Research Article Open Access
Is there Any Relationship Between Diabetic Gastroparesis and Mucosal Ä°njury?
Ozlem Ozer Cakir*Department of Gastroenterology and Hepatology, Konya Education and Research Hospital, Konya, Turkey
- *Corresponding Author:
- Ozlem Ozer Cakir
Department of Gastroenterology and Hepatology
Konya Education and Research Hospital, Konya, Turkey
Tel: +903322214107
E-mail: mailto:tansozlem@yahoo.com
Received date: June 4, 2016; Accepted date: August 2, 2016; Published date: August 9, 2016
Citation: Cakir OO (2016) Is there Any Relationship Between Diabetic Gastroparesis and Mucosal İnjury? J Gastrointest Dig Syst 6:461. doi:10.4172/2161-069X.1000461
Copyright: © 2016 Cakir OO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License; which permits unrestricted use; distribution; and reproduction in any medium; provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objective: We aimed to show the relationship between diabetic gastroparesis and mucosal injury by comparing the findings of upper gastrointestinal system endoscopy between patients with and without diabetic gastroparesis. Material and method: Upper gastrointestinal system endoscopy and gastric emptying scintigraphy were performed on the same day in 51 volunteer type 2 Diabetes Mellitus patients. 4-hour gastric emptying scintigraphy with 500 mCi Technetium (Tc) 99 m diethylene triamine pentaacetic acid (DTPA) in semisolid foods for the determination of gastroparesis was performed in the patients. Upper gastrointestinal system endoscopy was compared between patients with and without gastroparesis by gastric emptying scintigraphy. Results: The mean ages of patients with diabetic gastroparesis (20 Female (F)/10 Male (M)) and without (12F/9M) diabetic gastroparesis were 57.04 ± 9.41 and 53.35 ± 6.98 years, respectively. There was no statistically significant difference between groups in terms of age and gender distribution (p=0.192, p=0.489, respectively). There were no significant differences with respect to age, gender, body mass index (BMI), fasting blood glucose (FBG) and Hb A1c between groups (p=0.192, p=0.489, p=0.529, p=0.653, and p=0.707, respectively). Erosive (E) findings were more frequent in patients with (22 E/8 Nonerosive (NE)) than without (1 E/20 NE) diabetic gastroparesis, and the difference was statistically significant (p<0.05). Conclusion: Erosive findings were significantly more frequent in patients with than in those without diabetic gastroparesis. This also suggested that mucosal injury results from motility disorder.
Keywords
Diabetic gastroparesis; Endoscopy; Gastric emptying scintigraphy; Mucosal injury
Introduction
Gastroparesis is a disorder characterized by delayed gastric emptying in the absence of mechanical obstruction of the stomach [1]. It was shown in several studies that, diabetes mellitus (DM) accounted for almost one third of gastroparesis cases [2-4]. Symptoms of gastroparesis usually include early satiety, nausea, vomiting, bloating, and upper abdominal pain. Diabetic gastroparesis can result in nutritional compromise, impaired glucose control and a poorer quality of life, independent of other factors such as age, tobacco and alcohol use, or type of diabetes [4-6]. Diabetic gastroparesis is a clinical syndrome that occurs in both type 1 and type 2 diabetes. It is associated with considerable morbidity among these patients and with the resultant economic burden on the health care system [6].
According to the studies, the pathogenesis of gastroparesis is still not well understood but involves abnormalities in multiple interacting cell types including the extrinsic nervous system, enteric nervous system, interstitial cells of Cajal (ICCs), smooth muscles and immune cells. The primary diagnostic test is gastric scintigraphy, although other modalities such as breath test, capsule, ultrasound, magnetic resonance imaging (MRI), and single-photon emission computerized tomography (SPECT) imaging show promise as alternative diagnostic modalities [7].
We aimed to compare the findings of upper gastrointestinal system endoscopy between patients with and without diabetic gastroparesis. To the best of our knowledge, no similar prospective randomized study has been published previously in the literature.
Material and Method
The study was approved by the local ethics committee and all subjects provided written informed consent. A total of 51 type 2 DM patients consecutively, who were agreed to be included in our study and admitted to Endocrinology and Metabolism polyclinic for glucose monitorisation, were enrolled in this study from December 2011 to February 2012.
