speech. Hence, they concluded that these implanted children will continue to do well at further follow up and get actively integrated into the normal society [1]. In our study a similar trend of gradual improvement in outcomes is noted although this assessment is early at the end of one year of CI use and long term outcomes will have to be further judged in this cohort.
In a similar study in literature, the outcomes of 168 CI patients implanted in Denmark were studied [11]. Evaluation was performed 6-24 months after implantation for seventy-four participants and 56 (76%) received a CAP score ≥ 5 whilst 37 (50%) received a SIR score ≥ 3. In various other studies the share of implantees achieving a SIR score ≥ 3 one year post-implantation ranges from 11-16% [9,12,13] and one study reports a share of 26% of the implantees achieving a CAP score ≥ 5 [10]. Both the present study and the previously mentioned report on Danish implantees show substantial progress in both CAP and SIR scores, although a larger number of the children implanted at Chennai achieved high CAP and SIR scores. One explanation to this difference could be that implant age is twelve months for all patients included in the present study, while implant age of the Danish implantees ranged from 6-24 months.
In the present study high CAP and SIR scores (median scores; 5 and 3.5, respectively) were observed for participants with a duration of hearing loss of more than five years. In contrast, previous studies have consistently found relatively poor results for this patient group compared to patients with shorter duration of hearing loss [4,14]. In the current study, four (10%) patients out of 41 with impaired hearing for more than five years, were post-lingually deaf which may partly explain the high scores observed in this group. Ten (8.6%) of the 115 patients in the present study experienced complications during the first year postoperatively and, of these, five patients developed major complications requiring surgical intervention. This complication rate is similar to that reported in other studies on outcome after CI [15].
Cultural differences could be another explanation to the better outcome observed in Indian patients. As Indian children mostly live in joint families consisting of several generations in the same household, the entire family becomes involved in the rehabilitation process.
Thus, the child is under constant and intense training. In contrast, a household in Western Europe (i.e. Denmark) typically consists of two working parents, leaving less time for intensive rehabilitation. Also, the two study populations differ socioeconomically. CI’s performed in India are mostly Government health scheme funded, some privately financed and some others sponsored by charity. Most of these children come from low socio-economic background. In Denmark, on the other hand, CI’s are covered by public health insurance and Danish patients are therefore more likely to represent the general population of all social statuses. But since CI is an expensive procedure, the motivation to succeed is probably greater for Indian than Danish patients, resulting in increased focus on rehabilitation. The two studies were carried out in different periods of time, which probably contributed to the observed differences in outcome. All participants in the present study underwent implantation between 2012 and 2014, while the Danish implantees received their CI between 1993 and 2004.
During recent times cochlear implants, as well as surgical techniques, rehabilitation strategies and criteria for patient selection, have all improved, thereby also improving outcome. As defined by Black et al. in 2012 the overall success of the implant program is achieved when the outcome matches or exceeds the pre-operative expectations of the well-counselled family, without any adverse side effects [16]. But, differing outcome evaluation techniques have impeded direct comparison of studies, particularly in the speech and hearing domains till date. Most recently, and Jiang , have investigated this issue and have proposed a rational approach to facilitate future outcome research [17].
By using two different sets of parental questionnaires, Goh and colleagues in Malaysia have been able to document reasonable success of their 25 year long CI program [18]. Their first questionnaire contained methods to assess the children’s usage of CI, their types of education placement and their modes of communication in conjunction with the habilitationist. The second questionnaire was the validated Parent’s Evaluation Of Aural/Oral Performance of Children (PEACH) to evaluate the children’s auditory functionality as reflected by parents. Such a method is more useful especially while comparing the results in a child without inter-observer bias (as provided by the habilitationist versus the parent). This study showed that among the implantees, 97.6% were still using their CI, 69.8% were communicating fully orally and 58.5% were attending mainstream education. This non-biased approach will reduce the heterogeneity of results and precisely evaluate the functional outcomes, more so when dealing with implantees requiring special needs due to multiple handicaps, syndromic associations, cognitive and higher mental disabilities etc. Vlahovic et al. recommend that the evaluation of success, especially in such children should also include subjective indicators of positive life changes, which are not directly associated with hearing alone [19]. This is a future direction for the current study and has been included into the ongoing project.
In summary, all the implantees in the present study, showed significant improvement in listening and communication skills, as documented over time using globally applied outcome measures. The variation in outcome data is largely attributed to social, economical and cultural differences within India. Since all children in India live in a multilingual environment, long term data generated by using appropriate indigenous assessment tools are most essential to measure the speech and language outcomes [12,13]. The future holds on development of such validated indigenous outcome measures for the South Indian population, which will optimally reflect on the performance of implantees while addressing their phonetic, linguistic and socio-cultural ethnicity.
