Research Article Open Access
In vitro Evaluation of Centratherum anthelminticum Seeds for Antinephrolithiatic Activity
| Varsha J. Galani* and Rital R. Panchal | |
| M.Pharm (Pharmacology), A. R. College of Pharmacy & G. H. Patel Institute of Pharmacy, Gujarat, India | |
| Corresponding Author : | Varsha J. Galani, M. Pharmacy, Ph.D Department of Pharmacology A. R. College of Pharmacy & G. H. Patel Institute of Pharmacy 388120-Vallabh Vidyanagar, Gujarat, India Tel: +91-9429161203 Fax: +91-02692-230788 E-mail: vrp173@yahoo.com |
| Received January 04, 2014; Accepted January 27, 2014; Published January 29, 2014 | |
| Citation: Galani VJ, Panchal RR (2014) In vitro Evaluation of Centratherum anthelminticum Seeds for Antinephrolithiatic Activity. J Homeop Ayurv Med 3:145. doi: 10.4172/2167-1206.1000145 | |
| Copyright: © 2014 Galani VJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Traditional Medicine & Clinical Naturopathy
Abstract
Antinephrolithiatic activity of 70% methanolic extract of Centratherum anthelminticum seeds (CAE) was evaluated in vitro on nucleation and aggregation of calcium oxalate crystallization. Calcium oxalate crystallization was induced by the addition of 0.01 M sodium oxalate solution in synthetic urine. The effect of CAE (100, 200, 300, 400, 600, 800 and 1000 μg/ml) was studied by the measurement of turbidity in presence or absence of extract at 620 nm of a spectrophotometer. The rates of nucleation and aggregation were evaluated by comparing the turbidity of a control system with that of one exposed to the extract. Crystals in the urine were also analysed by light microscopy. From photomicrograph, it is confirmed that CAE inhibited the nucleation of calcium oxalate crystals, decreasing their number
and size. Also percentage inhibition of crystals aggregation increased as the concentration of CAE increased. The results of the present study indicated that 70% methanolic extract of C. anthelminticum seeds has the higher capacity to inhibit the crystal formation and aggregation. These suggested possible antinephrolithiatic activity of C. anthelminticum seeds against calcium oxalate stones.
| Keywords |
| Anti- nephrolithiatic activity; Centratherum anthelminticum (L) kuntze; Calcium oxalate |
| Introduction |
| Urolithiasis denotes presence of one or more stone in any location within the urinary tract, is one of the oldest and wide spread diseases known to man [1]. It is a serious, debilitating problem in all societies throughout the world, affecting approximately. 12% of the population and men are three times more prone than women [2]. It is more prevalent between the ages of 20 and 40 in both sexes [3]. The overall probability of forming stones differs in various parts of the world, and is estimated at 1–5% in Asia, 5–9% in Europe, and 13% in North America. The recurrence rate of renal stones is approximately 75% in a 20-year span [4]. Etiology is multifactorial and is strongly related to dietary lifestyle habits or practices [5]. The most common (more than 80%) renal stones are calculi of calcium oxalate crystals followed by uric acid, struvite cystine and other stones [6,7]. |
| The crystallization of the calcium oxalate begins with increased urinary supersaturation, with the subsequent formation of the solid crystalline particles within the urinary tract. This is followed by nucleation, by which stone-forming salts in supersaturated urinary solution coalesce in to clusters that then increase in size by the addition of new constituents [8]. These crystals then grow and aggregate with other crystals in solution, and are ultimately retained and accumulated in the kidney [9]. Renal injury promotes crystal retention and the development of a stone nidus on the renal papillary surface, and further supports crystal nucleation at lower supersaturation levels [10]. Therefore, levels of urinary supersaturation correlate with the type of stone formed, and reducing supersaturation is effective in preventing stone recurrence. Therefore, if this progression of crystallization can be prevented, then lithiasis could also be prevented. |