Inclusion criteria were as follows: not usage of drugs that affect gastric motility or that are gastroprotective, absence of gastric outlet obstruction, absence of previous stomach and/or bowel surgery, absence of ileus and/or motility disorder, absence of hypothyroidism and/or psychological distress and absence of pregnancy or breastfeeding. Patients not fulfilling one of the above-mentioned criteria were excluded from the study. Each patient determined suitable for inclusion in the study underwent a physical examination, and detailed medical histories were recorded. The patients did not mention any dispeptic complaints at first, but when we questioned symptoms of gastroparesis(early satiety, nausea, vomiting, bloating, and upper abdominal pain) and reflux (heartburn, pyrosis) in voluntary patients, it was shown presence of some dispeptic complaints in these patients. Information including sex, age, height, body weight, body mass index (BMI), systolic and diastolic blood pressure, triglyceride (TG) level, low-density lipoprotein (LDL), hemoglobin (Hb)A1c, post-prandial glucose, total cholesterol (T-chol), high-density lipoprotein (HDL), serum creatinine and fasting blood glucose (FBG) values were collected for each patient. The height (cm) and weight (kg) of the patients were measured and BMI (kg/m²) was calculated. Blood pressure was measured in all patients with conventional sphygmomanometer after 10 minutes of rest; two measurements from both arms were taken with a 15 minute interval, and the average of these values was recorded. Gastric emptying scintigraphy was performed in 51 volunteer type 2 DM patients by a single nuclear medicine physician. Normal value of 4-hour gastric emptying scintigraphy with 500 mCi technetium (Tc) 99 m diethylene triamine pentaacetic acid (DTPA) in semisolid foods was T1/2: 55 ± 15 minutes; any higher values were evaluated as gastroparesis [8,9]. Patients were divided into two groups as with or without gastroparesis. Upper gastrointestinal system endoscopy was performed on the same day in 51 volunteer type 2 DM patients by a single gastroenterolog using conventional gastroscope. Upper gastrointestinal system endoscopic findings were compared in the two groups by gastric emptying scintigraphy. Endoscopic biopsies were taken from pre-pyloric and plica angularis region for evaluation of Helicobacter pylori (HP) based on histology. These biopsies were evaluated by a single patholog. Erosive esophagitis was classified by Los Angeles classification system [10]. Gastritis and ulcers were classified by Sydney and Forrest classification system, respectively [11,12]. Upper gastrointestinal system endoscopic findings and HP status were compared in patients with and without gastroparesis by gastric emptying scintigraphy. The findings of upper gastrointestinal system endoscopy were divided into two groups as erosive or non-erosive findings. Erosive findings were evaluated as erosive esophagitis, erosive gastritis, erosive duodenitis, and stomach and/or duodenal ulcer. Non-erosive findings were evaluated as normal endoscopy, erythematous gastritis and alkaline reflux gastropathy (without mucosal injury).
Biochemical parameters
Venous blood samples were taken after 12 hours of fasting and then at 2 hours post-prandial. Biochemical assays were done in ABBOTTARCHİTECT C16000 devices. Glucose, T-chol, TG, HDL, and creatinine were assayed with original kits (GAGO-20 SIGMA, MAK043 SIGMA, TR 0100 SIGMA, L 2014 SIGMA), respectively, using enzymatic UV (Hexokinase), enzymatic colorimetric (cholestrooxidase), enzymatic colorimetric (Glycerol-P oxidase), enzymatic colorimetric, and colorimetric (kinetic alkaline picrate) methods. HbA1C was assayed with original kit (794-0412 SIGMA) using High Performance Liquid Chromatography (HPLC) method in TOSOH G8 device. LDL-chol was calculated from T-chol, HDL-chol and TG levels using Friedewald formula [LDL-chol=(T-chol) - (HDL-chol) - (TG/5)].
