Herbicide Bioavailability Determinant Processes in the Soil
Received: 13-Dec-2018 / Accepted Date: 09-Jan-2019 / Published Date: 14-Jan-2019 DOI: 10.4172/2155-6199.1000458
Abstract
Despite their crop protection benefits, herbicides may produce a wide range of possible environmental implications due to their persistence in the ecosystem. They can enter the soil from direct spraying onto the soil surface, irrigation runoff, or release from the dead vegetation. Microbial degradation is the main mechanisms responsible for the transformation and detoxification of most herbicidal compounds in soil. The microbial availability of herbicides for biodegradation in the soil is primarily determined by factors like adsorption, desorption, biodegradation and non-extractable residue formation. Exploring these processes can lead to better understanding the efficacy and fate of the herbicide in the environment. Here we present a short review of the processes affecting the bioavailability of herbicides in the soil and their subsequent influence on the environmental fate of these compounds.
Keywords: Adsorption; Desorption; Biodegradation; Nonextractable residues
Introduction
Herbicides, which are used to control the growth of weeds, can eventually end up in the soil through different ways. Herbicide's interaction with soil begins when the spray hits the soil surface or when herbicide or its metabolites are released from decomposing plants tissues. Biodegradation mediated by microbial enzymes is the primary route for their metabolism in soils [1]. Whether a herbicide compound can be successfully biodegraded in soil or not is determined by a myriad of factors such as microbial biota in the environment, concentration of the substrate, nutrients, environmental parameters, and so on. Given all other factors being favorable; substrates must be in a form readily available for microorganisms to act upon.
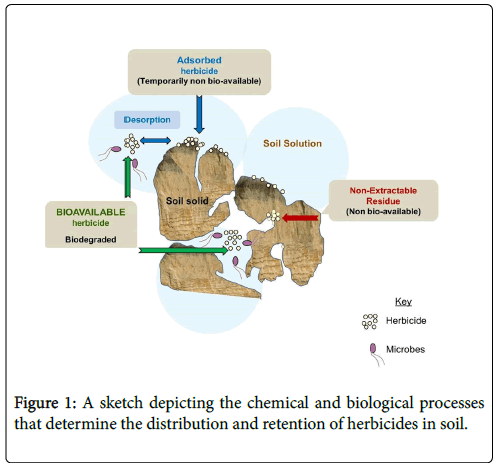
The bioavailability of a herbicide is the measure of its accessibility to microorganisms in the environment [2]. It is a major limiting factor for bioremediation of some polluted soils. Bioavailability frequently accounts for the persistence of soil contaminants that are otherwise biodegradable and thus merits consideration in the risk assessment of soil contaminants. Herbicide distribution and retention in the soil are determined by both chemical and biological processes (Figure 1). These processes are adsorption, desorption, biodegradation, and nonextractable residue formation. While these processes are interdependent on one another, they often occur in parallel and regulate the availability of the herbicide in the environment for use by microorganisms [3]. Understanding such factors is the first step in developing an efficient herbicide program in crop production systems or tailoring a remediation strategy for herbicide contaminated soils. The purpose of this short review is to briefly discuss these processes that influence the fate and bioavailability of herbicides in the soil.
Literature Review
Adsorption: The herbicide binding process onto soil surface
Adsorption is the retention of solutes present in the soil solution by the surfaces of soil colloids via ion exchange. Soil colloids are extremely important for influencing the behavior and potential biodegradation of herbicides since they can not only alter the bioavailability of herbicides but also retain nutrients and microbes. Adsorption plays a critical role in the regulation of herbicide persistence throughout the soil environment by affecting the transport of these compounds in soil. The importance of this process is determined by the physical and chemical properties of the herbicide (i.e., carbon rings, ionic form, etc.) and in part by soil characteristics such as soil texture, mineral composition, organic matter content, specific surface area, and soil solution chemistry. Not a single herbicide property or soil characteristic can predict adsorption of an herbicide in the soil.