| The stone formation requires supersaturated urine which also depends on urinary pH, ionic strength, solute concentration and complexions. Various substances in the body have an effect on one or more of the above processes, thereby influencing a person’s ability to promote or prevent stone formation [8]. |
| Management of stone disease depends on the size and location of the stones. Stones larger than 5 mm or stones that fail to pass through should be treated by some interventional procedures such as extracorporeal shock wave lithotripsy (ESWL), ureteroscopy (URS), or percutaneous nephrolithotomy (PNL) [11]. The recent treatment procedures are very costly for common man as well as recurrence of kidney stone and numbers of side effects are associated with these procedures [12]. Hence, search for new antinephrolithiatic drugs from natural sources assumed greater importance as herbal drugs are cost effective and they confer lesser side effects. |
| Traditional herbal medicines provide many opportunities for the development of potential therapeutic drugs, in the form of either extracts alone, in combination with other herbs, or in the form of phytochemical compounds isolated from them. There are several reports related to anti-crystallization compounds extracted from medicinal plants. |
| Centratherum anthelminticum (L.) Kuntze (compositae), commonly known as Kaligiri. It is highly reputed in Hindu medicine as remedy for leucoderma and other skin diseases. The seeds have a hot sharp taste, acrid, astringent to the bowels, anthelmintic and cure ulcers. The seeds are used as purgative, for asthma, kidney troubles and hiccough, applied in inflammatory swelling, remove blood from liver, good for sores and itching of the eyes. In Punjab, it is considered as antipyretic. The seeds are also credited with tonic, stomachic, and diuretic properties [13,14]. Different organic solvent and aqueous extracts of these seeds were scientifically evaluated for antifilarial, antibacterial, larvicidal, antiviral, antifungal, anticancer, anthelmintic, antidiabetic, antioxidant, analgesic, antipyretic, anti-inflammatory, diuretic, wound healing activities [15]. There is no scientific evidence regarding nephroprotective action of this plant. Based on that, aim of the present study is to evaluate nephroprotective activity of Centratherum anthelminticum using in vitro experimental models of nephrolithiasis. |
| Materials and Methods |
| Preparation of extract |
| Dried seeds of Centratherum anthelminticum were procured from Anand Agriculture University, Anand. Plant material was authentified by Dr. Jina Patel, Department of Botany, Gujarat University, Ahmedabad. The voucher specimen (Authentication reference number: RRP/CA-1/7/ARGH -11-13) was deposited at the pharmacognosy department of our institute. The seeds were air-dried and ground to fine powder. About 0.5 kg powdered sample was defatted with petroleum ether (40-60°C). The remaining part was extracted with (70:30) methanol and water by cold maceration for 4 days with frequent shaking. (Yield -11.49% w/w). Hydromethanolic extract after evaporating the solvent, was dried under vacuum and stored in an airtight container at 4°C. The dried extract was dissolved in distilled water and used for further study. |
| Preliminary phytochemical screening |
| The qualitative chemical investigation of hydroalcoholic extract was carried out to check the presence of various phytoconstituents [16]. |
| Preparation of synthetic urine: The artificial urine was prepared according to the method of Burns and Finlayson [17] and had the following composition: sodium chloride 105.5 mmol/l, sodium phosphate 32.3 mmol/l, sodium citrate 3.21 mmol/l, magnesium sulfate 3.85 mmol/l, sodium sulfate 16.95 mmol/l, potassium chloride 63.7 mmol/l, calcium chloride 4.5 mmol/l, sodium oxalate 0.32 mmol/l, ammonium hydroxide 17.9 mmol/l, and ammonium chloride 0.0028 mmol/l. The synthetic urine was prepared fresh each day and pH adjusted to 6.0. |