Gastric emptying scintigraphy
After fasted overnight, Test Meal consisting of egg substitue (equal to 2 large egg), two slices bread, strawberry jam (30 gm), 120 ml water with 500 mCi Tc-99m DTPA ingested within 10-15 min,and then gastric empting time was performed on supine position by 1 min images at time 0, 1, 2, and 4 hrs and geometric mean attenuation correction. Gamma camera images were recorded in 4 hours in Siemens Scintiview SP device, after patients were given semisolid foods with Tc 99 m DTPA. Gastric emptying time-activity curves were obtained, and gastric emptying time was calculated by a single blinded nucleer medicine specialist. Normal value of 4-hour gastric emptying scintigraphy was T1/2: 55 ± 15 minutes.
Upper gastrointestinal system endoscopy: Upper gastrointestinal system endoscopy (Esophago gastro duodenoscopy (EGD)) was performed in Fujinon device by a single blinded endoscopist.
Statistical analysis
Data entry and analysis were performed using the Statistical Package for the Social Sciences (SPSS) for Windows version 17.0 program. Chi-Square and Mann-Whitney-U tests were used for comparisons of means of the groups. Results having a p value lower than 0.05 were accepted as significant.
Results
The mean ages of patients with (20 F/10 M) and without (12F/9M) diabetic gastroparesis were 57.04 ± 9.41 and 53.35 ± 6.98 years, respectively. There were no significant differences with respect to age, gender, BMI, FBG, and HbA1c between groups (p= 0.192, p=0.489, p=0.529, p=0.653, and p=0.707, respectively) (Table 1).
| Demographic and clinical features | Patients without diabetic gastroparesis | Patients with diabetic gastroparesis | P |
|---|---|---|---|
| Gender (Female (F)/ Male (M)) (%) | 12/9 (57.1%/42.9%) | 20/10 (66.7%/33.3%) | 0.489 |
| Age (years) | 53.35 | 57.04 | 0.192 |
| Duration of diabetes mellitus (day) | 233.5 ± 25.3 | 265.8 ± 12.2 | 0.916 |
| SBP (mmHg) | 131 ± 4 | 128 ± 3 | 0.422 |
| DBP (mmHg) | 84.37 ± 2.57 | 80 ± 1 | 0.114 |
| BMI (kg/m2) | 32.25 | 31.48 | 0.529 |
| FBG (mg/dl) | 162 ± 17 | 159 ± 14 | 0.653 |
| HbA1c (%) | 8.1 ± 0.5 | 7.8 ± 1.5 | 0.707 |
| PPG (mg/dl) | 235 ± 25 | 228 ± 13 | 0.582 |
| Cre (mg/dl) | 0.73 ± 0.05 | 0.71 ± 0.03 | 0.922 |
| TG (mg/dl) | 140 ± 13 | 169 ± 17 | 0.33 |
| T-Chol (mg/dl) | 192 ± 9 | 184 ± 7 | 0.582 |
| HDL (mg/dl) | 44 ± 2 | 40 ± 2 | 0.174 |
| LDL (mg/dl) | 118 ± 9 | 111 ± 5 | 0.77 |
| HP status (positve/negative) | 3/18 (14.2%/85.7%) | 5/25 (16.6%/83.3%) | 0.818 |
| Scintigraphy | 21/0 | 0/30 | p<0.05 |
| (Normal/ Delayed) | (100%/0%) | (0%/100%) | |
| Endoscopy | 1/20 | 22/8 | p<0.05 |
| (Erosive/ Non-Erosive) | (4.8%/95.2%) | (73.3%/26.7%) |
Table 1: Demographic and clinical features. SBP: Systolic blood pressure, DBP: Diastolic blood pressure, FBG: Fasting Blood Glucose, BMI: Body mass index, Hb A1c: Hemoglobin A1c, PPG: Post prandial glucose, TG: Triglyceride, T-chol: Total cholesterol, HDL: High density lipoprotein, LDL: Low density lipoprotein, Cre: Creatinine, HP: Helicobacter pylori.