It is well known that soil texture refers to the percentage of sand, silt or clay present in any soil. Research has shown that in general, the bioavailability of a compound has been found to be lowest in soils high in organic matter and high clay content and highest in those soils in the light textured (coarse) soils [4]. Soil texture is often directly linked to mineral composition of the soil. Furthermore, the mineral composition of soil colloids influences the adsorption of herbicides. Soil minerals such as clays, iron (Fe), aluminum (Al), and magnesium (Mg), complex carbonaceous materials, as well as organic fractions (humic acid, fulvic acids, etc.) of soil and sediments provide active surfaces or surfaces containing active sites for herbicide adsorption.
Some soil chemical mechanisms that allow herbicides to adsorb to these soil colloids involve the formation of surface complexes, electrostatic and hydrophobic interactions, and ion exchange. For example, cationic organic molecules usually adsorb to cation exchange sites in clay particles, organic matter/humic surfaces or both. Anionic organic molecules may be moderately retained by organic surfaces but poorly adsorbed by clay minerals. Non-ionic, neutral and non-polar organic compounds tend to be adsorbed through hydrophobic interactions associated with organic materials [5]. These exchange reactions are the basis for ions moving into and out of the soil solution phase.
.Although varied depending on the soil type, particle size and aggregation, typically soils contain pore space where the soil solution resides [6]. The solid phase of the soil is formed by primary and secondary minerals (briefly described above) along with the organic matter. These materials provide specific sites for herbicide adsorption [7]. Between the soil colloids is the space forming pores that play a major role in the movement of water, solutes, and air. The herbicide bioavailability in soil depends on the interaction of herbicide between the soil solution and the soil colloid phase. The appropriate term for defining this movement is adsorption equilibrium, which is used to describe the partitioning of any herbicide between the soil solid components and soil solution.
Characterizing the bioavailability of an herbicide requires a thorough understanding of its strength of interaction with particular soil colloids and herbicide concentration in the soil solution. Bioavailability is characterized using adsorption isotherms. An adsorption isotherm shows the relationship between the herbicide’s concentration in the soil solution and the amount adsorbed to the soil surface sites at a constant temperature after equilibrium was reached. The fit of experimental data with theoretical and/or empirical equations for adsorption isotherms is beneficial for providing information on the strength of the soil-herbicide interaction, which will give an idea of the bioavailability of the herbicide in a particular soil type. However, isotherms are only a description of macroscopic data and do not conclusively shed any light on the specific adsorption [8].
Desorption is the reversibility of the adsorption where the soilsurface retained herbicide is released back into the soil solution. Not all herbicides will be desorbed from the soil colloids, and therefore adsorption can be considered irreversible in some cases. Both properties of the herbicide and soil play a role in the process of herbicide adsorption reversibility. In addition, desorption can be estimated for an individual herbicide in continuation with the adsorption isotherm coefficient test where the amount of adsorbed herbicide released back into the soil solution is calculated.
Adsorbed herbicides are assumed less accessible to attached or suspended microorganisms, which preferentially utilize herbicides in the soil solution phase. In this view, the soil-bound herbicide is available for biodegradation only after desorption into the soil solution. The adsorbed fraction remains protected from microbial degradation because of physical sequestration of the herbicide into the soil matrix and/or their chemical stabilization on the soil surface sites [4]. Reduction of herbicide concentration from the soil solution to levels that do not sustain microbial growth, emphasizes the importance of herbicide desorption in the microbial bioavailability of herbicides. Although desorption has been considered a pre-requisite for biodegradation of soil-bound herbicides, there is increasing evidence that adsorbed herbicides may still be degraded by the attached microbial cells [9]. However, considerable work is ahead for researchers to understand the mechanisms and populations intervening in these processes.
Biodegradation: The primary herbicide dissipation route in soil
Biodegradation is the primary attenuation process for herbicides in soil and is principally facilitated through the enzymatic transformations by living microbial cells. Biodegradation is controlled by biotic factors, for instance, microbial activity and several physiochemical processes such as adsorption and desorption, diffusion and dissolution [10]. Generally, herbicide biodegradation can be limited by its bioavailability in soil. As the compounds may be adsorbed to soil solids, physically entrapped in micropores, or simply unevenly distributed through the pore volume of the soil solution. Thus, leading to diffusion limited degradation kinetics in the soil [11,12].