Conclusion
The study has highlighted the success story of auditory verbal habilitation provided in a large implant centre in South India. The cohort studied has shown substantial progress in the various outcome measures after one year of intensive auditory verbal habilitation provided in-house by a dedicated team of skilled professionals. It is of note that more than 90% of these children showed optimal improvement in their communication skills and were able to understand common open-set phrases with nearly 80% of them having developed some connected intelligible speech, which is in keeping with the standard set by other reputed centres across the world. The study reinforces the emphasis on providing intensive auditory verbal habilitation for a minimum of one year to paediatric implantees with continuous monitoring in order to achieve optimal outcomes. Such a transparent and self-appraising implant program will succeed in providing the ideal platform for these deserving children to acquire education and integrate into the society as normal individuals.
References
- Beadle EAR, McKinley DJ, Nikolopoulos TP (2005) Long-term functional outcomes and academic-occupational status in implanted children after 10 to 14 years of cochlear implant use. Otol Neurotol 26: 1152-1160.
- NICE technology appraisal guidance TAG166 (2009) Cochlear implants for children and adults with severe to profound deafness.
- Hehar SS, Nikolopoulos TP (2002) Surgery and functional outcomes in deaf children receiving cochlear implants before age 2 years. Arch Otolaryngol Head Neck Surg 128: 11-14.
- O’Neill C, O’Donoghue GM, Archbold SM (2001) Variations in gains in auditory performance from pediatric cochlear implantation. Otol Neurotol 23: 44-48.
- Allen C, Nikolopoulos TP, Dyar D (2001) Reliability of a rating scale for measuring speech intelligibility after paediatric cochlear implantation. Otol Neurotol 22: 631-633.
- Archbold S, Lutman ME, Marshall DH (1995) Categories of auditory performance. Ann Otol Rhinol Laryngol Suppl 166: 312-314.
- Robbins AM, Renshaw JJ, Berry SW (1991) Evaluating meaningful auditory integration in profoundly hearing-impaired children. Am J Otol 12: 144-150.
- Eisenberg SL, Johnson KC, Martinez AS (2006) Speech recognition at 1-year follow-up in the childhood development after cochlear implantation: Study methods and preliminary findings. Audiol Neurotol 11: 259-268.
- Allen C, Nikolopoulos TP, O’Donoghue GM (1998) Speech intelligibility after cochlear implantation. Am J Otol 19: 742-746.
- Nikolopoulos TP, Archbold SM, O’Donoghue GM (1999) The development of auditory perception in children following cochlear implantation. Int J Pediatr Otorhinolaryngol 49: S189-S191.
- Percy-Smith L (2006) Danske Born Med Cochlear Implant: Undersøgelse af Medvindsfaktorer for Børnenes Hørelse, Talesprog og Trivsel.
- Calmels MN, Saliba I, Wanna G (2004) Speech perception and speech intelligibility in children after cochlear implantation. Int J Pediatr Otorhinolaryngol 68: 347-351.
- Inscoe J (1999) Communication outcomes after paediatric cochlear implantation. Int J Pediatr Otorhinolaryngol 47: 195-200.
- Waltzman SB, Roland JT Jr, Cohen NL (2002) Delayed implantation in congenitally deaf children and adults. Otol Neurotol 23: 333-340.
- Ovesen T, Johansen LV (2009) Post-operative problems and complications in 313 consecutive cochlear implantations. J Laryngol Otol 123: 492-496.
- Black J, Hickson L, Black B (2012) Defining and evaluating success in paediatric cochlear implantation-an exploratory study. Int J Pediatr Otorhinolaryngol 76: 1317-1326.
- Driver S, Jiang D (2017) Paediatric cochlear implantation factors that affect outcomes. Eur J Paediatr Neurol 21: 104-108.
- Goh BS, Fadzilah N, Abdullah A, Othman BF, Umat C (2018) Long-term outcomes of universiti kebangsaan Malaysia cochlear implant program among pediatric implantees. Int J Pediatr Otorhinolaryngol 105: 27-32.
- Vlahovic S, Sindija B (2004) The influence of potentially limiting factors on paediatric outcomes following cochlear implantation. Int J Pediatr Otorhinolaryngol 68: 1167-1174.
Citation: Kumar RS, Kameswaran M (2018) Habilitation Outcomes in Paediatric Cochlear Implantation-A South Indian Experience. Otolaryngol (Sunnyvale) 8: 354. DOI: 10.4172/2161-119X.1000354
Copyright: © 2018 Kumar RS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 4757
- [From(publication date): 0-2018 - Oct 16, 2025]
- Breakdown by view type
- HTML page views: 3831
- PDF downloads: 926