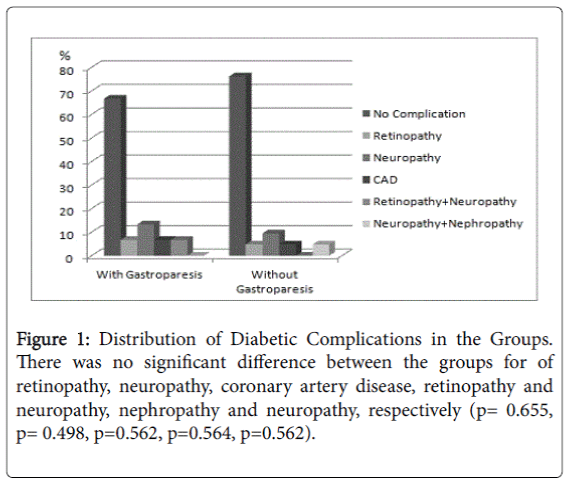
Regarding diabetic complications, nephropathy and neuropathy (n: 1 patient), retinopathy (n: 1), coronary artery disease (n: 1) and neuropathy (n: 2) were present in the patients without gastroparesis, while retinopathy (n: 2), neuropathy (n: 4), coronary artery disease (n: 2), and retinopathy and neuropathy (n: 2) were noted in patients with diabetic gastroparesis. There was no significant difference between the groups for retinopathy, neuropathy, coronary artery disease, retinopathy and neuropathy, nephropathy and neuropathy, respectively (p=0.655, p=0.498, p=0.562, p=0.564, p=0.562) (Figure 1). There was no significant difference between the groups for presence of retinopathy, neuropathy, coronary artery disease, retinopathy and neuropathy, nephropathy and neuropathy, respectively (p=0.655, p=0.498, p=0.562, p=0.564, p=0.562)). There was no statistically significant difference between the groups in terms of education level (p=0.082). None of the patients consumed alcohol. There was no statistically significant difference between the groups regarding smoking habit (p=0.194), familial history of DM (p=0.602) (Figure 2).
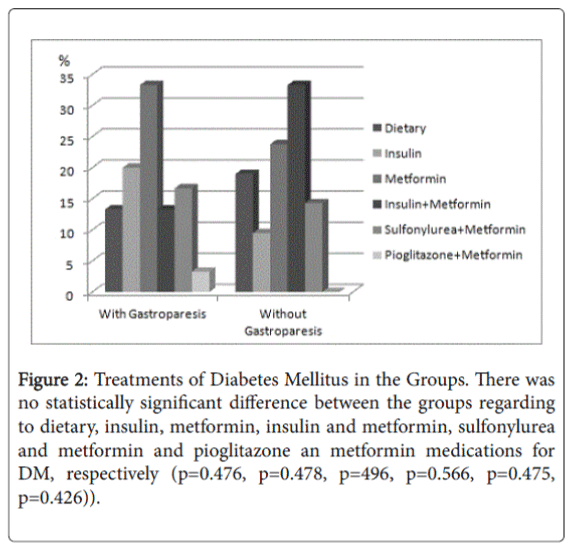
Figure 2: Treatments of Diabetes Mellitus in the Groups. There was no statistically significant difference between the groups regarding to dietary, insulin, metformin, insulin and metformin, sulfonylurea and metformin and pioglitazone an metformin medications for DM, respectively (p=0.476, p=0.478, p=496, p=0.566, p=0.475, p=0.426)).