| Experimental protocol: The classical model for the study of oxalate crystallization was chosen because of its simplicity and satisfactory reproducibility. According method reported by Sasikala et al. [18] which involves crystallization without inhibitors and with it, in order to assess the inhibiting capacity of test material used was suitably modified for the study. |
| Nucleation assay: The inhibitory activity of the extracts on the nucleation of calcium oxalate crystals was determined by a spectrophotometric assay [19]. Solution of calcium chloride and sodium oxalate were prepared at the final concentrations of 5 mmol/l and 7.5 mmol/l respectively in a buffer containing Tris 0.05 mol/l and NaCl 0.15 mol/l at pH 6.5. 950 μl of calcium chloride solution mixed with 100 μl of herb extracts at the different concentrations (100 μg/ml to 1000 μg/ml). Crystallization was started by adding 950 μl of sodium oxalate solution. The temperature was maintained at 37ºC. The rate of nucleation was determined by comparing the induction time of crystals (time of appearance of crystals that reached a critical size and thus became optically detectable) in the presence of the extract and that of the control with no extract. The absorbance (optical density) was recorded at 620 nm using spectrophotometer (Shimadzu). The growth of crystals was expected due to the following reaction: |
| CaCl2 +Na2C2O4→ CaC2O4+ 2NaCl |
| Aggregation assay: The method used was similar to that described by Atmani and Khan [20] with some minor modifications. Calcium oxalate crystals were prepared by mixing calcium chloride and sodium oxalate at 50 mmol/l. Both solutions were equilibrated to 60°C in a water bath for 1 hour and then cooled to 37°C overnight. The crystals were harvested by centrifugation and then evaporated at 37°C. Calcium oxalate crystals were used at a final concentration of 0.8 mg/ml, buffered with Tris 0.05 mol/l and NaCl 0.15 mol/l at pH 6.5. The absorbance (optical density) was recorded at 620 nm spectrophotometer (Shimadzu). Experiments were conducted at 37°C in the absence or presence of the plant extract. The percentage aggregation inhibition rate (Ir) was then calculated by comparing the turbidity in the presence of the extract with that obtained in the control using following formula: |
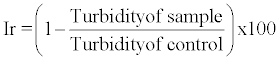 |
| Results |
| The in vitro inhibitory effect of CAE on various phases of calcium oxalate crystallization was determined by the time course of turbidity measured in synthetic urine at extract concentrations of 100, 200, 400, 600, 800 and 1000 μg/ml. Incubating the metastable solutions of Ca+2 and oxalate without extract resulted in the formation of bipyramid calcium oxalate crystals (Figure 1A). The respective crystals, observed under the light microscope (100×), in solutions incubated with CAE at 100 – 1000 μg/ml are shown in Figures 1B-G. CAE also caused a morphological change in calcium oxalate crystals, which was not fully grown as bipyramid calcium oxalate crystals that were inhibited in nucleation phase. The optical density decreased with the increase in concentration of CAE indicating that decreased the nucleation of calcium oxalate particles (Table 1). The optical density was highest (0.931) of positive control i.e. in the absence of herb extract and it was lowest (0.654) at the highest concentration of CAE (1000 μg/ ml). The crystals formed in the presence of CAE were less than that in the control, showing that crystals were less aggregated. As shown in Table 2, the percent inhibited aggregation associated with the CAE at concentration of 100 μg/ml was found to be 57.96 while percent was maximum i.e. 70.04 at highest concentration of CAE (1000 μg/ml). |