Microorganisms do exceptionally well by utilizing herbicides as a supply for nutrients and energy. Many herbicides have the much-needed carbon and/or nitrogen sources for soil microorganisms [13] to exist. Evidence of microorganisms remarkable range of degradative abilities can be seen in the recycling rather than the accumulation of vast quantities of biomass that have been produced throughout the history of life on earth [14]. This re-utilization of the chemical components of a herbicide by microorganisms is called mineralization [6]. Mineralization results in the complete aerobic or anaerobic degradation of the herbicide compounds to form carbon dioxide, water, and some other inorganic elements.
Mineralization has been used as a sensitive indicator of pesticide and heavy metal bioavailability in the soil. In addition, it is generally accepted as a measure of total soil microbial activity [15]. Conversely, the incomplete transformation of a herbicide by microorganisms that yields no carbon or nutrients and energy is called co-metabolism. In most co-metabolic processes, a co-substrate provides carbon and energy for microbial growth. The herbicide itself is transformed concomitantly without supporting microbial growth. While some herbicides may be partially transformed by co-metabolism, other soil microorganisms may completely mineralize their intermediate metabolites. The utilization of a consortium of microorganisms to enhance the degradation of herbicides has increased recently in bioremediation efforts, mainly owing to their potential for synergistic metabolism [16]. The transformation product formed by one microbe’s metabolism can be utilized by another degrader for efficient biodegradation, thus speeding up the bioremediation process.
The biodegradation rate of the herbicide in the soil can be described in terms of their halflife T1/2. This is the time required for 50% of the compound to degrade or dissipate. In addition to degradation, nonbiological field dissipation of the herbicide includes leaching losses, photodegradation, and volatilization. However, the presence of microorganisms in the soil system is crucial as they can significantly affect the T1/2 of herbicides in soil. Thus, an active soil microbial population is considered a key component of good soil quality [15] and an indirect indicator of biodegradation.
Microbes are expected to be more effective indicators of the soil quality and health than physical and chemical parameters as they can respond immediately to environmental changes. When predicting the potential in situ biodegradation of herbicides, additional factors other than presence of potential degraders and bioavailability must be considered. These include the presence of other contaminants that can compete for adsorption sites and the availability of nutrients and cofactors necessary for the degrader’s growth and activity [17]. Intrinsic soil environmental factors like oxygen concentration, water availability, carbon availability, temperature and pH can also have a great impact on the biodegradation of herbicides in the agroecosystem.
Soil amendments are also among the key factors that influence the biodegradation of herbicides. Various organic amendments and bioprocessed materials including compost, manure, sludge, biochar, etc. have been examined as potential agents for enhancing the degradation of herbicides [18,19]. Upon addition to the soil, these amendments stimulate the indigenous soil microbial activity and consequently enhance the degradation and mineralization of soil-applied herbicides. In some cases, these amendments altered the herbicide bioavailability determinant processes such as adsorption in soil [19]. Furthermore, the possible impacts of soil nutrients on the degradation of herbicides has been studied [18]. The addition of various forms of nitrogen (N), phosphorous (P), sodium (Na) and magnesium (Mg) has significantly increased the microbial transformation of herbicides in soil [20].