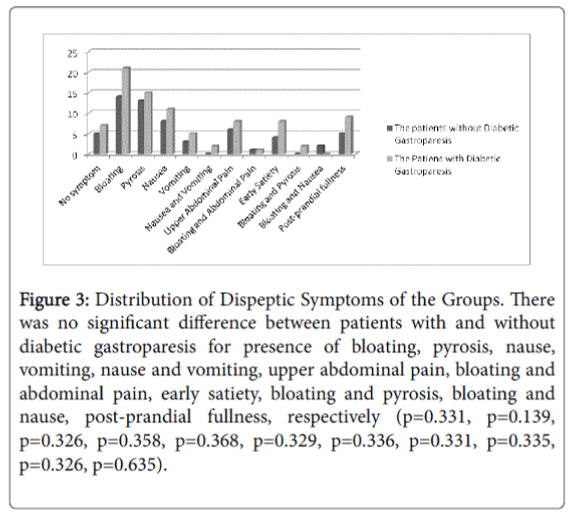
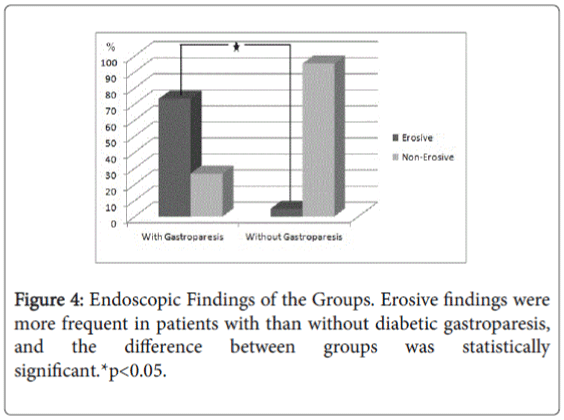
There was no statistically significant difference between the groups regarding to dietary, insulin, metformin, insulin and metformin, sulfonylurea and metformin and pioglitazone an metformin medications for DM, respectively (p=0.476, p=0.478, p=496, p=0.566, p=0.475, p=0.426)). There was no significant difference between patients with diabetic gastroparesis and patients without diabetic gastroparesis for presence of dispeptic complaints (p:0368). Nause (n: 8/11, 38.1%/36.7%), vomiting (n:3/5, 14.3%/16.7%), bloating (n:14/21, 66.7%/70%), early satiety (n:4/8, 19%/26.7%), upper abdominal pain (n:6/8, 28.6%/26.7%), post-prandial fullness (n:5/9, 23.8%/30%) and pyrosis (n:13/15, 61.9%/50%) were seen in patients without diabetic gastroparesis/ with gastroparesis (Figure 3). There was no significant difference between patients with and without diabetic gastroparesis for presence of bloating, pyrosis, nause, vomiting, nause and vomiting, upper abdominal pain, bloating and abdominal pain, early satiety, bloating and pyrosis, bloating and nause, post-prandial fullness, respectively (p=0.331, p=0.139, p=0.326, p=0.358, p=0.368, p=0.329, p=0.336, p=0.331, p=0.335, p=0.326, p=0.635). The mean values of 4- hour gastric emptying scintigraphy of patients with and without diabetic gastroparesis were T1/2: 49.4 ± 11.3 and T1/2: 118.8 ± 36.8 minutes, respectively. Regarding endoscopic findings, normal endoscopy (n:5), bile reflux gastropathy (n:5), erythematous gastropathy (n:10) were noted in patients without diabetic gastroparesis, and ulcers in the antrum (n:2), ulcers in the bulbus (n:4), erosive gastritis (n:11), esophagitis (grade A-C) (n:5), erythematous gastropathy (n:5) and bile reflux gastropathy (n:3) were noted in patients with diabetic gastroparesis. Erosive findings were more frequent in patients with than without diabetic gastroparesis, and the difference between groups was statistically significant (p<0.05) (Figure 4). Erosive findings were more frequent in patients with than without diabetic gastroparesis, and the difference between groups was statistically significant.*p<0.05).
Figure 3: Distribution of Dispeptic Symptoms of the Groups. There was no significant difference between patients with and without diabetic gastroparesis for presence of bloating, pyrosis, nause, vomiting, nause and vomiting, upper abdominal pain, bloating and abdominal pain, early satiety, bloating and pyrosis, bloating and nause, post-prandial fullness, respectively (p=0.331, p=0.139, p=0.326, p=0.358, p=0.368, p=0.329, p=0.336, p=0.331, p=0.335, p=0.326, p=0.635).
HP positivity (n: 3/5) were detected in patients without and with diabetic gastroparesis, respectively. While Upper gastrointestinal biopsy material for HP status were investigated, it was showed that there was no statistically significant difference between patients with and without diabetic gastroparesis (p=0.818).
In this study, we showed that erosive findings in patients with diabetic gastroparesis were as statistically significant more than in patients without diabetic gastroparesis (p<0.05) (Figure 4).
Discussion
Diabetic gastroparesis was described as “gastroparesis diabeticorum” in patients with type 1 diabetes with gastric retention by Kassander in 1958. Diabetic gastroparesis has been associated with advanced type 1 diabetes with poor glycemic control, it is increasingly being recognized in patients with type 2 diabetes. Gastroparesis is characterized by delayed gastric emptying in the absence of mechanical obstruction of the stomach [13]. Symptoms of gastroparesis are variable but may include early satiety, nausea, vomiting, bloating, and upper abdominal pain. Patients with gastroparesis admit to hospital and require more procedures [14].