| Discussion |
| Kidney stone function is a complex process that results from a succession of several physico-chemical events including supersaturation, nucleation, growth, aggregation and retention within renal tubules [21]. Thus if supersaturation or later steps in crystallization can be prevented, then lithiasis should be avoided. Nucleation is the formation of a solid crystal phase in a solution. It is an essential step in renal stone formation [22]. The main findings of the present study were that CAE inhibited the crystallization by inhibiting nucleation of calcium oxalate in solution; less and smaller particles were formed with increasing concentrations of the CAE. The results of the nucleation assay confirmed that the extract contained nucleation-preventing agents. The limiting factors in stone formation could be those processes that affect crystal growth, because particles may become large enough to occlude the urinary tract, leading to stone formation [23]. The herb extracts may contain substances that inhibit the growth of calcium oxalate crystals. This property of plants may be important in preventing the growth of kidney stone. Aggregation may be an important factor in the genesis of stones [24]. Crystal aggregation is the most critical step, as it occurs very rapidly and has a considerable effect on particle size, and aggregated crystals are commonly found in urine and renal stones [25]. Recurrent calcium stone formers excrete clusters of crystals in the urine, caused by aggregation, also named agglomeration, whereas urine from normal people contains mainly single crystals [24]. Again, percentage inhibition of crystals aggregation increased as the concentration of CAE increased indicating antinephrolithiatic activity. The efficacy of most herbal remedies is attributed to various active principles, in combination. Results of phytochemical screening showed the presence of carbohydrates, proteins, saponins, flavanoids, tannins and polyphenols in the seeds. Saponins are known to have anti-crystallisation properties by disaggregating the suspension of mucoproteins, the promoters of crystallization [26]. Antiurolithiatic activities also have been attributed to triterpenes, lupeol [27] and polyphenolic compound like quercetin [28]. It is therefore probable that the components that are present in abundance in the extract might exert their action directly on the calcium oxalate crystallization. |
| In conclusion, 70% methanolic extract of Centratherum anthelminticum seeds (CAE) have inhibitory effect on the nucleation and aggregation of calcium oxalate crystallization in vitro. Thus, this scientific evidence may rationalize the traditional use of Centratherum anthelminticum seeds for the treatment of nephrolithiasis. However, a detailed preclinical and clinical study is required to establish the use of plant as antinephrolithiatic agent. |
References
|
Tables and Figures at a glance
| Table 1 | Table 2 |
Figures at a glance
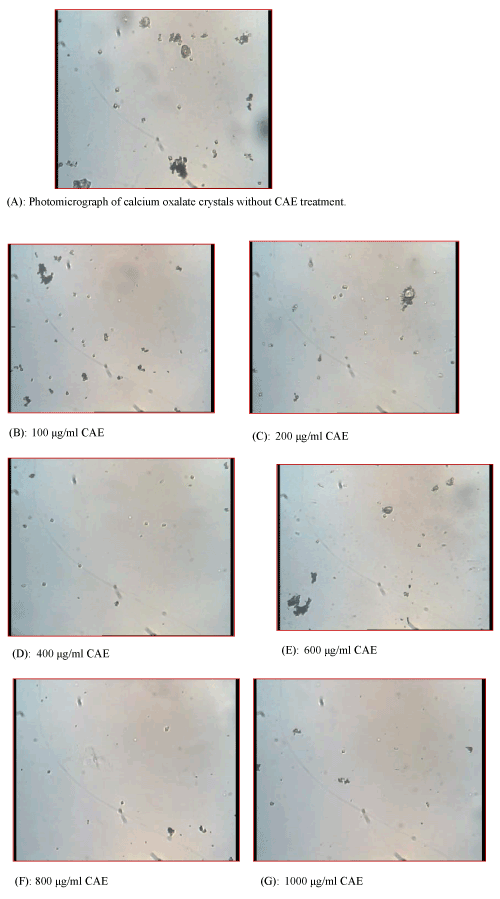 |
| Figure 1 |
Relevant Topics
- Acupuncture Therapy
- Advances in Naturopathic Treatment
- African Traditional Medicine
- Australian Traditional Medicine
- Chinese Acupuncture
- Chinese Medicine
- Clinical Naturopathic Medicine
- Clinical Naturopathy
- Herbal Medicines
- Holistic Cancer Treatment
- Holistic health
- Holistic Nutrition
- Homeopathic Medicine
- Homeopathic Remedies
- Japanese Traditional Medicine
- Korean Traditional Medicine
- Natural Remedies
- Naturopathic Medicine
- Naturopathic Practioner Communications
- Naturopathy
- Naturopathy Clinic Management
- Traditional Asian Medicine
- Traditional medicine
- Traditional Plant Medicine
- UK naturopathy
Recommended Journals
Article Tools
Article Usage
- Total views: 14102
- [From(publication date):
February-2014 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 9423
- PDF downloads : 4679