Non-Extractable Residue (NER) formation: An irreversible sink for herbicide sequestration
Some compounds may not be adsorbed in the solids but sequestered into soil micropores and nanopores. Microbes that are usually present on surfaces of particulate materials are restricted from access to those compounds because these pores are too small for microbes to penetrate. The primary mechanism for the contaminants to move out of the micropores is through diffusion, but it can be a very long process. In such cases where the herbicides and metabolites in soil have gone aging, they could not be removed from the environmental matrices utilizing extraction techniques [7]. NER represent compounds in the soil which persist in the matrix in the form of the parent substance or its metabolite(s) even after rigorous chemical extraction. NER formation is typically considered a process decreasing the bioavailability of the herbicides. Only a small percentage of the total amount of NER can be released from the soil solid surfaces. Hence, NER formation is often accounted as the irreversible sink in pesticide risk assessment procedures. Typically, strongly bound herbicide molecules on soil particles retard the microbial degradation and lead to its accumulation in soil. The most common explanation for why this occurs is the intact molecule becoming hidden or inaccessible to the microorganisms. Or the compounds may form complexes with humic materials forming a new molecular species. The linkages within the complexes are stable due to covalent bonding between the original chemical and humic polymers. Such complexes are not readily extractable with organic solvents. These irreversibly bound herbicide molecules are considered as the non-extractable fraction. The factors governing the formation of NER are not clear-cut. However, herbicides or metabolites supporting free reactive chemical groups, such as aniline or phenol, tend to create a larger proportion of NER during degradation [21]. Furthermore, if it is the parent compound which is involved in the NER formation, then rapid degradation competes with the NER formation. On the contrary, if it is the metabolite which is involved in NER formation, then rapid degradation may lead to an extensive formation of NER [22]. From an eco-toxicological viewpoint, NER formation can reduce the toxicity of a compound by decreasing its interaction with soil biota [23].
Among the various factors affecting the formation of NER, microbial activity has a direct and significant effect. The accumulation of NER for most herbicides is usually correlated to the soil biological activity and to the amount of organic matter in the soil. The total microbial activity has a direct effect on the NER formation as observed from herbicide incubation experiments. NER formation was shown to be low from incubation experiments involving soil samples taken from deeper depths which usually have low microbial activity [24]. Most environmental factors affecting the microbial bioavailability of herbicide in the soil such as temperature or moisture content are likely to have an influence on the NER formation. Generally, NER formation varies with an increase in soil moisture, and in most cases it increased with soil saturation [25,26].
Discussion And Conclusion
The distribution and retention of herbicides in the soil affect the extent of its availability to microbial degraders. The chemical and biological processes that determine microbial bioavailability of herbicide compounds in soil include adsorption, desorption, biodegradation, and non-extractable residue formation. All these processes are interdependent, occurring in parallel and regulating the availability of the herbicide that can be used by microorganisms in the environment. The knowledge on these bioavailability determinant factors could be applied towards improving the herbicide efficacy or framing a viable strategy for the clean-up of herbicide contaminated soils.
References
- Van-Eerd LL, Hoagland RE, Zablotowicz RM, Hall JC (2003) Pesticide metabolism in plants and microorganisms. Weed Sci 51: 472-495.
- Wu XM, Li M, Long YH, Liu RX, Yu YL, et al. (2011) Effects of adsorption on degradation and bioavailability of metolachlor in soil. J Soil Sci Plant Nutr 11: 83-97.
- Cheng HH (1990) Pesticides in the soil environment: Processes, impacts, and modeling. Soil Science Society of America, Madison, Wisconsin, USA.
- Ainsworth CC, Frederickson JK, Smith SC (1993) Effect of sorption on the degradation of aromatic acids and bases: Sorption and degradation of pesticides and organic chemicals in soil. Soil Science Society of America, Madison, Wisconsin, USA, pp: 125-144.
- Alexander M (1999) Biodegradation and bioremediation. Academic Press, New York, USA.
- Zabaloy MC, Zanini GP, Bianchinotti V, Gomez MA, Garland JL (2011) Herbicides in the soil environment: Linkage between bioavailability and microbial ecology, Herbicides, Theory and applications. In Tech, London, UK, pp: 161-192.
- Dixon JB, Weed SB (1990) Minerals in soil environments. Soil Science Society of America, Madison, WI, USA.
- Sparks D (2003) Environmental soil chemistry. Academic Press, California, USA.
- Park JH, Kay DH, Zhao XD, Boyd SA, Voice TC (2001) Kinetic modelling of bioavailability for sorbed-phase 2,4–dichlorophenoxyacetic acid. J Environ Qual 30: 1523-1527.