The predictors for diabetic gastroparesis remain poorly defined, although duration of disease and the presence of diabetic complications are thought to be potentially important [4,15,16]. In one study, no significant association was shown between DM duration and the development of gastroparesis, and there was also no significant association between neuropathy and the development of gastroparesis [17]. Another study suggested that coexisting peripheral neuropathy was significantly associated with the development of gastroparesis, while autonomic neuropathy was not (18). Those researchers also showed that poor glycemic control measured both by self-report and HbA1c was an independent risk factor for upper gastrointestinal symptoms, but the duration and type of diabetes were not significant [18]. A two-year prospective follow-up study observed no clear association between gastrointestinal symptoms and autonomic neuropathy or glycemic control [19]. In our study, we also found no significant association between the two groups regarding duration of DM, presence of diabetic complications or HbA1c (p>0.05).
In a consortium study, cellular changes were shown in the pathogenesis of diabetic gastroparesis [7]. Results of our study suggested that these cellular changes can cause mucosal injury macroscopically. Thus, in one study showed that the pathogenesis of diabetic gastroparesis was as follows: autonomic neuropathy, loss of neuronal nitric oxide synthase leading to loss of nitric oxide, increased oxidative stress with loss of upregulation of protective enzymes such as heme oxygenase-1, loss of ICCs with resultant gastric arrhythmia and delayed gastric emptying, smooth muscle atrophy, loss of insulin-like growth factor (IGF)-1 from smooth muscle, and loss of macrophages expressing heme oxygenase-1 [7]. However, another study showed that there was also a significant correlation between loss of ICCs and enteric nerves in diabetic gastroparesis. In diabetic gastroparesis, loss of ICCs was associated with delayed gastric emptying. It was shown that ICC or enteric nerve loss did not correlate with symptom severity [20-27]. Overall clinical severity and nausea in idiopathic gastroparesis were associated with a myenteric immune infiltrate [28]. Thus, some authors have suggested that full-thickness gastric biopsies could help define specific cellular abnormalities in gastroparesis, some of which are associated with the physiological and clinical characteristics of gastroparesis [27,28]. In our study, it was shown that the spectrum caused by the cellular changes can lead to mucosal damage macroscopically.
HP associated gastritis, peptic ulcer and gastric malignancy are known diseases. When investigating the relationship between HP positivity and presence of gastroparesis, the studies were showed that there was not related to the HP positivity and presence of gastroparesis [29,30]. In another study suggested that there was no relationship between HP positivity and presence of dyspepsia or gastroparesis in uremic patients [31]. In Caballero-Plasencia’s study showed that, there was no relation neither symptoms of dyspepsia nor HP positivity and gastroparesis to solids [32]. On the other hand, Oida’s study showed that postoperative marginal ulcers strongly were associated with gastroparesis than HP positivity and suggested that prevention of gastroparesis was important to reduce the occurrence rate of postoperative marginal ulcer [33]. In present study showed that there was no statistically difference between patients with and without diabetic gastroparesis (p=0.818).
In Parkman’s study, upper gastrointestinal endoscopies were examined retrospectively in 20 patients with diabetic gastroparesis. Nine patients had normal upper gastrointestinal system endoscopy, while 11 patients had pathologic findings [34]. The aim of the present study was to evaluate the relationship between presence of diabetic gastroparesis and erosive findings on endoscopic examination. We found no similar prospective study in the literature in which endoscopic findings in patients with and without diabetic gastroparesis were compared.
In all the studies on the pathogenesis, management and treatment of diabetic gastroparesis, upper gastrointestinal endoscopic examination studies are lacking in patients with diabetic gastroparesis. We aimed to show the effect of diabetic gastroparesis on endoscopic findings, and we showed that diabetic gastroparesis may cause macroscopic mucosal injury. In that case, motility disorders such as diabetic gastroparesis can cause cellular changes and inflammatory process. In course of time, mucosal injury may lead to macroscopic findings.
In conclusion, the presence of HP positive may not be associated with diabetic gastroparesis. We suggest that, the patients with DM would be benefit from screening EGD for signs of developing gastroparesis. Finally, we would suggest that non-erosive findings at EGD may be a precursor to development of gastroparesis. High volume studies in the future must clarify the relationship between diabetic gastroparesis and mucosal injury.
Acknowledgement
Our study is presented as an abstract in American Diabetes Association scientific sessions in 5-9 June 2015 Diabetes vol.64, ADA, 2015.
References
- Parkman HP, Hasler WL, Fisher RS (2004) American Gastroenterological Association medical position statement: diagnosis and treatment of gastroparesis. Gastroenterology 127: 1589-1591.
- Park MI, Camilleri M (2006) Gastroparesis: clinical update.Am J Gastroenterol 101: 1129-1139.
- Horowitz M, Wishart JM, Jones KL, Hebbard GS (1996) Gastric emptying in diabetes: an overview Diabet Med 13: S16-22.
- Camilleri M (2007) Clinical practice. Diabetic gastroparesis.N Engl J Med 356: 820-829.
- Kong MF, Horowitz M, Jones KL, Wishart JM, Harding PE (1999) Natural history of diabetic gastroparesis.Diabetes Care 22: 503-507.
- Jones KL, Russo A, Berry MK, Stevens JE, Wishart JM, et al. (2002) A longitudinal study of gastric emptying and upper gastrointestinal symptoms in patients with diabetes mellitus. Am J Med 113: 449-455.
- Kashyap P, Farrugia G (2010) Diabetic gastroparesis: what we have learned and had to unlearn in the past 5 years Gut 59: 1716-1726.
- Chang TM, Passaro EP, Su DJ, Hwang CC, Law SL, et al. (1986) Technetium 99m-DTPA microcapsules: a new preparation for gastric emptying studies. Am J Surg 151: 722-724.
- Tougas G, Eaker EY, Abell TL, Abrahamsson H, Boivin M, et al. (2000) Assessment of gastric emptying using a low fat meal: establishment of international control values. Am J Gastroenterol 95: 1456-1462.
- Lundell LR, Dent J, Bennett JR, Blum AL, Armstrong D, et al. (1999) Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut 45: 172-180.
- Dixon MF, Genta RM, Yardley JH, Correa P (1996) Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol 20: 1161-1181.
- Forrest JA, Finlayson ND, Shearman DJ (1974) Endoscopy in gastrointestinal bleeding. Lancet 2: 394-397.
- Kassander P (1958) Asymptomatic gastric retention in diabetics (gastroparesis diabeticorum). Ann Intern Med 48: 797-812.
- Wang YR, Fisher RS, Parkman HP (2008) Gastroparesis-related hospitalizations in the United States: trends, characteristics, and outcomes, 1995-2004. Am J Gastroenterol 103: 313-322.
- Jones KL, Russo A, Stevens JE, Wishart JM, Berry MK, et al. (2001) Predictors of delayed gastric emptying in diabetes.Diabetes Care 24: 1264-1269.
- Samsom M, Bharucha A, Gerich JE, Herrmann K, Limmer J, et al. (2009) Diabetes mellitus and gastric emptying: questions and issues in clinical practice. Diabetes Metab Res Rev 25: 502-514.
- Choung RS, Locke GR, Schleck CD, Zinsmeister AR, Melton LJ, et al. (2012) Risk of gastroparesis in subjects with type 1 and 2 diabetes in the general population. Am J Gastroenterol 107: 82-88.
- Bytzer P, Talley NJ, Hammer J, Young LJ, Jones MP, et al. (2002) GI symptoms in diabetes mellitus are associated with both poor glycemic control and diabetic complications. Am J Gastroenterol 97: 604-611.
- Quan C, Talley NJ, Jones MP, Spies J, Horowitz M (2002 )Gain and loss of gastrointestinal symptoms in diabetes mellitus: associations with psychiatric disease, glycemic control, and autonomic neuropathy over 2 years of follow-up. Am J Gastroenterol 103: 2023-2030.