- Chen H, He X, Rong X, Chen W, Cai P, et al. (2009) Adsorption and biodegradation of carbaryl on montmorillonite, kaolinite and goethite. Appl Clay Sci 46: 102-108.
- Sims GK, Wolt JD, Lehmann RG (1992) Bioavailability of sorbed pesticides and other xenobiotic molecules. Proceedings of the International Symposium on Environmental Aspects of Pesticide Microbiology, Sigtuna, Sweden.
- Sims GK, Taylorâ€Lovell S, Tarr G, Maskel S (2009) Role of sorption and degradation in the herbicidal function of isoxaflutole. Pest Manag Sci 65: 805-810.
- Qiu Y, Pang H, Zhou Z, Zhang P, Feng Y, et al. (2009) Competitive biodegradation of dichlobenil and atrazine coexisting in soil amended with a char and citrate. Environ Pollut 11: 2964-2969.
- Dua M, Singh A, Sethunathan N, Johri A (2002) Biotechnology and bioremediation: successes and limitations. Appl Microbiol Biotechnol 59: 143-152.
- Anderson TH (2003) Microbial eco-physiological indicators to assess soil quality. Agric Ecosyst Environ 98: 285-293.
- Villaverde J, Rubio-Bellido M, Lara-Moreno A, Merchan F, Morillo E (2018) Combined use of microbial consortia isolated from different agricultural soils and cyclodextrin as a bioremediation technique for herbicide contaminated soils. Chemosphere 193: 118-125.
- Haws NW, Ball WP, Bouwer EJ (2006) Modeling and interpreting bioavailability of organic contaminant mixtures in subsurface environments. J Contam Hydrol 82: 255-292.
- Kanissery RG, Sims GK (2011) Biostimulation for the enhanced degradation of herbicides in soil. Appl Environ Soil Sci 2011: 843450.
- He L, Fan S, Müller K, Hu G, Huang H, et al. (2016) Biochar reduces the bioavailability of di-(2-ethylhexyl) phthalate in soil. Chemosphere 142: 24-27.
- De Lipthay JR, Sørensen SR, Aamand J (2007) Effect of herbicide concentration and organic and inorganic nutrient amendment on the mineralization of mecoprop, 2, 4-D and 2, 4, 5-T in soil and aquifer samples. Env Pollu 148: 83-93.
- Bollag JM, Liu SY, Minard RD (1980) Cross-coupling of phenolic humus constituents and 2, 4-dichlorophenol. Soil Sci Soc Am J 44: 52-56.
- Barriuso E, Benoit P, Dubus IG (2008) Formation of pesticide nonextractable (bound) residues in soil: Magnitude, controlling factors and reversibility. Environ Sci Technol 42: 1845-1854.
- Gevao B, Semple KT, Jones KC (2000) Adsorption of atrazine, simazine and glyphosate in soils of Gnangara Mound, Western Australia. Aust J Soil Res 34: 599-607.
- Schiavon M (1988) Studies of the movement and the formation of bound residues of atrazine, of its chlorinated derivatives, and of hydroxyatrazine in soil using 14C ringlabeled compounds under outdoor conditions. Ecotoxicol Environ Saf 15: 55-61.
- Rice PJ, Anderson TA, Coats JR (2002) Degradation and persistence of metolachlor in soil: Effects of concentration, soil moisture, soil depth, and sterilization. Environ Toxicol Chem 21: 2640-2648.
- Kruger EL, Somasundaram L, Coats JR, Kanwar RS (1993) Persistence and degradation of [14C] atrazine and [14C] deisopropylatrazine as affected by soil depth and moisture conditions. Environ Toxico Chem 12: 1959-1967.
Citation: Kanissery R, Gairhe B, McAvoy C, Sims G (2019) Herbicide Bioavailability Determinant Processes in the Soil. J Bioremediat Biodegrad 10:458. DOI: 10.4172/2155-6199.1000458
Copyright: © 2019 Kanissery R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 5336
- [From(publication date): 0-2019 - Dec 17, 2025]
- Breakdown by view type
- HTML page views: 4349
- PDF downloads: 987