- Wang XY, Huizinga JD, Diamond J, Liu LW (2009) Loss of intramuscular and submuscular interstitial cells of Cajal and associated enteric nerves is related to decreased gastric emptying in streptozotocin-induced diabetes. Neurogastroenterol Motil 21: 1095-e92.
- Zarate N, Mearin F, Wang XY, Hewlett B, Huizinga JD, et al. (2003) Severe idiopathic gastroparesis due to neuronal and interstitial cells of Cajal degeneration: pathological findings and management. Gut 52: 966-970.
- Ordög T, Takayama I, Cheung WK, Ward SM, Sanders KM (2000) Remodeling of networks of interstitial cells of Cajal in a murine model of diabetic gastroparesis. Diabetes 49: 1731-1739.
- Horvath VJ, Vittal H, Lorincz A, Chen H, Almeida-Porada G, et al. (2006) Reduced stem cell factor links smooth myopathy and loss of interstitial cells of cajal in murine diabetic gastroparesis. Gastroenterology 130: 759-770.
- Forster J, Damjanov I, Lin Z, Sarosiek I, Wetzel P, et al. (2005) Absence of the interstitial cells of Cajal in patients with gastroparesis and correlation with clinical findings.J Gastrointest Surg 9: 102-108.
- He CL, Soffer EE, Ferris CD, Walsh RM, Szurszewski JH, et al. (2001) Loss of interstitial cells of cajal and inhibitory innervation in insulin-dependent diabetes.Gastroenterology 121: 427-434.
- Iwasaki H, Kajimura M, Osawa S, Kanaoka S, Furuta T, et al. (2006) A deficiency of gastric interstitial cells of Cajal accompanied by decreased expression of neuronal nitric oxide synthase and substance P in patients with type 2 diabetes mellitus.J Gastroenterol 41: 1076-1087.
- Grover M, Farrugia G, Lurken MS, Bernard CE, Faussone-Pellegrini MS, et al. (2011) NIDDK Gastroparesis Clinical Research Consortium. Cellular changes in diabetic and idiopathic gastroparesis. Gastroenterology 140: 1575-1585.
- Grover M, Bernard CE, Pasricha PJ, Lurken MS, Faussone-Pellegrini MS, et al. (2012) NIDDK Gastroparesis Clinical Research Consortium (GpCRC). Clinical-histological associations in gastroparesis: results from the Gastroparesis Clinical Research Consortium. Neurogastroenterol Motil 24: 531-539.
- Güvener N, Akcan Y, Paksoy I, Soylu AR, Aydin M, et al. (1999) Helicobacter pylori associated gastric pathology in patients with type II diabetes mellitus and its relationship with gastric emptying: the Ankara study. Exp Clin Endocrinol Diabetes. 107: 172-176.
- Caballero-Plasencia AM, Muros-Navarro MC, Martín-Ruiz JL, Valenzuela-Barranco M, de los Reyes-García MC, et al. (2012) Dyspeptic symptoms and gastric emptying of solids in patients with functional dyspepsia. Role of Helicobacter pylori infection. Hepatogastroenterology 59: 899-902.
- Schoonjans R, Van VB, Vandamme W, Van HN, Verdievel H, et al. (2002) Dyspepsia and gastroparesis in chronic renal failure: the role of Helicobacter pylori. Clin Nephrol 57: 201-207.
- Caballero-Plasencia AM, Muros-Navarro MC, Martín-Ruiz JL, Valenzuela-Barranco M, de los Reyes-García MC, et al. (1995) Dyspeptic symptoms and gastric emptying of solids in patients with functional dyspepsia. Role of Helicobacter pylori infection. Scand J Gastroenterol 30: 745-751.
- Oida T, Kano H, Mimatsu K, Kawasaki A, Kuboi Y, et al. (1995) Gastric marginal ulcer after pancreaticoduodenectomy with pancreaticogastrostomy due to delayed gastric emptying and Helicobacter pylori infection. Scand J Gastroenterol 30: 745-751.
- Parkman HP, Schwartz SS (1987) Esophagitis and gastroduodenal disorders associated with diabetic gastroparesis. Arch Intern Med 147: 1477-1480.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 12587
- [From(publication date):
August-2016 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 11621
- PDF downloads : 966